Abstract
Healthcare services rely on large amounts of disposable medical devices, even though similar reusable devices exist. This retrospective audit of operating theatre and endoscopy procurement data across two hospitals aimed to examine the number and mass of simple single-use devices and cost (purchasing and disposal) of such devices in operating theatres where a reusable device alternative exists. This was assessed at one health service across two hospitals with a total of 19 operating theatres and five endoscopy procedure rooms. The main outcome was the number of single-use devices procured and disposed of, mass of devices procured, cost of procurement, cost of disposal, and estimated environmental impact of device procurement and disposal. In a five-year period of operating at two hospitals, 2,683,412 simple single-use devices that have a reusable alternative were purchased and disposed of, with a combined mass of 748 t, an approximate purchasing cost of A$16.1m, and an approximate disposal cost of around A$1.2m. The associated environmental impact of these devices was estimated at 3690 tonnes carbon dioxide equivalent. Simple disposable medical devices in operating theatres such as bowls, jugs, drapes, gowns, scissors, towels and anaesthesia equipment produce tonnes of waste per annum, cost millions to procure and dispose of, and produce greenhouse gas emissions from production to disposal. Replacing some single-use devices with reusable alternatives presents an opportunity for health services to save money from purchasing and disposing of single-use devices, as well as reducing their waste and greenhouse gas footprint. Replacing disposable equipment with reusable equipment can divert tonnes of waste from landfill and save on procurement and disposal costs.
Introduction
Healthcare is a major contributor of greenhouse gas (GHG) emissions in Australia at around 7% of national emissions, and also produces a large amount of non-recyclable waste that must be disposed of in landfill or higher-emissions waste streams. 1 In the 2022 to 2023 financial year, it is estimated that around 42,000 t of solid waste was produced by Victorian public health services, of which only 20% was recycled. 2 Disposal costs for the 35,000 t produced in the 2017 to 2018 financial year were close to A$17m. 2 Approximately 15% of healthcare waste such as clinical and sharps waste is deemed hazardous and must be disposed of by higher-cost and higher-intensity waste streams. 3 One reason for large amounts of waste is the reliance on single-use medical devices and personal protective equipment such as sterile and non-sterile gowns. Hospital operating theatres are highly resource-intensive and utilise large volumes of medical devices, producing around 25% of hospital waste by mass.4,5 A single invasive neurointerventional procedure can produce over 13 kg of waste, up to 70% of which might require disposal through hazardous waste streams such as clinical waste owing to contamination from the procedure. 6 A hip arthroscopy procedure can produce an average of 9.4 kg of waste which does not include the reusable gowns and towels utilised for the procedure. 7 In the UK’s National Health Service, medical devices might contribute to around 10% of national healthcare emissions and 15% of hospital GHG emissions.2,6
