Abstract
To improve understanding of secondary treatment failure in migraine patients, we evaluated ‘headache return’ as a novel endpoint to assess returning headaches according to their severity, expanding on current standard assessments of overall recurrence or relapse rates, in a six-month observational study of triptan-treated migraineurs. A total of 359 patients (91% female; mean age, 42.5 years) recorded data for 2168 headaches in electronic diaries. Two-thirds of headaches responded to triptan treatment (improved-to-mild or no pain two hours post-dose); 34% of headaches had a pain-free response. By 48 hours post-dose, 19% of all responding headaches returned; 24% of headaches achieving a pain-free response returned, predominantly to mild pain. More severe baseline headache, short duration since diagnosis of migraine, and female gender were associated with increased likelihood of headache return. Treatment satisfaction declined with increasing severity of headache return, demonstrating the value of assessing headache return by severity to fully evaluate its impact.
Introduction
Triptans (5-HT1B/1D receptor agonists) are recommended for treatment of migraine in patients who respond poorly to non-specific analgesics, or whose headaches are moderate -to -severe (1). Triptans provide effective relief of migraine pain for approximately 50%–70% of patients (2,3). Relief is not always sustained, however, and substantial numbers of patients experience secondary treatment failure (return of pain to any level following initially successful treatment). Secondary treatment failure is a frequent source of patient dissatisfaction (4), and often leads to discontinuing or switching treatment (5–7). The returning headache can be as disabling as the initial headache, often necessitating the use of rescue medication. Thus, headache return increases the burden of migraine, treatment costs, and risk of medication overuse (8). Female gender, age over 35 years, and severe baseline headache have been identified as risk factors for secondary treatment failure (9,10).
Migraine secondary treatment failure is commonly measured using ‘recurrence rates’, often defined as worsening of headache pain from absent or mild two hours post-treatment to moderate or severe by 24 hours post-treatment. However, definitions, and estimates of recurrence rates, vary widely: a review of 27 triptan trials found recurrence rates of 7%–57% (11). Furthermore, recurrence rates may underestimate the impact of secondary treatment failure by focusing on return to moderate or severe pain, although even mild returning headaches may be a source of concern to patients and require rescue medication. The International Headache Society (IHS) recommends use of ‘relapse rates’ to encompass return of headache of any severity over a 48-hour period (12). This more inclusive approach is consistent with recognition of the importance of treating mild headaches to improve pain-free-response rates (13–15). The terms ‘relapse’ and ‘recurrence’ are often used interchangeably and are not well distinguished in practice; therefore, reported rates of secondary treatment failure are difficult to interpret and cannot be reliably compared among clinical trials that used different criteria to determine recurrence/relapse rates, diminishing the utility of these measures in clinical practice. It is desirable to establish a clear and comprehensive measure of secondary treatment failure to permit better understanding of this phenomenon.
Observation of migraineurs using triptans to treat their headaches informs this issue. We performed exploratory analyses of data collected in an outpatient clinical setting using electronic diaries (eDiaries). Headaches returning within 48 hours of treatment were assessed according to severity, to quantify and qualify secondary treatment failure in this population. The term ‘headache return’ is proposed to describe the measure used in this study to capture information about both incidence and severity of returning headaches over 48 hours. Headache return was broadly defined as any increase in headache severity following an initial treatment response, but was expressed not as a binary endpoint (presence or absence of headache return), but rather as worsening by 1, 2, or 3 points, to reflect return of headache to mild, moderate, or severe intensity. The goals of our analyses were to explore headache return as a measure of secondary treatment failure, and to evaluate the occurrence and severity of headache return in triptan-treated migraineurs. We also assessed the impact of headache return on treatment satisfaction and explored baseline clinical and demographic factors as potential risk factors for headache return.
Because headache return, by definition, occurs in the context of a treatment response, it is important to specify the nature of the treatment response. For the purposes of simplicity and brevity in these initial analyses of a large data set, we focused primarily on a pain-free responder population for the majority of the analyses reported herein. Furthermore, we included patients with mild pain at baseline in our assessment of treatment response, expanding on the ‘traditional’ definition of treatment response often used in clinical trials (improvement from moderate or severe pain to mild or no pain), to reflect the fact that many patients in this observational study would be expected to take medication when their headache was mild, consistent with the paradigm of early intervention (15).
