Abstract
Introduction: Behavioural animal studies are critical, particularly to translate results to human beings. Cortical spreading depression (CSD) has been implicated in migraine pathogenesis. We aimed to investigate the effects of CSD on the behaviour of freely moving rats, since available CSD models do not include awake animals.
Materials and methods: We developed a new model to induce single CSD by applying topical N-methyl-
Results: NMDA induced single CSD in ipsilateral cortex, evoked freezing behaviour (P < 0.01) and increased the number of wet dog shakes (WDS; P < 0.01). Grooming, locomotion, eating, drinking, and circling were not significantly altered among groups. Ultrasonic vocalisations compatible with pain calls (22–27 kHz) were only detected in 3 out of 25 rats. Sumatriptan did not significantly reduce the freezing behaviour. CSD induced significant c-fos expression in ipsilateral cerebral cortex and amygdala (P < 0.01).
Conclusions: CSD induces freezing behaviour by invoking anxiety/fear via amygdala activation in freely-moving rats. Single CSD is unlikely to lead to severe pain in freely-moving rats, though the development of mild or vague pain cannot be excluded. The relevance of rat behavioural responses triggered by CSD to migraine symptoms in humans needs further evaluation.
Introduction
Cortical spreading depression (CSD) indicates an extreme excitability state of the grey matter with massive redistribution of ions. The hallmark of CSD is a propagating wave of brief and massive neuronal excitation followed by prolonged inhibition (1,2). Recovery of spontaneous neuronal activity, as studied by electrocorticogram (ECoG), takes a few minutes, whereas recovery of evoked/synaptic neuronal activity takes much longer (up to 30 min) (1,2). As the induction and propagation of CSD depends strictly on glutamatergic neurotransmission, glutamate or N-methyl-
There has recently been an increased interest in CSD and a hypothesis that ‘CSD could also underlie migraine without aura attacks’ has been proposed (7,8). As the literature regarding CSD depends primarily on anaesthetised animal models, it is unclear whether the symptoms are fully comparable with those experienced by humans (9). Bolay and colleagues previously showed that CSD was a sufficient stimulus to activate the ipsilateral trigeminovascular system in the rodent brain by inducing neurogenic oedema and blood flow increase in the dura mater, as well as c-fos expression in the brainstem trigeminal nuclei (trigeminal nucleus caudalis, TNC) (10). A single event of CSD-induced blood flow increase in the dura mater was a trigeminal nerve mediated brain stem reflex which lasted up to 60 min in anaesthetised rats. The neurogenic blood flow increase in the middle meningeal artery that peaks around 20 min could reflect the possible time course of head pain following CSD (10). Depending on the previous data obtained in anaesthetised animals, we hypothesised that any symptom related to headache should be observable within 15–20 min following induction of CSD in freely moving rats.
As behavioural studies in animals are gaining importance as research tools, consistent behavioural correlates of headaches in rodents will improve the reliability of any migraine model. CSD in the gyrencephalic brain cannot propagate through the entire hemisphere due to limitations by major sulci (11,12); therefore, symptoms due to the neuronal dysfunction of the entire hemisphere are not seen as an aura in humans (4,13,14). This is one potential limitation to compare the behaviour of rodents having CSD with symptoms in humans. Although the effects of spreading depression in the cerebral cortex are well characterised, its direct (propagation of CSD) or indirect (due to transient disconnection from cortex) influences on subcortical and limbic structures in awake rats remain unclear. The link between CSD and pain behaviour has not been extensively evaluated in freely moving rats. Koroleva and Bures (15) concluded that spreading depression was not an aversive experience, as awake hooded Long–Evans rats did not display avoidance behaviour of the dark compartment when multiple CSD waves were induced by KCl.
We aimed to investigate behavioural alterations caused by CSD in order to address: (i) does CSD cause behaviour, suggestive of head pain, which can be observed and quantified in freely moving rats; and (ii) are there any behavioural characteristics of CSD in rats to employ for future migraine studies? We have developed a novel experimental model of single CSD in freely moving rats. We induced CSD by applying topical NMDA, and used a combination of an automated behavioural analysis system, video camera recording, and ultrasonic vocalisation (USV) calls to record behavioural changes.
Materials and methods
Animals
The study was performed in the Neuroscience Laboratory, Neuropsychiatry Centre, Gazi University. Ethical approval was obtained from Gazi University Animal Studies Ethical Committee. Male Wistar rats weighing 200–250 g were used. Animals were housed in a controlled environment where the room temperature was 22 ± 2°C and dark:light cycle was 12 h daylight and 12 h darkness. Rats received food and water ad libitum.
