Abstract
Objectives: We aimed to further elucidate the functional and attentional regulation of cortical excitability in migraine patients.
Methods: We investigated the cortical silent period (CSP) after transcranial magnetic stimulation as a measure of cortical inhibition under three conditions: resting condition, cortical preactivation during reaction preparation, and during the post-processing of a motor response using a visual contingent negative variation paradigm in adults with migraine with aura, migraine without aura and healthy controls.
Results: CSP was reduced in individuals with migraine with aura and unaffected in those with migraine without aura under resting conditions. Along with the intensity of transcranial magnetic stimulation, CSP increased equally in all groups (equal slopes). Furthermore, the functional challenge by a contingent negative variation task requiring focused sustained attention led to a comparable reduction of CSP duration in all groups.
Discussion: Our data provide further hints towards the conclusion that a specific cortical inhibition deficit in migraine with aura but not migraine without aura is due to a tonic imbalance and not related to increased reactions to phasic stressors. Given that CSP at rest is related to GABA-ergic inhibition whereas the CSP reduction during late contingent negative variation is thought to be related to dopaminergic disinhibition in the basal ganglia, our results point towards reduced GABA-ergic cortical inhibition related to dysfunctional thalamo-cortical loops, especially in migraine with aura.
Keywords
Introduction
Migraine is a very common kind of primary headache (1) leading to great suffering of the affected patients. At present the most widely accepted pathophysiological migraine model states that neuronal dysregulation leads to a secondary vascular reaction which causes the headache (2). This dysregulation is also present during the interictal interval (the interval between migraines) shown by, for example, exaggerated responses to light or noise (3–4). Coexistence of deficient habituation and reduced mitochondrial energy reserves may lead to an imbalance of the brain metabolic homeostasis (5). Positron emission tomography (PET) studies indicate that there might be an anatomical structure in the brain stem or midbrain involved as a possible migraine generator (6,7). The specific role of single anatomical components within a pathophysiological cascade has not been fully elucidated. Abnormal cortical excitability has also been supposed to play a central role in etiopathogenesis of migraine (8). A large number of neuropsychological studies investigated cortical activation in individuals suffering from migraine (9,10). The principal and most reproducible finding was a lack of habituation to repeated sensorial stimulation in migraine patients (11–16). It still remains a controversial issue whether this effect is associated with cortical hyper-excitability or a decreased cortical pre-activation level (17). Previous studies have resulted in contradictory findings of cortical hyper-excitability versus cortical hypo-excitability (17–19).
Hypo-excitability and a reduced pre-activation level have been primarily observed in studies assessing sensory evoked potentials such as auditory (AEP), visually (VEP) or somatosensory (SEP) evoked potentials (12,20–22), especially during the first few trials when long-lasting habituation paradigms with several trial blocks were applied.
Furthermore, some studies reported increased phosphene thresholds and accordingly a lower prevalence of phosphene perception in individuals with migraine (23–25), again indicating cortical hypo-excitability. An excitatory high frequency (10 Hz) repetitive transcranial magnetic stimulation (rTMS) was able to normalize VEP habituation in migraineurs for up to several hours (15). On the other hand, numerous neurophysiological studies suggested cortical hyper-excitability as a pivotal feature in migraine. Some studies found reduced phosphene thresholds in patients with migraine with aura (MA) (26–28) and in children suffering from migraine without aura (MO) (29), as well as an increased amplitude of contingent negative variation in the headache-free interval (30–32).
Multiple intrinsic and extrinsic factors may influence the level of cortical excitability in individuals with migraine. Contradictory findings might be caused by ‘functional hyper-excitability’ related to stressors and increased attention challenge accompanied by hypo-excitability under resting conditions with reduced habituation during sensorial activation (21). Similar to conditions of sensory de-afferentiation, changes in cortical excitability may be caused by impaired inhibitory circuits (9).
The cortical silent period (CSP) is a TMS parameter to measure cortical inhibition (33–35). Previous studies on the CSP in migraine found a reduced CSP at rest in individuals with MA (36–39), although this finding was not always replicated (24,29,40–42).
To test the idea of a ‘functional cortical hyper-excitability’ in migraine, we hypothesized that cortical inhibition will be reduced only under conditions of stress or focused attention in individuals with migraine. Therefore, we investigated the CSP after TMS as a measure of cortical inhibition under resting conditions, cortical pre-activation during orienting (early or initial contingent negative variation, iCNV) and focused attention (late CNV) as well as during the post-processing of a motor response (postimperative negative variation, PINV) (43–45) in adult individuals with MA and MO and healthy controls (CO) to obtain differential information about the functional regulation of cortical excitability in individuals with migraine.