Many medical devices are used in operating theatres and examples include personal protective equipment such as gowns, drapes and gloves, surgical instruments such as scissors, ‘hollow-ware’ such as bowls and jugs, absorbent towels and packs, syringes and needles. Some devices, such as syringes and scalpels, are single-use (disposable) and some devices are only reusable (such as specialised surgical instruments), while some items come in single-use or reusable options. Single-use devices produce more waste than reusable devices, might cost more and have higher GHG emissions.8–15 Utilising reusable invasive medical devices can reduce GHG emissions by up to 47% compared with the emissions of an equivalent single-use device. 16 Using reusable non-invasive medical devices such as bowls and dishes can reduce emissions by 63% compared with single-use alternatives. 16 Choosing a reusable medical device in place of a single-use device also reduces healthcare waste by avoiding landfill or hazardous waste disposal, both of which come with financial costs.16–18 Despite the financial and environmental benefits of reusable medical devices, simple single-use devices are routinely used across Australian operating theatres. Products such as bowls, jugs, trays, surgical drapes, sterile gowns and many anaesthesia devices are available as single-use devices or reusable devices and are considered for this study as ‘simple’ because they are not cannulated (more challenging to clean) and do not contain complex technology (such as endoscopes, for example). Single-use devices that could be prioritised for replacement with reusable alternatives are those that: (i) are used in the largest amounts, (ii) are the heaviest (because greater mass is associated with greater GHG emissions), (iii) have the highest financial cost, (iv) have a material composition with high environmental impact, or (v) have safety concerns (so therefore their replacement should be prioritised). 18
Australia’s share of renewably sourced electricity (35%) is increasing steadily, and now approximates the world’s average of 38% (2021 data). 19 Thus, increasingly in Australia, renewables make reusables better because the emissions produced when reprocessing reusable medical devices (decontamination, disinfection and sometimes sterilisation) are less when energy is sourced from non-fossil fuel sources. 20 Yet, it is unlikely (though unknown) that many Australian healthcare centres are actually reducing their use of single-use devices. One priority area that can reduce operating theatres’ environmental impacts is to reduce the amount of disposable devices, so reducing the volume of waste produced by operating theatres.21,22 Little is known about the devices used in operating theatres in Australia. In this article we refer to devices used in operating theatre contexts and in doing so have excluded any prefixes to describe these (e.g. ‘scissors’ rather than ‘surgical scissors’). The primary aim of this study was to describe the disposable anaesthetic and surgical devices used in operating theatres in a large Melbourne health service that could be replaced with reusable equipment alternatives, and to describe the physical waste that could be avoided. A secondary aim was to estimate the financial impacts of reliance on single-use medical devices in operating theatres.
Methods
This study occurred in a university-affiliated Melbourne health service with 19 operating theatres and five endoscopy procedure rooms across two campuses, where 28,121 surgical encounters were performed in the 2023 to 2024 financial year. The procurement data for both locations was acquired over a five-year period from July 2019 to June 2024 from three separate sources—sterile stores A, sterile stores B and from pre-packaged sterile surgical packs supplied by a third party.
