Abstract
Background: The causes underlying idiopathic intracranial hypertension (IIH) are poorly understood.
Methods: To identify disease-related biomarkers that could offer a new insight into IIH pathology, we analyzed the cerebrospinal fluid (CSF) of 18 patients with IIH and 18 controls using two-dimensional fluorescence differential in-gel electrophoresis (2-D DIGE).
Results: We found six proteins that were upregulated in IIH (sterol regulatory element-binding protein 1, zinc-alpha-2-glycoprotein, immunoglobulin heavy constant alpha 1 [IGHA1], alpha-1-antitrypsin [SERPINA1], serotransferrin, haptoglobin) and four proteins that were downregulated (hemopexin, angiotensinogen, vitamin-D-binding protein, transthyretin). The validity of our approach was confirmed for one candidate protein (angiotensinogen). To account for a dependency from blood-CSF barrier function, the ratio of angiotensinogen and albumin CSF-to-serum quotients (Qang/Qalb) was determined, which confirmed the downregulation of angiotensinogen in IIH (p = .04).
Conclusion: Previous studies showed the intrinsic renin-angiotensin system (RAS) to regulate choroid plexus blood flow and CSF production. Altered levels of angiotensinogen could indicate an imbalance of the RAS in IIH that may provide new targets for therapeutic intervention.
Keywords
Introduction
Idiopathic intracranial hypertension (IIH), also known as pseudotumor cerebri (PTC) or benign intracranial hypertension (BIH), is characterized by an increased intracranial pressure (ICP) without evidence of intracranial pathology (1). The typical patient is a slightly obese but otherwise healthy woman in her second to third decade who presents with cephalalgia and visual disturbances (1). So far, the causes of IIH remain poorly understood (2). Several studies sought to identify risk factors associated with IIH, though apart from obesity no convincing evidence exists (3–7). An imbalance between cerebrospinal fluid (CSF) and blood flow due to CSF hypersecretion, increased CSF outflow resistance or increased cerebral venous pressure has been suggested as a possible pathomechanism underlying the disease (1,2). The hypothesis that a disbalance in CSF-blood homeostasis may contribute to IIH encouraged the search for CSF biomarkers of disease pathology, though so far none has gained clinical relevance (8–11). To discover new candidate markers, unbiased approaches like proteomics have been applied in various neurological diseases (12). To date, no published study analyzing the CSF proteome in IIH exists. A well-established tool for CSF proteome analysis known for a high sensitivity and good reproducibility is 2-D DIGE, a two-dimensional fluorescence differential in-gel electrophoresis (13). This approach is based on the separation of proteins pre-labeled with fluorescent dyes according to their charge in a first dimension by isoelectric focusing (IEF) and according to their size in a second dimension by sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE) (14). We aimed to identify a characteristic CSF protein pattern of IIH that could provide biomarkers of disease and offer new insight into the pathomechanisms underlying the disease.
Methods
Demographic and CSF data of patients included for 2-D DIGE

CSF = cerebrospinal fluid. 2-D DIGE = two-dimensional fluorescence differential in-gel electrophoresis. IIH = idiopathic intracranial hypertension. Qalb = albumin CSF-to-serum quotient. S = statistical significance in Kruskal-Wallis Analysis of Variance on Ranks. NS = not significant. Paresthesia = controls who presented with transient paresthesia and received lumbar puncture to rule out chronic inflammatory CNS disease. These patients did not show any deficits in neurological examination, MRI or CSF diagnostics. * CSF pressure measured in lying (horizontal) position. **Included in enzyme-linked immunosorbent assay (ELISA) only. §Ratio of angiotensinogen CSF-to-serum quotient (Qang) and albumin CSF-to-serum quotient (Qalb).