Methods
Subjects
Clinical characterization of migraine patients

MO, migraine without aura; MA, migraine with aura.
All measurements were obtained during the headache-free interval with at least 3 days distance to the last or to next following migraine attack (subjects were interviewed 3 days after TMS and if necessary, the measurement was repeated or the subject was excluded). Three MA individuals had to be excluded for this reason because they did not agree to repeat the TMS; for two MO individuals the measurement could be repeated. Female subjects were measured during mid menstrual cycle. All subjects provided written informed consent according to the Declaration of Helsinki. The study was approved by the local ethics committee.
Experimental procedure
TMS was applied using monophasic single stimuli from a Magstim 200 (Magstim Ltd., Dyfed, UK) by a circular coil with 90 mm diameter. The coil was centred over the vertex and adjusted so that the MEP amplitude of the left first dorsal interosseous muscle (FDI) was maximal. The current flow was clockwise to excite the right motor cortex. The resting motor threshold was determined as the lowest intensity that produced a MEP-amplitude of not less than 50 µV in at least five of ten stimulations (49).
In the first part of our study we stimulated with 100%, 115% and 130% of the motor threshold under resting conditions (no task, pre-contraction of the left FDI with 20% maximum force under acoustic feedback) and recorded the resulting surface electromyography (EMG) response in the left FDI with Ag/AgCl electrodes (impedances <10 kΩ, filter 20–10,000 Hz, active electrode on the left FDI muscle belly, reference electrode on the basic phalanx of the middle finger). Fifteen stimulations at each intensity were performed and averaged.
In the second part of our study a visual CNV paradigm was applied (Figure 1, Gentask, Neuroscan Stim software package, TX, USA). A warning stimulus (S1) was followed by an imperative stimulus (S2) with a constant stimulus onset asynchrony of 3 s. Subjects had to respond as fast as possible by pressing a mouse button with their non-dominant left hand (index finger flexion). The CNV is characterized by a slow negative shift in the electroencephalography (EEG) between a warning and an imperative stimulus consisting of two components. The early part of the CNV wave (initial CNV, iCNV) appears immediately after the warning stimulus and reflects an orienting response. The later part of the CNV wave (late CNV, lCNV) corresponds to motor preparation and anticipation of the imperative stimulus. CNV is followed by another component, the post imperative variation (PINV), related to the evaluation of response control and the correctness of the response.
