Abstract
Keywords
Introduction
Pain is a personal, subjective experience that comprises sensory-discriminative, motivational-affective and cognitive-evaluative dimensions (1). Reliable and accurate pain assessment is a prerequisite for successful pain management (2). Reliable and valid measures of pain are also essential for conducting clinical trials of pain treatments. Perhaps the most important aspect of a pain measure’s validity is its sensitivity or ability to detect changes in pain over time and due to treatment. Without adequate sensitivity, the real effects of pain interventions cannot be estimated, and real effects of treatment can go undetected (3). Fortunately, in most situations, the most commonly used measures of pain intensity, including visual analogue scales (VASs), numerical rating scales (NRSs) and verbal rating scales (VRSs, also known as categorical scales, verbal descriptor scales, fixed descriptor scales or Likert scales) have been shown to have adequate sensitivity to changes in pain associated with treatment across many populations and settings (4).
Accurate evaluation of actual headache intensity is crucial in both clinical and research settings (5). Although pain is a multidimensional phenomenon, the severity of migraine headache is usually characterized by pain intensity, frequency, duration and disability (6). The evolution of verbal and non-verbal scales can be traced from the traditional ordinal scales which ask patients to rate their pain as none, mild, moderate or severe, or from 1 to 4, l to 10, etc., to the verbal descriptor tests and numerical scaling procedures (5). One of the most widely used non-verbal measurement techniques is the VAS. This measure is a form of cross-modality matching in which line length is the response continuum (5).
The VRS (4 grade scale) and the VAS are both recommended for assessing headache severity in the 2nd edition of the International Headache Society guidelines for controlled trials of drugs in migraine (7) and tension-type headache (8). It is reported that the VAS allows a greater sensitivity for determining treatment effects than a categorical scale such as the VRS (4 grade scale), because there is a greater range of possible scores (9), but it was discussed that the interpretation of statistically significant but small changes in VAS scores could be difficult (10). On the other hand, for analysis purposes, the VRS (4 grade scale), widely used during the development of triptans, is usually reduced to a binary scale (patients are categorized as responders or non-responders), which might be expected to further reduce its sensitivity compared with a continuous scale (9).
As part of the Thomapyrin Study that showed the significant superiority of the fixed combination of acetylsalicylic acid + paracetamol + caffeine over the combination without caffeine, the single preparations, and placebo in the treatment of headache (11), patients enrolled into the study were trained in the handling of the VAS by naming categories of a 6-point VRS. The same procedure was repeated in all patients at study conclusion.
These data were used:
to assess both scales with respect to consistency of rank order of the categories; to determine cut-off points on the VAS related to the categories of the VRS; to evaluate how the categories of the VRS are represented on the VAS (equidistant, non-equidistant, specific pattern); and to assess test-retest reliability after repetition of the complete training procedure at study conclusion.
Methods
Patients
The data for this analysis were collected as part of a randomized, placebo-controlled, double-blind, multi-centre parallel group trial with six treatment arms, conducted between September 1998 and January 2003 (11). The primary objective of the study was to investigate the efficacy, safety and tolerability of the fixed combination of acetylsalicylic acid + paracetamol + caffeine in comparison with the combination without caffeine, the single preparations, and placebo in patients who were used to treating their episodic tension type headache or migraine attacks with non-prescription analgesics.
The patients were enrolled by practitioners and specialists in general and internal medicine throughout Germany. Male or female patients (18–65 years) who were not consulting for headache were asked whether they had headaches that they treated with non-prescription analgesics. Usual headaches had to meet International Headache Society (2) criteria for episodic tension-type headache (2.1) and/or migraine with or without aura (1.1, 1.2.1) (12). Patients must have experienced these headaches for at least 12 months with a minimum of two headache episodes within the previous 3 months.
Patients were excluded if previous or concomitant diseases or medication could interfere with one of the study drugs or influence headache symptoms. Drug overuse connected with the headache and alcohol or drug abuse was also an exclusion criterion, as was pregnancy, lactation or participation in another clinical trial within 4 weeks of entering this study.
