Abstract
Aims: Preliminary reports regarding injections in the neck of onabotulinum toxin A have been positive in cervicogenic headache (CeH). The aim was to perform the first methodologically rigorous trial.
Methods: A randomised, placebo-controlled, patient-, injector- and evaluator-blinded crossover study included 28 adult patients with a long-standing and treatment-resistant CeH. After a baseline period, injections of either onabotulinum toxin A or placebo were given in fixed sites in the neck muscles on the pain side. Second injections were given after ≥8 weeks. Patients were thereafter followed for another 8 weeks. A detailed headache calendar was filled in, and patients were followed with quality-of-life (QoL) questionnaires, algometry and neck mobility measurements.
Results: There was no significant difference between verum and placebo in a mixed linear model analysis (p = 0.084) with regard to the primary end-point, reduction of days with moderate to severe headache. Six patients withdrew from the study before the second injections, but an intention-to-treat (ITT) analysis gave a similar result (p = 0.27). There were no significant differences favouring verum in any of the secondary efficacy measures. Side-effects of onabotulinum toxin A were minor and short-lasting.
Conclusion: Onabotulinum toxin A in neck muscles does not seem to be beneficial in CeH.
Introduction
Although cervicogenic headache (CeH) is still a somewhat controversial disease entity, and the prevalence shows large variation in epidemiological studies (0.17–17.8%) (1–3), it is considered to be a cause of substantial disability in the general population and one of the major headaches types in clinical practice (4–6).
Bogduk has characterised this unilateral syndrome as pain perceived in the head but whose source lies in the cervical spine (7). Nociceptive afferents in the trigeminal nerve synapse to some degree on the same second-order neurons in the trigeminal nucleus caudalis as nociceptive afferent neurons from cervical structures (5,8). The anatomical and physiological mechanisms are therefore in place to refer pain from the neck to the head (5,7). However, the insight into the putative pain-producing loci in CeH leaves much to be desired. All cervical structures with a sensory innervation are potential pain generating candidates, and it is possible that a central predisposition is also required for the picture to fully develop (9).
According to a recent review by Becker, there is a probable role for neck muscles in CeH (5). This assumption is supported by several clinical and laboratory findings. Firstly, a significantly higher occurrence of muscle tightness in the neck has been found in patients with CeH compared to patients with migraine and controls (10,11). Secondly, electromyographic activity from the trapezius muscles on the symptomatic side in patients with CeH is higher compared with the non-symptomatic side and controls (12,13). A mental load test led to an asymmetric response of non-voluntary muscle activity which was significantly more increased on the symptomatic side compared with the non-symptomatic side (12). Thirdly, palpation of tender spots in tight bands of muscle in the neck of patients with CeH resulted in referred symptoms similar in quality and location to their usual headache (14,15). It is not clear whether the muscular findings mostly imply central or peripheral mechanisms, and it is questionable whether they are a cause or consequence of the headache. However, a common conceptual model is that of a self-sustaining, lateralised algogenic loop involving nociceptive input from neck muscles producing continuous afferent bombardment of the trigeminal nucleus caudalis with a lowering of the pain threshold (16).
Although there have been many treatments suggested for CeH, few have been scientifically tested and proven successful (7). More than 20 years ago, it was hypothesised that therapy aimed at breaking up the ‘fixation of tissue’ might help, allowing the muscles to stretch to a fuller range of movement (ROM) and reducing trigger point activity (14). Botulinum toxin A significantly reduces pain associated with craniocervical dystonia – an effect that has long been considered secondary to its muscle relaxant action. Intriguingly, the pain alleviation in craniocervical dystonia frequently sets in before the muscle relaxation can be observed. This together with the recently shown effect of onabotulinum toxin A in chronic migraine supports a mechanism for pain reduction that is more complex than a simple secondary effect of muscle relaxation (17–20).