Transcranial magnetic stimulation (TMS) during a visual contingent negative variation paradigm: schematic overview. A visual imperative stimulus S2 follows a visual warning stimulus S1 with a constant interstimulus interval of 3 s. Subjects were required to respond as fast as possible by an unilateral button press. Following S1, the contingent negative variation (CNV) can be divided into an early (iCNV) and a late (lCNV) component while the postimperative negative variation (PINV) following S2 is small or absent for responses with the non-dominant hand. Time points of TMS are marked with flashes.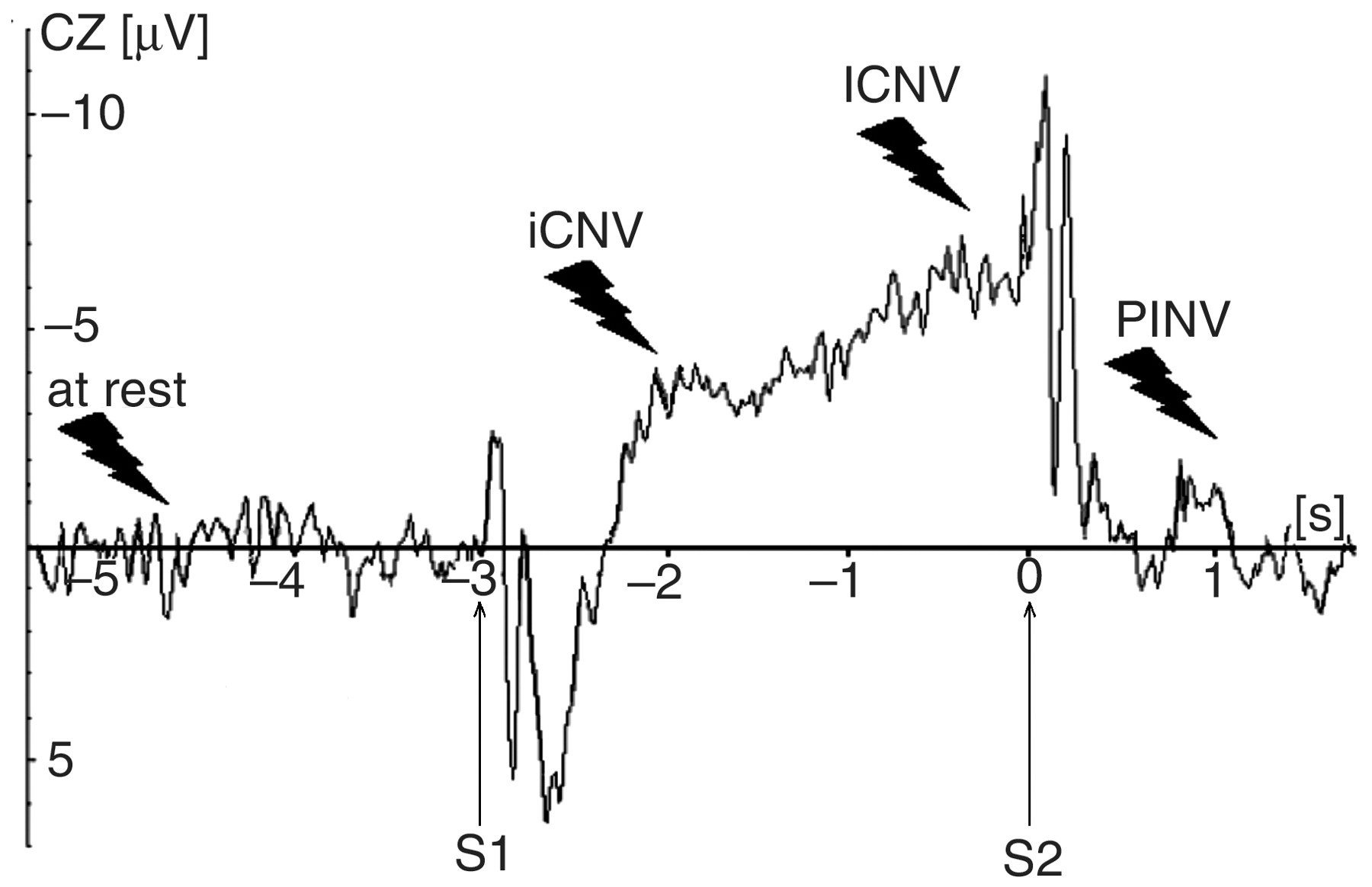
The inter-trial intervals varied randomly between 10 s and 14 s. All participants were asked to keep the FDI constantly tensed at 20% maximum force throughout the whole CNV recording session, which was supported by acoustic EMG feedback. Seventy-five TMS stimulations with 100% resting motor threshold followed in a pseudo-randomized order under resting conditions during the inter-trial intervals, during early contingent negative variation (iCNV, 700 ms after the warning stimulus), during late contingent negative variation (lCNV, 200 ms before the imperative stimulus was expected) and during postimperative negative variation (PINV, 850 ms after the imperative stimulus): Fifteen stimulations were performed at rest and 20 each during iCNV, lCNV and PINV.
In trials with TMS during late CNV, the imperative stimulus was delayed for 0.3 s in order to assure that between TMS and the imperative stimulus there would be 0.5 s distance to avoid any possible influences of the imperative stimulus on TMS-related measurements.
CSP was determined according to the standardized method described by Garvey et al. (50): EMG was rectified and averaged. CSP started when the rectified EMG amplitude dropped below 2.2 standard deviations from the baseline mean for five consecutive data points and ended when it reached this border again and for the next 5 ms at least 50% of the data points were above this limit. This procedure was controlled visually to assure that the same criteria were applied for all intra-individual comparisons.
EEG data were recorded using a Synamps DC EEG amplifier (Neuroscan Inc., Texas, USA) and 20 Ag/AgCl electrodes, which were named according to the extended international 10-20 system. Linked mastoids served as recording reference; the sampling rate was 500 Hz. Electrode impedances were kept below 5 kΩ. Data were filtered offline with a 16 Hz high cutoff filter (24dB/octave) and divided in segments of 7.5 s duration (from –4 s to +3.5 s relative to imperative stimulus).
iCNV was determined by detecting the local maximum in the interval from 550 ms to 750 ms after the warning stimulus and then calculating the average amplitude in the time segment from a 200 ms window centred on this peak (51). Owing to the artefacts produced by TMS in the EEG recordings and the limited duration of the iCNV component, no analysis of iCNV amplitude was possible in trials with TMS during iCNV.