We used 2-D DIGE, allowing a simultaneous co-separation of multiple samples, as previously described (14,17). For each of the three 2-D-DIGE experiments we labeled 50 µg protein of each sample pool (IIH, controls, internal standard) with 400 pmol of the appropriate CyDye. Gel images were analyzed with commercially available DeCyder software (version 5.0, Amersham Biosciences, Uppsala, Sweden) using DIA (Difference In-gel Analysis) module and BVA (Biological Variation Analysis) module. Protein spots that showed a significant difference between the groups over three independent 2-D-DIGE gels were selected for further analysis with matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) mass spectrometry. Protein spot digestion and mass spectrometry were performed by TOPLAB GmbH (Martinsried, Germany). Differentially expressed protein spots of interest were manually picked and trypsinated and RP C18–enriched (ZipTip RP C18, Millipore, MA, USA). For MALDI-TOF MS analysis, enriched trypsin peptide solutions were spotted on stainless-steel MALDI sample plates, mixed with matrix solution and analyzed with a 4800 MALDI-TOF analyzer (Applied Biosystems, Darmstadt, Germany). Each spectrum was internally calibrated using the monoisotopic protonated masses of trypsin autolysis peptides. The observed m/z values between 700 and 4200 were submitted to MASCOT and ProFound (version 2004.01.26, Genomic Solutions, Huntingdon, Cambridgeshire, UK) for peptide mass-fingerprint searching, and spectra were analyzed by searching the non-redundant protein database of the National Center for Biotechnology Information (NCBI) (Bethesda, MD, USA). For database search, a mass range of 5–200 kDa and a pI range of 2–14 was applied. The chosen taxonomy was Homo sapiens and a mass tolerance of 100 ppm was used.
To validate the proteomic findings, further analysis was performed for one selected candidate protein. Angiotensinogen was determined using a sandwich enzyme-linked immunosorbent assay (ELISA) (IBL, Hamburg, Germany). Data analysis was performed using SPSS (Version 15.0 SPSS Inc., Chicago, IL, USA). Because of non-normal data distribution, the medians and interquartile ranges are shown. All correlations were studied using Spearman’s rank order correlation. Differences between multiple groups were compared using the Mann-Whitney rank sum test. Differences between multiple groups were compared using Kruskal-Wallis One Way Analysis of Variance on Ranks. p values < .05 were considered significant.
Results
We observed no significant difference regarding routine CSF analysis (cell count, total protein content, albumin CSF/serum quotient Qalb, lactate concentration) between the groups.
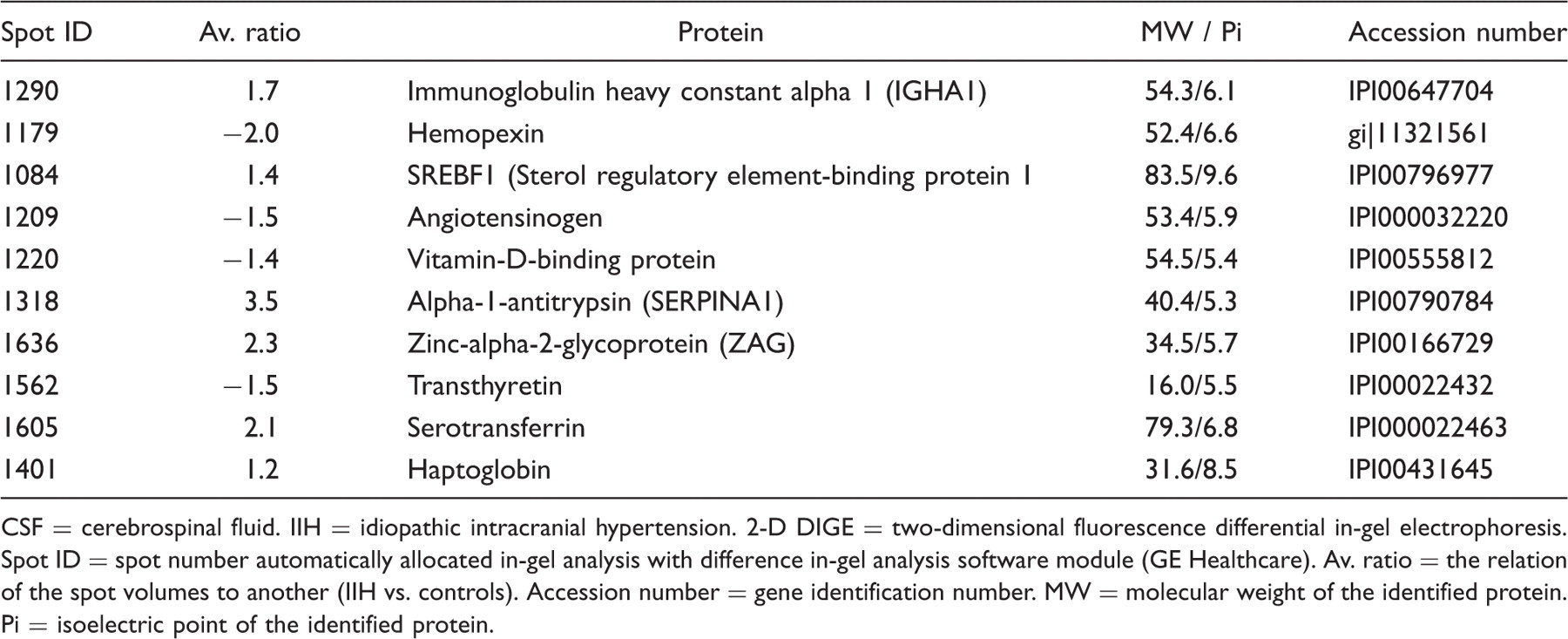
