Abstract
The long-term course of migraine with aura (MA) has been poorly explored. The present 11-year follow-up study assessed the long-term natural history and possible prognostic factors of MA with onset in childhood or adolescence. Patients were recruited from the original case records of our department, which are specifically designed to report all headache characteristics, aura symptoms and electroencephalogram (EEG) findings. A total of 77 patients (47 females; 30 males) whose records contained detailed descriptions of both headache and aura symptoms apparently meeting the International Classification of Headache Disorders (ICHD)-II criteria for MA (i.e., 1.2.1, 1.2.2, 1.2.6) underwent structured face-to-face follow-up headache interviews, all of which were conducted by the same neurologist, who has particular expertise in this field. A multivariate model (logistic regression analysis) was used to investigate the association between possible prognostic factors and the remission of both aura and headache at follow-up. The results of our study showed that 23.4% of the MA patients were headache-free at follow-up, 44.1% still had MA and 32.5% had a transformed headache diagnosis (i.e., fulfilling the criteria for ICHD-II 1.1. or 2). Patients with basilar-type migraine (1.2.6) showed the highest headache remission rate (38.5%).
Our study seems to show that migraine with typical aura (1.2.1–1.2.2) is associated with a favourable evolution of aura symptoms over time (remission of aura in 54.1% of patients). Subjects experiencing only visual aura had a lower remission rate compared with those with visual ± sensory ± aphasic aura symptoms (36.8% vs. 61.5%, p = 0.054). A short headache duration (<12 hrs) and the presence of EEG abnormalities at baseline were the only significant predictors of aura remission at follow-up (odds ratio [OR] = 9.12, 95% confidence interval [CI]: 1.79 ± 46.51, and OR = 4.76, 95% CI: 1.18 ± 19.15, respectively). No significant predictors of headache remission were found.
In conclusion, our results suggest that MA shows a favourable course. Further prospective studies with detailed EEG analysis both at baseline and at follow-up are needed in order to confirm the possible prognostic role of EEG abnormalities in MA. That said, it would, in our opinion, be highly premature at present to submit children with MA to EEG examinations for prognostication purposes.
Introduction
Although migraine is a common and disabling condition in children and adolescents, its long-term course has so far been poorly explored in this population. This is particularly true of migraine with aura (MA). Better knowledge of the natural history of headache (i.e., persistence, transformation, remission) and the role of prognostic factors is important in order to identify children at risk of chronic headache and to evaluate the need for and effect of treatments.
The nautral history of migraine in children has been investigated in several studies, which have given considerably varying results. This is mainly due to methodological differences several areas.
Sampling method: clinic-based (1–5) versus population-based (6–9)
Diagnostic criteria: use of nonstandardized definitions (3,6) the International Headache Society’s (HIS)1988 criteria (1,4,7,9,10) or the IHS 2004 criteria (ICHD-II) (5)
Headache type considered: MA (1,4,11), both MA and migraine without aura (MwA) (3,6,7,10), migrainous disorders (MDs) (9), and/or tension-type headache (TTH) (5,12)
Outcome measures: absence of MA attacks for 18 (1) or 24 (2,4) months, migraine-free period of at least 12 (5,9,2) or 24 (6) months, or reduction in frequency, intensity or duration of migraine attacks (3,7,10)
Age at onset: childhood and adolescence (1,3,5–7,10,12), only adolescence (9), or both the paediatric and the adult populations (2,4)
Duration of follow-up: from three to 10 years (1,3,5,7,9,10,12), or more than 10 years (2,4,6)
Bille’s epochal follow-up examinations of children with migraine (6) revealed a considerable variability in the long-term course of the disease, with pain-free years and subsequent migraine relapse occurring in about 20% of cases. This author also showed a considerable recall bias with regard to MA (i.e., in middle age, 41% of the subjects could not remember that they had had aura symptoms at younger ages), highlighting a possible need for prospective follow-up studies.
Prospective studies have shown that migraine headaches remit in 17% to 34% of subjects, persist in 20% to 48%, and transform into other types of headache in 11% to 37% (12,13). Studies providing detailed headache diagnoses suggest that up to one-quarter of patients may evolve from migraine to TTH or vice versa (7,12,13).
Among the prognostic factors, an early age at onset (3), psychosocial stressors (7) and psychiatric comorbidity (14) may be related to an unfavourable outcome, whereas the importance of gender remains unclear (7,14).
Studies regarding the natural history of MA

The aim of this follow-up study is to describe the long-term natural history of MA with onset in childhood and adolescence, and to identify possible prognostic factors by using a multivariate statistical method.
Materials and methods
Study population and design
The clinical records of 137 patients discharged from our department between 1983 and 1999 with a diagnosis of MA were analysed (Figure 1). The case records were specifically designed to report headache and aura symptoms, and other demographic, clinical and EEG characteristics (Table 2).
