Abstract
Background: Clinical studies indicated that nitric oxide (NO) donors cause regional changes in cerebral blood flow (CBF), similar to those reported in spontaneous migraine. Systemic nitroglycerin (NTG), a NO donor, is a well-accepted experimental model of migraine. In this study we have examined the effects of NTG on the meningeal and cortical blood flow in rats.
Methods: Regional blood flow was monitored in male Sprague-Dawley rats using laser Doppler flowmetry before and after NTG/saline injection over 150 minutes. The effect of pre-treatment with Nω-nitro-L-arginine ester (L-NAME) or 7-nitroindazole (7-NI) on NTG-induced changes on blood flow was also investigated.
Results: In the dura NTG caused a biphasic response represented by an initial decrease in blood flow followed by a significant increase. At variance, in the cortex NTG caused only an increase in blood flow. Pre-treatment with either L-NAME or 7-NI prevented NTG-induced increase in blood flow in both districts, while only L-NAME also prevented NTG-induced decrease in dural blood flow.
Conclusion: The present findings provide additional information on the timing of effects of NTG on blood flow at both the meningeal and cortical levels. These effects seem to be related to vasoregulatory mechanisms and/or metabolic activity in response to the synthesis of endogenous NO.
Introduction
Nitric oxide (NO) is a potent vasodilator that increases CBF (1). In experimental animals, NO causes meningeal vessel dilation when given intravenously (2).
Changes in the diameter of cranial blood vessels as well as changes in CBF have been linked to the pathophysiology of migraine (3,4). However, mechanisms other than the purely vascular one have become more and more important. An increasing body of evidence suggests that NO may act directly on the trigeminal system neurons, and several reports demonstrate that trigeminal perivascular nerve fibers in dura mater are immunoreactive for NO synthase (NOS) (5–7). Nitroglycerin (NTG) administration evokes migraine-like attacks in migraineurs but not in healthy subjects (8,9), and has been used as a clinical model of migraine. In animal studies, NTG injection induces neuronal activity in several cerebral nuclei (10–12) and regional changes of CBF that are similar to the changes reported in spontaneous migraine (13). The migraine-provoking effect of NTG depends, at least partly, on the local formation of NO in intra- and extra-cranial blood vessels, although it is not clear whether this effect depends upon exogenous NO, endogenous NO or both (14,15). Previous reports from our group and others support the idea that de novo synthesis of NO—induced by NTG—is likely one of the NTG-mediated effects in specific neuronal areas (16–18).
In order to further investigate the pathophysiological mechanisms underlying migraine attacks, we investigated in rats the changes in blood flow induced by systemic NTG simultaneously at the meningeal and cortical levels. Furthermore, we evaluated the effect of different inhibitors of NO synthase (NOS) upon these vascular changes for elucidating the possible contribution of endogenous NO.
Materials and methods
Animals
Male Sprague-Dawley rats (Charles River, Calco, LC, Italy), weighing 250–350 g, were housed at 20–22°C on a 12-hour light-dark cycle in cages where they had access to food and water ad libitum. All the experiments were performed in accordance with the European Convention for Animal Care and Use of Laboratory Animals and were approved by the local Animal Care Committee.
Drugs
Experimental groups and treatments

CBF monitoring
Rats were anesthetized with sodium thiopental (50 mg/kg) IP. Depth of anesthesia was regularly controlled during the experiments by checking a nociceptive reflex (paw pinching); additional thiopental was administered IP as necessary. Arterial blood gas levels and pH were maintained within normal limits throughout the experiments. Core temperature of the animals was kept at 37°C by an electronically regulated heating pad. Blood pressure was monitored non-invasively throughout experiments with a cuff system applied at the tail (NIBP 23934, IITC Life Science, Woodland Hills, CA, USA).
