Abstract
Introduction
Idiopathic intracranial hypertension (IIH) is characterized by increased intracranial pressure leading to headache and visual symptoms and signs (1). Its strongest and most consistent risk factors are obesity and female gender. The pathophysiology of IIH is still not completely understood, although several mechanisms, such as parenchymal oedema, increased cerebral blood volume, excessive cerebrospinal fluid (CSF) production, venous outflow obstruction or compromised CSF resorption, and lastly a possible contribution of inflammatory factors have been proposed (1, 2). It has also recently been suggested that anti-aquaporin-4 (AQP-4) water channel antibodies may be involved in IIH pathogenesis by affecting CSF secretion and absorption and thus leading to parenchymal oedema (3). AQPs are a family of water channels expressed in plasma membrane of many cell types in the central nervous system (CNS) and eye (4). AQP-4, situated in astrocytes, particularly at the blood–brain and brain–CSF barriers, is involved in bidirectional water transport (5).
Our aim was to investigate the presence of anti-AQP-4 antibodies as a possible causative factor in IIH patients and their clinical associations.
Methods
All patients seen in our headache outpatient clinics in the past 2 years fulfilling the modified Dandy’s diagnostic criteria for IIH were included after their informed consent. All had diagnostic lumbar punctures, MRIs, complete blood counts and blood biochemistry analyses. Their demographics, clinical, neuroimaging and CSF findings were all investigated from their files. Patients were interviewed when there were missing data.
AQP-4 antibodies were investigated in the sera of 29 IIH patients and 30 healthy controls. Furthermore, 27 patients with neuromyelitis optica (NMO), in which AQP-4 antibodies have been suggested to have a role in pathogenesis (6), and the sera of 283 consecutive patients with autoimmune/paraneoplastic or viral encephalitis were used as other control groups.
We used an immunofluorescence detection kit comprising AQP-4-transfected and non-transfected HEK-293 cells (Euroimmun, Lübeck, Germany). The antibody binding was visualized using a fluorescence microscope by two independent experienced observers, who were blinded to patients’ identity. The antibody binding intensity was scored by two investigators on a scale from 0 to 4, with 0 corresponding to negative and 4 corresponding to very strong staining.
In addition, an indirect immunohistochemistry test was performed to determine serum samples with the NMO immunoglobulin G (IgG) binding pattern. Whole rat brain was first treated with 4% paraformaldehyde overnight at 4°C, then immersed in 40% sucrose overnight at 4°C, and then snap frozen in liquid nitrogen. Seven micrometre-thick frozen sections were serially incubated with 0.3% H2O2 for 20 minutes, 10% goat serum for 1 hour and serum samples (1:200) overnight at 4°C. They were then incubated with biotinylated goat anti-human IgG (1:2000, Vector Laboratories, Burlingame, CA), and the immunoreactivity developed by serial incubation with avidin–biotin peroxidase (Vector Laboratories) for 1 hour and diaminobenzidine (7). The IgG binding patterns were classified as NMO IgG binding pattern, neuronal nuclear, neuronal cytoplasmic and neuropil (immunolabelling of the neuronal axons and dendrites located in cerebellar and/or hippocampal molecular layers) staining.