As a result of three independent 2-D-DIGE gels, 2175 total spots could be detected (Figure 1). Analysis of only those spots with a significant difference over all three gels between IIH and controls revealed 71 spots, corresponding to 10 different proteins and their isoforms (Table 2). Of these, six proteins were upregulated in IIH as compared to controls (immunglobulin heavy constant alpha 1 (IGHA1), alpha-1 antitrypsin (SERPINA1), sterol regulatory element–binding protein 1 (SREBF1), zinc-alpha-2-glycoprotein (ZAG), serotransferrin, haptoglobin) and four proteins were downregulated (hemopexin, angiotensinogen, vitamin D–binding protein, transthyretin).
Difference in-gel analysis of cerebrospinal fluid (CSF) from patients with idiopathic intracranial hypertension (IIH) and controls analyzed with two-dimensional fluorescence differential in-gel electrophoresis (2-D DIGE) and DeCyder two-dimensional difference analysis software (GE Healthcare). Only spots with a >1.2-fold difference between IIH and controls are shown. Upregulated spots in IIH: shown in blue. Downregulated spots in IIH: shown in red. Proteins identified in CSF of IIH by 2-D DIGE that showed a significant difference to controls over three independent gels CSF = cerebrospinal fluid. IIH = idiopathic intracranial hypertension. 2-D DIGE = two-dimensional fluorescence differential in-gel electrophoresis. Spot ID = spot number automatically allocated in-gel analysis with difference in-gel analysis software module (GE Healthcare). Av. ratio = the relation of the spot volumes to another (IIH vs. controls). Accession number = gene identification number. MW = molecular weight of the identified protein. Pi = isoelectric point of the identified protein.
We found the CSF-to-serum concentration ratio of angiotensinogen in controls to be 0.02. Based on a molecular weight of 49.8 kDa and a corresponding hydrodynamic radius of 30 Å, the estimated CSF-to-serum ratio according to Felgenhauer would be 0.01 if angiotensinogen found in CSF originated from the blood compartment via passive diffusion only (18). Consequently, approximately 50% of the angiotensinogen detected in CSF seems to be of intrathecal origin. We observed CSF angiotensinogen to correlate with increasing age (p = .03, rho = 0.4). Furthermore, a weak correlation of CSF and serum concentrations of angiotensinogen was observed (p = .04, rho = 0.04) and CSF angiotensinogen was found to correlate with blood-CSF barrier function as measured by Qalb (p = .003, rho = 0.5). No significant difference of angiotensinogen CSF or serum concentrations was found between IIH and controls (p = 0.9, p = 0.4). To allow for the dependency of CSF angiotensinogen from blood-CSF barrier function (Qalb), the angiotensinogen index (angiotensinogenCSF/angiotensinogenSerum/Qalb) was determined (19), showing a significant difference between IIH and controls (p = .04). The CSF and serum angiotensinogen levels observed in the second control group (transient paresthesia) were similar to those observed in TTH patients, and the significant difference of the angiotensinogen- ndex to IIH could be confirmed (p = 0.04).