CBF was measured with laser Doppler (LD) flowmetry with a two-channel flow monitor by means of an Oxyflo instrument (Oxyflo 2000, Oxford Optronix Ltd, Oxford, UK). The head of the animals was fixed in a stereotaxic head holder. After exposing the skull, a burr hole (1.5 mm in diameter) was opened using a micro drill, 2.5 mm posterior and 5.0 mm lateral to the bregma, corresponding to the primary somatosensory cortex. The position of the probe was determined by using the stereotaxic coordinates of Paxinos and Watson (Paxinos 1982). For the measurements of the meningeal flux, a needle-type LD probe (0.8 mm in diameter) was gently lowered on the dura, while for the measurements of cortical flow in the primary somatosensory cortex, the dura was opened and the same LD probe was inserted 3 mm deep into the brain parenchyma.
Microvascular blood perfusion was recorded in blood perfusion unit (BPU), which is a relative unit scale defined using a carefully controlled motility standard, and which is obtained as the product of mean red blood cell velocity and mean red blood cell concentration in the volume of tissue under illumination from the probe.
Experimental design
Experimental groups and treatments are described in Table 1.
All measurements started at least 30 minutes after surgery to allow stabilization of LD-flux signals. Signals were recorded for 30 minutes at baseline and subsequently for 150 minutes after the administration of NTG or vehicle. Subsequently animals were euthanized by means of the injection of an excess dose of thiopental. Observation periods longer than 150 minutes were attempted, but prolongation of anesthesia beyond that limit was associated, in the majority of animals, with significant changes in blood gases and pH.
Data analysis
All data are presented as mean ± standard deviation (SD). CBF was measured every 30 minutes for a total period of 150 minutes. Data were expressed as percentage changes from the baseline value (100%) and they represent the mean value of blood flow calculated within a range of 5 minutes.
As regards systemic blood pressure, values were recorded as mean blood pressure (MBP) at baseline and, subsequently, at intervals of 30 minutes. Data were expressed as percentage changes from the baseline value, which was equal to 100. To quantify the relationship between MAP and variations of CBF, the Pearson correlation coefficient (r) was evaluated 30 and 150 minutes after NTG injection.
Statistical analysis of the effects of NTG on dural or cortical blood flow and on systemic blood pressure was carried out using the one-way analysis of variance (ANOVA) for repeated measures followed by post hoc Tukey’s test; the effect of different pre-treatments was evaluated with two-way ANOVA followed by Bonferroni test. A value of p < .05 was considered as statistically significant.
Results
Effects of nitroglycerin on blood flow
Baseline values of dural and cortical blood flow in the different experimental groups are illustrated in Table 2. Neither dural nor cortical blood flow changed significantly over time in the group of animals treated with vehicle (Figure 1a and b). In the dura mater, NTG administration caused an initial decrease in the blood flow, followed by a progressive increase, which reached a significant level at 90 minutes and the maximal value at 150 minutes, when it was about 30% more than baseline. In the cortex, we detected an increase that reached a significant level at 90 minutes and the maximum amplitude at 150 minutes, when it was nearly 48% of the baseline.
Dural (a) and cortical blood flow (b) following systemic administration of nitroglycerin (NTG) or vehicle. Data are shown as mean percentage changes from baseline (baseline = 100%) ± SD. One-way analysis of variance (ANOVA) for repeated measures p < .0001 F = 25.38 followed by Tukey's test. ^p < .05, Basal values of cerebral blood flow in the different experimental groups Data are shown as mean of blood perfusion unit (BPU) ± SD.