First, an inventory of disposable devices that have a known and available reusable alternative was created using data retrieved from the operating theatres’ procurement data. A disposable device was defined as a device that is used once (or not at all) and discarded into waste without being reused or reprocessed, either on-site or by a third party. A reusable device can be reused by standard disinfection and sterilisation pathways in Australia and for which the manufacturer states is a reusable device. As such, complex equipment such as laparoscopic devices, endoscopes and bronchoscopes and cannulated equipment were excluded. Devices were considered sufficiently ‘simple’ if they were a container such as a jug or bowl, were a drape or gown, or were contained within a single-use surgical pack and therefore deemed non-specialised, such that they would routinely be disposed of after a single use. Devices to be included in the disposable devices list are listed in Table 1 along with exclusion criteria.
Inclusion and exclusion criteria for selecting disposable devices to be included in the equipment inventory and the associated anaesthesia and surgical devices.
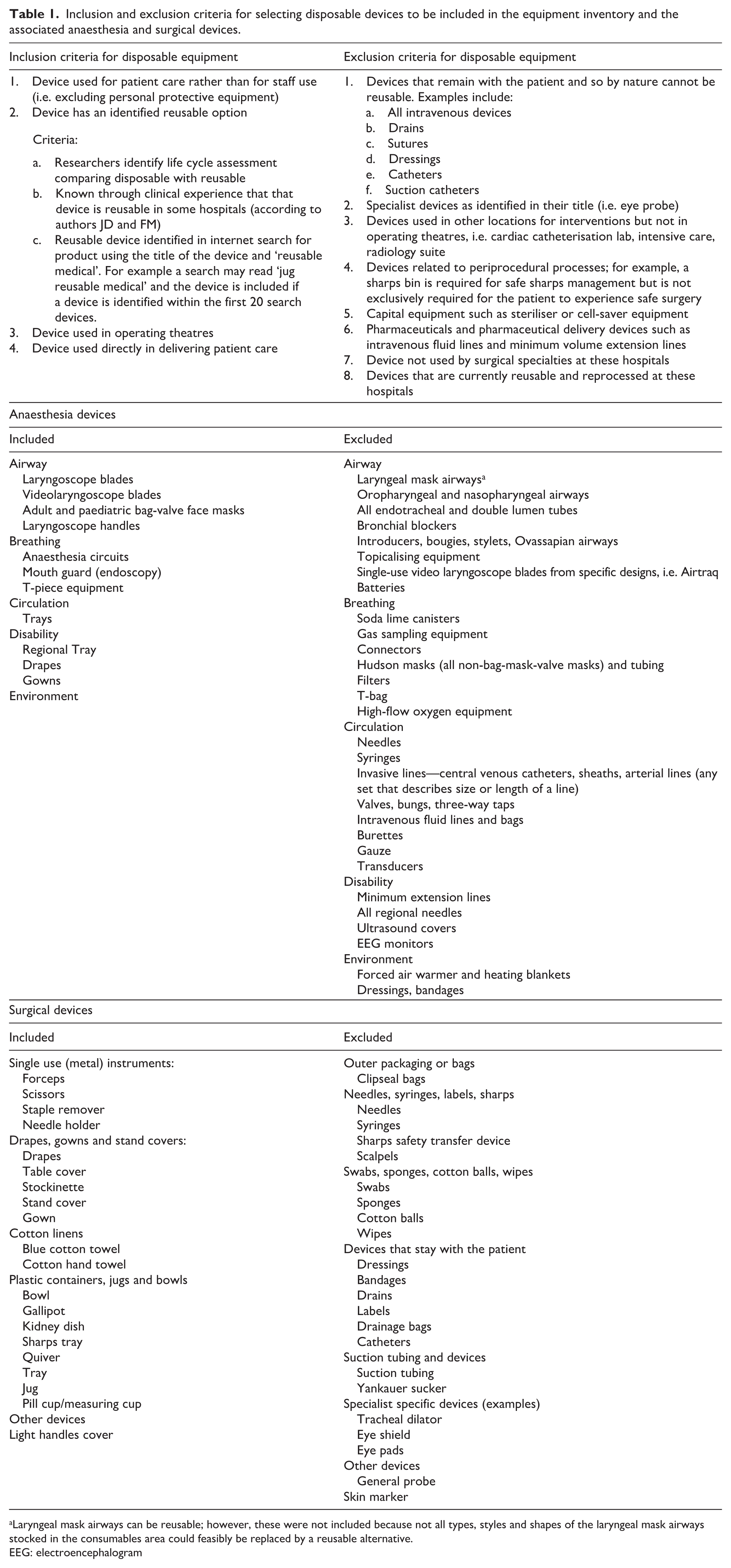
Laryngeal mask airways can be reusable; however, these were not included because not all types, styles and shapes of the laryngeal mask airways stocked in the consumables area could feasibly be replaced by a reusable alternative.
EEG: electroencephalogram
Process of identifying potentially reusable devices
Consumable medical devices used in operating theatres were derived from three sources; first, the pre-packed surgical packs that contain drapes, gowns, bowls and some single-use metal instruments that were fully disposable and came pre-packaged by an external company. This external company provided contents of each pack and numbers of packs purchased by both hospital A’s and B’s operating theatres combined for each financial year from July 2019 through to June 2024. The contents of some packs changed over the period, and these changes were accounted for. Each device within each pack was then assessed according to the inclusion and exclusion criteria, and the total number of devices purchased during the period was summed and then summarised according to type of single-use product: drape, gown, plastic containers, other sterile plastics (quivers, light handles), cotton towels, metal instruments, and trays. Of note, the majority of metal surgical instruments are reusable—only metal disposable scissors and forceps were included.
The second and third sources were from the sterile stock area of hospital A and hospital B, which contained 1934 freely packaged sterile and non-sterile devices that can be selected by the user and ‘scanned’ out of the area according to a cost-centre for the operating theatre of that hospital. Only single-use devices are stored in these areas and only devices used in operating theatres are stored. The area ‘operating theatres’ includes the anaesthetic bays, operating rooms and post-anaesthesia recovery unit. Data for these cost-centres were obtained for each financial year from July 2019 through to June 2024 for both locations A and B. Each device within the database was assessed according to the inclusion and exclusion criteria, then grouped into type of product, with additional categories for anaesthesia equipment and denture cups and lids (because of the number of these two devices). Additional devices were identified through line analysis of each product. Several devices were removed because although their description matched the inclusion criteria it became apparent that the device could not be replaced with a reusable alternative when the device had been identified and weighed. Examples thus excluded were an iodine impregnated sticky drape (no reusable drape technology available) and products described as a face mask but were instead surgical face padding for patient positioning.