Results
The clinical findings of the patients with idiopathic intracranial hypertension
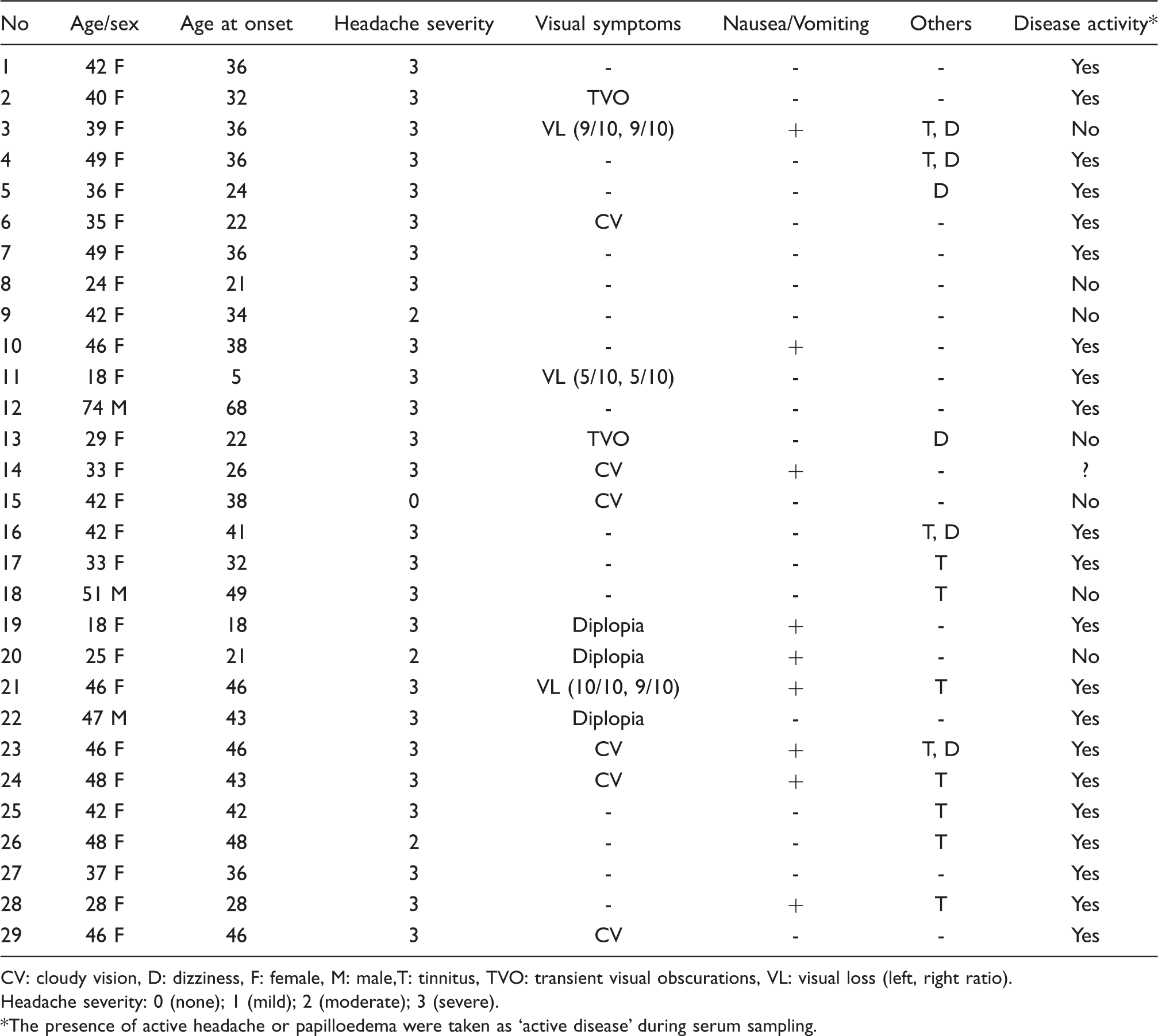
CV: cloudy vision, D: dizziness, F: female, M: male,T: tinnitus, TVO: transient visual obscurations, VL: visual loss (left, right ratio).
Headache severity: 0 (none); 1 (mild); 2 (moderate); 3 (severe).
The presence of active headache or papilloedema were taken as ‘active disease’ during serum sampling.
MRIs were normal in all patients and MR venography done in 15 patients showed normal results. CSF analysis did not reveal any abnormalities except increased opening CSF pressure (mean ± standard deviation, 282 ± 147 mm H2O; median: 345, range 200–840). Among all, two patients had minimal vision loss (9/10), whereas another one had vision loss of 5/10, necessitated optic nerve fenestration. Three of 29 patients were followed without any medication because of rapid remission of headache and papilloedema. Whereas 13 of the remaining 26 patients were taking only acetazolamide, 13 patients additionally received topiramate, an antidepressant medication, or both.
We could not detect AQP-4 antibodies in any of our IIH patients’ or healthy controls’ sera (score 0). Whereas 17 NMO patients of the control group were found out to be AQP-4 antibody positive, the remaining ten NMO patients were seronegative. The scores of patients with AQP-4 antibodies ranged between 2 and 4 (moderate to very strong staining). None of the tested sera showed reactivity with non-transfected HEK-293 cells.
Distribution of immunohistochemistry (IHC) staining patterns among groups

IIH: idiopathic intracranial hypertension, NMO: neuromyelitis optica, NMO IgG: vascular staining pattern observed in AQP-4 antibody-positive patients. Note that some patients showed more than one staining pattern.