Effects of nitroglycerin on mean blood pressure
Mean blood pressure values in the different experimental groups
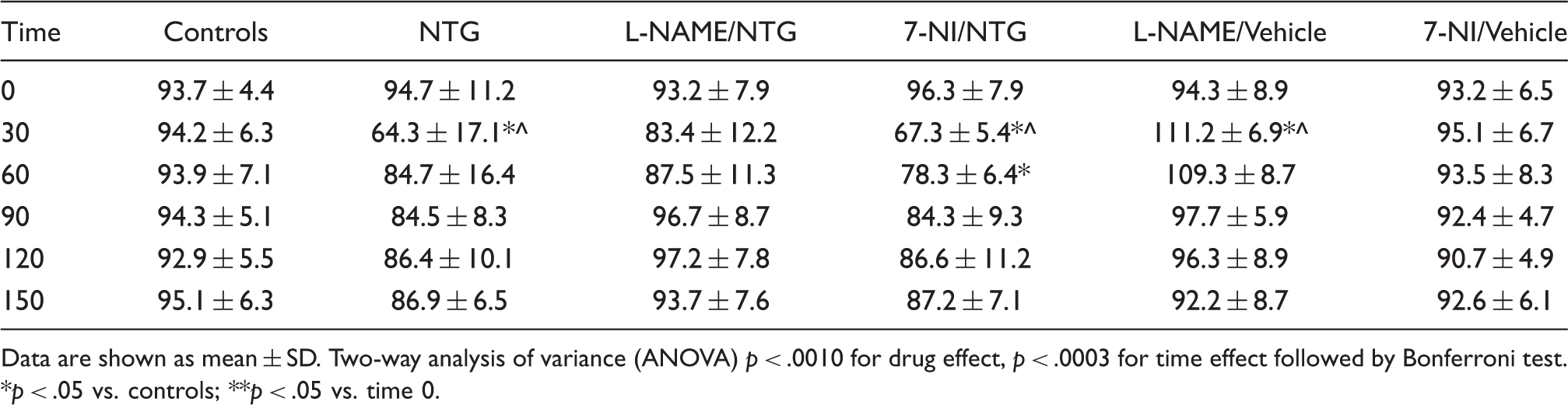
Data are shown as mean ± SD. Two-way analysis of variance (ANOVA) p < .0010 for drug effect, p < .0003 for time effect followed by Bonferroni test. *p < .05 vs. controls; **p < .05 vs. time 0.
Correlation between mean blood pressure and CBF in NTG-treated animals
The relationship between MBP and changes in CBF was evaluated 30 and 150 minutes after NTG injection (Figure 2). A trend toward a positive correlation between dural blood flow and MBP (r = 0.7399; p = .0572) was observed 30 minutes after NTG injection, while a significant inverse relationship was observed between cortical blood flow and MBP changes (R2 = 0,676; r = −0.796; p = .031).
Relationship between NTG-induced changes on dural (left column) or cortical (right column) blood flow (BF) and mean blood pressure (MBP) 30 and 150 minutes after nitroglycerin (NTG) administration. Data are shown as mean percentage changes from baseline (baseline = 100%) ± SD.
Effects of pre-treatment with L-NAME and 7-NI on CBF
L-NAME completely inhibited NTG-induced changes in blood flow at both the dural and cortical levels (Figure 3a and b).
Effect of L-NAME on nitroglycerin (NTG)-induced changes on dural (a) and cortical (b) blood flow. L-NAME 50 mg/kg body weight (bw) (or vehicle) was injected intraperitoneally (IP) 30 minutes before the administration of NTG 10 mg/kg bw IP. Data are shown as mean percentage changes from baseline (baseline = 100%) ± SD. Two-way analysis of variance (ANOVA) p < .0001 for drug effect, p < .0001 for time effect followed by Bonferroni test. *p < .05; **p < .01; ***p < .001 vs. controls; #
p < .05; ###
p < .001 vs. NTG.
The selective inhibitor for neuronal NOS, 7-NI, did not interfere with the initial reduction in dural blood flow provoked by NTG, but it did prevent the subsequent increase observed at 120 and 150 minutes. Pre-treatment with 7-NI also prevented NTG-induced increase in cortical blood flow at 90, 120 and 150 minutes (Figure 4a and b).
Effect of 7-NI on nitroglycerin (NTG)-induced changes on dural (a) and cortical (b) blood flow. 7-NI 20 mg/kg body weight (bw) (or vehicle) was injected intraperitoneally (IP) 30 minutes before the administration of nitroglycerin (NTG) 10 mg/kg bw IP. Data are shown as mean percentage changes from baseline (baseline = 100%) ± SD. Two-way analysis of variance (ANOVA) p < .0010 for drug effect, p < .0003 for time effect. *p < .05; **p < .01, ***p < .001 vs. controls; #
p < .05; # #
p < .01; # # #
p < .001 vs. NTG.
Effects of pre-treatment with L-NAME and 7-NI on mean blood pressure
As regards MPB values, it is noteworthy that L-NAME pre-treatment seemed to prevent an NTG-related fall in systemic blood pressure, while 7-NI pre-treatment did not influence NTG-induced hypotension (Table 3).