Discussion
Identified proteins
To the best of our knowledge this is the first study analyzing the CSF proteome of patients with IIH. We identified a panel of proteins that were altered in CSF of patients with IIH as compared to controls, including angiotensinogen and hemopexin (Table 2). Humoral mechanisms were suggested to play an important role in the regulation of blood flow to the choroids plexus and CSF production (20,21). Consequently, a possible association of angiotensinogen with IIH pathology seemed intriguing, and this candidate marker was the first we selected for further validation. Hormones like renin and angiotensin were found to be synthesized by brain neuronal and glial cells (22), indicating the existence of an intrinsic renin-angiotensin system (RAS) (23). Studies in rodents and humans demonstrated the RAS to influence cerebral blood flow autoregulation (24). Furthermore, the choroid plexus, which produces the bulk of CSF, was shown to contain receptors for angiotensin II as well as high concentrations of renin and angiotensin-converting enzyme (ACE) (25,26). Both angiotensin I and angiotensin II were shown to selectively decrease blood flow to the choroid plexus as well as CSF production, an effect that seems to be mediated by angiotensin II receptors (21,27,28). Downregulation of the RAS in IIH as observed in our study could contribute to an increased CSF production. On a speculative level, it seems intriguing that modulation of brain angiotensinogen by estrogens observed in animal models (29) could contribute to the great preponderance of women among IIH patients, which is so far poorly understood.
Hemopexin is synthesized in the brain by ventricular ependymal cells and was suggested to play an important role in controlling iron distribution within the brain (30). Though alterations of iron metabolism have been discussed as possible risk factors for IIH (31), no direct link to IIH pathology is known so far. Alteration of hemopexin levels seems to be unspecific to IIH, as it was also observed in proteomic studies analyzing the CSF of patients with Alzheimer’s disease (32,33). As is the case of hemopexin, the disease specificity of some of the proteins identified here seems to be rather low. ZAG is widely distributed in different body fluids and has been observed to be altered in neurodegenerative diseases such as amyotrophic lateral sclerosis and frontotemporal dementia (34,35). Vitamin D–binding protein is best known for binding of vitamin D and its metabolites but also has immune regulatory functions implicated in chronic inflammatory CNS disease (36). Transferrin, transthyretin, alpha-1-antitrypsin and haptoglobin have been described in previous studies investigating the CSF proteome in inflammatory as well as neurodegenerative diseases (14,17,19,37). For example, transferrin was also found to be altered in CSF of patients with relapsing-remitting multiple sclerosis (RRMS) and Guillain-Barré syndrome (GBS) (14). The low disease specificity of many candidate proteins may be due to common pathological mechanisms underlying different neurological diseases as well as to methodological aspects of 2-D DIGE. On a methodological level these markers, which are all part of the acute or negative acute phase inflammatory reaction, may be proteins that are comparatively easy to detect using the specific setting of 2-D DIGE, which would account for their frequent observation in proteomic studies on very different neurological diseases (38).
SREBF1 is part of a family of transcription factors that regulate the synthesis of enzymes involved in sterol biosynthesis (39). Though the pathological relevance in IIH so far is unclear, elevated SREBF1 could indicate changes in lipid homeostasis and may offer a link to the observation of obesity as a risk factor of IIH.
Methodological considerations
Though 2-D DIGE is a well-established approach to CSF proteome analysis, some methodological aspects need to be considered to evaluate the results of our study. A basic problem in CSF proteome analysis is that concentrations of the brain-specific proteins are much lower than those of extra-cerebral proteins, such as albumin and immunoglobulins (40). To avoid an overlay of brain-specific proteins by blood-derived high-abundance proteins, CSF must be pre-processed by extracting the bulk of extra-cerebral proteins (14). However, potentially interesting proteins may remain bound to albumin or IgG and could thus be extracted from the CSF. To allow for the pre-processing steps, large quantities of CSF are necessary, a demand which was answered by pooling of CSF samples. However, pooling of CSF is controversial, as it may hinder the detection of markers of different subtypes of neurological disease that may be prominent only in individual patients (40). We included one single IIH patient with CSF opening pressure of 15 mmHg, which is nondiagnostic according to modified Dandy criteria (15). However, as pooled CSF was used in this proteomic approach and as the patient met all other criteria, it was included in this study. As both the control groups included in this study were not strictly normal controls, further studies including healthy volunteer samples are necessary.
Conclusion
Our study should be regarded as a pilot study to identify new biomarkers of IIH. The validity of the proteomic results could be confirmed for one of the identified proteins (angiotensinogen) using a quantitative assay. The intrinsic RAS of the brain may offer new targets for therapeutic intervention in IIH, though further studies on a larger cohort of patients are necessary to confirm the relevance of angiotensinogen as well as of the other candidate proteins identified here.
