Abstract
Objective: The objective of our study was to field test different chronic migraine (CM) criteria and compare CM epidemiological profiles, which include demographic, personal, and lifestyle characteristics, with high-frequency episodic migraine (HFEM) and low-frequency episodic migraine (LFEM).
Methods: Questionnaires were mailed to a random sample of 18,000 18–65-year-olds in demographically diverse regions of Germany. The epidemiological data for the three classifications of CM, LFEM and HFEM were assessed using descriptive statistics, Pearson Chi-square, and analysis of variance tests.
Results: Among 9350 respondents, CM_I was the most restrictive (N = 37, 0.4%), followed by CM_II (N = 45, 0.5%) and CM_III (N = 185, 2.0%). CM groups did not differ in distribution by age, gender, body mass index, education or smoking and alcohol consumption. Compared to those with LFEM and HFEM, those with CM (CM_III) had significantly different epidemiological profiles.
Conclusions: CM prevalence varies by case definition. The epidemiological profiles of the three CM groups are similar but differ significantly from those of HFEM and LFEM. Optimal definitions for clinical practice and epidemiological research require additional field testing.
Introduction
The common primary headache disorders, migraine and tension-type headache (TTH), affect up to 80% of the general population worldwide (1). Both migraine and TTH headache are usually classified based on frequency of days with headache, and divide into two categories: episodic (<15 headache days per month) and chronic (≥15 headache days per month) (2). Primary chronic headaches (including both chronic migraine (CM) and chronic TTH) are common, affecting 3%–4% of the general population (3–8), and CM in particular is associated with a significant burden of illness for individuals, their families and society (9,10). There is growing evidence that headache chronicity in migraine as well as in TTH is not simply an increased quantity of headache days, but rather a qualitative shift based on different clinical and epidemiologic profiles, greater disability, increased co-morbidities, different pathophysiology and differences in treatment response (11). Clinical, neurological and functional studies of CM suggest a pathophysiological state in which the brain exhibits complex and pervasive alterations (12), in contrast to the intermittent changes seen in episodic migraine (EM). CM is also associated with a greater degree of impairment in cortical processing of sensory stimuli than EM, perhaps due to more pervasive or persistent cortical hyperexcitability (13).
Despite the recognition of CM and the proposed pathophysiological differentiation from EM, the nomenclature and diagnostic criteria of CM have been the subject of discussion during the past 30 years, leading to publication of a number of revisions to the CM classification (2,14–16). The majority of the data for field testing of CM criteria have been acquired from patients in specialized headache clinics and may therefore be biased, because the clinical population is not always representative of the disorder within the general population (17).
In this manuscript, we present the data from the population-based survey of the German Headache Consortium (GHC) study. We aimed to focus on the prevalence and epidemiology of CM using three common case definitions. Additionally, we have compared the epidemiology profile, including demographic, personal and lifestyle characteristics, for those who have CM with the profile of those who have EM. Bigal et al. conducted a study to field test the CM diagnostic criteria within a clinic-based population and identified overlap between those meeting the proposed criteria (17–19). Additionally, Buse et al. (20) utilized population-based data to compare those with EM to those with CM and identified significant differences between the two migraine populations. As suggested by these published studies, we hypothesized that there is significant overlap in the profiles between the varying CM classifications and that the population with chronic migraine will differ from that with EM.
Methods
The GHC study is a population-based, longitudinal cohort study designed to investigate the prevalence and incidence of headaches within the general population of Germany; data were collected in the years 2003–2005. The study was approved by the ethics committee of the University Duisburg-Essen, Germany. Informed written consent was obtained via mail from all subjects. Herein, we report the results of the baseline cross-sectional part of the study.
Study sample
The study population consisted of a random sample of 18,000 inhabitants drawn from three regions in Germany: the city of Essen, a large town (585,481 residents) in the region of North Rhine-Westphalia in the western part of Germany; the city of Münster, a medium-sized town (272,890 residents), also in the western part of Germany; and Sigmaringen, a rural area, consisting of a small town with 16,501 inhabitants and 20 surrounding villages in southern Germany. Inclusion criteria were: (i) age between 18 and 65 years, and (ii) German citizenship, to ensure proper knowledge of the German language.