Induction of CSD
Animals were anaesthetised with ketamine (50 mg/kg) and xylazine (8 mg/kg). The depth of anaesthesia was adjusted according to the heart rate and hind leg pinch reaction. During all the surgical procedures, body temperature was kept constant at 37 ± 0.5°C with a heating pad. The animals were placed in a stereotaxic frame (Stoelting CO, USA). The skull was exposed by a median incision of the scalp. A burr hole with a diameter of 2 mm was opened over the right parietal cortex (3 mm lateral and 7 mm posterior to the bregma) by means of a mini-drill (FHC, USA). The dura mater was kept intact. Plastic tubing covering the burr hole was placed on the skull (8 mm height, 2.5 mm inner diameter, 3.5 mm outer diameter; Bicakcilar®, Turkey) and was fixed to the skull by dental acrylic (Meliodent, Heraus Kulzer®). To avoid drying of the dura mater, the tubing was always kept sealed with a cap. A stainless steel screw (1 mm diameter; PlasticsOne, USA) was attached to the occipital bone (2 mm posterior to lambda, 1 mm lateral) in order to secure the dental acrylic firmly. The animals were kept in the stereotaxic frame for another 30 min until the preparation was stable. A schematic illustration of the experimental preparation is shown in Figure 1. For CSD induction or in control experiments, 20 µl of NMDA solution or isotonic saline, respectively, were injected into the tubing with a pipette, without touching the dura mater. The animals were kept in their cage and were not handled. Before the drug or saline application, the fluid accumulated in the tubing was removed using a piece of blotting paper. The cap was only removed during NMDA or saline application, and the tubing was gently filled with solution, taking care to avoid air bubbles. All behavioural experiments were conducted during the day-time (12:00 pm ± 2:00 h) following 72 ± 5 h of recovery from surgery.
Schematic illustration of experimental rat preparation showing the locations of the ECoG recording electrodes and the burr hole for NMDA application. NMDA was applied into the tubing placed over a burr hole on the posterior parietal cortex and ECoG was recorded from 9 mm anterior to induction site.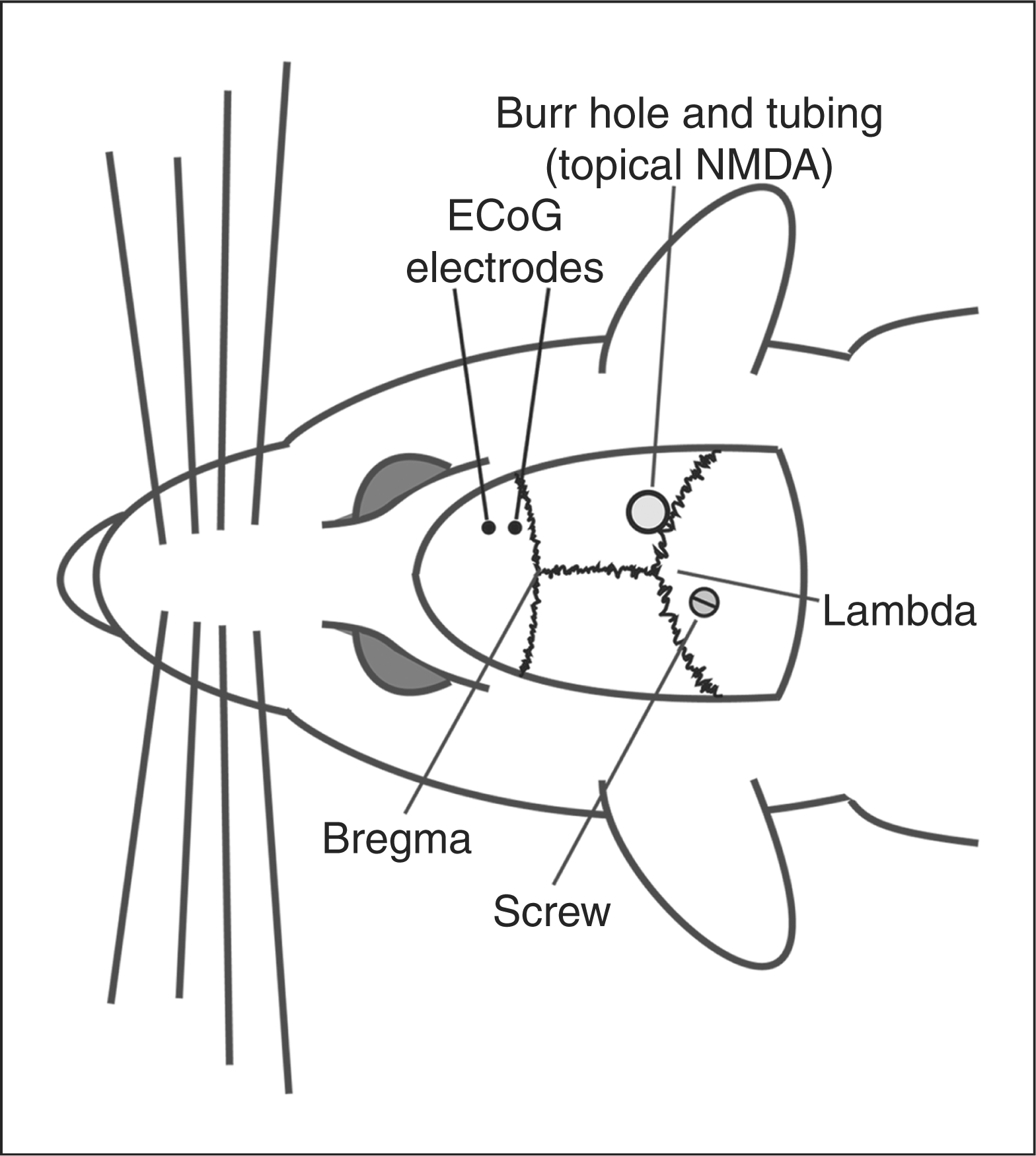
Behavioural experiments were conducted in three separate groups: (i) saline group (control, n = 9); (ii) NMDA group (n = 9); and (iii) sumatriptan succinate (SUMA) + NMDA group (n = 7).