Before enrolment the patients gave their written informed consent according to paragraphs 40 and 41 of the German Drug Law (AMG) and ICH GCP (International Conference on Harmonization, Guidance for Good Clinical Practice, E6) standards. Patients were allowed to terminate participation in the trial at any time, without giving reasons. The study was conducted in accordance with the Declaration of Helsinki, the German Drug Law (AMG), and ICH GCP standards and did not start before Independent Ethics Committee approval was obtained.
Training of the VAS
Patients recorded pain intensity on a 100 mm VAS by drawing a vertical line on the horizontally positioned VAS before and at several time points after drug intake during the course of the study in a diary. The left end of the VAS represented ‘no pain’ and the right end ‘most severe pain imaginable’ with no intermediate divisions or descriptive terms. During the screening visit (visit 1), the investigator gave a standardized explanation of the VAS using a written explanatory text. He handed a VAS to the patient and named one of the categories of a 6-point VRS used to describe pain intensity (the original German wording is given in brackets): 0 = no pain (kein Schmerz), 1 = mild pain (leichter Schmerz), 2 = moderate pain (mäßiger Schmerz), 3 = severe pain (starker Schmerz), 4 = very severe pain (überaus starker Schmerz), 5 = most severe pain imaginable (stärkster vorstellbarer Schmerz). Within 30 seconds, the patient was to draw a vertical line on the horizontal scale, where, according to their assessment, the mentioned pain intensity would be represented, based on the experience of their own headache. This procedure was repeated until all six categories had been named randomly by the investigator according to a randomization list provided (PROC PLAN, SAS Institute Inc., Cary, NC, USA). Before naming the next category, the patient returned the page to the investigator so that the next evaluation was made without reference to the previous one. The patient was not informed on the number or the wordings of the VRS categories. The procedure was repeated at the patient’s final conclusion visit (visit 4). The data from patients who concluded at least one of the two trainings as planned for were included in the analyses.
Statistical analysis
Descriptive statistics and frequency distributions of the assessments on the VAS recorded in the training sessions were calculated, sorted by the scores of the VRS. The level of order consistency between the VAS and the VRS was analysed by evaluating within patients the permutations of the assessment on the VAS in comparison to the order of the scores of the VRS.
For rescaling the continuous VAS into a discrete ordinal scale, cut-off points were determined on the VAS at which the categories of the VRS could best be represented. These cut-off points were deduced from receiver operating characteristic (ROC) curves (13). A cut-off point on the VAS was to be defined where patients irrespective of the actual VRS were assigned to the higher pain category if the mark on the VAS was above the cut-off point or to the lower pain category if the mark was below. If the patient named the higher category on the VRS and their mark on the VAS was above the cut-off point, the outcome was called true higher. The ROC curve displays the true higher (sensitivity) versus the false higher (one minus specificity) rates for the range of possible cut-off points. As the sensitivity is a decreasing and the specificity an increasing function of the cut-off values, it is not possible to select a cut-off maximizing both. Instead we chose the cut-off point at which the sensitivity and specificity are closest assuming equal importance of sensitivity and specificity. The area under the ROC curve was calculated as a summary measure of its discriminatory ability. It ranges from 0.5 (no discriminatory accuracy, distribution of VAS for both categories identical) to 1.0 (perfect discriminatory accuracy, no overlapping of distribution of VAS for both categories). The AUC provides an estimation of the probability of correctly ranking a pair of randomly selected categories from the VRS on the VAS. The cut-off points were determined for each pair of the VRS scores separately using logistic regression analysis.
The repetition of the training procedure at the patient’s final conclusion visit enabled a test-retest evaluation several weeks or sometimes even months after the first training. Based on the cut-off points determined, the number of concordant and discordant pairs of classifications was summarized to evaluate a possible shift in the assessments on the VAS.
Results
Patient characteristics
Patient disposition

Differences between treatment groups
The superior efficacy of the triple combination acetylsalicylic acid + paracetamol + caffeine could be shown for all efficacy endpoints such as the time to 50% pain relief (primary endpoint), time until reduction of pain intensity to 10 mm, pain intensity difference, percentage sum of pain intensity difference weighted, extent of impairment of daily activities, and patient’s global efficacy assessment. Further details on the efficacy, tolerability and safety results were reported previously (11).