For stimulations during lCNV, a segment of 7.8 s was chosen (from –4.3 s to +3.5 s relative to imperative stimulus). The baseline was determined during the 1000 ms preceding the warning stimulus. Late CNV was calculated using the mean amplitude at vertex (Cz) in the time interval 200 ms prior to the imperative stimulus (43,52) in trials without TMS and as the mean amplitude 200 ms prior to TMS in trials with TMS. We analysed data from 15 CNV trials without TMS pulses to avoid any biases caused by TMS stimulation artifacts. Because CNV trials with and without TMS occurred in random order, subjects did not know beforehand whether a TMS stimulation would follow during the CNV interval. PINV was very small because of the simultaneous muscle contraction and the use of the non-dominant left hand (53) and thus no detailed analysis is presented.
EMG was recorded by a Jaeger-Toennies Neuroscreen (Germany) with a bandpass filter of 20–10,000 Hz, which was synchronized with TMS and EEG recordings by Transistor-Transistor Logic (TTL) pulses (49). Epochs of 800 ms duration (240 ms baseline before to 560 ms after the TMS pulse) were stored for offline analysis. In addition to CSP duration, rectified background EMG (extent of muscle pre-contraction) was calculated as the mean amplitude 240 ms before the TMS pulse.
Because of technical triggering problems that produced additional EMG sweeps, which made a proper assignment of EMG and EEG recordings impossible, for this part of the study we had to exclude two CO, two MO and one MA individuals.
Statistical analysis
For statistical evaluation of the first part of the study, CSP at rest (tonic baseline inhibition level), a repeated-measures analysis of variance (ANOVA) with the between-subject factor diagnosis (CO, MO, MA) and the repeated measurements factor stimulation intensity (100%, 115% and 130% of the resting motor threshold) was calculated. Moreover, linear TMS-intensity slopes were calculated and compared between the three diagnostic groups in an ANOVA. For the second part of the study, CSP under attentional challenge, a repeated measures ANOVA with the between-subject factor diagnosis and the repeated measurements factor functional state of activation (rest, early CNV, late CNV, PINV) was applied. Greenhouse-Geisser correction was applied where appropriate. Newman Keuls post hoc tests were used to further differentiate main effects and interactions. We checked for both parts of the study (TMS at different intensities, TMS during the CNV task), whether any effect of diagnosis would still be found when age was added as a linear predictor in order to control for age effects. A repeated-measures ANOVA with the between subjects factor diagnosis and the combined repeated measurements factors functional state of activation or stimulation intensity was used to examine whether a main effect of diagnosis was present in both parts of the study.
Results
The resting motor threshold (RMT) of subjects was 45 ± 10% of the maximum stimulator output for CO (mean ± standard deviation), 47 ± 9% for MO and 46 ± 9% for MA. There were no significant differences between the groups (F(2,45) = 0.2; p = 0.81).
Experiment 1. Stimulation under resting conditions with different intensities
Representative samples of CSP are shown in Figure 2. For an overview of the group CSP data at different stimulation intensities see Table 2 and Figure 3.
Cortical silent period (CSP) at rest. Representative sample EMGs of a migraineur without aura (MO, a), a migraineur with aura (MA, b) and a healthy control subject (CO, c). For better visualization CSP onset and offset are marked by arrows. Cortical silent period (CSP) duration at different stimulation intensities. Migraineurs with aura (MA) had significantly shortened durations of CSP compared with healthy controls (CO) and migraineurs without aura (MO). Note the parallel increases in CSP duration with increasing transcranial magnetic stimulation (TMS) intensity in all groups. Error bars indicate 95% confidence intervals. RMT, resting motor threshold. CSP duration (ms) at different stimulation intensities CO, healthy controls; MO, migraine without aura; MA, migraine with aura; RMT, resting motor threshold.