Methods
Study design, population and procedures
This was a six-month observational study of an outpatient migraineur population using triptans to manage their headaches and recording headache data in eDiaries. Patients were recruited at 19 study centers in the United States in 2004–2005. A sample size of approximately 500 patients was chosen; this sample size was expected to provide the precision for estimating the incidence of migraine relapse and recurrence. Assuming that 300 to 500 patients have a migraine headache that responds, an observed recurrence rate of 0.20 would have a two-sided 95% confidence interval (CI), extending in the range of 0.045 to 0.035 above and below the observed proportion. Furthermore, a sample size of 500 patients (resulting in approximately 400 patients with responding migraines) when divided equally into two groups by a demographic or clinical characteristic would have 80% power to detect an increase in a recurrence rate from 0.20 to 0.33 using a continuity-corrected Chi-square test with a 0.05, two-sided significance level.
Inclusion and exclusion criteria
Male and female outpatients aged 18–65 years who were diagnosed with migraine headache (International Classification of Headache Disorders, 2nd ed., criteria (16)) for ≥1 year, and were prescribed any triptan for the acute treatment of their migraine attacks, were included in this study. Excluded from the study were migraine patients who also experienced frequent non-migrainous headache (≥six attacks per month); patients with atypical migraine attacks that had consistently failed to respond to medical therapy, ophthalmoplegic migraine, or chronic migraine (≥15 migraine days per month); and patients with a history of familial hemiplegic migraine, sporadic hemiplegic migraine, basilar migraine, or migrainous infarction. Eligible patients were issued an eDiary to record headache data.
Study procedures
At screening, investigators took medical and migraine histories, including typical frequency, duration and severity of migraine attacks and associated functional impairment, and obtained patients’ written informed consent. Additional baseline assessments included the Migraine Disability Assessment (MIDAS (17)) to assess headache-related disability and functional consequences; a migraine recurrence history questionnaire, relating to their five previous triptan-treated attacks; and a cutaneous allodynia questionnaire to assess increase in tenderness/pain in response to touch or pressure to different areas of the head. Females were questioned about the relationship of attacks to their menstrual cycle.
The observation period was up to six months or 10 migraine attacks. During the study, patients continued to use their usual triptan migraine medication as prescribed. Patients attended clinic visits at months 1, 2, 4 and 6 (or after 10 attacks), where investigators reviewed eDiaries for compliance with completion.
For each migraine attack, patients recorded the time at which the headache began, rated the severity of headache pain on a 4-point scale (0 = absent; 1 = mild; 2 = moderate; 3 = severe), and recorded the presence or absence of associated symptoms of nausea, photophobia and phonophobia. Pain severity and associated symptoms were recorded again at 1, 2, 4, 12, 24 and 48 hours after triptan treatment. Additional eDiary entries included functional ability, rated on a 4-point scale (0 = requiring bed rest; 1 = able to do nothing/very little; 2 = able to do something; 3 = active as normal) at headache baseline, and treatment satisfaction, rated on a 7-point scale (1 = completely satisfied; 2 = very satisfied; 3 = somewhat satisfied; 4 = neither satisfied nor dissatisfied; 5 = somewhat dissatisfied; 6 = very dissatisfied; 7 = completely dissatisfied) 24 and 48 hours post-treatment.
Rescue medication was permitted for the treatment of non-responding or returning headaches; use was recorded in the eDiary. As no investigational medications were used, no safety parameters were measured and adverse events were not routinely collected but, if noted, were reported in accordance with the Food and Drug Administration Code of Regulations. This study was compliant with the International Conference on Harmonization Good Clinical Practice Guidelines, and the protocol was approved by Institutional Review Boards for the participating study centers.
Efficacy assessments
Efficacy endpoints were derived from headache data recorded in the eDiaries. In order to fully establish headache characteristics among study participants, treatment response and the overall incidence of headache return were assessed for the whole study population.
Response rates were calculated for two overlapping groups of responders. ‘Overall’ response rates used a broad definition that included improvement of headache from mild, moderate or severe pain to no pain, or from moderate or severe pain to mild pain, at two hours post-treatment. ‘Pain-free’ responders were limited to the segment of the population with improvement from mild, moderate or severe pain at baseline to no pain two hours post-treatment.
Headache return was derived from pain evaluations at 4, 12, 24, and 48 hours post-treatment. For each set of responders, the total incidence of headache return was determined, based on a broad definition of any worsening of pain by at least one point (from no pain to mild, moderate or severe pain, or from mild pain to moderate or severe pain). For pain-free responders, returning headaches were further analyzed according to their intensity: a return value of 1, 2, or 3 was assigned based on the worst pain severity score recorded at any evaluation 4–48 hours post-treatment.