Data collection
Figure 1 illustrates the study design. All potential study participants received a questionnaire via postal mail. Individuals who did not respond to the questionnaire were mailed a reminder two weeks later. Individuals who still did not respond via mail were called and asked to participate in a telephone interview, which was to be conducted by trained medical students based on the same questionnaire. After no response to the mailed survey and eight unsuccessful follow-up calls, individuals were considered as nonresponders. Additionally, individuals who refused the interview, either by postal response or by phone, were considered nonresponders.
Study flow and sample.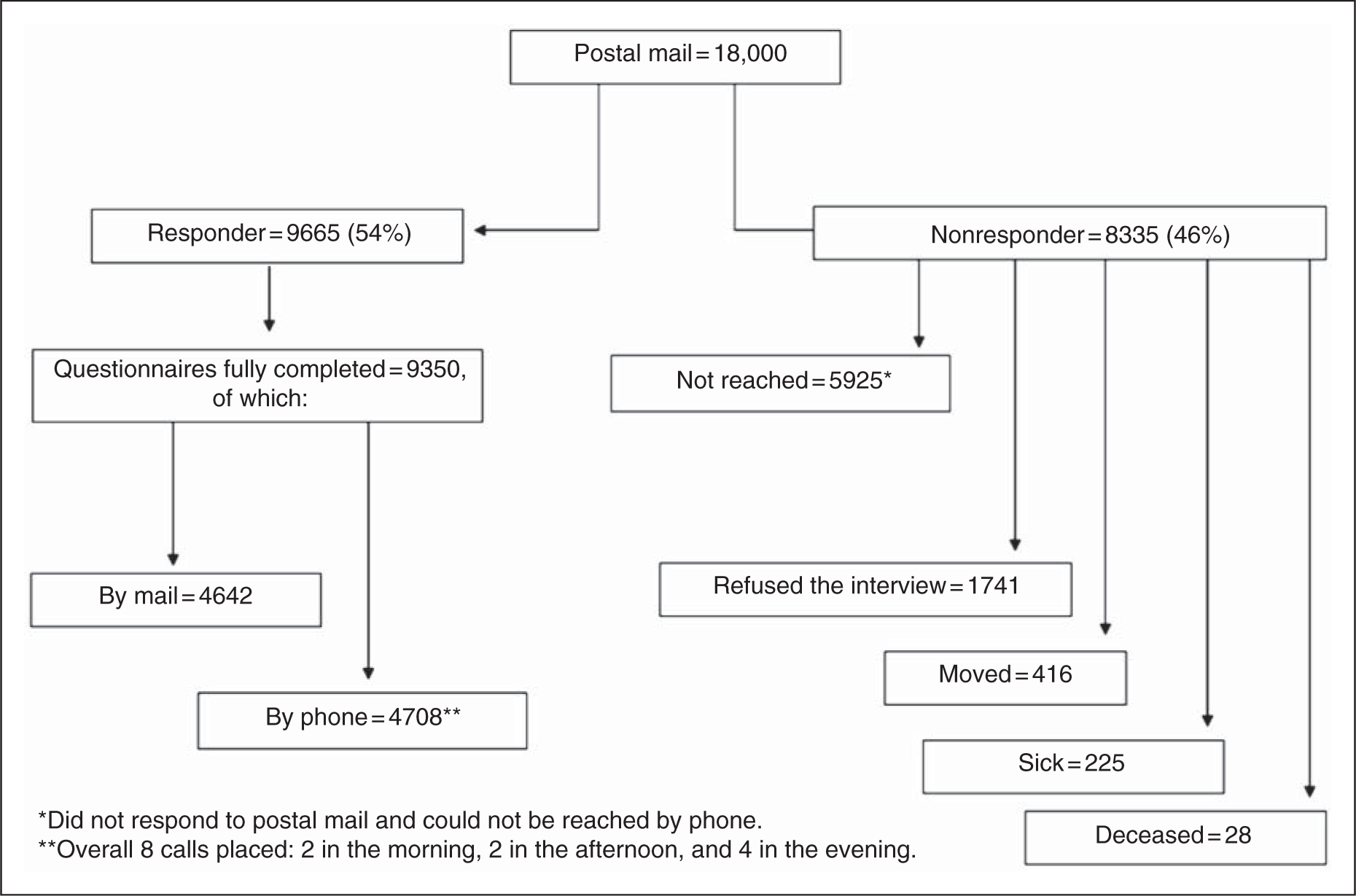
Description of questionnaire
A detailed description and validation of the questionnaire has been previously published (21,22). In summary, the questionnaire was based on International Classification of Headache Disorders (ICHD)-II classification criteria for headaches (2) and was designed to collect demographic data; information regarding migraine, TTH and trigeminal autonomic cephalalgias, including questions to ascertain the number of days associated with the different headache types; intake of acute and preventive headache or migraine drugs; intake of any medications; comorbidities; and socioeconomic status (based mostly on education and without any direct questions about income). Notably, the questionnaire allowed for the diagnosis of each headache type. For example, in a person with more than one headache type, one headache might be classified as migraine and another as TTH. The questionnaire asked a series of questions that allowed for the diagnosis of ICHD-II migraine and then asked on how many days per month one suffered from this type of headache. Another series of questions allowed for diagnosis of ICHD-II TTH and then asked on how many days per month one suffered from this type of headache. The questionnaire did not allow for the diagnosis of each headache attack; this type of data is best ascertained through a daily headache diary.
Classification of chronic migraine
Summary of chronic migraine classifications and diagnostic criteria


CM = chronic migraine. PREEMPT = Phase III Research Evaluating Migraine Prophylaxis Therapy. ICHD = International Classification of Headache Disorders. IHS = International Headache Society. aMedication overuse is defined as the revised 2006 ICHD medication overuse headache bHistory and physical and neurologic examinations do not suggest any of the disorders listed in groups 5–12, or history and/or physical and/or neurologic examinations do suggest such a disorder, but it is ruled out by appropriate investigations, or such disorder is present, but headache does not develop in close temporal relation to the disorder.