Frequency distribution of the assessment on VAS by VRS categories
The two extreme categories of the VRS (no pain and most severe pain imaginable) resulted in assessments very close to the anchor points of the VAS with very low inter-individual variability (Table 2). The frequency distribution of the VAS assessments for the other four scores of the VRS showed somewhat higher variability. The shape of the distribution was nearly symmetrical and did not markedly deviate from the normal distribution fitted to the data (Figure 1). Substantial overlapping of the VAS assessments could be seen for the categories mild and moderate pain as well as the categories severe and very severe pain. Except for a trend to slightly higher average values (less than 1 mm on the 100 mm VAS), inter-individual variability and overall frequency distributions of the VAS assessments were very similar for all categories of the VRS at the first training at the study entry and at the repetition at the study conclusion (Table 2, Figure 1).
Frequency distribution of the assessment on the visual analogue scale (VAS) sorted by the scores of the verbal rating scale (VRS) and the two visits. The observed frequencies and the fitted normal distribution are presented. Dotted lines mark the cut-off points on the VAS in the calibration on the VRS at visit 1. Descriptive statistics of the assessment on the visual analogue scale (VAS) sorted by the scores of the verbal rating scale (VRS)

Order consistency between VRS and VAS
Number and percentage of patients with permutations of the assessment on the visual analogue scale (VAS) in comparison to the order of the scores of the verbal rating scale (VRS)

Number of patients and percentages do not add up to total and 100%, respectively, as some patients may have several permutations.
Calibration of VAS on VRS
Cut-off points on the visual analogue scale (VAS) in the calibration on the verbal rating scale (VRS)

The corresponding ROC curves are displayed in Figures 2 and 3 and the calculated AUC summarized in Table 4. The ROC curves corresponding to cut-off points separating none and mild, moderate and severe, and very severe and most severe pain imaginable approached very closely the upper left corner of the coordinate system, indicating excellent discrimination between the respective categories. This was confirmed by the calculated AUCs very close to 1 (Table 4). The ROC curves corresponding to the other two cut-off points were also far from the diagonal of the coordinate system, with all AUCs above 0.832 also indicating very good discrimination between the VRS categories.
ROC curve for cut-off points of data from training at visit 1. ROC curve for cut-off points based on assessment at visit 4.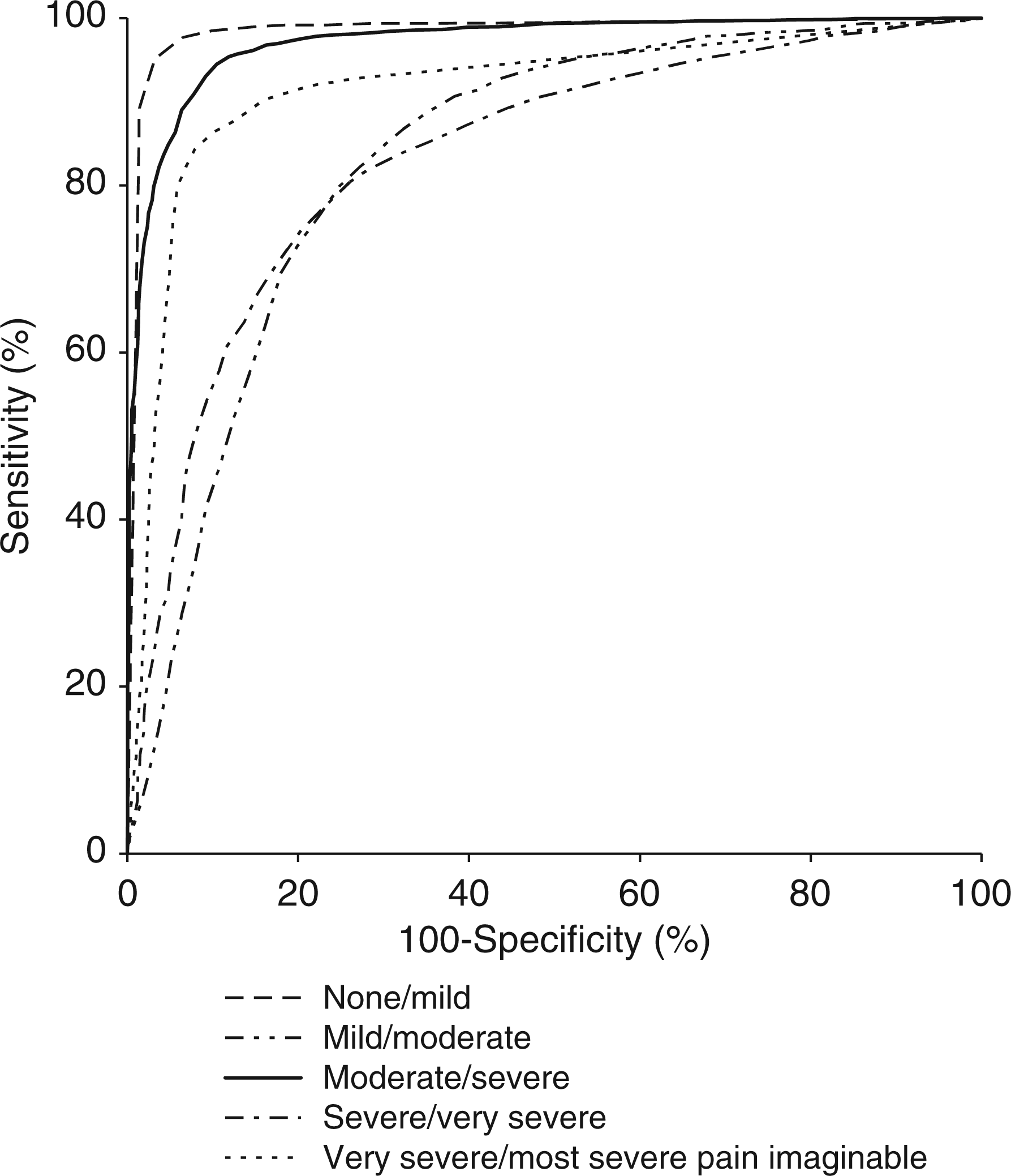

Test-retest assessment
Descriptive statistics of the difference between visit 1 and visit 4 in the assessment on the visual analogue scale (VAS) sorted by the scores of the verbal rating scale (VRS)
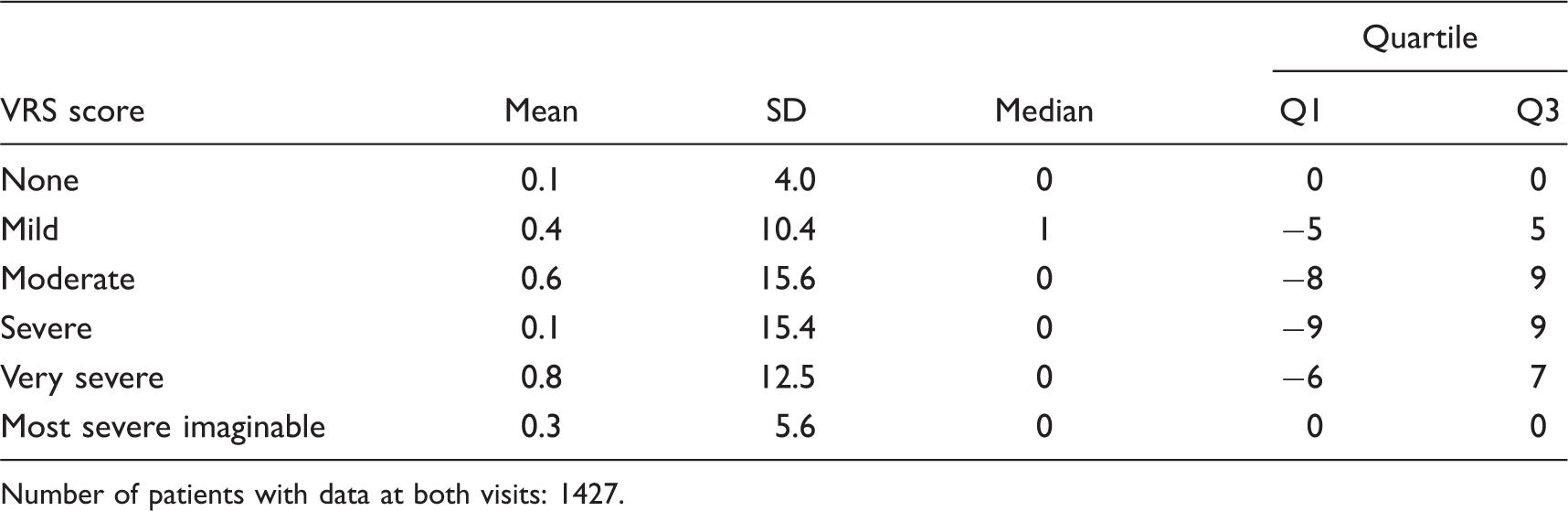
Number of patients with data at both visits: 1427.