Flow diagram of the study. Clinical and demographic data collected at baseline. (In brackets the dichotomisation used in the logistic regression model to study their possible prognostic role)

During hospitalization, all the patients were submitted to an awake EEG performed in accordance with the International 10-20 System. No patient underwent an EEG during a headache attack or within 24 hours of the last one. Epileptiform abnormalities (i.e., spikes, sharp waves, spike-wave complexes or polyspike waves) and generalized or focal slowing were considered EEG abnormalities (15).
We selected all the patient records containing detailed descriptions of both headache and aura symptoms apparently meeting the ICHD-II criteria for a typical aura with migraine headache (1.2.1), typical aura with non-migraine headache (1.2.2) or basilar-type migraine (1.2.6) diagnosis (16).
Thirty-three patients did not meet the ICHD-II diagnostic criteria; thus the selected sample was made up of 104 subjects.
All the selected patients were contacted by phone and invited to attend our department for a clinical evaluation. Eight refused to take part in the survey, and 19 could not be traced (either because they failed to answer phone calls or because their current address and phone number were not available). This left 77 patients for the current analysis.
To describe the natural history of MA, all the patients were evaluated at follow-up by the same neurologist, who has specific expertise in the field of MA. A detailed, structured, face-to-face headache interview was used to collect headache and aura characteristicsand to make an ICHD-II diagnosis at follow-up.
On the basis of the evolution of their headache attacks over a mean of 11 years, the subjects were clustered into the following groups: persistent, transformed and headache-free. Persistent headaches were defined as those that still presented an MA diagnosis (i.e., typical aura with migraine headache [1.2.1]), typical aura with non-migraine headache (1.2.2), basilar type migraine (1.2.6) or probable migraine with aura (1.6.2) at follow-up. Transformed headaches were those that had changed to MwA (ICHD-II code: 1.1) or TTH (ICHD-II code: 2). Subjects who had been entirely free of headache attacks for at least 12 months prior to the follow-up evaluation were considered headache-free. All the clinical and demographic characteristics collected at baseline (Table 2) were studied as possible prognostic factors.
Written informed consent was provided by all the participants or their relatives. The study was approved by the local ethics committee.
Statistical analysis
Continuous data were analysed using the two-tailed Student’s t-test and categorical data using the Chi-square test (Fisher’s exact test was applied when at least one expected frequency was less than five).
Logistic regression analyses were implemented to investigate prognostic factors. For this purpose, subjects were dichotomized according to the presence versus absence of aura at follow-up. In a second model, subjects were dichotomized according to the presence versus absence of headache (any form) at follow-up.
The study of prognostic factors for long-term persistence of aura was limited to subjects with typical aura at baseline (i.e., 1.2.1 and 1.2.2). Those with basilar-type migraine (i.e., 1.2.6) were excluded because it is still unclear whether 1.2.6 is a subtype of migraine with typical aura or a distinct phenotype or genotype. Basilar-type migraine is currently included as a distinct diagnostic entity in ICHD-II.
The results are presented as ORs with 95% CIs. Univariate ORs were calculated for all dichotomized baseline characteristics (Table 2) considered possible prognostic factors on the basis of their significance in previous studies.
The variables that differed significantly in univariate analysis were included in the multivariate model. The variable “follow-up duration” (
Results
Headache diagnoses at follow up according to MA diagnoses at baseline

Data are expressed as number of patients and (%).
Natural history of MA
Natural history of MA

Data are expressed as number of patients and (%).
ICHD-II 1.2.1 + 1.2.2 + 1.2.6 + 1.6.2.
ICHD-II 1.1 + 2.
Exploring the natural history of the headache phase, we found that 50% of patients with longer attacks at baseline (>12 hours) showed a significant reduction of headache duration at follow-up (p = 0.015). No significant differences were found between baseline and follow-up data (not shown) for headache frequency and intensity.
Evolution of typical aura characteristics

Data are expressed as number of patients and (%).
The sample is limited to patients with ICHD-II 1.2.1 and 1.2.2 diagnoses at baseline.