The role of medication overuse within the diagnosis of CM differs in these proposals. Silberstein and Lipton criteria stratify CM into those with or without medication overuse (16). The ICHD-II revised classification requires the exclusion of medication overuse headache as defined by ICHD-II under “8.2 medication-overuse headache” in order to definitively diagnose CM (2).
For the purposes of this study, each case definition included subjects with and without overuse of headache medication, which was defined as intake of acute headache medications within the previous month as defined by the ICHD-II (2) A limited sample size for CM within the study population did not allow a separate analysis of those with and without medication overuse.
Additionally, and in order to better evaluate the headache syndromes in these groups, we applied the ICHD-II criteria of TTH (2) to each case definition in order to assess if those who met CM also met criteria for chronic tension-type headache (CTTH).
Data analyses
Descriptive statistics were used to compare the epidemiological profiles for the three CM case definitions, and prevalences with 95% confidence intervals (CIs) were calculated by the number of cases per the number of total responders. The profile included variables of gender (males vs. females); age (in years); body mass index (BMI) (as an interval scaled variable); education (low level defined as duration of education ≤10 years vs. high level of education); smoking status (currently smoking vs. nonsmoking); and alcohol intake (daily intake of any kind of alcoholic beverages vs. less frequent intake).
Based on previous studies, we hypothesized that the epidemiological profiles for CM_I, CM_II and CM_III were similar. To test this hypothesis within these nested populations, we calculated the differences between CM_II and CM_I and between CM_III and CM_II and then compared “CM_I” to “CM_II minus CM_I” and to “CM_III minus CM_II.” Statistical differences were detected by Pearson Chi-square tests for nominal variables (e.g. gender, education) and analysis of variance (ANOVA) for comparisons of variables of continuous measures (e.g. age, BMI). If the global significance was achieved, we further performed two-by-two comparisons or Student’s T-tests with the subsequent Bonferroni correction for the triple comparisons.