Each individual included device from sterile store A and sterile store B was weighed (OXO scales, SKU 48296, New York, USA). Each device from each pack was weighed except for the drapes, because not all drapes could be directly weighed without wasting many sterile packs. Instead, each drape that came separately packaged and stored within the sterile stores areas was weighed and that weight was applied to the same drape contained within a surgical pack. Where a drape could not be directly weighed, the surface area of the drape was estimated using the drape description, and a weight was estimated based on the gram per square centimetre that had been calculated from the drapes that had been directly weighed. Finally, for drapes that existed only in pre-packaged surgical packs and that could not be directly weighed without opening a fresh pack, the weight of a drape of the closest surface area was applied to that drape. Each device was weighed three times to ensure accuracy, and the same scales were utilised throughout.
The purchase cost of each individual device was available for sterile supply area devices only, calculated as a total cost using the individual cost price in 2024. The purchase costs of devices contained within packs (drapes, gowns and plastic containers) were approximated by taking the individual purchase cost of the device from sterile stores and applying that cost to the device in the pack. For drapes that existed only in the packs, the mean cost of drapes in the sterile stores area was used to calculate an approximate cost for all drapes in the surgical packs.
All devices, given that they were disposable, were assumed to be disposed of after use. In Australia, if a medical device marketed as single use is reprocessed for reuse by a hospital, the hospital is deemed the manufacturer and is potentially liable for device failure. At this hospital no single-use devices were reused, and were disposed of as general waste (landfill), clinical waste or sharps waste. The cost of disposal per kilogram or tonne was obtained from the hospital waste services for each waste stream in 2024. An estimate of the cost of disposal for each product category was determined by assuming that 57% of waste is disposed of via clinical waste streams, based on a study of waste disposal after neurointerventional procedures at the same institution that found clinical waste was 57–70% of all waste streams. 6
An estimate of GHG emissions associated with each product group was made using generic carbon dioxide equivalent (CO2e) emissions for each material type. The number of surgical encounters per annum at both sites was utilised to check the reliability of the calculated numbers of devices.
The health service’s Health Human Research Ethics Committee waived ethics approval for this study as it did not involve research on humans and was observational.
Results
In a five-year period of operating at two hospitals with 19 operating theatres and five endoscopy procedure rooms, 2,683,412 simple single-use devices that have a reusable alternative were purchased and disposed of, with a combined mass of 748 t. These had an approximate financial purchasing cost of A$16.1m and a disposal cost of around A$1.2m. The associated environmental impact of these devices was estimated at 3690 t CO2e. Table 2 shows the largest product by mass was surgical drapes, 97% of which were located within pre-packaged surgical packs and were 25% of the total number of devices purchased. The largest number of devices by individually purchased devices was the plastic containers category, comprising sterile bowls, jugs, trays and dishes, and when combined with the other sterile plastic devices, such as quivers and light handles, this totalled 968,492 devices. A total of 46,913 stainless steel disposable metal instruments were purchased (1.7% of total). Fifteen percent of all devices were cotton ‘huck’ towels, which also had higher emissions per kilogram due to the impact of cotton. Anaesthesia equipment was 8% of the total number of devices, assuming no anaesthesia use of non-anaesthesia devices such as towels or gowns, which underestimates the impact of anaesthesia. One hospital used reusable laryngoscope blades and made a small number of disposable purchases during the financial years July 2020 through to June 2022, presumably because of the perceived impacts of COVID-19.
Summary of the disposable devices examined between July 2019 and June 2024 by one health service. Financial data are rounded to the closest thousand.