Subsequent analyses focused on the pain-free responder population only, for investigation of relationships between intensity of headache return and treatment satisfaction or rescue medication usage. We also investigated relationships between headache return and several clinical and demographic characteristics assessed on entry to the study (age, gender, duration since migraine diagnosis) or recorded at the onset of the headache (headache severity, nausea, photophobia/phonophobia, functional impairment), in the pain-free responder population.
Statistical methods
Exploratory efficacy analyses
Headaches were included in post-hoc efficacy analyses if they were treated with a triptan and had pain severity assessments at baseline, two hours post-treatment, and at least once thereafter (described as a fully qualified headache). Response and return rates were calculated at the attack level (all fully qualified headaches included) or at the patient level (all patients with at least one fully qualified headache included). For patient-level analyses of headache return intensity (pain-free responder population only), each patient’s attack with the most severe returning headache was selected for inclusion in the analysis. Where multiple headaches were equal in severity upon return, selection criteria were the greatest baseline severity and then chronological order of attacks.
To assess the association between headache return and treatment satisfaction in pain-free responders at the patient level, hour 48 satisfaction level (defined as satisfied [score 1–3], neutral [score of 4] or not satisfied [score 5–7]) was tabulated against severity of the selected returning headache. For attack-level assessments, mean hour 48 satisfaction score (0–7) was calculated for each level of headache return.
The effect of baseline clinical and demographic characteristics on headache return following a pain-free response was assessed using a repeated measure ANOVA (Analysis Of Variance) model with a contrast statement (Statistical Analysis System [SAS Institute, Cary, NC, USA]). The response variable was number of headache returns following pain-free response. Gender, age, duration since diagnosis, baseline (pre-treatment) headache severity and functional impairment, and presence/absence at baseline of nausea and photophobia/phonophobia were included in the model as independent variables.
Results
Patients
A total of 411 patients enrolled in this study; 359 patients had at least one fully qualified headache and were included in the efficacy analyses (evaluable patients). A total of 246 patients completed the study by treating 10 headaches or fulfilling the six-month study period. Of the 165 patients that discontinued the study, 69 had not completed the assessment period when the study was terminated by the sponsor, 56 were no longer willing to participate and the other 40 withdrew for other reasons, were lost to follow-up or violated the protocol.
Patient demographics and baseline characteri-stics
Patient demographics and baseline migraine characteristics a (N = 359)

Evaluable patients.
N/A, not applicable.
MIDAS, Migraine Disability Assessment.
