Abstract
We studied the excitability of the motor cortex using transcranial magnetic stimulation (TMS) in 12 patients with migraine with aura (MA) and nine patients with familial hemiplegic migraine (FHM). Motor thresholds at rest, the duration of the cortical and peripheral silent period and intracortical inhibition and facilitation using paired-pulse TMS at intervals of 2 to 15 ms were measured with patients free of attacks for at least 48 h. In contrast to previous reports we could not find any significant differences between patient groups and compared to controls (n = 17) in the parameters tested. The results suggest that there are no interictal changes of excitability of the motor cortex in migraine. This study does not support the concept of general cortical hyperexcitability in migraine secondary to a genetic predisposition or a structural alteration of inhibitory interneurones in the cortex due to repeated parenchymal insults during attacks.
Keywords
The pathogenesis of the cerebral dysfunction in migraine is the subject of controversial discussions. Neurological symptoms like visual auras might be due to mechanisms similar to spreading depression (1), caused by ictal hypoperfusion (2, 3), or due to generalized hyperexcitability followed by an inhibition of the cortex (4, 5). Parallel to this a decrease in cerebral blood flow can be observed. As one possible mechanism favouring hyperexcitability a selective damage to inhibitory γ-aminobutiric acid (GABA)-ergic interneurones by recurrent hypoxaemia during the migraine attack was hypothesized (6) since this cell type has been shown to be particularly sensitive to hypoxia (7). Alternatively repetitive spreading depression might lead to a selective suppression of GABA-ergic function (8). Evoked potential studies (9, 10) revealed hyperexcitability of the occipital cortex. An increased excitability of the visual system was also suggested by a study showing that the threshold for phosphenes evoked by TMS in patients with migraine is decreased (11). However, another study could not confirm these results (12). Studies investigating motor cortical excitability in patients with migraine with or without aura revealed controversial results (12–14).
Familial hemiplegic migraine is an autosomal-dominant inherited subtype of migraine characterized by transient hemiparesis during the attack. In roughly half of the families the disorder is assigned to a gene on chromosome 19 (15, 16), coding for a P/Q-type calcium channel with distinct expression in the central nervous system (17). Mutation in this gene leads to a dysfunctional protein. Given the regular sensorimotor dysfunction during a typical attack, one would expect an alteration of motor cortical excitability to be more likely in FHM than in MA. One report measuring motor thresholds and amplitudes of the motor evoked potentials (MEP) using TMS hypothesized that motor cortical excitability is increased in general in FHM, but decreased on the affected side (18).
There are only few experimental techniques that allow the investigation of inhibitory and excitatory mechanisms of the cortex in vivo in humans. At low intensities TMS activates the cortico-spinal system transsynaptically through activation of excitatory or inhibitory interneurones leading to excitation or inhibition as judged by the amplitude of the EMG activity elicited by a test shock to the ipsilateral sensorimotor cortex (19–21). This type of intracortical inhibition and facilitation can be altered in particular by drugs acting on the GABA metabolism (22), suggesting that this technique can be used to study changes of GABA mediated excitability in vivo within the motor cortex.
To the best of our knowledge this study is the first to systematically analyse parameters of cortical excitability like intracortical inhibition and facilitation and silent period duration using TMS in patients with FHM. If hyperexcitability is the cornerstone in the pathophysiology of the cerebral dysfunction in migraine one would expect to find changes of the excitability of motor cortex especially in FHM. To test this hypothesis we measured motor thresholds, duration of the cortically evoked silent periods and intracortical inhibition and facilitation in patients suffering from migraine with aura and patients with familial hemiplegic migraine.
Methods
With the approval of the local ethical committee and after informed written consent
according to the declaration of Helsinki we studied 12 (mean age
(±
Clinical data of patients

Values are means ± 1
In the MA group surface EMG recordings were made from the right and left first dorsal interosseus muscle (FDI) using bipolar silver/silver chloride electrodes, placing the active electrode over the muscle belly and the reference over the second metacarpophalangeal joint. In the FHM group EMG was registered from the FDI bilaterally in five patients. In the remaining patients with FHM, recordings were made from the hand affected by clinical signs during the attacks. We were not able to record from FDI bilaterally in all FHM patients because some patients disagreed. In the controls recordings were made from the left FDI only. The raw signal was amplified, filtered (time constant of 10 ms, high cut filter of 1 kHz), and recorded onto a PC using a 12-bit AD interface (sampling rate 5–10 kHz) for off-line analysis. TMS was performed using a figure-of-eight magnetic coil (external loop diameter, 9 cm; peak magnetic field, 2.2 T) connected with two Magstim 200 stimulators via a Bistim module (The Magstim Company, Dyfed, UK). This device allows two magnetic stimulators to discharge through the same coil but attenuates the maximum power output by about 30% (peak magnetic field 1.5T).