Electrocorticogram recordings
Electrophysiological recordings during CSD were obtained in two different groups of rats. The first group of experiments were performed on four rats to determine the optimum NMDA dose for CSD induction. Anaesthesia was induced (sodium thiopental, 30 mg/kg) and the animals placed in a stereotaxic frame. An anterior craniotomy was performed (2 mm lateral and 2 mm anterior to bregma) and a side-by-side bipolar electrode (FHC, USA) was inserted in the cortex. The silver–silver chloride ground electrode was placed into the neck. Signal amplification and data analysis were performed via an EEG amplifier and a data acquisition system (Bioamplifier and Powerlab/8SP, AD Instruments, Australia).
Since the design of the automated behavioural analysis set-up did not allow simultaneous ECoG recordings, a second group of ECoG experiments were conducted on awake rats (n = 5). To monitor the cortical activity, bipolar electrodes (MS303, PlasticsOne, USA) were implanted in the frontal lobe (2 mm lateral, 1–2 mm anterior to bregma, 1 mm deep into the cortex) in addition to the parietal tubing. The rats were electrically grounded via a stainless steel screw electrodes fixed in the occipital bone. The tubing for NMDA or saline application was placed as in the behavioural experiments. Animals were connected to an AC amplifier (Biopac MP30, USA) with a thin cable that allowed the animals to move freely in a transparent observation box. The electrical signals from the electrodes were filtered between 0.05 Hz and 100 Hz, and sampled at 1 kHz using the Biopac MP30 system.
Drugs administered
NMDA was purchased from RBI, sumatriptan succinate was kindly provided by Glaxo Smith Kline. Sumatriptan succinate (300 µg/kg) was given subcutaneously 30 min before the topical NMDA administration. NMDA was dissolved in saline to a final concentration of 10 mM. The concentration of sumatriptan and topical NMDA (16,17), in addition to the timing of sumatriptan administration before the induction of CSD, were comparable with the literature (10,18).
Behavioural analysis
In this study, we evaluated animal behaviour such as locomotion, immobility, grooming, rearing, eating, drinking, purposeless chewing, clockwise (CW) and counter clockwise (CCW) turning, head shakes (HS) and wet dog shakes (WDS). Rat behaviours were automatically recorded by a non-invasive behavioural analysis system. This system is composed of a standard rat cage fixed on a platform with several force-displacement transducers, connected to a personal computer (Laboras®, Metris, The Netherlands). The platform detects and classifies the behaviours by using the vibrations created by movement (such as rearing, swallowing, circling, etc.) of the animal (19–21). Rats were free to access food or water located as in standard rat cages, and needed to rear to feed or drink.
All experiments were simultaneously recorded by a video-camera system in order to confirm the data obtained from automated analysis system, and to differentiate freezing periods from immobility.
Ultrasonic vocalisation (USV) calls
Many vertebrates use vocalisations to communicate in mother–offspring interactions, mating, affective or mood status (fear, pain or aggression), the presence of predators or the location of food. Most small rodent species, including laboratory rats and mice, emit USV calls (>20 kHz) (22). These vocalisations are inaudible to humans without the use of specialised equipment. Ultrasonic sounds (within a range of 15–100 kHz) of laboratory animals can be monitored and analyzed by a USV detector system (Sonotrack®, Metris, The Netherlands). Sonotrack uses a hardware bandpass filter (10th order Butterworth filter) with sharp cut-offs at 15 kHz and 100 kHz. This filter prevents aliasing and it also removes almost all environmental sounds. The data are presented without further filtering or smoothing. In Sonotrack, the dB (decibel) scale is relative to a 1 mV (RMS) signal. In the spectrogram, red indicates the strongest signal value (the 50 mV or 35.3 mV RMS or 31 dB) and black indicates the background noise (this is approximately 10 mV in Sonotrack or 7 mV RMS or 16 dB). The shift in the frequency at the beginning and at the end of the vocalisation is characteristic of a biological sound. The 22–27 kHz range of vocalisations in juvenile and adult rats indicates a negative affective state and is seen during, pain, startling, distressing events and exposure to predators. The 50 kHz vocalisations usually represent positive affective state.
Immunohistochemistry (IHC)
The animals were anaesthetised with a lethal dose of thiopental sodium 2 h after topical NMDA or saline application. They were perfused transcardially by heparinated saline, followed by 4% 0.1 M paraformaldehyde solution. The brains were prepared for c-fos immunohistochemistry, as previously described (5), and cerebral cortical sections of the whole cortex, 50 µm thick coronal sections at every 150 µm, were evaluated for c-fos immunoreactivity. Wide-spread (in all posterior to anterior sections) and intense c-fos immunoreactivity within all layers of cerebral cortex in the ipsilateral hemisphere was considered a characteristic feature of CSD. To test whether the freezing behaviour is related to activation of the amygdala, c-fos expression was also studied in amygdala. c-fos positive cells were counted in the amygdala (basolateral, central and medial nucleus were all included) bilaterally from bregma −1.4 to −3.8 mm, and expressed as the total number of cells.
Statistical analysis
The results are presented as mean ± SEM. Statistical analysis was performed using a one-way analysis of variance (ANOVA) and followed by a post-hoc (Tukey–Kramer) test, or a two-way ANOVA (locomotion) with repeated measures and Bonferroni post-hoc tests (Figure 3B), as appropriate. P < 0.05 was considered statistically significant.