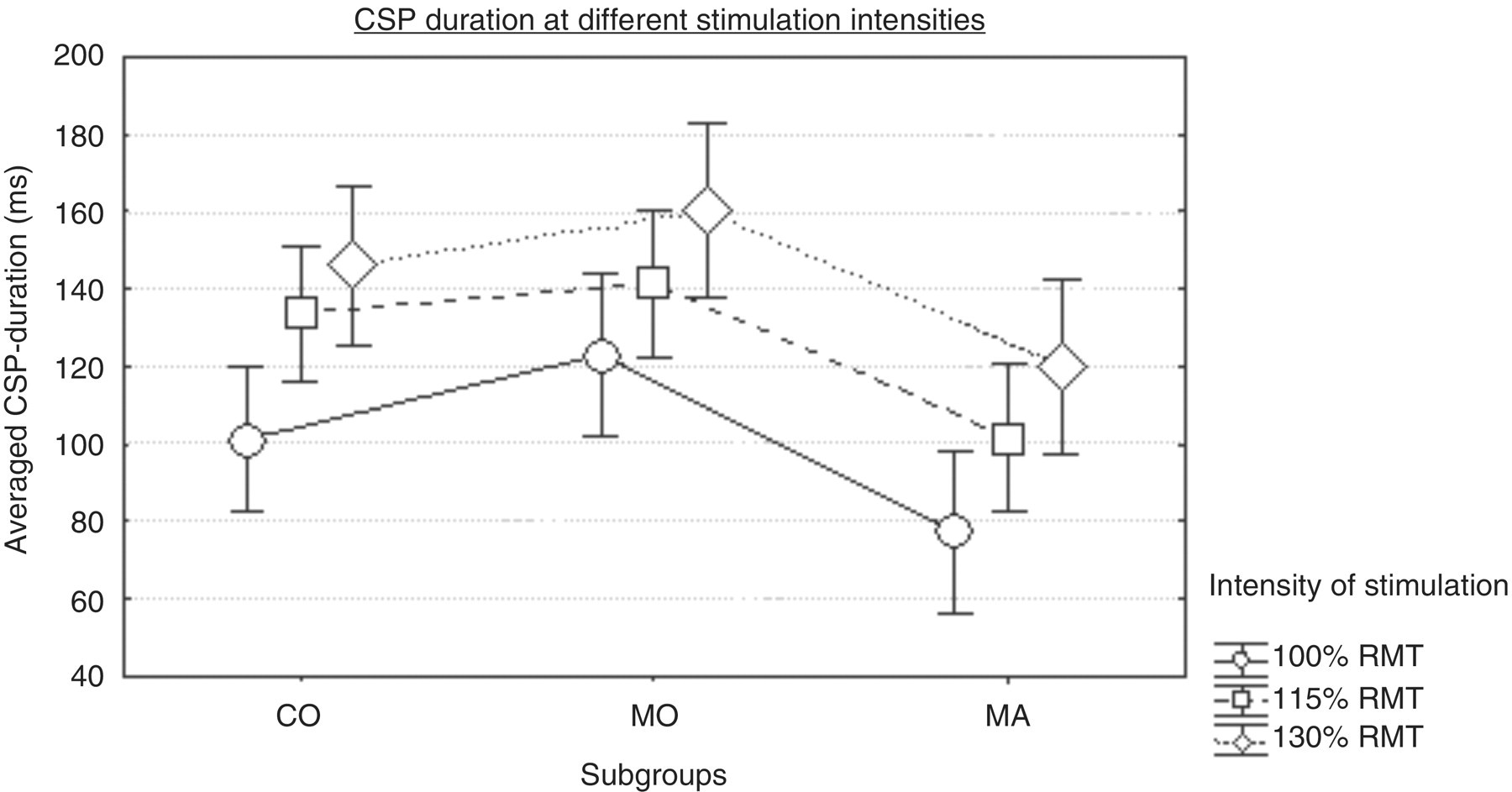

Higher stimulation intensity led to longer duration of CSP in all groups (main effect stimulation intensity F(2,90) = 48.7; p = 0.00001). Moreover, MA showed a reduced CSP compared with CO and MO (main effect diagnosis F(2,45) = 5.1; p = 0.01; post hoc analysis with Newman-Keuls test: MA versus MO p = 0.01; MA versus CO p = 0.04). An ANOVA showed that the factor diagnostic group had no significant effect on the TMS-intensity slopes (F(2,45) = 0.2; p = 0.82; CO 22.5 ± 15.4 ms per 15% RMT; MO 18.7 ± 16.0 ms per 15% RMT; MA 21.3 ± 22.0 ms per 15% RMT). There were no differences in pre-contraction EMG level between the diagnostic groups (F(2,45) = 0.1; p = 0.89). The main effect of the migraine diagnosis remained significant also when we controlled for age effects by adding age as an additional linear predictor (F(2,44) = 3.8; p = 0.03). There were no significant overall age effects on CSP duration in the age range of our sample (F(1,44) = 0.03; p = 0.87). Although there was an interaction between age and stimulation intensity (F(2,88) = 4.9; p = 0.02, Greenhouse Geisser epsilon = 0.78), univariate ANOVAs showed that for no TMS intensity was there a significant influence of age (100% RMT F(1,44) = 2.1; p = 0.16; 115% RMT F(1,44) = 0.3; p = 0.60; 130% RMT F(1,44) = 0.2; p = 0.67). Controlling for gender by an additional covariate did not change any of the above mentioned results either. Disease duration did not correlate with CSP duration at any TMS intensity (all r < 0.08; p > 0.7).