In an open-label study, botulinum toxin A seemed to be an effective treatment for chronic headache in patients with cervical dystonia and a history of migraine (21). Regarding botulinum toxin A in CeH, all case reports have suggested a positive effect (22,23), as has the only randomised, placebo-controlled trial published so far (24). This pilot study had several methodological limitations. Anaesthetic blocks were not used for diagnosis, and all patients had chronic daily headache secondary to cervical whiplash injury, which is not the case in most CeH patient series. In addition, a statistically significant pre-injection difference was noted between the two groups with respect to head pain – probably due to the small sample size of the groups. Finally, pain was not registered daily, but only before treatment and after 3 or 4 weeks.
Despite the lack of high-quality evidence it has been concluded that therapy with repeated injections of botulinum toxin A may be the most safe and efficacious approach in CeH (25). We found that this needed to be further explored, and here report the findings of the first well-powered, randomised, placebo-controlled trial of botulinum toxin A for CeH.
Methods
Study design
This was a randomised, double-blind, placebo-controlled crossover study conducted in the Department of Neurology of the University Hospital of Trondheim, Norway. Patients were recruited from the outpatient clinic. After having signed an informed consent form, they entered a 28-day baseline screening phase (hereafter referred to as baseline). Eligible patients were then randomised (1:1) in double-blind fashion to onabotulinum toxin A (BOTOX®, Allergan, Inc., Irvine, CA, USA) or placebo (saline). Randomisation was stratified in blocks of four to either sequence A: first verum, thereafter placebo, or sequence B: first placebo, thereafter verum. Participants, those administering the interventions, and those assessing the outcomes were blinded to sequence assignment. Identical syringes containing verum or placebo were prepared and labelled in code by a hospital pharmacist not otherwise associated with the study, according to a computer-generated randomisation list delivered as a printout from the university’s Unit for Applied Clinical Research. The sequence was thus not concealed for the pharmacist when interventions were assigned, but to participants and those administering the treatments. Onabotulinum toxin A (in total 100 U = 2.0 ml) or the corresponding volume of placebo was administered via a 30-gauge needle as fixed-site injections across six specific head/neck muscle areas on the pain side: 20 U divided upon two points in the occipital muscle insertions, 20 U in the upper trapezius, 20 U in the splenius capitis, 20 U in the sternocleidomastoideus, and 20 U in the levator scapulae. The second injections were given when the patient had reached ≥50% of days with moderate to severe headache compared to baseline but not before 8 weeks after the first injections. Those who had not reached ≥50% of days with moderate to severe headache compared to baseline 8 weeks after the first injections were to be called up every second week to evaluate whether they fulfilled the criterion for getting the second injections. After the second injections, patients were followed for another 8 weeks. There were four visits to the physician: (1) enrolment, (2) randomisation and first injections, (3) second injections, and (4) finalisation.
The study was conducted in accordance with the Declaration of Helsinki ethical principles, good clinical practice, principles of informed consent, and requirements of public registration of clinical trials (ClinicalTrials.gov identifier: NCT00184197). Approval was obtained from the Regional Committee for Ethics in Medical Research, and by the Norwegian Data Inspectorate.
Study patients
Participants were prospectively enrolled by two neurologists at the Norwegian National Headache Centre. Eligible patients were men or women aged 18 to 70 years with a history of CeH of >1 year’s duration meeting the revised Cervicogenic Headache International Study Group (CHISG) diagnostic criteria (26) used in a simplified version (27):
Strictly unilateral headache without side-shift. At least two of the following indicating neck involvement:
Restriction in range of motion (ROM) of the neck. Provocation of usual head pain by neck movement or sustained awkward neck positions. Provocation of usual head pain with external pressure over the upper cervical or occipital region on the symptomatic side. Irradiation of pain to ipsilateral shoulder or arm. Positive effect of blockade of the greater occipital nerve (GON) defined as any pain reduction on a VAS scale lasting at least 4 hours after the procedure.