Results
CSD induction by NMDA
NMDA in the 1 µM to 100 mM range was applied to the burr hole in the anaesthetised animals. Simultaneous electrophysiological recordings were also obtained to confirm CSD. While NMDA up to 1 mM did not induce CSD at all, NMDA 10–100 mM consistently evoked CSD. The 10 mM dose was chosen to induce CSD in awake animals. NMDA always induced a single CSD (Figure 2A), while KCl (3 M) induced multiple CSDs in the ipsilateral cortex.
ECoG recordings obtained from anaesthetised and freely moving rats following CSD induction with NMDA. (A) In anaesthetised rats, topical NMDA administration induced suppression of ECoG amplitude (lower trace) and an AC analogue of the DC shift (upper trace). NMDA application caused a single CSD. (B) In freely moving rats, NMDA suppressed ECoG amplitude (lower trace) and an AC analogue of the DC shift were also observed (upper trace). No activity resembling epileptiform discharges were detected in any of the experiments in particular during the freezing episodes. Movement artefacts obscured the ECoG activity. F, g, wds, and l denote ‘freezing’, ‘grooming’, ‘wet dog shake’ and ‘locomotion’, respectively.
ECoG and behaviour
In the freely moving rats, topical NMDA application temporarily suppressed the ECoG amplitude in 5.1 ± 0.9 min (Figure 2B). AC analogue of the DC shift was also observed by filtering the ECoG data with a bandpass filter (0.05–0.5 Hz; Figure 1B). The time period for full recovery of ECoG amplitude was found to be 14.2 ± 2.7 min. In all animals studied, NMDA-induced reduction in ECoG activity occurred only once. Freezing episodes were never observed before the NMDA application. The freezing episodes did not coincide with the CSD-induced ECoG amplitude suppression. Epileptiform discharges were not detected in any of the recordings, including freezing periods. Since behaviour during freezing episodes displays similarities to absence seizures, we specifically analyzed ECoG data during these episodes. Spike-wave discharges of 7–11 Hz specific to the absence type of epilepsy were not detected. In the period of ECoG amplitude suppression, animals were not paralysed, since they exhibited several motor activities like walking, grooming, WDS, and rearing. When depression of ECoG amplitude was detected (9 mm anterior to induction site), CSD had already invaded several cortical areas such as sensorimotor hindlimb and forelimb areas (bregma −3 mm and bregma +1 mm, respectively (21)) which are associated with behavioural reactions such as grooming, WDS or freezing. Those movement artefacts mostly obscured background neuronal activity and the AC analogue of DC shift in the ECoG (Figure 2B).
Behavioural results
CSD evoked a set of behavioural responses, of which freezing was the most prominent behavioural change. In another group of animals (n = 4), we prolonged observation period up to 12 h following CSD induction. We did not find any significant behavioural alterations beyond 60 min. Therefore, the experiments were designed to evaluate the first hour following NMDA application.
Freezing behaviour was recognised when rats suddenly became immobile and developed a fixed stare (staring empty), the latter being particularly important for brief freezing episodes lasting 2–3 s. Freezing episodes interrupted any on-going activity such as walking, rearing, grooming or eating. Immediately after the cessation of the brief freezing episodes, animals resumed their activity. The first freezing episode following NMDA application appeared at 3.9 ± 0.5 min. The duration of each freezing period was variable and ranged from seconds to minutes (2 s to 10 min). Prolonged freezing periods were differentiated from sleeping by observing open and blinking eyes. Freezing can easily and temporarily be disrupted by external stimuli such as touch, light and sound. Increased muscular tonus was not detected during freezing. Freezing was the only consistent behaviour in all animals with CSD, particularly during the first 15 min following CSD induction and sleeping became dominant after 30 min.
The total freezing time was significantly longer in the NMDA group compared to that of saline group (Figure 3A; P < 0.001). Sumatriptan treatment did not significantly reduce the duration of freezing (Figure 3A). Locomotor activity was evaluated by constructing cumulative distance travelled curves (Figure 3B). Although a reduced activity was observed in the NMDA group during the first half of the observation, the difference was not statistically significant with respect to saline group. The SUMA+NMDA curve was similar to the saline group curve (Figure 3B).