Headache frequency, treatment response and headache return
Fully qualified headaches recorded during the observation period—overall headache frequency, response and return statistics
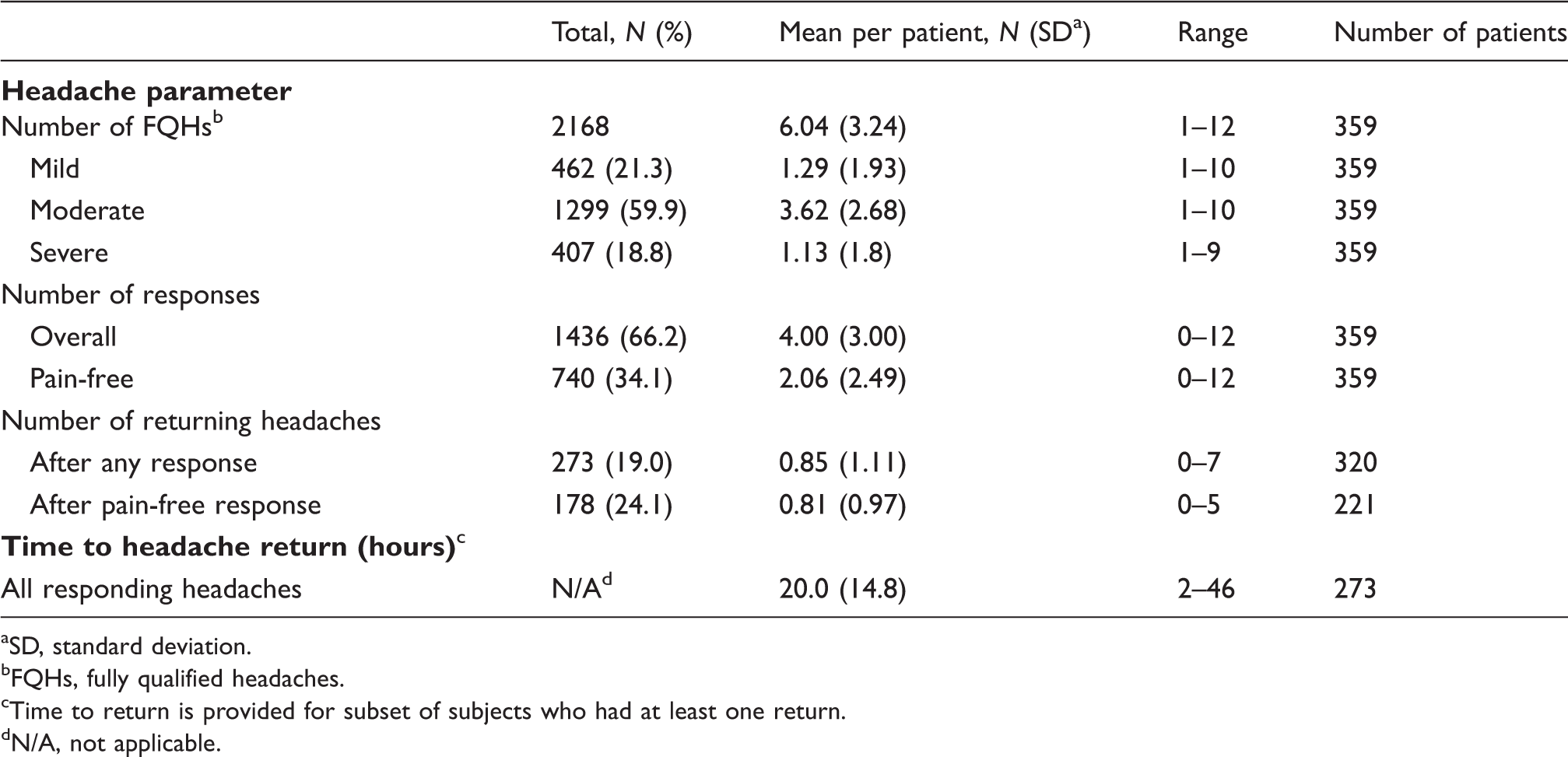
SD, standard deviation.
FQHs, fully qualified headaches.
Time to return is provided for subset of subjects who had at least one return.
N/A, not applicable.
Headache return following pain-free response: frequency and intensity by subject and by attack

Impact of headache return
Association of headache return with treatment satisfaction
The majority of patients achieving a pain-free response (58%) reported that they were satisfied with their treatment (Figure 1A). Satisfaction rates were highest among patients who did not experience headache return, and declined with increasing intensity of headache return (Figure 1B). At the attack level, the mean satisfaction score (on a scale where a higher score represents greater dissatisfaction) increased with increasing intensity of headache return (Figure 1C).
Treatment satisfaction following pain-free response, and association with headache return. (A) Overall distribution of satisfaction/dissatisfaction in all patients with ≥1 pain-free response. (B) Satisfaction rate according to headache return intensity (by-subject analysis: patients recording 1, 2, or 3 on the treatment satisfaction scale at hour 48 are included here as ‘satisfied’ patients). (C) Mean satisfaction scores associated with different levels of headache return intensity (by-attack analysis: all attacks with a pain-free response and an hour 48 satisfaction rating are included).
Rescue medication usage
Patients taking rescue medication to treat returning headache after pain-free response

Excludes patients who did not record whether or not they used rescue medication.
Predictors of headache return
Association of baseline factors with headache return following pain-free response

p values derived from generalized estimating equations model contrast statement.
Discussion
This study monitored treatment responses during an observation period of up to six months, among 359 patients who treated at least one migraine headache with a triptan. Approximately two-thirds of headaches (66%) responded to initial treatment within two hours of triptan administration (34% with a pain-free response). This value is within the range of two-hour response rates for most members of the triptan class (2,3), suggesting that observations in this study were representative of treatment responses typically seen in triptan-treated migraine patients. In addition, the predominantly female (91%), white (91%) population with a mean age around 40 years is typical of migraine clinical trial populations (18–20).
Headache return was associated with approximately one-fifth (19%) of all headaches that responded to treatment, and approximately one-quarter (24%) of headaches that had a pain-free response. At the patient level, more than one-half of patients achieving a pain-free response at some point during the study period were affected by headache return. The majority of returning headaches were mild in intensity.