Experiment 2. Focused attention challenge (CNV paradigm)
For group mean CSP durations and standard deviations during iCNV (‘orienting’), during lCNV (‘focused attention’), during PINV (‘response evaluation’), and during the inter-trial intervals (‘rest’) see Table 3 and Figure 4.
CSP duration during the contingent negative variation (CNV) paradigm. Transcranial magnetic stimulation (TMS) was performed at rest (baseline, BL) during the inter-trial intervals, and during early contingent negative variation (iCNV), late contingent negative variation (lCNV) and postimperative negative variation (PINV). Migraineurs with aura (MA) tended towards shortened CSP durations under all conditions. Furthermore a significant shortening of CSP during late CNV independent of the diagnostic group was found. Error bars indicate 95% confidence intervals. CO, healthy controls; MO, migraineurs without aura. CSP duration (ms) during CNV paradigm CO, healthy controls; MO, migraine without aura; MA, migraine with aura; iCNV, initial contingent negative variation; lCNV, late contingent negative variation; PINV, postimperative negative variation.
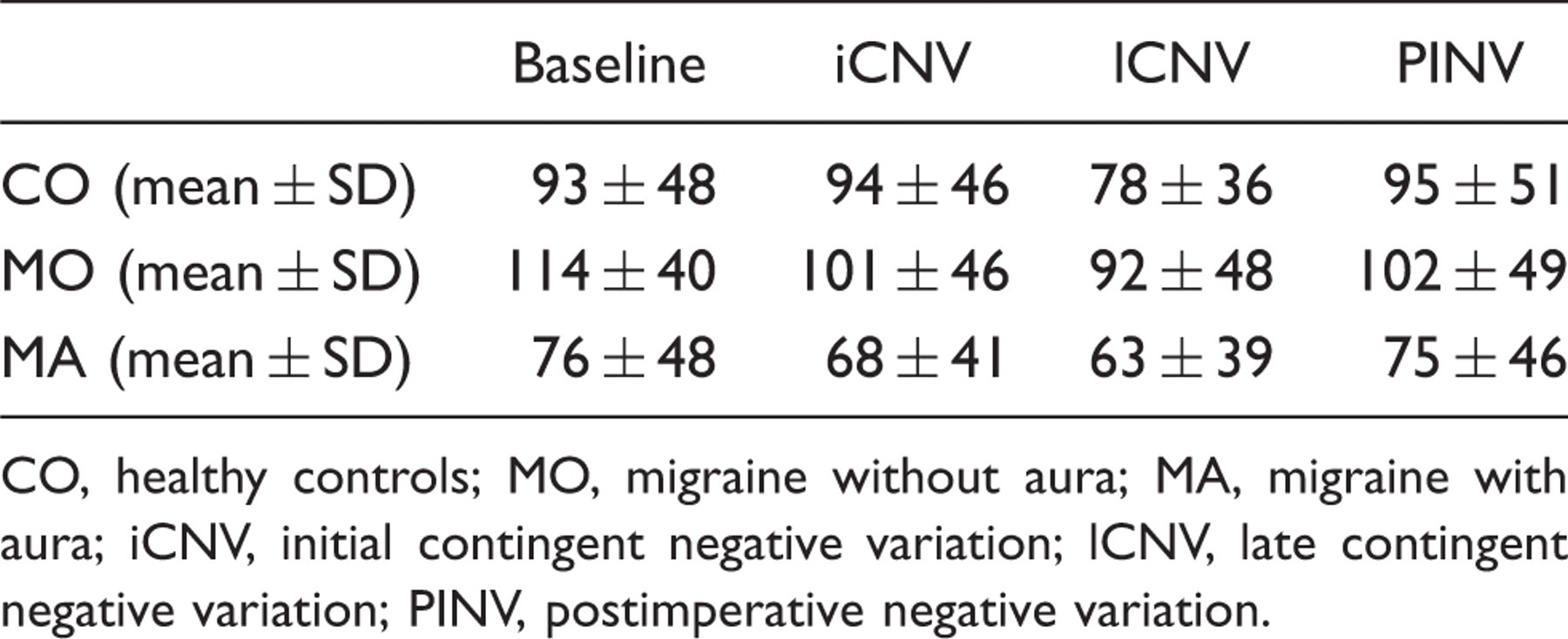
CSP was reduced during lCNV independent of diagnostic group (main effect functional state F(3,120) = 11.5, p = 0.00002, Greenhouse-Geisser epsilon = 0.7). When we controlled for age effects by adding age as a covariate, there was an interaction between the diagnosis and the repeated measurement factor functional state of activation (F(3,38) = 3.4; p = 0.03). Newman Keuls post hoc tests showed that CSP during iCNV was significantly shorter than CSP at rest in MO (p = 0.03), but not in COs (p = 0.92) or MA (p = 0.28). Further significant effects were not observed, although MA showed a reduced CSP in all states of activation.