Total freezing durations and cumulative distance travelled over an hour indicating the locomotor activity. (A) NMDA application significantly increased total freezing period and sumatriptan pre-treatment did not alter NMDA-induced freezing. (B) All animals were tested for locomotor activity following topical drug administration. Cumulative distances travelled in 60-min period did not reveal any statistically significant difference. SAL, saline (control group). SUMA, sumatriptan pretreated group. Freezing data of each animal are shown in grey symbols, and mean ± SEM shown as dark lines. **P < 0.001, ***P < 0.0001.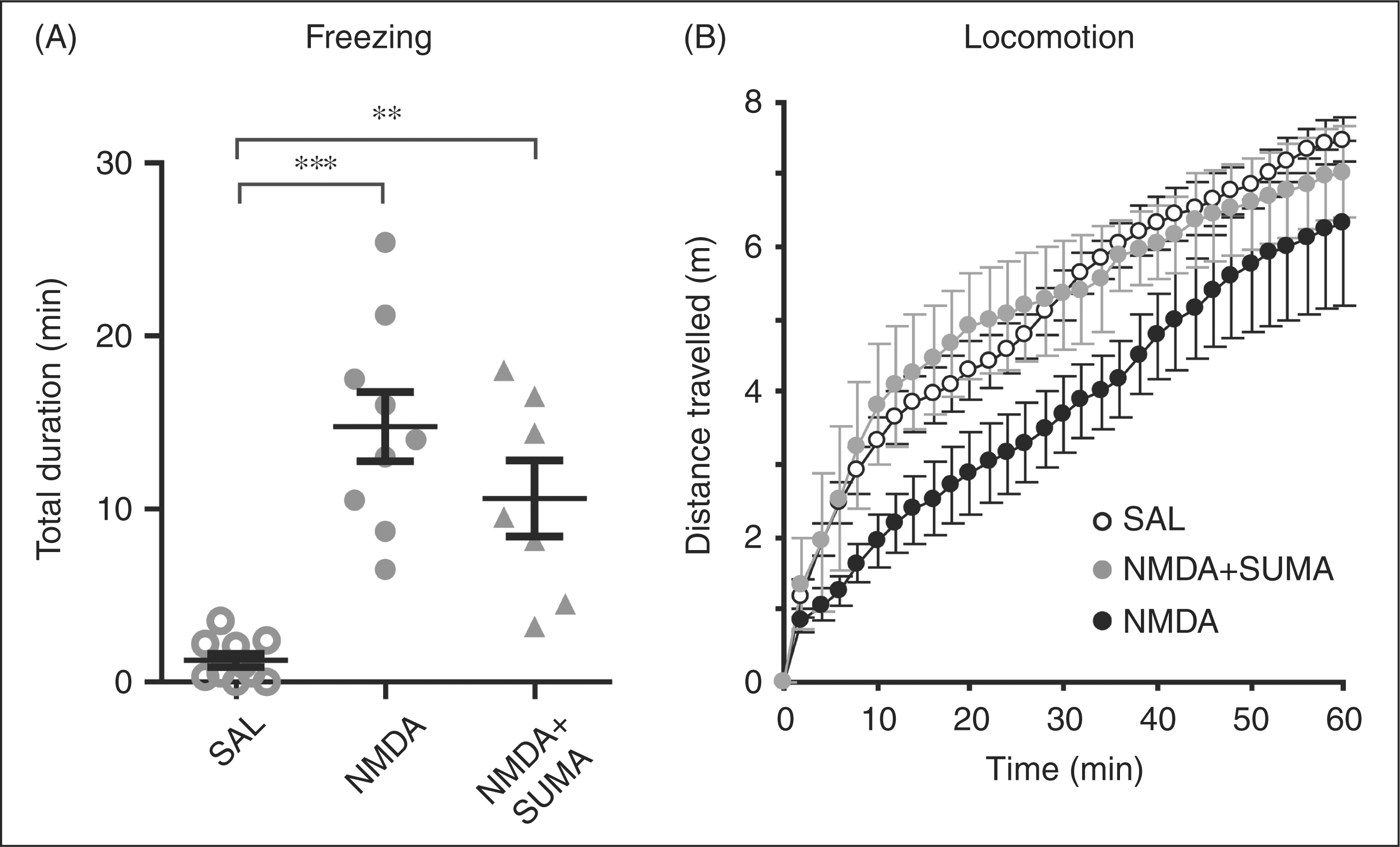
The total duration of body grooming (including head) was slightly increased in both the NMDA and SUMA+NMDA groups. However, the difference did not reach statistical significance (Figure 4A). When head grooming was analysed separately, there was no significant difference between groups and no lateralisation was found.
(A) Total duration of body grooming (including head grooming) is not significantly different among groups. (B) The total number of head shake/wet dog shake (HS/WDS) episodes are significantly increased in the NMDA group. Sumatriptan pretreatment did not significantly reduce the WDS number. SAL, saline (control group). SUMA, sumatriptan pretreated group. Data of each animal are shown in grey symbols, and mean ± SEM shown as dark lines. *P < 0.05.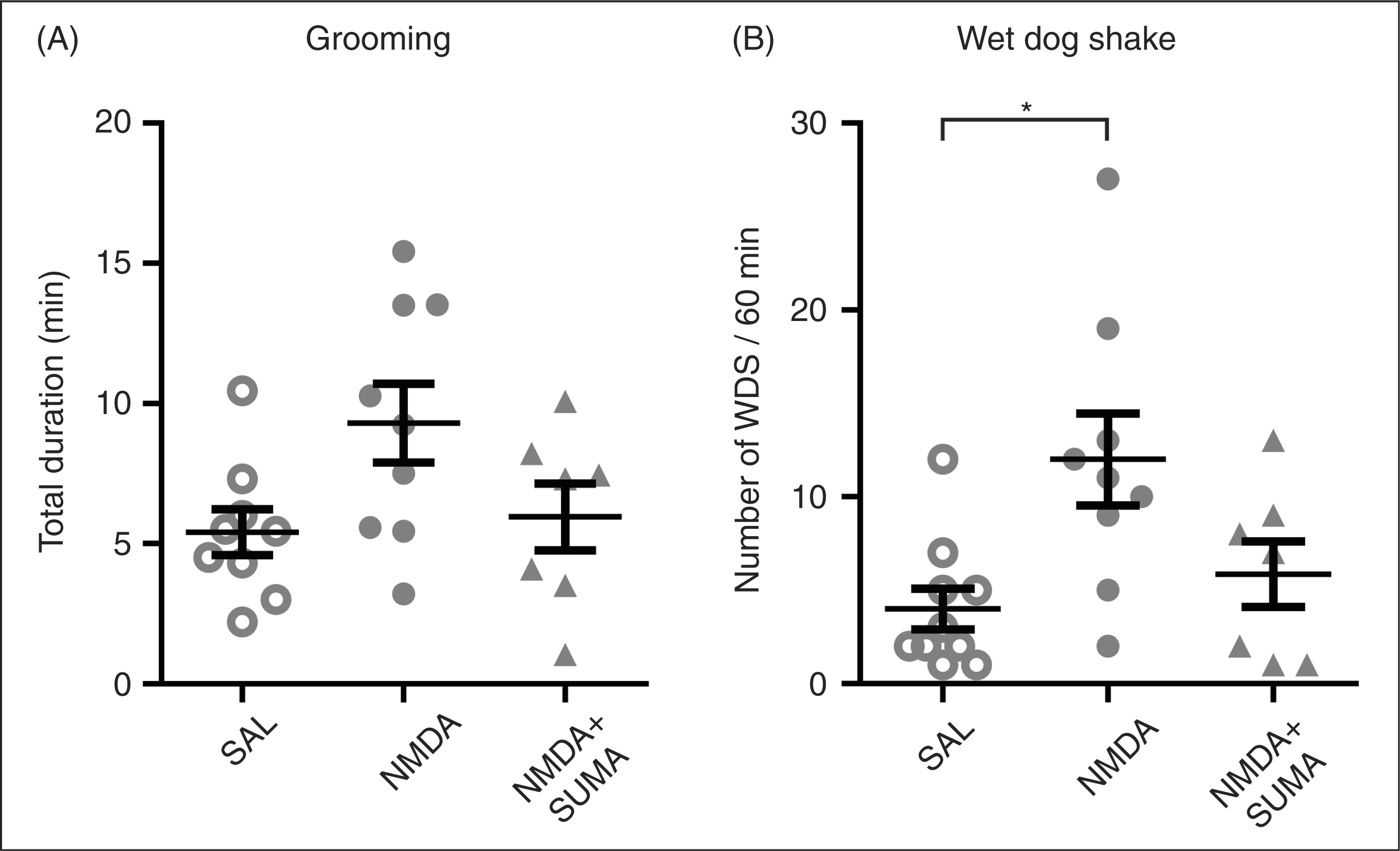
In our study, WDS was always preceded by HS and associated with grooming behaviour. The number of combined WDS–HS episodes within one hour was increased in the NMDA group compared to the control (Figure 4B; P < 0.01). Sumatriptan pretreatment did not significantly reduce the number of WDS/HS responses.
Eating, drinking (Figure 5), clockwise or counter-clockwise rotation and rearing were not significantly different among groups. Although video recordings revealed a tendency to turn left (CCW) in some animals during the first 5–15 min, or difficulty in raising fore paw during rearing or paw preference for getting food, none of those observations reached statistical significance amongst the groups. Behavioural data of each animal are shown in Figures. 3–5 as the variability was high in groups. Additionally, the possible propagation of CSD with variable speed in awake animals, as shown by Koroleva et al. (24), may have influenced behavioural variability in our study.