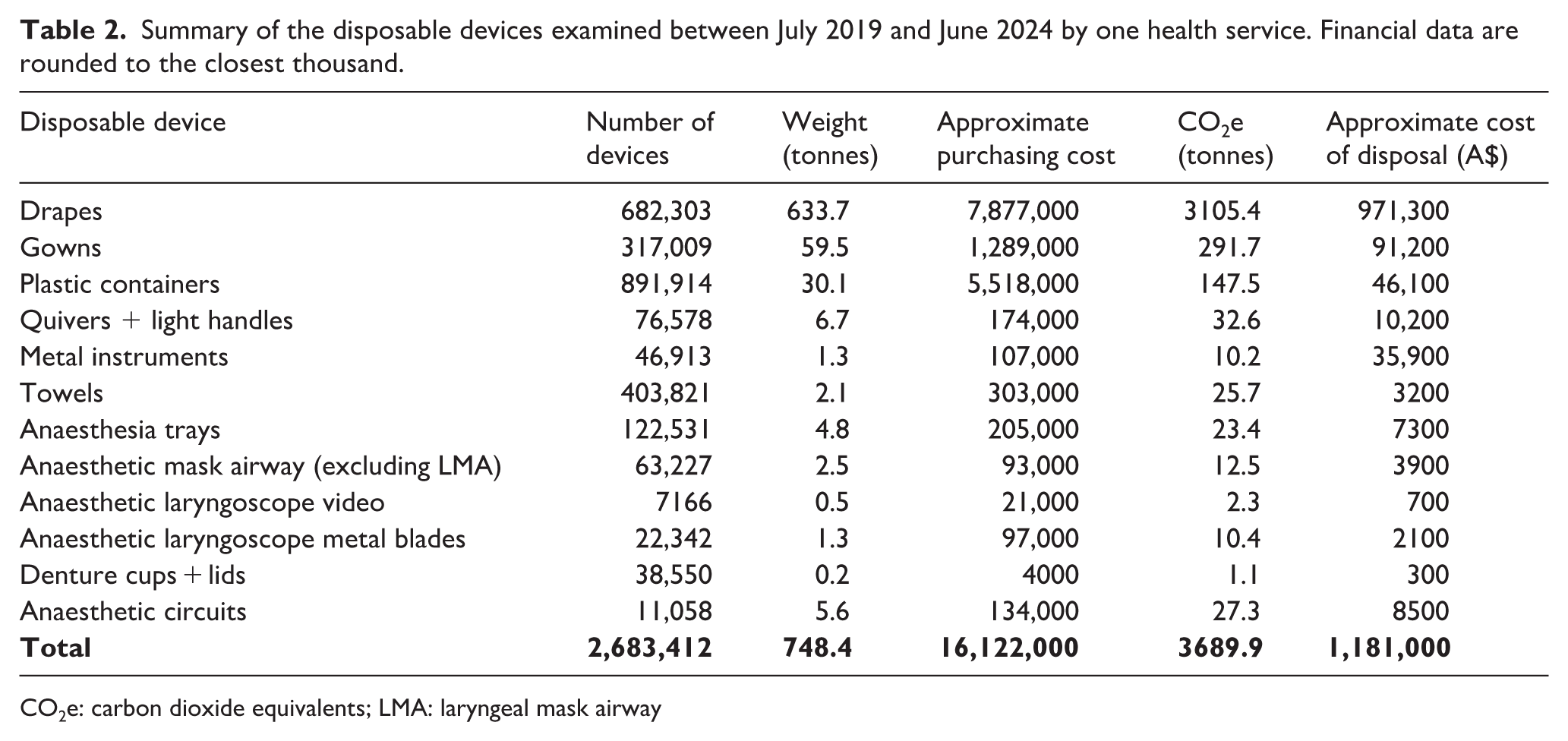
CO2e: carbon dioxide equivalents; LMA: laryngeal mask airway
These data come from 167,867 surgical packs that were purchased over the five-year period and from two sterile supply areas with a total of 1934 single-use devices, of which 102 met the inclusion criteria but 17 were removed upon further inspection because they were not easily replaced by a reusable alternative, as previously noted. A further 23 laryngeal mask airways of various sizes and types could have been considered; however, these were excluded because of functional and safety differences between products. Figure 1 shows the number of devices purchased, Figure 2 shows the mass of devices purchased and Figure 3 shows the purchasing and the disposal costs of these devices.
