Abstract
Introduction: Impairment of visual contrast sensitivity is a well-known phenomenon in adult migraineurs. Little is known, however, about whether contrast sensitivity deficits are already present in children with migraine.
Methods: We conducted an exploratory study with 18 children with migraine without aura, in which we tested our subjects’ visual contrast sensitivity. Eighteen age- and sex-matched healthy children served as controls.
Results: Among the youngest subjects (6–10 years) we found no significant differences at any of the spatial frequencies tested, as compared to the controls, whereas from the age of 10 on, migraineurs exhibited significantly poorer contrast sensitivity, especially at the lower spatial frequencies.
Conclusion: To our knowledge, we are the first to report on such a deficit in children, and we conclude that our findings might be interpreted as reflecting an increased vulnerability of the visual system to migraine attacks as part of the migrainous endophenotype.
Introduction
It is established that migraine is often accompanied by various types of visual impairment, beyond the well-known transitory phenomena in the aura phase (1–4). Altered visual contrast sensitivity (CS) is one of the frequently reported abnormalities (5–8). Psychophysical data suggest that in healthy children CS is progressively developing even between 5 and 14 years of age (9). However, although repeated migraine attacks have been reported to strongly interfere with the development of cortical functions (10,11), no study, to our knowledge, has previously directly addressed the development of CS in child migraineurs. We therefore conducted an exploratory study to investigate whether CS alterations are already found in children suffering from migraine. Here we report photopic dynamic CS data from child migraineurs (without aura) aged between 6 and 14 years, as compared to age- and sex-matched controls.
Participants
Characteristics of the studied migraineurs

M = male. F = female.
Methods
We followed the method of Benedek and colleagues (9), except that we measured only the photopic dynamic condition. One reason for this was that the dynamic condition was one of those exhibiting the most protracted development in that study. Also, we wanted to keep testing sessions short, in order to make the length of the procedure tolerable for even the youngest participants. Avoiding the relatively long adaptation period of the scotopic condition, studied by Benedek et al. (9), served the same end. This design allowed us to ensure that a testing session lasted no more than 35 minutes.
Dynamic CS was measured in the photopic condition, at nine spatial frequencies (0.5, 1.2, 1.9, 2.9, 3.6, 4.8, 5.7, 7.2 and 14.3 cycles/degree [c/d]) with a computerized test (Venus, NeuroScientific Corporation, Farmingdale, NY, USA). Stimuli were luminance-contrast gratings with a sinusoidal luminance profile. Contrast was defined according to the Michelson formula (C = [L max − L min]/[L max + L min]). The pattern was reversed at an 8 Hz rate. The display subtended a visual angle of 13 × 13° and was viewed from a distance of 1 m. The maximum contrast was 70.7%. For the measurement of the contrast threshold at each spatial frequency, we used a method of adjustment. First, the contrast was set at 15 dB above the mean normal value. If the participant was unable to detect the grating at this contrast level, the contrast was increased until the child was able to detect the stimulus. The contrast level was then decreased stepwise by 3 dB every 5 seconds until the subject reported that the stimulus disappeared (descending method). Next, contrast was set at 15 dB below the threshold, and then increased until the subject was able to detect the stimulus (ascending method). The whole procedure was repeated five times to obtain a mean contrast threshold at a particular spatial frequency. Different spatial frequencies were tested in a random order between participants. CS was defined as the reciprocal of the contrast threshold.
Data analysis
Given the small number of observations, we applied a non-parametric test (Mann-Whitney U [MWU]) to see if there was a statistically significant difference in CS at each tested spatial frequency between the migraineurs and the control cohort, by age groups. Data analysis was carried out with Statistica for Windows (StatSoft, Tulsa, OK, USA).