Total duration of eating and drinking are not significantly different among experimental groups. SAL, saline (control group). SUMA, sumatriptan pretreated group. Data of each animal are shown in grey symbols, and mean ± SEM shown as dark lines.
Ultrasonic vocalisation calls
Distress or pain calls (22–27 kHz) were recorded in 3 out of 25 animals (Table 1), which were emitted when animals were displaying freezing behaviour. None of the control animals exhibit distress calls and only two rats from the SUMA+NMDA group and one rat from the NMDA group emitted distress calls (Figure 6). Pain calls were long-lasting (808 ± 25 ms) and the total duration of pain calls lasted up to a minute (Table 1). The 50 kHz vocalisations reflect a positive affective state of the animal and with a frequency range of 32–96 kHz. The so-called ‘50 kHz vocalizations’ are referred to as ‘chirps’ because of their brief duration (30–50 ms) (25). USV calls reflecting positive affect (55 ± 9 ms duration) were only recorded in control animals during eating (Figure 6) and not detected in the NMDA or SUMA+NMDA groups. The sonotract software also converts USV calls into frequencies audible to humans; listening to these converted USV calls revealed that 50 kHz vocalisations sounded like a whistle/chirp, while pain calls sounded like a scream.
Sonograms of ultrasonic vocalisations (USV calls) emitted by the rats. Frequency shifts at the beginning and at the end of the vocalisations are characteristic of a biological sound. No statistically significant increase in the duration and/or number of pain calls were detected in NMDA, SUMA+NMDA and SAL (control) groups. (A) USV pain call was detected in only one of the rats in NMDA group, and this call coincided with freezing episode. (B) The panel demonstrates the pain/distress call emitted during freezing by a rat in the SUMA+NMDA group. Both USV calls in the 22–27 kHz range are compatible with a negative affect state such as pain or anxiety. (C) Example recordings exhibiting positive mood state obtained from the control animals during eating. Vocalisations with high frequency and brief duration reflect a positive affective state (chirp sound). Y-axis denotes frequency spectrum of the USV calls and the power of the spectrum was shown by using colour coding. Pain- or distress-induced ultrasonic vocalisations (USV) in experimental groups Number of animals emitting (E) or not emitting (NE) USV calls. Onset, the time period between NMDA application and the first USV call. Concurrent behaviour, animal behaviour accompanying the USV call.