Patients should have documented a headache frequency >15 days/month during at least 1 month. Exclusion criteria were:
Cervical spinal stenosis, cervical disc herniation, or other neck pathology that should be treated surgically. Malignant disorder. Rheumatic disorder or other disorders which induce analgesic use. Patients with other clinically relevant disease (neuromuscular, liver, kidney, endocrinological, metabolic, systemic, psychiatric). Pregnancy, breastfeeding, or lack of reliable means of contraception in fertile women (women of childbearing potential had a negative urine pregnancy test before each injection). Frequent other headache that cannot be distinguished from CeH. Pending or ongoing litigation for head or neck trauma. Having used any headache prophylactic medication within 4 weeks prior to start of baseline. Previous exposure to any botulinum toxin serotype.
Eligible patients underwent physical and neurological examinations. GON blockades were performed as previously described (28). To exclude disorders or lesions that could be treated, magnetic resonance imaging (MRI) of the brain and neck were performed if they had not been done previously during the last 5 years.
Efficacy and safety measures
Throughout the study, participants were required to provide daily diary data indicating the maximum intensity of head pain (scale 0–3), presence of neck pain, total duration of pain in head and neck (hours), analgesic medication (number of tablets), and sick leave. Patients were telephoned by the study nurse 4 weeks after each round of injections to log adverse events (AEs) and as a reminder to fill in the diary.
At visit 2 (baseline), visit 3 (after first injections) and visit 4 (after second injections), the QoL, neck ROM, and pain pressure thresholds (PPTs) were assessed. QoL was measured using the Short-Form 36 questionnaire (SF-36, Norwegian Acute version 1.2a, Copyright© Medical Outcome Trust, 1992) consisting of 36 questions concerning the last week. Neck mobility and PPT were measured by the same study nurse, who was blinded to the patient’s pain side. As described in detail previously, measurements of ROM were performed in a relaxed sitting position with a baseline inclinometer (Cybex instrument EDI320) (29). Active movements were carried out in three dimensions: forward flexion + dorsal extension, lateral flexion to the right + lateral flexion to the left, and rotation to the right + rotation to the left. The average of three measurements was recorded for each type of movement. PPT measurements were performed with a pressure algometer (Model PTH-AF2, Pain Diagnostic and Thermography Corporation, Italy) (11,30). With the patient sitting in a chair, five specified points were studied on each side: intermediate part of the temporal muscle, mastoid process, superior insertion of the trapezius muscle in the occiput, sternocleidomastoid muscle over the transverse processes of C2–C3, and inferior trapezius muscle over the transverse processes of C5–C6. The PPT was defined as the minimum amount of pressure where the perception of pressure first changed to discomfort or pain. Three readings were taken over each site and averaged for analysis.
The null hypothesis was stated as no difference between the two treatment regimes. The same hypothesis was used for all efficacy variables. The primary efficacy measure was the change in the mean weekly frequency of moderate to severe headache (intensity grade 2–3). Secondary efficacy variables from diaries were headache intensity (on days with headache), headache index (intensity × days/week), duration of pain in head/neck, frequency of neck pain, analgesic use and sick leave. For all these, the mean change from baseline during the period 3–8 weeks after injections was examined. The incidence of any AEs during the first 4 weeks after injections was used as a safety measure. Other secondary efficacy measures were the change from baseline at 2 months after injections in QoL (a commando file was used to convert the results into eight domains), neck ROM (three dimensions) and PPTs (mean difference between the sites in the dominant side compared to the other side, and the total tenderness score calculated by summarising data from both sides).
Statistical analysis
Estimated enrolment was 24 patients. This figure was based on the variance of days with moderate to severe headache in an earlier pre-treatment material of CeH and the judgment that a 30% reduction was the minimal effect which has clinical relevance (27). A power calculation was done post hoc based on the primary end-point in a linear mixed model. The retained sample size of 26 participants with SD 0.4 has a power of 0.9996 to detect a 22% (1.0 days/week) between-intervention difference in mean change from baseline, using a two-sided alpha = 0.05. The power for detecting a 13% difference (0.6 days/week) was 0.89.