It is known that response, and particularly rapidity of response, are important determinants of patient satisfaction (21), and that secondary treatment failure contributes to dissatisfaction with treatment (4). We sought to investigate further the impact of secondary treatment failure on treatment satisfaction using the headache return endpoint to assess the influence of intensity of the returning headache. For the purposes of this initial exploratory analysis, we focused on the pain-free responder population. There was a trend for a decline in treatment satisfaction with increasing intensity of headache return. Interpretation of treatment satisfaction data in this study is somewhat confounded by the uneven distribution of data across groups with different magnitudes of headache return. The majority of returning headaches reached mild intensity upon return, and were not associated with a substantial impact on treatment satisfaction. Thus, if all returning headaches were considered together (as is typical in assessment of relapse), any impact of a minority of headaches returning with greater intensity could have been masked. Therefore, it was useful to consider the severity of returning headaches, by derivation of the headache return endpoint, to detect the impact of more severe returning headache.
Nonetheless, it is important to capture mild returning headaches, as per IHS recommendations for the assessment of relapse (12). The similar usage of rescue medication across different levels of headache return in this study suggests that patients are as likely to require rescue medication when a headache returns with mild intensity as when it returns with moderate-to-severe intensity, further supporting the inclusion of mild headaches in assessment of secondary treatment failure. Our definition of ‘headache return’ facilitates assessment of the full extent of secondary treatment failure by capturing headaches that return with mild intensity as well as those of moderate or severe intensity, but differs from standard measures of relapse by distinguishing among returning headaches of differing severities. In this study, information regarding the intensity of returning headaches enhanced the utility of ‘headache return’ as a measure of secondary treatment failure, by increasing sensitivity to an impact on treatment satisfaction.
Clinical factors associated with headache return were largely consistent with factors previously identified as predictors of headache recurrence (9,10). Baseline headache severity was a significant risk factor, and female patients were at significantly greater risk of headache return than males. Time since the diagnosis of migraine was inversely related to risk of headache return. We suggest two possible explanations for this finding: the risk of headache return may decline with duration of illness; alternatively, effective treatment may have been optimized for long-term migraine patients over time. Identification of factors that place patients at increased risk of headache return should help to guide treatment decisions when considering properties such as speed of onset versus secondary treatment failure rate. Our analysis of predictors of return did not include properties of different triptans that could affect headache return, but a previous study found that the rate of headache recurrence following triptan treatment was influenced by the pharmacological and pharmacokinetic properties of the drug (22).
In conclusion, we recommend the routine clinical assessment of headache return to characterize returning headaches. Headache return always occurs within the context of a treatment response, and we have focused here largely on headache return following a pain-free response. However, headache return is also applicable to headaches that respond by improving from moderate or severe to mild pain, and subsequently worsen again. The ‘headache return’ endpoint permits thorough assessment of outcomes that vary with intensity of the returning headache, and thus has potential to improve understanding of the phenomenon of secondary treatment failure and thereby provide physicians with information to further optimize treatment for migraine patients.
Author conflicts of interest
Dr Sheftell has worked with GlaxoSmithKline, Merck, MAP, NuPath, Pfizer and Optinhose in one or more of the following capacities: speaker, consultant, advisory board member.
Dr Lipton is funded by National Institutes of Health grants PO1AG03949, PO1AG027734 RO1AG25119 and is a mentor for K23AG030857, K23NS05140901A1 and K23NS47256. He receives research funding from the National Headache Foundation and the Migraine Research Fund. He serves as an editorial board member for Neurology and Cephalalgia and as senior advisor to Headache. He holds stock options in Neuralieve, Inc., and Minster, Inc., and consults for, conducts studies funded or has received lecture honoraria from Advanced Bionics, Allergan, Inc., Boehringer-Ingelheim, Bristol-Myers Squibb, Endo, GlaxoSmithKline, Johnson & Johnson, Kowa, Minster, Merck, Neuralieve, OrthoMcNeil and Pfizer.
Disclosure information
This study was sponsored by Pfizer, Inc. Drs Sheftell, Lipton and Matthew have received research grants from Pfizer. Drs Weeks and Matthew have received honoraria from Pfizer (not relating to development of this manuscript). Drs Pitman and Almas are employees of Pfizer, and Dr Pitman has stock in Pfizer. Editorial assistance in preparing the manuscript was also funded by Pfizer.
Footnotes
Acknowledgements
Editorial assistance with the preparation of this manuscript was provided by Samantha Stanbury, PhD, a medical writer at FireKite, UK.