In the second analysis, cross-sectional data were analyzed to compare the epidemiological profile of CM in study subjects with definitions of low-frequency EM (LFEM: 0–8 headache days per month) and high-frequency EM (HFEM: 9–14 headache days per month). For this purpose, the CM_III (not CM_I or CM_II) group was chosen, since it was the most inclusive. Descriptive statistics were utilized to compare the epidemiological profiles. Additionally, and similar to the CM case definition comparisons, each epidemiological variable was compared using either ANOVA or Pearson Chi-square tests. A p value of 0.05 was considered statistically significant. All analyses were performed using SPSS 15.0 (SPSS, Inc., Chicago, IL, USA).
Results
Study sample
Of the 18,000 subjects contacted, 9665 returned the questionnaire (54%). Of these, 9350 questionnaires were completed, with 4642 persons responding by mail and 4708 by phone interview (Figure 1). No differences were found in demographic characteristics when comparing study subjects who responded via mail to those who were interviewed by phone. The demographic characteristics of responders versus nonresponders differed slightly: responders were generally older (43.0 ± 13.1 vs. 40.6 ± 11.9 years) and included more women (52.5% vs. 49.4%).
Prevalence of chronic migraine according to the different definitions
The prevalence of any headache occurring on ≥15 days per month (chronic daily headache) was 2.9% (95% CI 2.6%–3.3%). All the CM case definitions identified were subsets of chronic daily headache, although the prevalence of CM varied by case definition. The smallest number of subjects was found to be in the CM_I classification (N = 37; 0.4%; 95% CI [0.27–0.52%]). This was followed in prevalence by CM_II (N = 45; 0.5%; 95% CI [0.34–0.62%]), and then CM_III (N = 185; 2.0%; 95%CI [1.7–2.2%]). CM_I, which was the most restrictive, was a subset of CM_II, which in turn was a subset of CM_III (Figure 2). The percentage of CM sufferers overusing acute headache medication varied by case definition: CM_I (11 of 37, 29.7%), CM_II (15 of 45, 33.3%), and CM_III (22 of 185, 11.9%).
Chronic migraine using different definitions.
Coexistence of tension-type headache and chronic migraine
TTH was diagnosed in 8 of 37 (21.6%) subjects with CM_I, in 10 of 45 (22.2%) subjects with CM_II, and in 54 of 185 (29.2%) subjects with CM_III. Interestingly, of 140 subjects who were diagnosed with CM_III and who did not meet the criteria for CM_I or CM_II, only 44 subjects (31.4%) were found to have a TTH, thus meeting criteria for CTTH.
Epidemiological profiles of chronic migraine
Comparison of the epidemiological profile by chronic migraine case definition*

CM = chronic migraine. SD = standard deviation. BMI = body mass index. df = degrees of freedom. ANOVA = analysis of variance. *Chronic migraine case definitions include those with and without medication overuse.
Current use of preventive medication (i.e. beta-blockers, anticonvulsants, antidepressants, and/or combination therapy) was not common in any of the CM case definition groups (CM_I = 2.7%, CM_II = 2.2%, CM_III = 7%). As the definitions became more inclusive, use of preventive medication increased. Patterns of acute headache medication use differed between case definitions; CM_I and CM_II represented similar patterns, but those classified as CM_III were more likely to report not using acute headache medications or using single analgesics only, and less likely to use combination analgesics compared to CM_I and CM_III.
Comparing prevalence and epidemiology profiles for chronic migraine with LFEM and HFEM
Prevalence varied by migraine groups (CM_III = 2.0%, 185 of 9350; HFEM = 2.4%, 228 of 9350; LFEM = 25.2%, 2356 of 9350) and was highest in the LFEM group. Women predominated in each of the migraine groups and ranged between 67% and 72% of the respective populations.
Comparison of the epidemiological profiles of the chronic migraine* and episodic migraine groups

ANOVA = analysis of variance. BMI = body mass index. HA = headache. CM = chronic migraine. HFEM = high-frequency episodic migraine. LFEM = low-frequency episodic migraine. df = degrees of freedom.