We first determined the optimal coil position over the left and right motor cortex using suprathreshold stimulus intensities. With the stimulating coil connected to the Bistim module and the coil held over the optimal site the stimulus intensity was adjusted by 1% and relaxed motor thresholds (RMT) were defined as the stimulus intensity – expressed as a percentage of the maximum stimulator output – needed to produce a response of 50 µV or more in more than five of 10 consecutive trials.
Intracortical excitability was tested at rest as described elsewhere (21). In brief, two magnetic stimuli were given through the same coil over the motor cortex and the effects of the first (conditioning) stimulus on the second (test) stimulus were investigated. The conditioning stimulus was set at an intensity of 25% below RMT. The intensity of the second (test) stimulus was adjusted to evoke muscle responses of 1–2 mV peak-to-peak amplitude. The timing of the conditioning shock was varied in relation to the test shock. Single test stimuli and paired stimuli with interstimulus intervals (ISI) of 2, 3, 4, 7, 10, 12 and 15 ms were delivered in random order generated by the computer. In the FHM group an ISI of 3 and 12 ms was tested in all patients, and the remaining intervals in four patients. The time interval between each sweep was randomly varied by the computer between 7 and 9 s. Ten trials were recorded for each ISI. Subjects and patients were asked to stay awake and keep a constant level of vigilance throughout the recordings. The EMG silence was monitored by continuous EMG at a high gain (50 µV/Div). In addition, signals of the EMG at high gain were used for audio-visual feedback for the subjects and patients. Trials with background EMG activity were rejected on-line. Off-line measurements were made of individual responses, and the peak-to-peak size of the conditioned responses at each ISI was expressed as a percentage of the size of the test response alone.
The cortical-stimulation-induced silent period (CSSP) was elicited in all subjects, 11 patients with MA and seven patients with FHM, while they held a tonic voluntary contraction of approximately 30% maximum voluntary contraction as judged by audio-visual feedback. Trials consisted of 20 stimuli, which were delivered at the optimal scalp site using a stimulus intensity of 30% above RMT. In individual trials the duration of the CSSP was measured off-line from stimulus onset to the end of the CSSP, which was defined as the point where tonic EMG activity following the period of EMG silence could first be noticed when the EMG was displayed at high magnification.
The silent period induced by peripheral nerve stimulation (PSSP) was studied in six controls and four patients with FHM with the first dorsal interosseus contracted at about 30% of maximal force. Ten electrical stimuli were delivered through a Digitimer D180 electrical stimulator (stimulus width 100 µs, stimulus intensity 375 V). At this intensity a maximal M-response was elicited in all patients and subjects studied. PSSP duration was assessed from the time of stimulation until the time of continuous EMG activity at the same level as prior to the stimulus.