All data is described using descriptive statistics, i.e. n, mean and SD for continuous variables and frequency and percentage for categorical variables. Those assessing the outcomes were blinded to sequence assignments when feeding data into the analytical software. Statistical testing was done for all primary and secondary efficacy end-points comparing verum to placebo. A linear mixed models procedure based on all data generated by the subset of subjects (n = 26) reporting the effect of the first injections was used to analyse end-point variables from diaries, SF-36 validations, ROM measurements and algometry for as long as they complied with the protocol. A supporting ITT analysis of the primary variable was done for all randomised participants (n = 28) where missing data were imputed using last observation carried forward (LOCF) methodology (assuming no change for non-completers) and the paired samples t-test (31). The McNemar matched pairs test was used to compare proportions (AEs and >30% reduction of moderate to severe headache). Two-tailed estimations of significance were used, and the level of significance was set at p < 0.05. Data analysis was performed with the Predictive Analytics SoftWare (PASW) Statistics version 17.0 by SPSS Inc. (Chicago, IL).
Results
Participants
Inclusions were performed from May 2005 to August 2009. The flow of participants through each stage of the study is described in Figure 1. Of the 29 patients screened, one (male, 43 years, duration of CeH 6 years, no trauma) was excluded because he did not fill in the calendar during the pre-treatment period. Twenty-eight patients were randomised to treatment. During the treatment periods, there were six drop-outs, four after the first round of injections and two after the second round. Of these six, four were lost to follow-up (two after each round of injections). The other two filled in calendars but did not return for a second injection. The reason for drop-out was reported by one case only (heart operation not related to study medication). All randomised patients were finally included in the statistical analyses for the primary outcome.
Flow of patients through the study.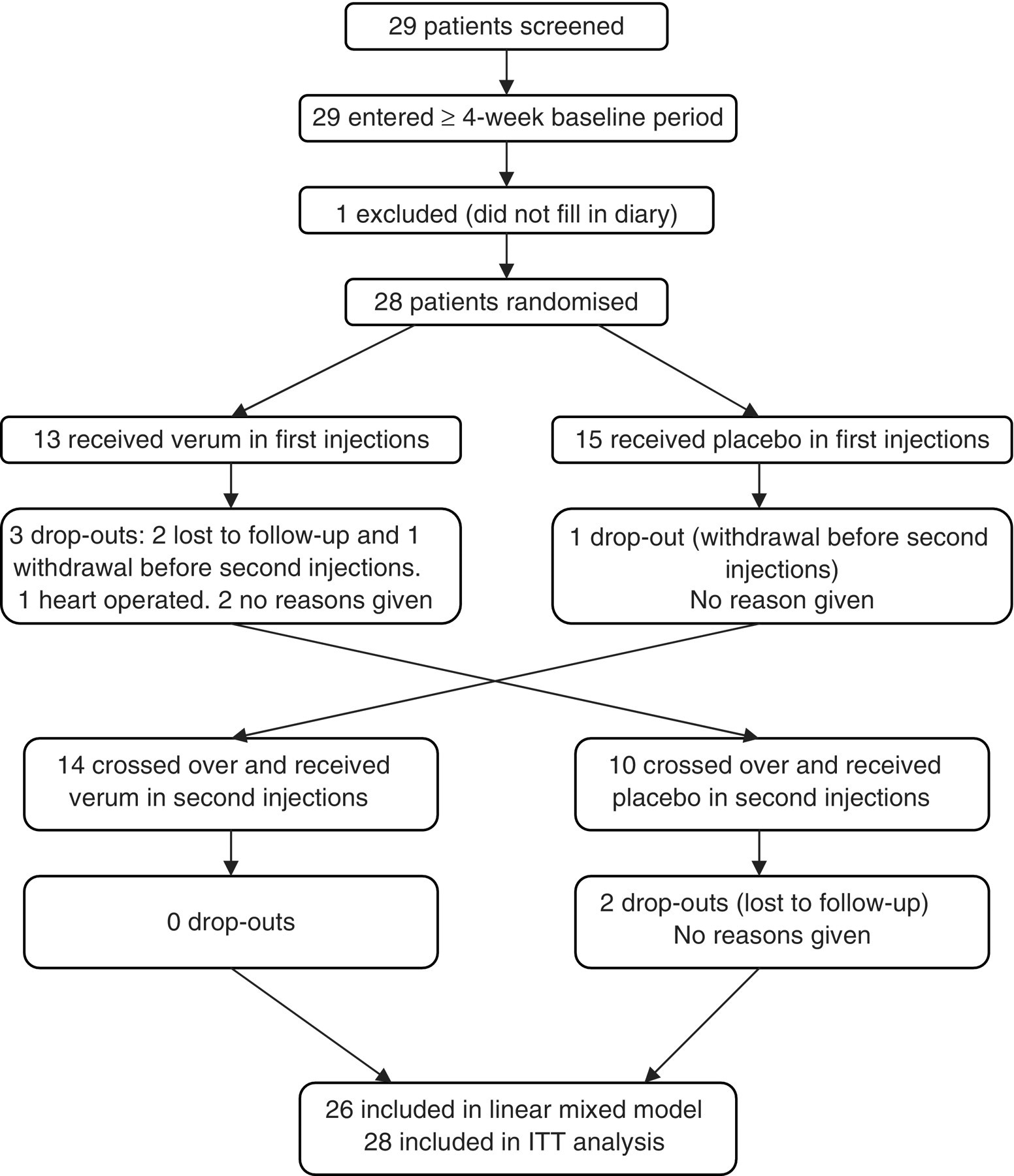
Effects of onabotulinum toxin A and placebo on outcomes based on diaries

The mean change from baseline during the period 3–8 weeks after injections was examined.