Remission vs persistence in the natural history of aura
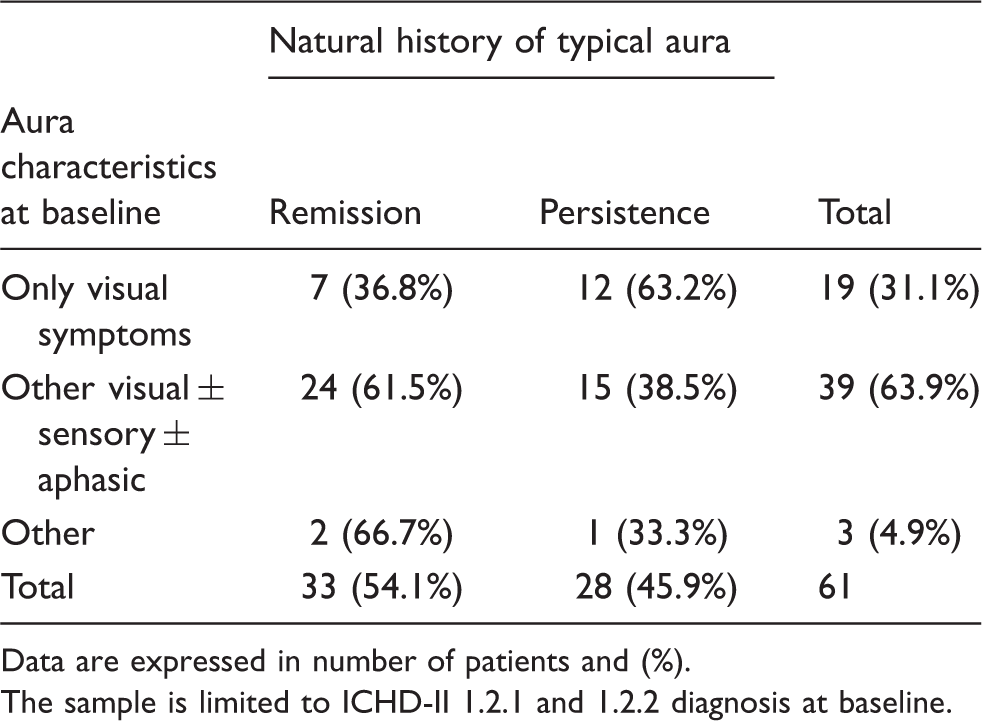
Data are expressed in number of patients and (%).
The sample is limited to ICHD-II 1.2.1 and 1.2.2 diagnosis at baseline.
Prognostic factors
Prognostic factors for aura persistence at follow up

Data are expressed as number of patients and (%).
Control variable in the multivariate model (see text for details).
The sample is limited to patients with ICHD-II 1.2.1 and 1.2.2 diagnoses at baseline.
The second multivariate model, used to study possible prognostic factors for the persistence of any form of headache at follow-up, did not identify any significant predictors (data not shown).
Discussion
Natural history
In our sample the natural history of MA (1.2.1, 1.2.2, 1.2.6) was found to be characterized by an overall remission rate of about 25%, similar to that observed in other headache forms by different authors in studies in which the percentage of subjects who became headache-free varied considerably, ranging between 5% and 45% (7,12,13). Considering in detail the different diagnoses at baseline, the patients with basilar-type migraine (1.2.6) showed the highest remission rate (38.5%) and the lowest percentage of persistence of any aura symptoms at follow-up (23% in 1.2.6 patients vs. 48.4% in those diagnosed as 1.2.1 and 1.2.2). It remains to be established whether basilar-type migraine is a subtype of migraine with typical aura or a distinct phenotype or genotype. In a recent study, the clinical, epidemiological and genetic features of basilar-type migraine were analysed in a large, population-based sample of familial migraine (17) The authors demonstrated that basilar-type migraine is the combination of migraine with typical aura attacks and basilar-type symptoms, which suggests that basilar-type migraine should no longer be regarded as a separate diagnostic entity, but rather as the description of a subtype of migraine attacks that would be more appropriately included in the Appendix of ICHD-II.
In line with the hypothesis advanced by Kirchmann et al., (17) the results of our study suggest that, among MA patients, those experiencing basilar-type attacks at onset show the best prognosis. To our knowledge, our study is the only published report providing specific data about the long-term natural history of basilar-type migraine with onset in childhood and adolescence. In fact, the study conducted by Sturzenegger and Meienberg (18) on 82 cases of “basilar artery migraine” was not really a follow-up study. The authors retrospectively interviewed their patients to evaluate clearly remembered symptoms with the purpose of diagnosing previous ‘basilar artery migraine’, however, no data on the natural history of the disease were given.
In subjects with typical aura with onset in childhood and adolescence (i.e., 1.2.1 and 1.2.2 at baseline), our study seems to show a favourable evolution of aura symptoms over the mean 11-year follow-up (remission of aura in 54.1% of patients).
A favourable evolution of MA was documented by all the previously published reports (1, 2, 4). As shown in Table 1, our results are similar to those obtained by Balottin et al., (1) who examined a cohort of 45 MA subjects aged 7 to 17 years (16 males; 29 females) over a seven-year follow-up period. They reported that 62% of patients, at follow-up, had been attack-free for at least 18 months. Conversely, Cologno et al. (2) and Eriksen et al. (4) reported a lower MA remission rate (about 30%). Cologno et al. conducted a 10–20-year follow-up of 81 subjects aged 11 to 63 years (26 males; 55 females) with an IHS 1988 diagnosis of MA. At follow-up, 35% of this sample had been attack-free for at least a year.