The effects of paired TMS were analysed using an analysis of variance with either a
multivariate or repeated (ISI within-subjects) measures model (
Results
Resting motor thresholds and silent period durations
The threshold for activation of the FDI at rest was 54.5 ± 12.2% of
the maximal stimulator output in healthy volunteers, 60.8 ± 12.4
(P = 0.092) and 57.1 ± 14.4 (P
= 0.305) for right and left hemispheres in MA, and 59.8 ± 9.8
(P = 0.121) and 59.7 ± 10.9 (P
= 0.142) in FHM (means ±
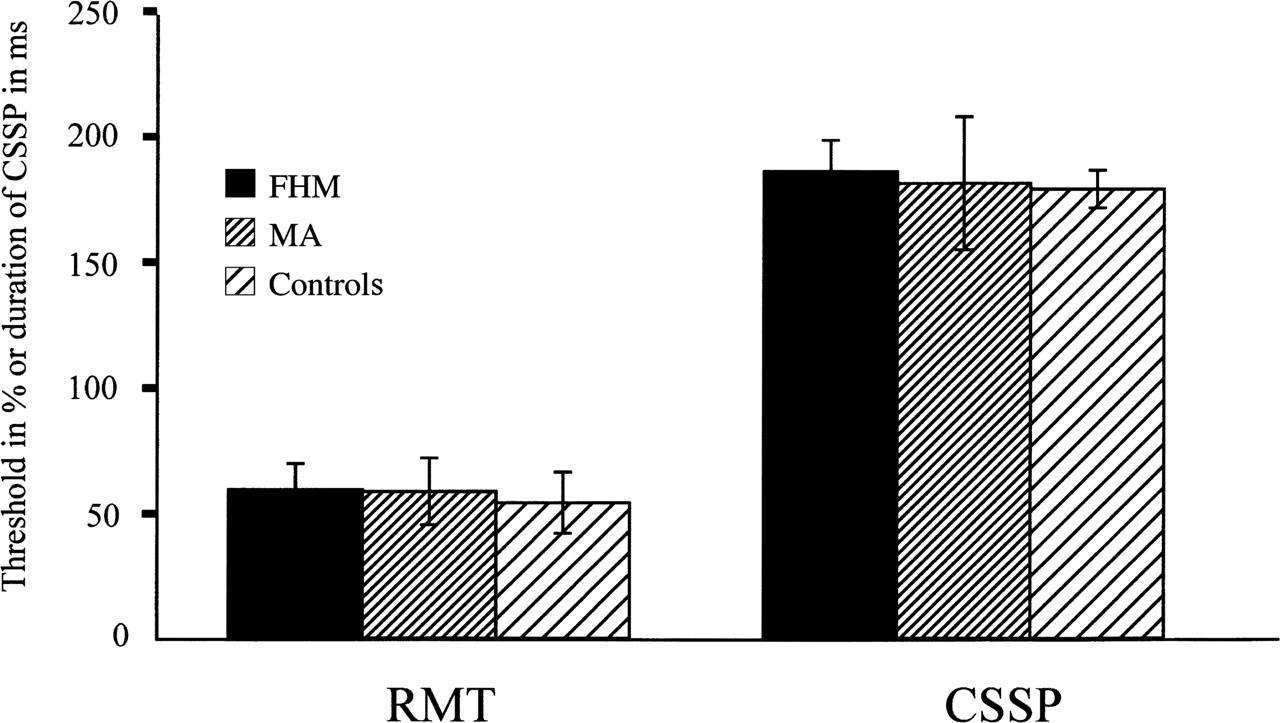
Resting motor threshold and CSSP duration as a mean (± 1
There was no statistically significant (unpaired t-tests)
difference in the duration of the CSSP between subjects and patient groups
(Fig. 1).
The CCSP duration was 178.9 ± 29.9 ms in the controls, 183.2
± 29.8 (P = 0.35) and 179.5 ± 23.3 ms (P = 0.48) for right
and left hemispheres in MA, and 178.8 ± 5.5 (P = 0.49) and 192.3
± 19.5 ms (P = 0.08) in FHM (means ±
Intracortical inhibition and facilitation following paired stimuli
The magnitude of a MEP response was reduced if the test shock was preceded by a sub-threshold conditioning stimulus at an ISI of 2–4 ms and it was facilitated at ISIs of 7 to 15 ms (Fig. 2). The time course illustrated in Fig. 2 shows that there was no difference between the patients groups and controls. In FHM inhibition and facilitation tend to be somewhat more variable than in MA; however, none of these differences were significant. We also grouped the patients either according to the side of head affected by headache in the MA group or according to the hemisphere contralateral to the clinical signs in FHM if both phenomena were consistent in laterality in between attacks and could not find significant differences to the controls or between patient groups.