The mean number of years since debut of CeH was 10.8 ± 1.9 (range 1–43 years). Twelve patients (43%) had a history of head or neck trauma. The specific mishaps were, in order of appearance: car accident (n = 3), hitting the head in a fall (n = 3), heavy object in head or neck (n = 2), skiing accident (n = 2), bicycle accident (n = 1), and kick-boxed in head (n = 1). Nine of these patients reported debut of headache the same year as the trauma. Among the other three, the headache started 2 years before, or 4 and 27 years after the accident. Two patients had neurological deficits, in both cases ipsilateral to the pain; one slight hypalgesia on the chin and the radial part of the hand, the other had tongue deviation, pharyngeal weakness and hypesthesia, and atrophy of the sternocleidomastoid muscle after operation for a giant intracranial aneurysm.
Fifteen patients had left-sided pain and thirteen had pain on the right side. The mean frequency and intensity of headache before study treatment is shown in Table 1. The mean individual reduction of pain after GON blockade was 61.0 ± 5.4%. Six patients had also had other headache (tension-type headache, TTH n = 2, migraine n = 4). Among participants with a history of migraine, the current attack frequency was very low. Only one patient reported self-considered migraine (on 1 day) during the 4-week baseline period.
Primary efficacy variable
The mean frequency of moderate to severe headache was 4.48 days/week during baseline, and the mean changes were −0.72 ± 0.20 days/week after onabotulinum toxin A and −0.40 ± 0.20 days/week after placebo (Table 1). Neither a mixed linear model analysis (p = 0.084) nor an ITT analysis using the LOCF (p = 0.27) revealed a statistical difference between verum and placebo. There was no period effect (i.e. no significant difference in the effect of onabotulinum toxin A or placebo when given as first or second injection) (p > 0.2). There was no significant difference (p > 0.2) in the proportion of patients that reported a ≥30% improvement after onabotulinum toxin A (n = 10) compared to placebo (n = 7). Two months after the first injection, all patients that were followed (onabotulinum toxin A n = 11, placebo n = 15) had at least 50% of days with moderate to severe headache as compared to baseline.