c-fos immunochemistry
The c-fos immunoreactivity, a reliable marker of neuronal activity, was used to confirm CSD at the end of each experiment. An intense and wide-spread c-fos expression was detected in all cortical layers and pia-arachnoid membranes in the NMDA and SUMA+NMDA groups (Figure 7). The c-fos expression in the anterior sections (bregma +1 mm) indicated propagation of CSD from posterior (bregma −7 mm) cortical area where CSD was induced. As the animals were awake and freely moving, some patchy c-fos expression was also observed in the contralateral cortex, but the magnitude and the intensity was not comparable to that of ipsilateral cortex. Three animals were not included to the analysis as c-fos expression was not detected in the ipsilateral cortex. In the saline group, c-fos expression characteristic of CSD was not detected in the cerebral cortex. c-fos expression in the amygdala was also analyzed in all groups. The mean number of c-fos positive cells per section was significantly greater in the ipsilateral amygdala when compared to that of contralateral (non-CSD) amygdala in NMDA group (Figure 7; P < 0.01). Sumatriptan treatment did not affect the CSD-induced c-fos expression pattern (Figure 7). The c-fos immunoreactivity in ipsilateral amygdala of the NMDA and SUMA+NMDA groups were significantly greater with respect to that of saline group (P < 0.0001 and P < 0.0001, respectively).
Immunohistochemical coronal sections showing c-fos expression in cerebral cortex and amygdala. (A) The c-fos immunoreactivity of cerebral cortical sections obtained from 8 mm anterior to NMDA application site. In contrast to topical saline application (SAL), topical NMDA application induced wide-spread and intense c-fos expression in the ipsilateral cerebral cortex (NMDA) as a characteristic feature of CSD. Sumatriptan pretreatment did not inhibit NMDA-induced c-fos expression pattern (NMDA+SUMA). Scattered c-fos positive neurons are also seen in the contralateral hemisphere since animals are free to move, rear, groom, eat or drink. (B) NMDA application significantly induced c-fos positive cell expression in ipsilateral amygdala nuclei (basolateral and central nucleus are shown) with respect to that of saline group. Sumatriptan treatment did not alter the CSD induced c-fos activation in amygdala. (C) Bar graphs show mean c-fos positive cell numbers (mean ± SEM) in all amygdala nuclei. CSD induced significant increase in c-fos cell number in ipsilateral (I) amygdala compared to that of contralateral (C) amygdala and saline group. Sumatriptan did not reduce the CSD induced c-fos expression *P < 0.01, ***P < 0.0001.
Discussion
In the present study, we investigated the behavioural correlates of CSD/aura in freely moving rats. To mimic pathophysiological conditions of migraine aura, we developed a single CSD model using NMDA which is specific to the glutamatergic system, instead of non-selective depolarising agents such as potassium. We employed an automated behavioural analysis system to record the behavioural responses and USV vocalisations simultaneously during CSD for the first time. Our data showed that: (i) a single episode of CSD, verified by ECoG suppression and ipsilateral c-fos cortical expression, induced freezing episodes and wet dog shakes; (ii) there was no association between CSD and grooming, eating, drinking, locomotion and ultrasonic vocalisations; and (iii) sumatriptan pretreatment did not change the total freezing duration or the number of wet dog shakes.
Freezing behaviour in rodents is usually related to anxiety reactions, and it is plausible that freezing could reflect a fear or anxiety reaction induced by the involvement of amygdala during CSD (25,26). We observed a significant increase in c-fos expression in ipsilateral amygdala in rats with CSD and sumatriptan did not alter the CSD-induced c-fos activation. In a recent study, it was reported that, in amygdala–hippocampus–cortical slices, >75% of CSD waves invaded the lateral amygdale (27). The early onset of freezing (∼4 min) could support its relation to CSD propagation and ensuing anxiety in our study. Sensorial information related to noxious stimuli enters the amygdala through its lateral nucleus which, in turn, projects to other amygdala areas to control defence reactions (25,26). Recent evidence suggests that the basolateral and central amygdala are involved in the expression of freezing behaviour in the rat (28,29). The latter is also implicated in emission of USV distress calls (29). Significant c-fos expression in both basolateral and central nucleus (Figure 7) further supported the 3 notion that freezing behaviour was probably mediated through amygdala activation in freely moving rats. The activation of amygdala by the invasion of the CSD wave seems a more likely explanation; however, this issue should be confirmed by the simultaneous electrophysiological recordings from both amygdala and cortex. The amygdala is the crucial structure in the affective assessment of sensorial inputs and the association of fear and anxiety responses (30). Scintillations and/or scotomas interfering with visual perception, intense sensory loss or hemiplegia cause fear and anxiety in migraine patients (13). The functional interactions of the amygdala with other brain regions associated with vision, pain, emotion and autonomic centres could be important in the development of migraine symptoms.
An alternative explanation is that the freezing may be a manifestation of basal ganglia involvement. CSD invasion terminates synaptic activity in cortex and this could functionally disconnect cortical glutamatergic projections to the cauda-putamen. CSD has been shown to inhibit dopamine release in the cauda-putamen (31). Additionally, CSD may directly propagate into striatum with a variable rate depending on rat strain, anaesthesia and application route of KCl (32–34). Although, we cannot exclude the contribution of the basal ganglia to freezing behaviour that occurred in rats with CSD, our observations of an unchanged muscular tonus and the temporary interruption of freezing by any sound or visual stimuli were not consistent with cataleptic freezing (35).