Discussion
The discovery of the aquaporin (AQP) family of membrane water channels has provided new insights into the pathophysiology of brain water homeostasis. Aquaporin-4 (AQP4) has been presumed to have an important functional role in the transport of water in and out of the brain, owing to its wide distribution within the CNS, including the choroid plexus and ependymal cells of the ventricles, and its critical localization in astrocytic foot processes along the blood–brain barrier (BBB) and brain–CSF interface (8). Many studies of transgenic mice with a complete deficiency or altered expression of AQP4 suggest a prominent role for AQP4 in cerebral water transport (9). AQP4 seems to have a significant role in the development of cytotoxic oedema and the absorption of excess brain water resulting from vasogenic oedema. However, these important preclinical results have not been translated to human clinical diseases, except the association with neuromyelitis optica (6).
On the other hand, the pathogenesis of IIH remains still unclear, although it has been recognized for more than a century. Some patients experience a chronic disabling course with headache for years that limits their capacity to work and to participate in social life. Experimental pathophysiological studies are needed to optimize the biological understanding and therapy for this disease (10). Increased water content of the brain tissue in patients with IIH reported in some previous studies may indicate the involvement of brain water transport system in its pathogenesis (11). Another recent study suggested that brain lesions in some patients with NMO spectrum disorder may be accompanied by vasogenic oedema due to AQP-4 autoimmunity-related water flux impairment (12). The first published study hypothesizing that a dysfunction of AQP-4 expression could affect CSF secretion or absorption, and potentially contribute to the pathogenesis of IIH, investigated the AQP antibodies in ten female patients with IIH (3). To look for AQP-4 antibodies in the serum and CSF, the authors used a recently described quantitative fluorescence immunoprecipitation assay (FIPA) for each sample. As in our study, however, none of the IIH patients had detectable anti-AQP-4 antibodies.
One drawback of our study was that AQP-4 and anti-neuronal antibodies could not be investigated in the CSF of IIH patients. However, because of the substantial lack of intrathecal synthesis, AQP-4 antibodies are almost always detected in CSF of patients with high serum AQP-4 antibody levels and therefore testing for CSF AQP-4 antibodies is not expected to improve the sensitivity and specificity of the assay (13). We identified antibodies to the cerebellar molecular layer in most of our IIH patients (9/11 patients) with anti-neuronal antibodies. This staining pattern has often been associated with antibodies reacting with the conformational epitopes of neuronal cell surface antigens (7). Therefore, future studies using antibody screening assays that demonstrate conformational anti-neuronal antibodies (e.g. immunoprecipitation with cultured neuronal cells) would be expected to identify potential IIH-specific target autoantigens. Certain anti-neuronal antibodies, particularly those directed against cell membrane antigens, tend to be detected in the earlier stages of the disease (14). Therefore, a possible explanation for the absence of AQP-4 antibodies in our study could have been the sampling of the sera during the late and chronic stages of the disease. However, our IIH cohort consisted of several patients in the first few years of the disease and majority of the patients had active IIH symptoms during serum sampling, reducing this possibility.
In our previous studies, we have detected anti-neuronal antibodies with staining patterns that were identical to those of IIH patients in several diseases such as neuro-Behcet’s disease (established as an aetiology of cerebral venous thrombosis) and also Bickerstaff encephalitis (15,16). These antibodies might plausibly be the byproducts of an ongoing CNS inflammation that might have been produced following exposure of the CNS antigens to the immune system as a consequence of BBB disruption. The origins of the anti-neuronal antibodies detected in the sera of IIH patients, whether they have any pathogenic significance and whether they can be used as indicators of CNS inflammation or BBB disruption should be elucidated by future experiments.
Our study with a larger sample of IIH patients confirmed that anti-AQP-4 antibodies that are associated with cerebral water balance mechanisms do not have a causative role in IIH. However, we identified anti-neuronal antibodies in the sera of several IIH patients at rates comparable to those of encephalitis patients. Our results might suggest that inflammatory pathogenic mechanisms are in play at least in some IIH patients, and thus further investigation of potential neuronal target autoantigens is warranted to fully understand the pathophysiology of raised intracranial pressure in this mysterious condition.
Footnotes
Acknowledgements
This study was presented in a preliminary form as a poster in the 20th World Congress of Neurology, 12–17 November 2011, held in Marrakesh, Morocco.