With regard to the primary efficacy variable, five patients improved (any degree) after onabotulinum toxin A but not after placebo (Figure 2). All of these ‘responders’ were women but did not differ apparently from the other participants in age (mean 45 years), duration of disease (mean 16 years since debut), proportion with triggering trauma (3/5, 60%), or effect of GON blockade (−52% change in pain). Two of them had concomitant migraine.
Scatterplot demonstrating the relative change (%) from baseline in the number of days with moderate to severe headache after injections of onabotulinum toxin A and placebo for all patients (n = 28) in the ITT analysis using LOCF. The outlier reported a change from 0.8 days/week during baseline to 3.7 days/week after verum and 1.5 days/week after placebo.
Secondary efficacy variables
There was no significant difference between onabotulinum toxin A and placebo regarding the effect on headache intensity, headache frequency, headache index, frequency of neck pain, duration of pain (head and/or neck) or analgesic use (Table 1). Among the 60% of patients who were occupied (any degree), the frequency of sick leave doubled after verum treatment, resulting in a significant difference (p < 0.001) favouring placebo (Table 1).
Effects of onabotulinum toxin A and placebo on QoL according to SF-36

Effects of onabotulinum toxin A and placebo on neck ROM and PPT

Adverse events
All AEs reported after injections of onabotulinum toxin A and placebo (some patients had >1 AE)

One serious AE (hospitalisation for heart operation) was reported during the study (Figure 1). This was considered to be unrelated to the study medication (onabotulinum toxin A).
Discussion
This is the first clinical trial of botulinum toxin A in CeH applying rigorous methodological standards. Efficacy was not observed for the primary end-point, or for any of the secondary efficacy measures.
The reduction in frequency of moderate to severe headache after onabotulinum toxin A compared to placebo was estimated at 0.33 ± 0.19 days/week. This difference was not significantly different from zero. Although the data for the primary end-point do not exclude the possibility of a small effect of verum, it would be much lower than what we define as clinically meaningful. Furthermore, none of the secondary end-points indicated an effect of onabotulinum toxin A. Improvement from baseline was often observed after both treatments. Possible explanations are regression towards the mean, spontaneous improvement and placebo response. In headache research, invasive procedures have a higher placebo response compared to oral drugs (32). Placebo was significantly better than onabotulinum toxin A for one end-point: days of sick leave (p < 0.001). It remained significant even after a highly conservative Bonferroni adjustment modifying the critical level from 0.05 to 0.002 (i.e. dividing by 22 variables). Side-effects, more frequent after onabotulinum toxin A, may be one cause of this, but this variable should be interpreted with caution, because fewer than half of patients were occupied at ≥50%, and the answer ‘not applicable’ to the question on work absence in the calendar was common.
Previous physiological studies have found mechanical allodynia in CeH, which is most pronounced on the symptomatic side and most likely central in origin (8,33). Onabotulinum toxin A is thought to block peripheral sensory signals to the central nervous system and indirectly inhibit central sensitisation (17,34). It is therefore noteworthy that the algometry findings did not reveal a difference between verum and placebo. Particular attention was paid to avoiding variation in circumstances around the procedure in order to reduce variability (30). Because earlier research implies that a bilateral sensory dysfunction may be present in CeH (8), we not only studied the PPT difference between the sides, but also the total tenderness score. In rats, local peripheral injection of botulinum toxin A significantly reduced formalin-induced pain with the absence of obvious muscle weakness. This anti-nociceptive effect was associated with the inhibition of release of glutamate or inflammatory neuropeptides from primary afferent terminals (17,35). Inhibition of neurogenic inflammation induced by the peripheral release of inflammatory neuropeptides may account for the (modest) effectiveness of onabotulinum toxin A in the treatment of chronic migraine (17,18). Because such a neurogenic inflammation is absent in CeH (36), it may be logical that onabotulinum toxin A does not help for CeH or TTH (37,38).