Number and percentage of patients with a shift between visit 1 and visit 4 in the category defined by the cut-off points of the assessment on the visual analogue scale (VAS) at visit 1

Number of patients with data at both visits: 1427.
Discussion
Pain is a personal, subjective experience, which makes it difficult to define and measure (14).
The continuous VAS and discrete categorical VRS are the two most commonly used scales to assess pain intensity/severity (1,15,16,17). The most common VAS consists of a 10 cm horizontal line (used in this study) or vertical line (range 5–20 cm) (18–20).
Usually, the VRS consists of 3–11 adjectives reflecting degrees of pain severity/intensity; the specific adjectives, such as ‘none’ or ‘no pain’, ‘little pain’, ‘slight pain’, ‘mild pain’, ‘moderate pain’, ‘severe pain’, ‘very severe pain’, ‘extreme pain’, ‘unbearable pain’, and ‘worst pain ever’, vary from version to version (21–24). Certain terms, such as little, slight, or mild pain, are often used synonymously in VRS, even though it has been shown that VAS scores differ significantly between, for instance, slight and mild (25). For headache pain, the necessary number of VRS categories has not been systematically examined. In migraine studies a 4-grade VRS (none, mild, moderate, severe) (‘Glaxo-’) scale of headache pain measurement was mostly chosen, as this was sufficient to depict the effect of sumatriptan and consequently that of other triptans in terms of their clinical development (26,27). However, the question of whether, from the perspective of headache patients, these four categories are really sufficient to depict their perceived headache intensity remains open. After all, in the current Thomapyrin Study 65 out of 1,765 patients rated the intensity of their usual headache as ‘very severe’ (28). With only four VRS categories, these patients are forced to choose ‘severe pain’, even though this underestimates their pain intensity. Serlin et al. refute that patients should be limited to rating pain as mild, moderate or severe only, and determine that ‘three categories do not tell us all we wish to know about pain severity for clinical trials or for clinical use’ (29). Moreover, a number of researchers have reported that reliability and respondent preferences for categorical scales are maximized with 7–11-grade scales (30–32).
Patients who preferred VAS over VRS justified their preference through the insufficient number of pain categories of the VRS, stating that the VAS was ‘more accurate,’ and that it ‘gave better indication of their pain’ (33,34). To be able to depict as many pain intensities as possible on the VAS, in the current study, a 6-grade VRS was chosen, which in particular also measures the ‘severe’ to ‘most severe pain imaginable’ pain intensities and which is becoming more frequently applied (35,36). As a VRS, with an uneven number of categories, can increase the risk of a regression towards the mean (37) and more than seven word descriptors are considered to decrease the reliability of the VRS (38), a 6-grade VRS was chosen, as was used in other pain and headache studies (39,40). A systematic investigation among chronic pain patients showed that 10 - and 11-point NRS, compared with a 101-point NRS, provide sufficient levels of discrimination, whereas a 6-point NRS proved to be slightly worse and a 2 - and 4-point NRS proved to be substantially worse (41). The 11-point NRS was 55% more sensitive than the 4-grade VRS (none, mild, moderate, severe) in detecting clinically important differences (6). The sensitivity was compared in a simulation model of postoperative pain. The 4-grade VRS was less sensitive than the VAS but the 11-point NRS and the VAS were approximately equally sensitive (42). However, it should be noted that the 11-point NRS and the 100 mm VAS are sometimes interchangeable and sometimes not (43,44).