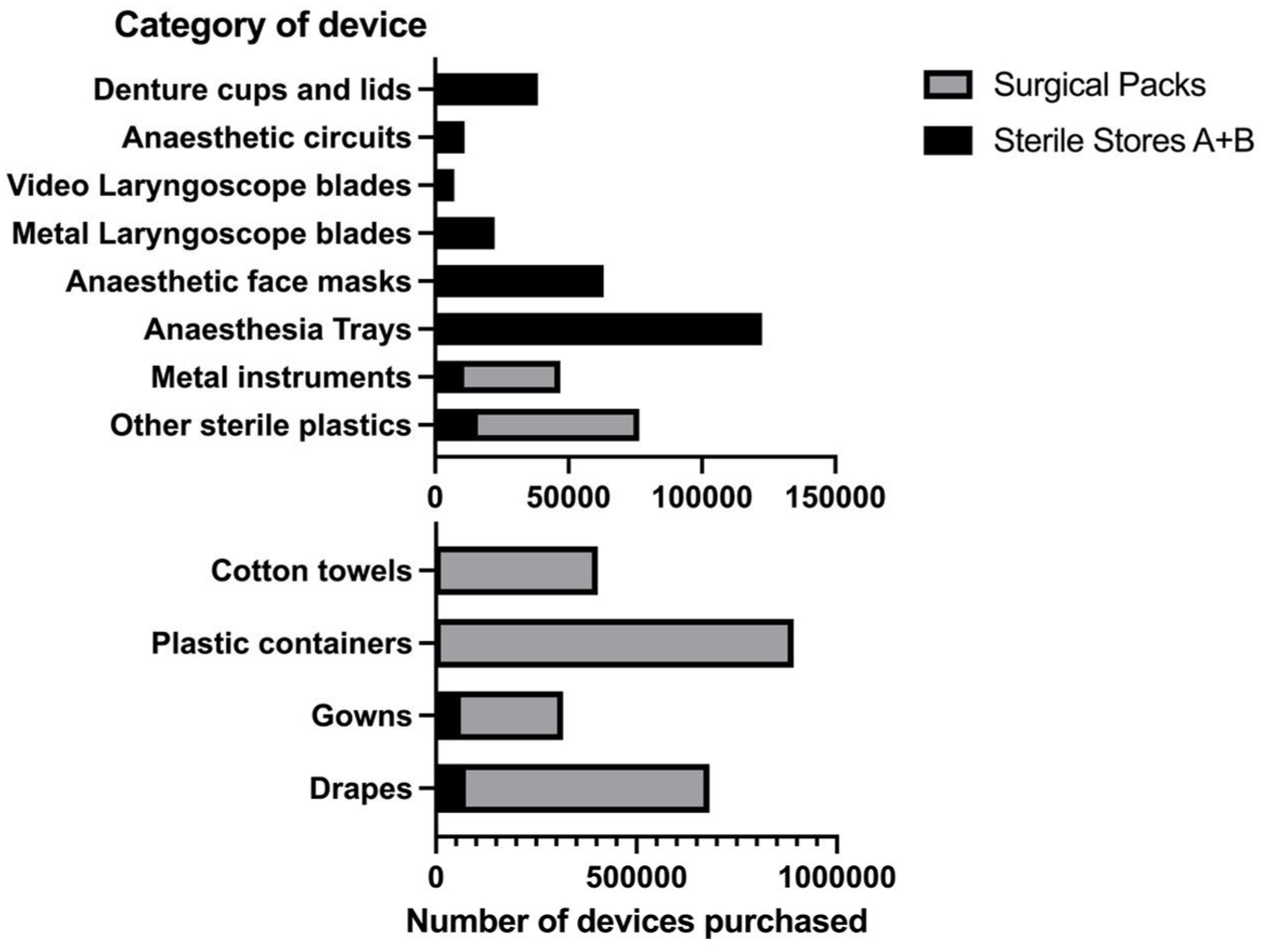
Number of single-use devices purchased by two operating theatre suites between July 2019 and June 2024, where the device had an available reusable alternative.