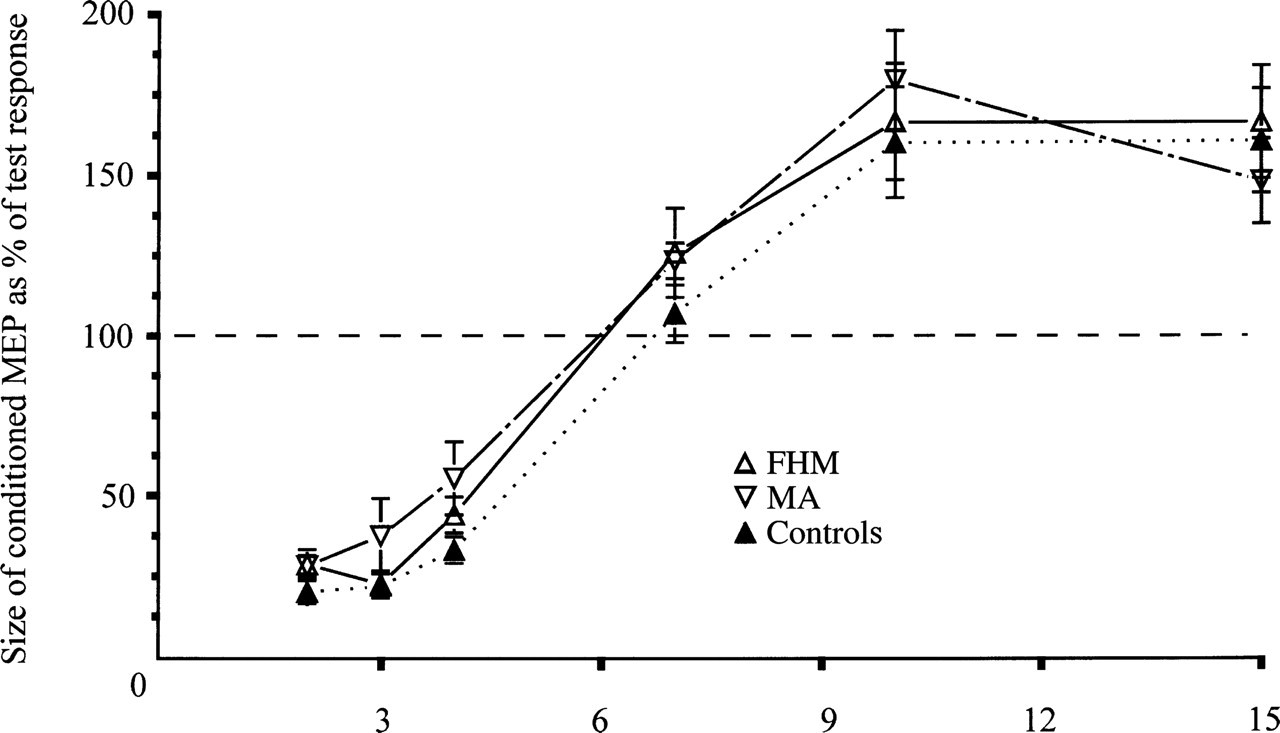
Time course of intracortical inhibition and facilitation as mean
(± 1
There was no significant correlation (r < 0.25) between RMT, CSSP, PPI and PPF and attack frequency. If patients were divided into those taking (n = 7) and those not taking (n = 14) prophylactic drugs, no differences in the TMS parameters could be found.
Discussion
By using TMS we have assessed the motor cortical excitability of patients with migraine with aura and familial hemiplegic migraine. We do not think that our results were affected by the age difference between patients and controls, since significant influence on TMS-induced motor conduction has so far only been shown in subjects over the age of 60 years (24). In addition, in patients with MA and FHM age was not found to be a significant covariant factor for cortical excitability (18).
Results of previous studies investigating cortical excitability using TMS and migraine were conflicting (13, 14, 24, 25); some suggesting hyper- (13), and some hypoexcitability (14, 25), while others found no differences (13). Our results in patients with MA seem to confirm those reported in the largest cohort of patients with migraine with and without aura (12), at least if only PPI, PPF and CSSP are concerned. Áfra and coworkers found no changes in intracortical inhibition or facilitation using paired stimuli, as well as in other parameters like CSSP duration or amplitudes of the motor evoked potentials. However, they concluded – based on an increased threshold only during isometric contraction in the MA, but not in the migraine without aura group – that the motor cortex might be hypoexcitable interictally (12). Most (14, 18, 25), although not all (13), previous studies reported changes in threshold at rest for the activation of motor evoked potentials in patients with migraine with and without aura as well in patients with menstrual migraine. In contrast, RMT in the present study was not different to controls. However, one has to consider the small sample size in our study. Indeed, if the RMT values we found are extrapolated to the same numbers of patients as reported by Áfra et al. (12), threshold differences approach the level of significance (P=0.065). On the other hand, this discrepancy might also be explained by methodological differences. Careful exclusion of background EMG activity by audiovisual feedback during threshold measurements has not been mentioned in most earlier reports and might have affected the results (19, 26).