At visit 1, for approximately 75% of patients, the ascending order of the six VRS categories was in accordance with a corresponding ascending order of the VAS ratings (increase in the mm values). According to Pilgrim (26), the requirement of an unambiguous translatability into a range of languages was a reason for choosing the 4-grade VRS as a measurement instrument for pain intensity in the clinical development of sumatriptan. However, at visit 1, 12.6% of the patients mixed up the terms ‘leicht’ (mild) and ‘mäßig’ (moderate) with regard to their order on the VAS, meaning that for these patients, the term ‘mäßig’ (moderate) represented a lower pain intensity than ‘leicht’ (mild). This does not suggest uniform terminology, and therefore argues against an easy translatability of these pain intensity-describing adjectives in German and English and vice versa. It is unclear whether the German VRS categories ‘mäßig’, ‘mittel’ or ‘mäßig stark’ can be equated with the English ‘moderate’ in terms of their meaning for pain patients. Other researchers regarded the terms ‘mäßig’ and ‘mittelstark’ in German as two different pain categories (45). In contrast to ‘leicht, mäßig, stark (strong) or sehr stark (very strong)’, the term ‘mittel’, in the sense of a medium pain intensity, calls for the notion of two anchor points or extremes, of which the pain intensity can then be judged as lying in the middle (at the same distance from the two). The term ‘mittel’, at least in German, does not fit ‘organically’ into a series of ‘kein’ (none), ‘leicht’ (mild), ‘mäßig’ (moderate), ‘stark’ (strong), ‘sehr stark’ (very strong), etc. Equally, the fact that the English term ‘moderate’ is used in both 4-grade VRS (cf. most triptan studies) and 5-grade VRS (34,45–49) also does not speak for its terminological clarity in English. In French, too, the term ‘modéré’ is used both in 5-grade VRS (‘absente’, ‘faible’, ‘modérée’, ‘intense’ and ‘extrément intense’ douleur (50,51) and in 4-grade VRS (‘pas du tout’, ‘un peu’,’modérée’, ‘sévère’ (52,53). In the SF 36, the English term ‘moderate’ and its official translations (Italian ‘moderato’; Danish ‘middelstærker smerter’) is also located above the middle in the 6-grade VRS (54).
Some researchers see an advantage of the VRS in the fact that the descriptors derive from general vocabulary and that this leads to category judgements on the basis of word meanings (55). By contrast, it has been pointed out on various occasions that it is difficult to specify exactly what will be taken as severe, mild, and so on (46); thus, this is also the case for headaches, where the definition of Tfelt-Hansen (27) was adopted with little modification into the ‘definition of terms’ of the ICHD-II.
Various studies showed a clear correlation between VAS and VRS (5,16,34,56–58). However, if solely the VAS is used, it is unclear which area on a VAS corresponds to the VRS categories mild, moderate or severe pain intensity, or, as Collins et al. put it in the title of their publication: ‘What is moderate pain in millimetres?’ (59). As clinical pain studies often require ‘at least moderate pain intensity’ as an inclusion criterion, on various occasions, and apparently arbitrarily so according to Collins et al. (59), the criterion has been operationalized on a 100 mm VAS with the guideline>30 mm (60), >60 mm (61) and >75 mm (62). Gursoy et al. (63) classify a VAS score of 0–4 mm as ‘no pain’, 5–44 mm as ‘mild pain’, 45–74 mm as ‘moderate pain’ and 75–100 mm as ‘severe pain’, while Page et al. classify <10 mm as mild pain, 10–50 mm as moderate pain and >50 mm as severe pain (64). Similarly, in migraine and headache studies, VAS areas have been allocated to VRS categories without further justification: for instance 0 cm as no pain, 1–3 cm mild pain, 4–7 cm moderate pain and 8–10 cm severe pain (65), or 1–3 cm mild, 4–6 cm moderate, 7–9 cm severe, 10 cm very severe pain (66), whereby in the latter, the reference to ICHD-II is incorrect, as it contains no such classification (67).