When the CSP data of both parts of the study (TMS at different stimulation intensities; TMS during the CNV task) were analysed together, a consistent reduction of the CSP in the MA group was observed despite the fact that five subjects could not be included in this analysis because of missing data for the CNV task (main effect diagnosis F(2,40) = 3.4; p = 0.04).
EEG data
iCNV and lCNV amplitude between groups

CO, healthy controls; MA, migraine with aura; MO, migraine without aura; iCNV, initial contingent negative variation; lCNV, late contingent negative variation.
Discussion
In this study we investigated the CSP after TMS under resting conditions, pre-activation (early and late CNV) and activation (postimperative negative variation) in MA, MO and CO to obtain further insights in the role of cortical inhibition in adults with migraine. The major finding of this study is a reduced CSP in MA compared with CO and MO. Increasing TMS intensity led to parallel increases in CSP duration in all diagnostic groups without any apparent slope differences. Task-related changes in alertness could most likely account for the slightly shorter CSP durations during experiment 2 (inter-trial interval of the CNV task as ‘rest’ condition) compared with experiment 1 (varying intensity of TMS at rest); that is, the subjects were waiting for the next trial during the inter-trial intervals in experiment 2. Most importantly, when we controlled for age effects, we found an interaction between functional state of activation and diagnosis, in that MO but not CO showed a reduced CSP duration as early as during the orienting reaction following the warning stimulus (iCNV) and not only during response preparation (lCNV). This is in line with previous findings of an abnormal orienting response in MO (29,52) and shows that abnormally strong orienting reactions in MO may go together with slight reductions in cortical inhibition levels. In our visual CNV task, however, we did not find abnormalities in iCNV amplitude, possibly because this CNV component is much more pronounced in auditory CNV tasks. We had decided to use a visual CNV task in this study in order to separate motor and sensory activity well, because auditory activation in the temporal cortex often projects to frontal or fronto-central scalp areas.
The CSP after TMS is a measure of cortical inhibition (54–56), which is modulated by the inhibitory neurotransmitter GABA (57). Reduced concentration of GABA leads to a shorter CSP (58) whereas increased concentration, for example by TMS stimulation (38) or GABA agonists (59), results in an extended CSP. Excitatory parameters such as motor threshold or motor evoked potentials are not influenced by GABA in this way (57,60). Thus a shortening of CSP indicates a dysfunction of inhibitory mechanisms and indirectly points towards a preponderance of excitatory effects (41).
Previous studies reported partly contradictory results. Some studies found no differences of CSP duration in either MO (24,29,40,61) or MA (24,42,61) compared with healthy controls under resting conditions, other studies reported a reduction of the CSP especially in MA (36–39). Only one study found an extended CSP in chronic migraineurs with relatively frequent migraine attacks (at least on 15 days per month during the previous 3 months) (41).
Differences between the studies might be explained by the differing diagnostic and inclusion criteria of individuals with migraine. For example, one study (41) measured the duration of CSP only in MO and chronic migraine but not in MA. Other studies (29,40) concentrated only on children and females with MO and did not observe differences in CSP between MO and CO, as in the present study. In contrast, studies on subjects with MA (37,38) reported a reduced CSP, a finding that was also replicated by our study. Therefore, our study provides further evidence that cortical inhibitory deficits are more specifically related to aura phenomena than to migraine headaches themselves.
A recent study (62) provides additional support for a functional hyper-excitability in MA using a TMS technique termed magnetic suppression of perceptual accuracy (MSPA). MA with aura had shallower MSPA profiles, indicating functional hyper-excitability caused by impaired inhibition, whereas MO showed regular inhibitory mechanisms, replicating findings of a previous study (63).