Results
The results of comparisons are illustrated in Figure 1. As can be seen, we found no significant differences between migraineurs and controls for either of the tested spatial frequencies in the first age cohort (6–10 years). It was in the second age group (10–12 years) that significant differences were first found by diagnosis, for two frequencies: 1.2 c/d (MWU = 3.5, N
1 = N
2 = 8, p < .05, two-tailed) and 1.9 c/d (MWU = 1.5, N
1 = N
2 = 8, p < .01, two-tailed). Analysis of the third age group (12–14 years) yielded similar results. Statistically significant difference was found at 1.2 c/d (MWU = 2.5, N
1 = N
2 = 8, p < .05, two-tailed) and 1.9 c/d (MWU = 2.5, N
1 = N
2 = 8, p < .05, two-tailed). To see if there might be any link between disease duration and CS performance in the migraineur group, we also calculated Spearman’s R for 1.2 and 1.9 c/d. The results suggest no link between disease duration and CS performance (1.2 c/d R = 0.09; 1.9 c/d R = 0.12). Age of participants, however, proved to be significantly correlated with the spatial frequencies of interest in controls (1.2 c/d R = 0.6; 1.9 c/d R = 0.54), whereas this relationship did not reach statistical significance in migraineurs (1.2 c/d R = 0.46; 1.9 c/d R = 0.4).
Contrast sensitivity at the tested spatial frequencies, test versus control groups, by age (a: 6–10 years; b: 10–12 years; c: 12–14 years).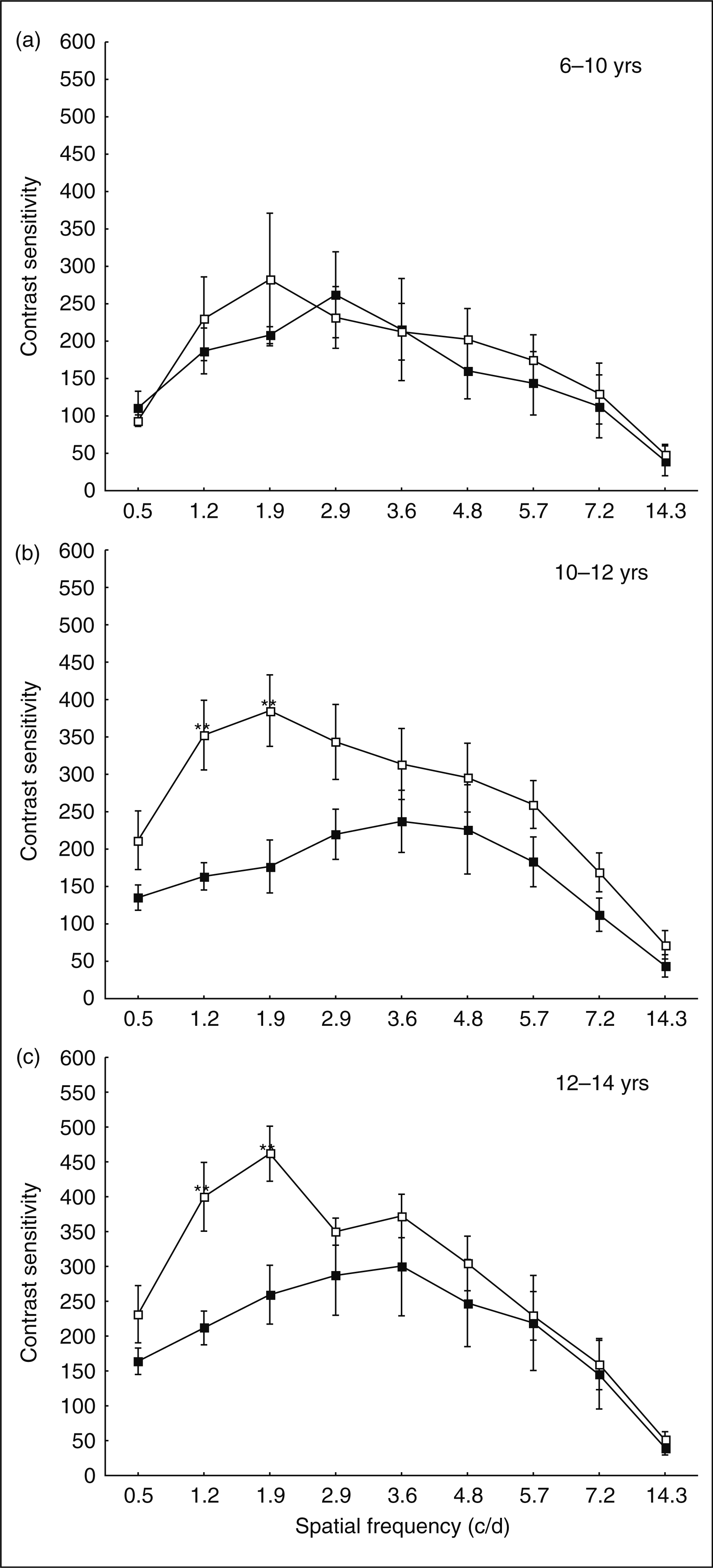
Discussion
To our knowledge, we are the first to report on data suggesting that the CS deficit described in adult migraineurs (6) is to be observed already in childhood, especially at low spatial frequencies.