The VAS mean (\pm SD in mm) of the VRS categories of our study (none 1.3 ± 1.6, mild 13.4 ± 8.5, moderate 28.6 ± 13.8, severe 67.3 ± 13.7, very severe 83.7 ± 11.2, and most severe imaginable 98.3 ± 5.4) differed in part from those found in other studies. In a study with post-operative pain on movement (4-grade VRS), the following results emerged (mean and range in mm): for no pain 0.5 (0–10), slight pain 20.3 (2–50), moderate pain 36.8 (0–92) and severe pain 59.7 (21–92) (68). A knee surgery study (4-grade VRS) showed the following: none 1.4 ± 2.6 (0–28), mild 27.8 ± 10.4 (2–60), moderate 55.8 ± 11.7 (22–88), and severe 86.6 ± 9.9 (52–100) (69); while a laparotomy study (4-grade VRS) provided the following: none 0.0 ± 0.0 (0–15), mild 26.8 ± 9.4 (2–60), moderate 58.2 ± 8.6 (32–84), and severe 82.5 ± 10.0 (62–100) (69). A study of 785 migraine patients with and without aura (4-grade VRS) showed the following median VAS scores: no headache 0 mm, mild pain 20 mm, moderate pain 55 mm, and severe pain 82 mm (9).
Central to the current study was the question of how the cut-off points should be selected to separate the overlaps of distributions of two consecutive pain intensity categories regarding sensitivity and specificity in as optimal a manner as possible. One procedure for simultaneously reaching as optimal a sensitivity and specificity as possible, ‘the point closest to perfect differentiation and the point farthest from none’, as Perkins and Schisterman (70) put it, is the construction of a ROC curve (13). In headache research, ROC analysis has rarely been applied to date (66,71).
To convert VAS into VRS scores, cut-off points are mandatory (72). In the current study, the cut-off points were determined on the basis of ROC curves as follows: for no pain 0–2 mm, mild pain 2–17 mm, moderate pain 17–47 mm, severe pain 47–77 mm, very severe pain 77–96 mm, most severe pain imaginable 96–100 mm. They show high sensitivity values for the certainty of decision-making between the VRS categories. In line with expectation, this was lower for the category pairs mild/moderate and severe/very severe than for the others. Jensen et al. indicate that 100 mm VAS ratings of 0 to 4 mm can be considered no pain; 5 to 44 mm as mild pain; 45 to 74 mm as moderate pain; and 75 to 100 mm as severe pain (69); these clearly differ from the results of the current study. In terms of methodology, in our study the patients were not aware of how many pain descriptors would be mentioned to them. The upper cut-off point of the category moderate pain, with 47 mm, almost corresponds to the upper cut-off point of the category mild pain of Jensen et al., with 44 mm. In 603 patients with groin hernia repair, on the basis of paired VRS and VAS data, Loos et al. found cut-off points (in mm) of 0–8 as no pain, 9–32 as mild, 33–71 as moderate, and >71 as severe pain (72). Serlin et al. identified the cut-off points between the categories of a 4-grade VRS on the basis of an 11-point NRS. They found that classifying a score of 1 to 4 as mild pain, 5 to 6 as moderate pain and 7 to 10 as severe pain (29). Paul et al. found that classifying 1–4 as mild pain, >4–7 as moderate pain and >7–10 as severe pain constituted the optimal cut-off points (11-point NRS and 4-grade VRS) (73), whereas Palos et al. reported the cut-off points 1.3 to 3.6 (mild), 4.3 to 6.5 (moderate) and 7.5 to 9.8 (severe) (74).
The test-retest agreement between visits 1 and 4 in the current study was good. By contrast, the test-retest reliability (stability) was lowest for 2-grade, 3-grade and 4-grade VRS and was significantly higher for scales with more response categories; the most reliable scores were derived from scales with 7, 8, 9, or 10 response categories (32).
There are several limitations of this study that should be noted. Although the calibration of VRS on VAS was performed with headache patients, it was done during a pain-free interval, which might be considered both a strength and weakness of the study. A strength might be seen in the fact that the patients did not only undertake one VRS category, but rather all six VRS descriptors in a relatively short time interval and under virtually identical conditions, and without knowledge of the number of descriptors to be rated. In each case, the information was gathered once, meaning that a possible influence through a multiple pain measurement by means of various scales (69) was ruled out. However, the rating in a pain-free interval might also be considered a weakness, if one assumes that during the sensation of pain, a verbal pain intensity descriptor would be assigned a different VAS score. Nevertheless, this is not necessarily likely. A more critical effect might be exerted by the differing pain history of the patients, that is, based on the intensity of the previously experienced pain, the terms, and thus also the anchor points of the VAS, might acquire a different meaning. Moreover, an indication dependency cannot be ruled out, according to which, for instance, headache patients have not yet experienced ‘very severe pain’ in comparison to cancer patients. Nevertheless, this applies to all scales for measuring pain intensity. For this reason, Dionne et al. suggest an individual responder analysis for pain (75), which takes into account this individual pain experience.