Eriksen et al., in a 16-year study, found at follow-up that approximately one-third of their sample had been free of MA attacks for more than 24 months. The above differences in remission rates may be related to different ages at onset of aura symptoms (i.e., childhood and adolescence in the sample of Balottin et al. and in the present study vs. young adulthood in the samples described by Cologno and Eriksen). Thus, patients with a younger age at onset may show a more favourable prognosis of MA.
As regards the natural history of the headache phase of MA attacks, our data did not confirm the improvement in headache frequency and intensity found by other authors (2,4); however, they showed a significant reduction in the duration of the headache phase over time.
Prognostic factors
None of the several possible prognostic factors of MA resolution described in previous studies (Table 1) appeared to be correlated with a favourable outcome in our sample following a multivariate analysis of the data, controlling for possible confounding biases (e.g., duration of follow-up). In particular, in line with reports by other authors (2,4), and in contrast with the findings of Balottin et al., (1) visual aura alone did not emerge as a significant favourable predictive factor in the prognosis of MA. Moreover, our results clearly confirmed the non-significance of the favourable predictive factors found by Cologno et al. and Ericksen et al. (2,4)
Our results showed that a long headache duration at baseline (>12 hours) significantly predicted the persistence of MA at follow-up (OR = 26.57) and was never associated with headache remission.
On the other hand, in our sample the presence of EEG abnormalities at baseline was found to be a predictor of a good outcome of MA (OR = 8.76).
The EEG findings in our sample of MA patients may contribute to the ongoing debate about the relationship between migraine and epilepsy (19). EEG was one of the first techniques used to discern physiological differences between migraine and controls. It is suggested, in a review of the literature, that EEG is not a valuable diagnostic tool in primary headache disorders (20). Nevertheless, the technique has provided useful information for use in the research setting. Migraine and epilepsy are considered quite different clinical entities and, in each case, diagnosis is typically based on well-defined and widely accepted international classification criteria (21,22). Conversely, it is well known that up to 60% of subjects with infantile idiopathic epilepsy (i.e., epilepsy with rolandic or occipital paroxysms, absence epilepsy) show concomitant migraine (23,24), that 4% to 8% of people with migraine suffer from epileptic seizures (25,26), and that seizures and specific interictal EEG abnormalities are significantly more prevalent in patients with MA as opposed to those with other headache types (27). Although clinical, epidemiological and therapeutic similarities may be coincidental and are no proof of a common aetiological background, a genetically determined dysfunction of ion channels seems to point to a common underlying mechanism for both these paroxysmal disorders. (28) In our study, the finding of EEG abnormalities in at least a subgroup of MA patients supports, albeit indirectly, the hypothesis of altered neuronal excitability. It has been suggested that the enigmatic epileptic-epileptoid EEG characteristics found in migrainueurs could be an expression of electrically hyperactive ‘quasi epileptic foci’ located mainly within the brainstem and generated by insufficient opioid inhibition of peptidergic neurons (29). Moreover, our data showed that the presence of EEG abnormalities at baseline was a predictor of a good outcome for MA. This allowed us to hypothesize the existence, in MA, of an age-dependent subgroup of patients (i.e., those with EEG abnormalities at onset) who show the best long-term prognosis; moreover, something similar has been observed in several well-known age-dependent epileptic syndromes (e.g., absences) (30). However, the design of our study did not allow us to verify this hypothesis: the recruited patients were not submitted to EEG recording at the end of the follow-up interview. Had they undergone this assessment, it might have proved possible to establish a positive prognostic value of the presence of EEG abnormalities at onset (i.e., if remitting patients had been found, at the end of follow-up, no longer to present these abnormalities).
We believe that the strengths of our research are the application of ICHD-II criteria in all the phases of the study and the use of multivariate methods (controlled for follow-up duration) to identify prognostic factors. Our study has some limitations: the baseline is retrospective and the sample of MA patients, being drawn from a clinical population, possibly presents a referral bias. Furthermore, we could not control for psychiatric comorbidity, which might play a role in headache persistence. In addition, EEG examinations were not reported by the same neurologist, both specific and non-specific EEG anomalies were considered, EEG examination was not performed at follow-up, and no data about past or current history of epilepsy were collected. Thus, further prospective studies with detailed EEG analysis both at baseline and at follow-up are needed in order to confirm the possible prognostic role of EEG abnormalities in MA. That said, it would, in our opinion, be highly premature at present to submit children with MA to EEG examinations for prognostication purposes.