Freezing could also indicate pain perception due to the trigeminovascular system activation by CSD. In humans, the throbbing pain during migraine attacks is aggravated by locomotion, so freezing might be a defence mechanism to limit head and body movements of the animal. Sumatriptan has been shown to be selectively effective for cephalalgia in Wistar rats without any systemic analgesic activity (36). Therefore, we administered sumatriptan, a 5-HT1D/B agonist, to inhibit activation of the trigeminovascular system, as described previously (10,18). However, sumatriptan did not appreciably reduce the duration of freezing behaviour in our study, nor significantly increase locomotor activity. Therefore, it is unlikely that freezing behaviour resulted from throbbing head pain aggravated by movement.
In addition, grooming, another stress or pain response (37–39), was not significantly increased in the NMDA group. We also observed increased WDS behaviour, which was typically associated with grooming episodes. Although its relation to CSD is not clear, the WDS has been shown to be increased during physical stress conditions and associated with 5-HT2A receptor activation (40).
In order to evaluate further whether CSD caused head pain, we analysed USV pain calls, reliable indicators of cephalalgia and somatic pain (36,41,42). We assumed that, if CSD-induced changes were perceived as painful, the rats would start emitting USV pain calls around 15 min, the peak time of increased middle meningeal artery flow (10). In the present study, typical pain or distress calls (22–27 kHz) were recorded in only three rats out of 25: one from the NMDA group and the other two from the SUMA+NMDA group. However, USV calls indicating a positive affect were only detected in the control animals. These results imply that the CSD-induced alterations were not painful. Increased feeding and drinking behaviour was reported to be the most prominent finding in previous studies investigating CSD in awake Lister hooded rats (32,43,44). By contrast, our data did not reveal any increase in eating and drinking following CSD. This discrepancy might be explained by the differences in rat strains and experimental methods. Lister hooded rats display locomotor bursts and convulsions to acoustic stimuli, while our rat strain responds with behavioural arrest (45,46). Moreover, in prior studies, several CSDs were induced per day for a period of weeks (32,43,44).
The propagation of the CSD waves through one cerebral hemisphere should cause a temporary sensorial and motor dysfunction. However, we did not observe any behaviour consistent with the hemiparesis during CSD. In rodents, it is challenging to detect sensory motor deficits and it was demonstrated that there was no neurological deficit following CSD in C57 mice by using the 5-point neurological scale (47). It is also possible that the freezing behaviour limited any movements of the animal and this, in turn, may have influenced the detection of paresis or circling behaviour in our study.
We mainly aimed to investigate behaviour, the ultimate outcome of the combination of perceived sensory inputs, evoked senses such as pain and emotions. Behaviour is a very complex process and does not necessarily reflect the activation of lower brain centres such as trigeminal nucleus caudalis. Therefore, the lack of observable pain behaviour in freely moving rats following single CSD does not exclude the possibility of neuronal activation in TNC or existence of mild pain. This statement would be more important where the pain behaviour was not pronounced as in our study. However, it was shown that repetitive CSD was capable of activating ipsilateral c-fos expression in TNC under anaesthesia either by pinprick or KCl (10,18). On the contrary, Ingvardsen et al. (48) found bilateral TNC activation by injecting KCl ipsilaterally and hyperosmolar NaCl (1 M) to the contralateral cortex, which was attributed to spill over of high amount of KCl to the contralateral side (49). Lambert et al. (50) did not find activation/sensitisation of upper cervical neurons by pinprick-induced single or several CSDs in the cat. In another study, Ebersberger et al. (51) did not observe increased neuronal firing rate in the brain stem trigeminal nuclei following multiple CSDs; however, they recorded from deeper lamina (lamina V) in the rostral TNC where the c-fos activation following CSD highly clustered in nociceptive lamina (I and II) of the caudal TNC (10,18). These conflicting results could be interpreted that a single CSD is capable of activating TNC, though its impact can be subtle in the rodent brain.
Is there any relevance to the migraine headache of humans? Migraineurs experience moderate-to-severe pulsatile pain aggravated by any movement that increases intracranial pressure. In addition, accompanying symptoms of decreased appetite, nausea, vomiting, photophobia or phonophobia are common. Triptans are specific drugs that attenuate not only headache but also these associated symptoms. In the present study, sumatriptan did not exert any significant effect on CSD-induced behavioural parameters. Freezing, grooming and WDS behaviour independent of trigeminal activation, and the lack of USV pain calls, suggest that severe head pain does not follow CSD in rats. Moreover, repetitive CSDs were not experienced as an aversive stimulus by rats (15).
Study limitations
The potential limitations of this study include:
We used a species with a lissencephalic cortex, which may limit extrapolation of our model to the gyrencephalic human brain. Species and sex differences that could influence behaviour were not studied. Techniques that may unmask mild or vague pain by stimulating sensory modalities were not employed.
Conclusions
Behavioural studies in experimental animals are important tools in developing treatments for humans. This study focused on species-specific behavioural patterns and vocalisations. Our results suggest that: (i) CSD induces freezing behaviour by invoking fear and anxiety via amygdala activation; (ii) a single episode of CSD is unlikely to lead to severe pain in freely moving rats, though the development of mild or vague pain cannot be excluded; and (iii) the relevance of CSD-induced behaviour in freely moving rats to migraine symptoms in humans needs further evaluation.
Footnotes
Acknowledgements
The study was supported by Gazi and Hacettepe University scientific research grants (01-2003-19, 01-2009-41, 05-01-101-012). The authors are thankful to GSK Turkey for kindly providing sumatriptan succinate and Dr Paul Firth for critically revising the manuscript. Part of the study was presented at the European Headache and Migraine Trust International Congress, London, 2008.