Reduced CSP and flattened MSPA profiles are both thought to be linked to impaired GABA-ergic intracortical inhibition. This GABA-ergic impairment may also have a crucial role in the initiation of cortical spreading depression (CSD), which is considered as the pathophysiological basis of the aura phenomenon in migraine (64–66). Taken together, an inhibitory deficit and a shortened CSP seem to be specifically related to spreading depression and the aura phenomenon at least in a considerable subgroup of MA patients. As some studies found no differences in CSP in MA (24,42,61), the GABA-ergic inhibitory deficit seems to not necessarily always affect the hand motor area but seems to be limited to the visual cortex in some individuals with MA (61). Furthermore, a variety of neurotransmitter systems, especially glutamatergic pathways (67,68), may also be implicated in the aura phenomenon and the initiation, spread and resolution of CSD.
We have observed a parallel increase in CSP with incremental stimulus intensities across diagnostic groups. Although an increase in CSP duration with higher TMS intensities has been reported before in healthy subjects (29,35,69–72) and in children suffering from migraine without aura (29), we have shown for the first time that the slopes of the CSP-intensity curves were very similar (if not equal) across MO, MA and CO subjects. This points towards the hypothesis that the number of inhibitory cells is not reduced in MA, but rather the baseline tonic inhibitory level seems to be affected (as indicated by the reduced baseline CSP in MA), altering the balance between excitatory and inhibitory systems. This might be caused by dysfunctionally hypo-active thalamo-cortical loops (36) related to a reduced thalamo-cortical facilitation of inhibitory motor interneurons from the basal ganglia.
The differences in resting state CSP between the diagnostic groups cannot be accounted for by differences in muscle pre-contraction: the CSP is thought to be largely independent of the preceding muscle pre-contraction (35,71). Furthermore, pre-TMS EMG amplitude was comparable in the three diagnostic groups. Beyond that, gender effects can also be excluded, as the two migraine groups (MO and MA) were gender-matched but clearly differed in their CSP. In agreement with our finding that adding gender as covariate did not change the results, a previous study had not found gender effects on CSP either (73).
Disease duration was not related to CSP duration in our study. In previous studies contradictory results have been reported (36,41).
Regarding age effects on CSP, shorter CSPs in elderly subjects have been described (74); however, these subjects were considerably older than the subjects in our sample (mean age 71 years). We did not find significant influences of the factor age in our adult subjects, which is in agreement with a previous study (73). We think that during adult age (about 20–60 years), age effects on CSP are very small, if there are any at all.
Other factors possibly influencing cortical excitability such as contemporary migraine attacks (75,76) or level of ovarian hormones (77) were ruled out by our study design (measurements only in the attack-free interval and always in the same phase of the menstrual cycle).
In the second part of our study we investigated the influence of a stress challenge by focused attention and response preparation using a visual CNV paradigm. CSP was reduced during late CNV compared with baseline, iCNV and PINV. MA again showed a shorter CSP duration compared with controls under all conditions. However, this was not due to the stress challenge or focused attention task. Reduced CSP during lCNV in all diagnostic groups might result from reduced cortical inhibition during motor preparation (54). Late CNV can be understood as motor preparation and sensory attention towards the imperative stimulus S2 (78), and thus as a phase of increased cortical excitability. According to a widely accepted model of CNV generation, this cortical pre-activation might be mediated by a phasic, dopaminergic-based disinhibition in the basal ganglia (71,79,80). This phasic disinhibition loop, which subserves focused attention and the regulation of cortical pre-activation, was not altered in MA. However, when we controlled for age effects, we found that patients suffering from MO did show a CSP reduction during the orienting reaction and not only when the response movement was imminent.
In summary, we observed a reduced tonic baseline inhibition level in MA at rest. This reduced inhibition was not influenced or produced by a stress challenge and a focused attention task. Because CSP at rest is related to GABA-ergic inhibition whereas the CSP reduction during late CNV is thought to be related to dopaminergic disinhibition in the basal ganglia, our results point towards reduced GABA-ergic cortical inhibition related to dysfunctional thalamo-cortical loops, especially in MA patients.
Footnotes
Acknowledgements
We thank Kai Kress, Heidelberg, for his support with manuscript editing.
Funding
This work was supported by the Young Medical Investor Award of the Medical Faculty of the University of Heidelberg to SB.
Conflict of interests
The authors declare that there is no conflict of interest.