Discussion
Systemic NTG administration is a well-accepted and extensively used experimental model of migraine. NTG is a potent vasodilator and its effect upon cortical and dural circulation may be relevant for further understanding the neurovascular mechanisms underlying migraine. Unfortunately, previous studies evaluating the effect of NTG on the cranial circulation in humans and animals yield inconclusive results, mostly due to the different experimental settings and protocols. A clinical study showed that intravenous (IV) infusion of NTG at different rates in patients with migraine without aura causes a decrease in blood velocity in the middle cerebral artery (MCA) without affecting cortical blood flow in the perfusion territory of the same artery (19). On the other hand, Bednarczyk et al. (2002), using positron emission tomography and transcranial Doppler, showed that in healthy subjects NTG causes an increase of regional blood flow in the cingulate cortex associated with a reduction of blood velocity in the MCA (13). In rats, NTG infusion produces a significant increase in cortical blood flow versus baseline, within five minutes of the initiation of IV infusion, subsequently remaining stable throughout for 80 minutes (20). Conversely, Srikiatkhachorn et al. (2002), showed that NTG infusion (10 mg/kg IV) in rats causes an initial, short-lasting hypoxemia, followed by a long-lasting increase in cortical blood flow (21).
Very few studies have investigated changes in blood flow at the dural level. Strecker et al. (2002) evaluated dural blood flow in the rat following the local application of different NO donors (but not NTG) and they observed dose-dependent increases in meningeal blood flow, that is in part the result of the release of calcitonin gene-related peptide (CGRP) (22). Similarly, Gozalov et al. (2008) showed that NTG causes a transient increase in dural blood flow associated with a significant dilatation of meningeal (dural and pial) arteries in rats (23). Taken together, these findings reasonably suggest that NTG administration dilates large cranial arteries as well as small pial vessels; however, the findings provide very little information on the timing of these events and on the potential specific effect of NTG on the small arterioles of the cortex.
In the present study, we show that NTG induces differential variations in the blood flow at the meningeal and the cortical levels. In the dura, we recorded a biphasic response consisting of an initial decrease in blood flow (at 30 minutes) followed by a significant increase. At variance, in the cortex we detected only an increase in blood flow, whose timing was similar to the increase observed at the dural level. The strength of the present findings resides in the fact that we evaluated both circulatory districts with the same technique, LD flowmetry, using the same experimental settings and for a quite long length of time (probably the longest allowed by the experimental conditions), with the simultaneous recording of blood pressure changes.
This evaluation allows us to hypothesize that the initial NTG-induced decrease in dural blood flow is in part secondary to NTG-induced hypotension, as suggested by the trend toward a positive correlation between dural flow and MBP 30 minutes after NTG injection (r = 0.7399; p = .0572). The presence of complex auto-regulatory mechanisms at the cortical level (24–26) may likely explain the absence of changes in cortical blood flow during the initial hypotension caused by NTG. Indeed, a significant negative correlation was reported between cortical blood flow and MBP 30 minutes after NTG administration (r = −0.796; p = .031). The subsequent and persistent increase in blood flow observed in the dura (up to 150 minutes) is unlikely related to the effect of a nitrovasodilator upon systemic blood pressure, as we demonstrated that, in this experimental paradigm, blood pressure had recovered baseline values as early as 60 minutes after the drug administration. Based on the data from the literature (10,11,15,18,19,27–30), we can hypothesize that the increase in dural cerebral flow observed 90 minutes after NTG infusion may be related either to NTG-derived NO that promotes the formation of soluble guanylyl cyclase (31) or to the possible sensitization of perivascular nociceptors and central nociceptive neurons in trigeminovascular system (14,15,33–35).