The finding that significant differences between the two groups were first to be seen in the 10–12-year-old age group came as no surprise, given that this is consistent with the finding that in healthy children the first significant developmental step in terms of low spatial frequencies occurs between the ages o 5 and 10 (9). Why in the age group of 6–10-year-olds we found no significant differences might be due to a number of possible reasons. First, although migraine does exist in such a young age (13–15), the diagnosis poses challenging dilemmas (16), even following the IHS criteria as closely as possible. Thus, it cannot be ruled out that 'false positives' were included in the sample, and in turn, an existing but not robust difference was rendered insignificant, given the small number of observations. Another factor is disease duration, which was quite short in the first cohort (∼0.9 years). We do not find it likely that such a short exposure to migraine attacks should result in significant deterioration. However, this is not to say that repetitive migraine attacks themselves offer the ultimate explanation for the described CS deficits, as there is some evidence indicating that the length of migraine history is a factor (5).
The results for the two older age groups are less ambiguous. That migraineurs are lagging behind at low spatial frequencies is obvious. However, it must be noted that even if statistically significant difference characterizes only two frequencies in both groups, robust differences are to be seen over a wide range of frequencies (see Figure 1). Why 1.2 c/d and 1.9 c/d are affected the most we consider a theoretically unimportant problem; possibly, this is an epiphenomenon of our measurement, reflecting a broader tendency of lower spatial frequencies in general being affected, as we found no reference in the literature to such specific differences.
Correlations show that CS as measured here does develop with age in both migraineurs and controls, even if the correlation in migraineurs is weaker, suggesting an impaired developmental course. That the length of migraine history and CS are not correlated we do believe to be a result of the low number of eligible patients, which prevents us from drawing a sound conclusion regarding this relationship.
What, then, might be the explanation for the deficit found in our study? Known evidence suggests a genetically determined vulnerability model. Repeated migraine attacks seem to interfere with CS in a progressive manner (6). This is corroborated by morphological alterations reported in migraine (17,18). As migraine is known to be a familial disease, it is possible that the explanation for the early appearance of this CS difference is that it reflects an endophenotype. Di Clemente et al. (17) make a similar argument in connection with the interictal habituation deficit of the nociceptive blink reflex in migraineurs. However, even if we consider the deficit described here as reflecting an endophenotype, it remains an open question whether increased vulnerability to insults or a relatively stable (fixed) degree of CS decrease constitutes that endophenotype. The former scenario, of course, predicts that the longer the migraine history is, the more pronounced the CS deficit, which—despite the relative paucity of research in this direction—seems to be the case indeed. Therefore, we propose that migraineurs might be extremely vulnerable to the neurovascular challenge posed by the attacks, and that this vulnerability might be genetically determined. Of course, if migraine attacks can bring about a progressive deterioration in adult CS performance (indicating at least some functional damage to the underlying neural structures), they must also interfere with the development of the immature visual system. Indeed, our results show a weaker age-performance correlation in migraineurs than in controls, suggesting greater variability and a less homogenous developmental pattern.
The above argument, of course, raises the question of the affected structures. Certainly, these must be structures with a protracted course of maturation, one reaching well beyond the onset of migraine in childhood. During the last decade the dogma of the early completion of visual development has been challenged, and by today it has been proven that different functional aspects of vision show different patterns of gradual development, sometimes reaching as far as into adolescence (19). These developmental differences are often discussed in terms of the different maturation patterns of the magnocellular/parvocellular pathways (5,19). Viewed in this framework, our results would suggest a magnocellular deficit. However, in agreement with recent criticism (6,7), we avoid linking our psychophysical findings to one particular pathway, and suggest that for such associations to be possible, testing methods need refinement.
In conclusion, our exploratory study has shown that visual contrast sensitivity in the tested child migraineurs exhibits similar deficits to those described in adults suffering from migraine headache. This finding is consistent with the widely accepted concept of migraine as a disease causing early functional deficits (10,11), and it can also be well explained in terms of genetics.