Zwart showed that cervical flexion/extension and rotation ranges were significantly less in subjects with CeH compared to those with migraine and TTH (29). This has later been supported by others (11,39). It is not clear whether this reduction in neck ROM is part of the pathophysiology of CeH or a function of the head pain. In a case report of CeH, full ROM in the neck was achieved after injection of onabotulinum toxin A in the trapezius muscle on the symptomatic side (22). Likewise, a positive effect on ROM was noted in the pilot study by Freund and Schwartz (24). The fact that we could not repeat these findings places doubt on the idea that limited neck movement is a consequence of reduced length and hyperactivity of the cervical muscles (40). It may be the other way around, because decreased ROM in any joint causes shortening and tightening of muscles (14).
AEs were consistent with the known safety profile of botulinum toxin A injected in cervical musculature (20,41).
There is a lack of an easily applied ‘gold standard’ for CeH diagnosis. The International Classification of Headache Disorders criteria (ICHD-II) (42) are considered provisional (5) because they require objective evidence of a lesion in the neck “known to be, or generally accepted as, a valid cause of headache”, although what exactly said lesions are has not been defined (43). Most lesions do not produce CeH, and data show that the majority of CeH patients have no demonstrable lesion in the neck (9). Furthermore, criterion D demands that pain has resolved within 3 months of successful treatment of a cervical lesion/disorder. This was obviously not designed for diagnosis before treatment (7), and the ICHD-II criteria are therefore not very useful in the context of clinical trials (6). For scientific purposes, the CHISG criteria (26) are functional and have therefore been applied in many clinical studies. They are able to differentiate most CeH from migraine and TTH, with some overlap (44). Positive effects of blockades of the GON and/or C2 root was an important but not obligatory criterion in the 1990 CHISG criteria (45). In the revised 1998 version it was made an obligatory criterion in scientific works (26). However, the present status of obligatory blockades is somewhat dubious (46), and standardisation remains to be defined (47). An important question is where to deposit the anaesthetic agent. CeH is understood as a syndrome that can result from many possible sources in the neck, and conceivably, not all these can be blocked. Even cervical facet blockades assess only some of the potentially painful structures in the neck, and when multiple facet joints are blocked simultaneously, it is possible that non-specific effects could emerge (5). GON blocks are the most widely examined in CeH (48), and the resulting pain reduction has been found to be significantly more marked in CeH compared to migraine and TTH (49). Anaesthetic blocks of the GON are therefore considered to favour the CeH diagnosis if positive (44). There are currently no data as to the optimal dose, volume or type of local anaesthetic to be used for GON blocks in headache management (43). A positive effect of GON blockade did not seem to predict a favourable outcome of onabotulinum toxin A in the present study.
The study population was highly disabled. Participants had in general suffered from CeH for over a decade and experienced an average of 26 days per month of mainly moderate to severe headache. This is representative of typical patients with CeH seen in clinical practice, as is the proportion with previous craniocervical injuries (50). In a clinic-based study, patients with CeH more often reported having had a severe trauma as compared to migraine and TTH patients (51). Recall bias may have interfered when trying to determine the aetiological role of the accident. It often simply modifies an existing pain (52). The treatment results in the present material were, however, not influenced by any compensation demands. CeH and migraine are not mutually excluding diagnoses (44), and the 14% prevalence of migraine is typical for the general population.
Clinical guidelines for injections to treat hyperkinetic cervical muscles with botulinum toxin are widely published. No one cervico-brachial muscle seems to predominate in the incidence of tightness in CeH (11). When choosing injection paradigm, we therefore took into consideration the way others had performed them in earlier publications regarding botulinum toxin A in CeH (22,53). The splenius capitis and trapezius muscles had been injected in earlier positive reports (22,24) and were therefore included. In one study (24), the semispinalis capitis and rectus capitis were also injected. The first is covered by the trapezius, and the injection must be given sufficiently deeply using EMG monitoring while contracting the trapezius. The latter muscle is more deeply situated and smaller, and seldom injected with botulinum toxin. Instead of these two, we chose also to inject the other superficial muscles of the dorsal neck, i.e. the levator scapulae, occipitalis and sternocleidomastoideus muscles. For standardisation reasons, there was no individual selection of trigger points. All subjects had strictly unilateral headache without side-shift, and we considered it as irrational to inject the non-symptomatic side.