As regards the effect of NTG on cortical blood flow, the blood flow increase observed in the cortex—starting 90 minutes after NTG administration and persisting up to 150 minutes—may be attributed in part to the local accumulation of NTG-derived NO in the brain tissue adjacent to the site of registration. Systemic NTG indeed accumulates in the brain parenchyma, reaching a maximal concentration two hours after its administration (36). An alternative explanation might be the activation of the trigemino-vascular-mediated release of vasodilatory substances following NO-induced trigeminal sensitization. However, it is uncertain whether cortical arterioles are innervated by the trigemino-vascular innervation. Arterioles in the cortex are innervated by the noradrenergic system, arising mostly from the locus coeruleus (37), which represents the major candidate for the neurogenic control of capillary and microarterioles in the cortex (38). In this framework it is worth considering that CGRP-mediated vasodilatation may theoretically occur also independently of the trigemino-vascular system as suggested by the increase in CGRP-like immunoreactivity observed in vascular districts devoid of trigeminovascular innervation, that is, the aorta and the heart (39). Additionally, the increase of blood flow in the cortex may be also a consequence of neuronal activity. Cerebral circulation indeed may be influenced by neuronal activity via pathways extrinsic and intrinsic to the central nervous system (40,41). NO is a likely mediator of the coupling between neuronal activation and CBF (40–44). This hypothesis seems particularly exciting when considering the old and recent findings regarding the talk between cortical flow and trigeminal neurons during experimental spreading depression (45,46).
Role of endogenous NO
The possible role of endogenous NO in the changes induced by NTG upon dural and cortical blood flow was investigated by probing the effect of the pre-treatment with L-NAME or 7-NI. Systemic administration of L-NAME completely inhibited NTG-induced changes in both cranial districts (cortex and dura mater), whereas 7-NI only prevented NTG-induced increase in blood flow in the dura and in cortex, but failed to inhibit the initial reduction in blood flow observed in the dura. Our finding suggest that the inhibitory effect of L-NAME on the decrease in dural blood flow is probably related to its counteracting effect upon NTG-induced arterial hypotension; this is further suggested by the parallel failure of 7-NI to inhibit the NTG-induced reduction in dural blood flow and NTG-induced hypotension. Based on these data, we suggest that the blocking effects of both NOS inhibitors on NTG-induced increase in dural and cortical blood flow may depend on de novo synthesis of both endothelial and neuronal NO, rather than on the availability of exogenous NO.
This, at least in the dura, is in keeping with the hypothesis presented above on the capability of NTG to sensitize trigemino-vascular terminals and to induce a state of inflammation mediated by NO-dependent mechanisms (15). Our idea is in agreement with a previous study where it was suggested that the neuronal (n)NOS and endothelial (e)NOS isoforms are particularly important in dural vessel vasodilator responses and that L-NAME administration antagonizes neurogenic activation of the trigemino-vascular system also via the inhibition of CGRP release (2). Indeed, NOS inhibitors have been found to attenuate CGRP endothelium-dependent relaxation of smooth muscle by restricting the eNOS activity in the endothelium (47). One of the main sources of innervation of the cortical arterioles is represented by efferents from the locus coeruleus. In a previous report, we described the existence of neurons containing NOS in this brainstem nucleus, as well as in the nucleus trigeminalis caudalis (27). Based on the present results it is tempting to speculate that the inhibitory effect of the different NOS inhibitors may be ascribed either to the inhibition of endothelial NOS in the vessels wall of cortical arterioles receiving terminals originating in the locus coeruleus or to the interaction with neuronal NOS in the locus coeruleus and in the nucleus trigeminalis caudalis.
Conclusions
Our findings show that systemic NTG induces an early and short-lasting decrease in dural blood flow, followed by delayed hyperemia in the cortex and in the dura mater. The differences observed in the two districts are likely related to the existence of autoregulatory mechanisms in the cortical district, while the simultaneous delayed hyperemia is likely to reflect the activation of vasoregulatory mechanisms or metabolic activity in response to the local release of exogenous NO as well as to the synthesis of endogenous NO, locally or in neuronal areas innervating cortical and/or dural blood vessels (nucleus trigeminalis caudalis for the dura, locus coeruleus for the cortex). The data obtained provide further insights into the role of endogenous NO, produced by nNOS and in part by eNOS, in the mediation of mechanisms that may be relevant for migraine in the dural and cortical circulatory districts, as well as in their neuronal suppliers.