Chronic migraine was defined as those who met who met criteria for CM_III (not CM_I or CM_II) in order to represent all individuals who reported migraine regardless of migraine day frequency.
Discussion
In this study, the prevalence of any headache occurring on ≥15 days per month was 2.9%. This study also shows that the prevalence of CM in the population is determined by its definition. The three CM groups consisted of subjects with migraine who, at the time of the survey, reported any headache on 15 or more days per month. The prevalence of CM_III according to this least restrictive definition was 2.0%. Two other definitions were more restrictive, and required an addition to the diagnosis of migraine and headache frequency of ≥15 days per month and the predefined number of headache days to be migraine days (50% for CM_I and >8 for CM_II). The prevalences of CM_I and CM_II were therefore lower, at 0.4% and 0.5%, respectively. As defined, CM_I was a subset of CM_II, which was a subset of CM_III.
A recently published systematic literature search summarizing 12 population-based studies using different definitions of CM reported prevalence estimates between 0 and 5.1%, with estimates typically in the range of 1.4%–2.2% (28). This review highlights the variability of the results of CM prevalence estimates resulting from the use of differing criteria to classify CM (28). The prevalence of CM in this current study is also variable, depending on the definition of CM, and thus aligns with previous population-based studies.
Based on statistical analyses that demonstrated the lack of difference between the groups, an important conclusion of the study was that there were similarities among the epidemiological profiles of the three CM case definitions. All three populations were approximately 70% women, were on average 44–46 years old, and had a mean BMI of approximately 26. The percentage of people with a low level of education ranged between 70% and 78%. Approximately 43%–45% of people in these groups were current smokers. Differences were found in patterns of use of acute headache medications: subjects in the CM_I and CM_II groups used single analgesics more frequently (27% in both groups vs. 19% in the CM_III group) and used combined analgesics less frequently (5% in CM_I and 7% in CM_II, vs. 27% in CM_III). This finding is not surprising, given that by definition those who are CM_I and CM_II have more frequent days with migraine and thus are more likely to seek relief with acute medications.
Additionally, there were statistical differences among subjects in the CM, HFEM, and LFEM groups. Those with LFEM represented the majority of migraineurs. Compared to LFEM and HFEM, the CM population consisted of subjects who were significantly older, had a higher BMI, and were more likely to smoke (Table 3).
With regard to the epidemiological profiles of CM versus EM, our findings are also in line with previous studies (20,29,30). According to results of the American Migraine Prevalence and Prevention (AMPP) study, when comparing CM with EM, there were no significant differences found with regard to gender, but there were differences with regard to age, as those with CM were significantly older. Those with CM reported lower household income levels, were significantly less likely to be employed full-time, and were nearly twice as likely to be occupationally disabled (20).
We found that 275 subjects (2.9%) in our study had headache on ≥15 days per month. Of those, 185 (67.3%) were chronic migraineurs, according to the CM_III criteria. Only 37 (13.4%) of these subjects fit the definition of CM_I, and 45 (16.4%) would be classified as CM_II. This means, however, that 140 subjects with CM were excluded from the CM_III category under the more restrictive definition CM_II (Figure 2). Since this criterion was designed to reflect the ICHD-II revised criteria, the main question here is: what do these individuals have if they do not have CM? We therefore applied the ICHD-II criteria for TTH, and found out that approximately 20% of subjects with CM_I and CM_II and 30% of subjects with CM_III were diagnosed with a coexisting TTH. Even more important, of the 140 subjects who were included in CM_III but were excluded from CM_I and CM_II, only 40 subjects could be diagnosed with CTTH according to the ICHD-II. This means that the remaining 100 subjects with chronic headache (≥15 headaches days/month), of whom 15 overused acute headache medication, cannot be classified using ICHD-II criteria. Therefore, the majority of subjects did not fit into the criteria of medication overuse headache. Additionally, subjects did not fit into the criteria of TTH. Other chronic daily headaches such as hemicrania continua or new daily persistent headache are so rare that they could be eliminated.