When the study was designed, the average doses of botulinum toxin used by clinicians for the treatment of headaches were 70–100 U, and lower doses were often tested in clinical headache trials (54). In the published positive reports regarding botulinum toxin A in CeH, doses ranged between 50 and 100 U (22–24). We thus chose a relatively high unilateral dosage (100 U), although this increased the cost and possibly the risk for AEs. In the recent PREEMPT clinical programme, doses between 155 and 195 U of onabotulinum toxin A were investigated for chronic migraine (18). This was distributed on both sides of the head and neck.
Whether the use of other injection sites or bilateral injections, a higher dosage, or a different injection regimen such as the follow-the-pain strategy could have been even more optimal is unclear, but we doubt it would have changed the outcomes. There are currently three major commercially available preparations of botulinum toxin A, which differ in their pharmacology, potency and other properties. Dose units are specific to the preparation and not directly interchangeable. Onabotulinum toxin A is the one most often used in headache research.
Strengths of this study are that it is the largest well-designed prospective controlled trial conducted to date on onabotulinum toxin A in CeH. The generalisability (external validity) of the trial findings is high due to the representative population. Because pharmacological effects provided from botulinum toxin injections are temporary and gradually wear off (41,55), each subject could be randomised to a sequence of two treatments, and act as his/her own control for treatment comparisons. This simple manoeuvre led to high statistical power to detect an effect which has clinical relevance, i.e. a power of > 99% and 89%, respectively, for a between-intervention difference in mean change from baseline of 1.0 days/week and 0.6 days/week. Non-compliance with treatment was not a major issue because study drugs were injected.
There are some sources of potential imprecision. The study design did not include a true washout period. The potential effect duration of onabotulinum toxin A in headache is uncertain, and the planning for sufficiently long washout periods requires knowledge of the pharmacodynamics of the treatment. Furthermore, there was a concern that patients would drop out and try other treatments if they had no positive effect from the first injections and the waiting time in between injections was too long. It was therefore decided not to have a long and fixed interval between treatments. As a result, a residual influence of the active drug in subsequent placebo periods (carryover effect) cannot be entirely dismissed. On the other hand, we could prove that there was no period effect. There is a risk that patients and/or investigators could have been unblinded to the study treatment because of physical changes due to muscle relaxation. This is, however, much less likely than in studies where botulinum toxin is injected in the frontalis muscle, where the effect is readily visible. Also, the presence of the placebo response suggests that the blind was maintained. Complications of analysis and interpretation arose from the loss of subjects. The ITT principle implies that the primary analysis should include all randomised subjects. Compliance with this principle would ideally necessitate complete follow-up of all randomised subjects for study outcomes (31), which is usually not possible to achieve in practice. A contemporary statistical approach using a linear mixed models procedure based on all data generated was used for the main analysis of the primary end-point and for all analyses of the secondary end-point. In a supplementary analysis of the primary end-point, we used the LOCF methodology despite underestimation of the standard error for the estimated effect and a potential bias in the estimation of the treatment effect. We conducted these two methodologically different primary analyses so that any differences between them could be the subject of explicit discussion and interpretation. They did, however, lead to the same conclusion.
In conclusion, this study does not support prior findings concerning the efficacy of onabotulinum toxin A in CeH.
Footnotes
Funding
Allergan, Inc. provided the study drug BOTOX® without cost. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
Hagen and Linde are both members of an Allergan international advisory board on chronic migraine. Honoraria are given in connection to that work. They are also both members of the Nordic Headache Innovators group which received sponsorship from Allergan for a two-day scientific meeting in Trondheim in March 2010.
Stovner has received lecture honoraria and travel grants from Allergan.
Helde, Salvesen and Bruvik Gravdahl: no conflict.