Which scale, then, is particularly recommended for measuring pain intensity? Both the 4-grade VRS and the 100 mm VAS are recommended for the measurement of headache intensity in the IHS guidelines for clinical trials for migraine und tension-type headache (7,8), but there is no discussion of their relative merits (9). In the comments on ‘intensity of headache’, it is merely established that ‘this verbal/numerical scale is simple and has been used successfully in RCTs [randomised controlled trials] for acute migraine treatment for many years. Visual analogue scales are probably most suitable for trials that include patients with mild and moderate headache’ (7). The comments on ‘headache intensity’ in the tension-type headache (TTH) guideline state that ‘experience with the VAS is limited in TTH trials. However, as mentioned above, the pain in TTH is usually mild or moderate. The categorical verbal scale, commonly used for migraine attacks, may thus not be sensitive enough’ (8). The medical textbook ‘The Headaches’ (3rd edition) notes only that ‘simple efficacy parameters’ or ‘a clinically relevant decrease of headache on a simple verbal scale’ should be used (76). In the ‘note for guidance on clinical investigation of medicinal products for treatment of nociceptive pain’ of the Committee for Proprietary Medicinal Products of the European Medicines Agency, it is stated regarding ‘pain measurement’ that: ‘From the regulatory point of view, no specific recommendation for choice of rating scale is made. The applicant should discuss and justify the reasons of his choice taking into consideration factors such as population demographics, characteristics (e.g. intensity, duration), associated pathology, and concomitant medication. There are several scales or methodologies to assess pain, but none of them are completely free of problems’ (77). However, in the gold standard of pain research, the ‘Textbook of Pain’, Melzack and Katz (1) and McGrath and Unruh (78) discuss in detail the advantages, limitations and disadvantages of the various pain measurement scales. Melzack and Katz conclude that ‘although VRS and NRS are simple to administer and have demonstrated reliability and validity, the advantages associated with VAS make VAS the measurement instrument of choice when unidimensional measure of pain is required’ (1). Besides limitations of this recommendation for older patients, there are also other opinions in this regard (79).
Clark et al. point out that ‘the choice for using either scale depends on several factors: the clinician’s goal and the setting in which the scale is to be used (clinical or research), the patient’s level of formal education, and the training time available in the setting’ (34). Chapman determines that ‘only a few pain researchers critically examine their own measurement practices. We normally follow the tradition within our parent disciplines’ (80), while Altman adds that ‘it may be sensible to use measures that are common in other trials in the field, although popularity alone is not sufficient reason to use a measure’ (81).
In conclusion, this study shows that the VRS categories cannot be presented in an equidistant manner on the VAS, and that, contrary to previous assumptions, pain intensity descriptors are less clear and can have different meanings in different languages. Therefore, both in the 3rd edition of the International Headache Classification (ICHD-III) and in the guidelines for clinical trials of patients with headache illnesses, rather than a 4-grade VRS, a 6-grade or higher level VRS or a VAS should be recommended, with correspondingly broadly defined anchor points.
Footnotes
Funding
This work was supported by Boehringer Ingelheim Pharma GmbH & Co. KG, Germany.
Conflicts of interest
BA and HP are employees of Boehringer Ingelheim Pharma GmbH & Co. KG, Germany. BP declares no conflict of interest. HCD received honoraria for participation in clinical trials, contribution to advisory boards or oral presentations from: Addex Pharma, Allergan, Almirall, AstraZeneca, Bayer Vital, Berlin Chemie, Coherex, CoLucid, Boehringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Grünenthal, Janssen-Cilag, Lilly, La Roche, 3 M Medica, Medtronic, Menerini, Minster, MSD, Novartis, Johnson & Johnson, Pierre Fabre, Pfizer, Schaper and Brümmer, SanofiAventis and Weber & Weber. HCD has no ownership interest and does not own stocks of any pharmaceutical company.