The boundary between CM and CTTH has been controversial, but is quite important because of its therapeutic implications. It is a complex task to distinguish between CM and CTTH. There are several reasons for this complexity, including the fact that most individuals with CM have at least some attacks that resemble TTH, creating an overlap of attack profiles. In addition, some of these individuals overuse acute headache medications, which can make the headache phenotypes indistinguishable, particularly in individuals who are using triptans.
Our study contributes to the discussion of definitions of CM. Existing criteria were developed based on data gathered at specialized headache clinics (17,18), and are therefore very likely biased by severity of disease. Recent large-scale population-based studies provide a slightly different picture, which is probably less biased and therefore closer to the “true” picture of the disease. On the other hand, criteria used in the epidemiological studies are more simple but probably less precise. The international headache community has a difficult task: to find an optimal balance between the needs of population-based research, everyday clinical use, and scientific studies in academic headache centers.
We are aware of the methodological limitations of the study. First, if CTTH and CM exist in the same individual, this stretches the limits of our questionnaire, which allowed for separate diagnosis of each headache subtype but not separate diagnosis of each headache attack. Second, even a headache specialist depends on his/her clinical evaluation when making a diagnosis, which can lead to some bias in the results, although he/she is likely to be more precise than a diagnosis made by a screening questionnaire. In both cases, the diagnosis depends on how precisely the patients or respondents are able to recall specific features of the headaches in question. Use of a daily headache diary can help resolve diagnostic ambiguities in clinical practice and research, but daily diaries can be difficult and expensive to implement in large population-based studies. Another important challenge was a limited number of people with CM, especially if using the restrictive definitions. Therefore, there is reduced statistical power to compare profiles between CM_I and CM_II groups, and a separate analysis of CM with and without medication overuse was not possible.
The study has several strengths. In contrast with previous field testing of CM criteria that was performed in specialized headache clinics, we studied a large population-based sample in three different regions in Germany that cover both large- and medium-sized towns and rural areas. Although the demographic characteristics of responders and nonresponders differed slightly, probably due to an interest bias, we do believe that the data reflect with a reasonable approximation the natural picture of the disease, given that the population was randomly selected and that the study had a high participation rate for a population-based sample (54%). The questionnaire we used was validated twice for migraine and TTH, in patients of a tertiary headache center and in a population-based setting. Notably, the questionnaire was not validated to diagnose CM or CTTH, which is difficult, considering the low prevalence of CM in a population-based sample. The findings on prevalence and epidemiological profile of CM are in line with the international literature. We were able to clearly demonstrate the differences between CM and both low- and high-frequency episodic forms.
These results lead us to conclude that, first, the prevalence of CM varies by definition; second, epidemiological profiles of the three tested definitions are fairly similar; and, finally, an international consensus should be reached to facilitate research, learn more about chronic forms of primary headaches, and provide evidence for possible better definitions.
Footnotes
Disclosure
This research project was funded by the German Federal Ministry for Education and Research. Additional analyses were supported by Allergan, Inc., Irvine, CA, USA. The authors would like to thank Allergan, Inc., for funding and IntraMed Educational Group, New York, NY, for providing editorial support in the preparation and styling of this manuscript.
Conflict of interest statement
ZK has received research grants and honoraria from Allergan and Bayer, and is an advisory board member for Allergan. Headache research at the Department of Neurology in Essen is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF), and the European Union. AM and CCT are employees of Allergan, Inc. MSY has nothing to disclose. MO has nothing to disclose. PD has nothing to disclose. RBL has received honoraria and research funding from Allergan, in addition to serving as a consultant to and on the advisory board of Allergan. HCD has received honoraria for participation in clinical trials, contribution to advisory boards, or oral presentations from Addex Pharma, Allergan, Almirall, AstraZeneca, Bayer Vital, Berlin Chemie, CoLucid, Boehringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Grünenthal, Janssen-Cilag, Eli Lilly, La Roche, 3M Medica, Minster, MSD, Novartis, Johnson & Johnson, Pierre Fabre, Pfizer, Schaper & Brümmer, Sanofi-Aventis and Weber & Weber; and has received financial support for research projects from Allergan, Almirall, AstraZeneca, Bayer, GlaxoSmithKline, Janssen-Cilag and Pfizer.