Studies in healthy subjects revealed that the EMG threshold for TMS impulses at rest reflects changes in membrane excitability rather than neurotransmission at synapses (22) while paired-pulse inhibition and excitation are due to activation of inhibitory GABA-ergic or excitatory interneurones (21). This was deduced from results showing that anti-epileptic drugs acting mainly on voltage-gated sodium channels tend to increase RMT while drugs acting through an increase of the action of GABA like Diazepam, Vigabatrin, Baclofen or Tiagabine have no effect on RMT but alter PPI or PPF (22, 27–29).
Previously, it was argued that the hyperexcitability of the occipital cortex (9, 10) in migraine is due to damage of GABA-ergic interneurones (6). The visual processing in MA was thought to be impaired through a GABA-ergic insufficiency due to selective damage by recurrent ictal ischaemia to spinous stellate cells in the occipital cortex since they might be particularly sensitive to the relative hypoxaemia during the attacks (6). In MA one could envisage a remote effect of migraine attacks affecting mainly parieto-occipital areas on the motor cortex since GABA is also the main neurotransmitter influencing spread of neuronal activity within the cortex (30, 31). In FHM one hypothesis of the present study was that motor cortical excitability might be changed since: (a) the sensorimotor deficit during the attacks suggests direct involvement of the same cortical areas which are activated by the TMS stimuli; (b) GABA is also the main inhibitory transmitter in the sensorimotor cortex (32); and (c) changes in motor cortical excitability were insinuated by a previous study in patients with FHM (18). We thought that Áfra and colleagues (12) did not find any changes because they studied only patients with migraine with visual aura in whom the occipital cortex is mainly involved during the attacks and assumed FHM as a neurophysiological model of migraine affecting mainly primary sensorimotor areas of the cortex. Hence, if GABA-ergic mechanisms were mainly altered in migraine one would expect changes in PPI and PPF and not in threshold. To our surprise, however, even in FHM, excitability as assessed by TMS was unchanged.
Our results therefore challenge the frequently cited theory that there is a GABA insufficiency and a general interictal hyperexcitability (4) of the cortex in patients with migraine.
We do realize that there are confounding factors in this conclusion. The pathophysiology of FHM and MA might not be comparable, excitability might undergo dynamic changes (33), being increased ictally and recuperating interictally, and the mechanisms tested by us might not be the same as those suggested to be affected in the occipital cortex. In addition, drugs successfully used in migraine attacks can alter intracortical excitability even if single dosages are given to healthy subjects (34). Even if no correlation could be found, given the small number of patients who took prophylactic drugs (n=7), we cannot rule out that our results were affected by the prophylactic medication. One important reason why we did not find any changes in the TMS parameters in our patients, however, might be the longer duration of the intervals to the last attack compared to previous studies (12) and the lower average attack frequency, mirroring a milder phenotype. If mentioned at all in earlier reports (11–13), patients were attack-free for a minimum of 3 days to a week. It has been suggested (12, 18, 33) that changes of cortical excitability depend on the duration of the time interval from the last attack. A positive correlation between attack frequency – which is comparable to the duration of attack intervals – and changes in excitability have been found in some reports for the motor (18) as well as the occipital cortex (33), but neither could be confirmed in the present study nor in the work of others (12). In addition, some investigators stress that the excitability might change in the last 24 h prior to an attack (12, 33). For the present work, post-testing attacks would have been of more crucial importance if we had found changes in cortical excitability, assuming an oncoming attack influences excitability. In addition, the fact that attack frequency on average was low and some patients were free of attacks for years makes it unlikely that our data do not reflect excitability in between attacks.
In conclusion, our finding of a normal interictal excitability of the motor cortex in patients with migraine with aura and patients with familial hemiplegic migraine compared to normal controls does argue against the theory of persisting hyperexcitability as a major mechanism of cortical dysfunction in migraine. Given the long attack-free time interval prior to the recordings in our patients it seems possible that short-lasting functional changes might have been missed. Further studies investigating the dynamic changes in the time period around the attack in FHM are therefore needed and might shed more light on a possible alteration of excitability as a cause for neurological deficits during migrainous attacks.
Footnotes
Acknowledgements
The authors wish to thank J. Ditterich for building part of the equipment and Ms J. Benson for carefully reading and editing the manuscript. Part of this work will be submitted as a doctoral thesis by K. Wiseman to the Faculty of Medicine at the Ludwig-Maximilians University, Munich.