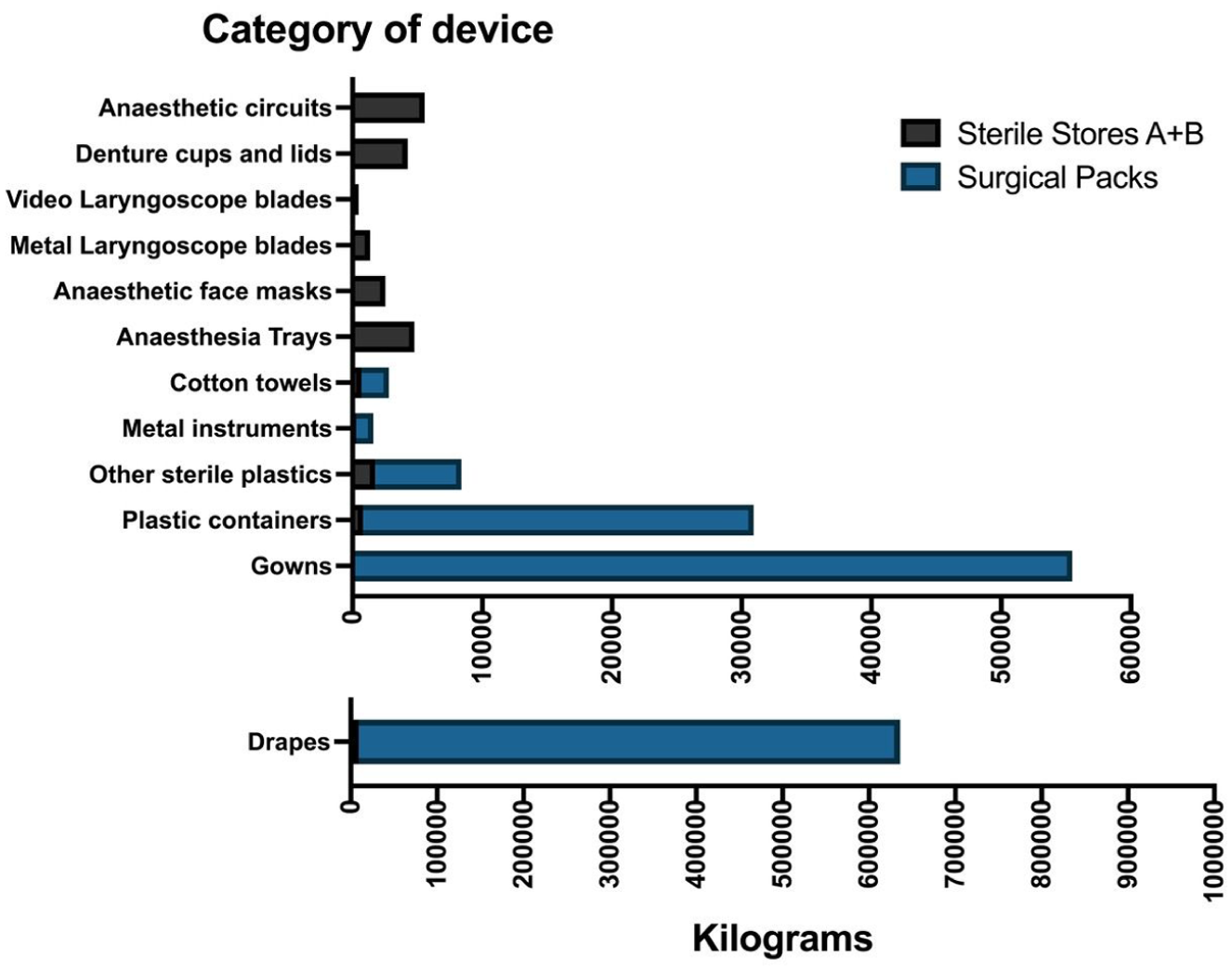
Mass of single-use devices purchased by two operating theatre suites between July 2019 and June 2024, where the device had an available reusable alternative.
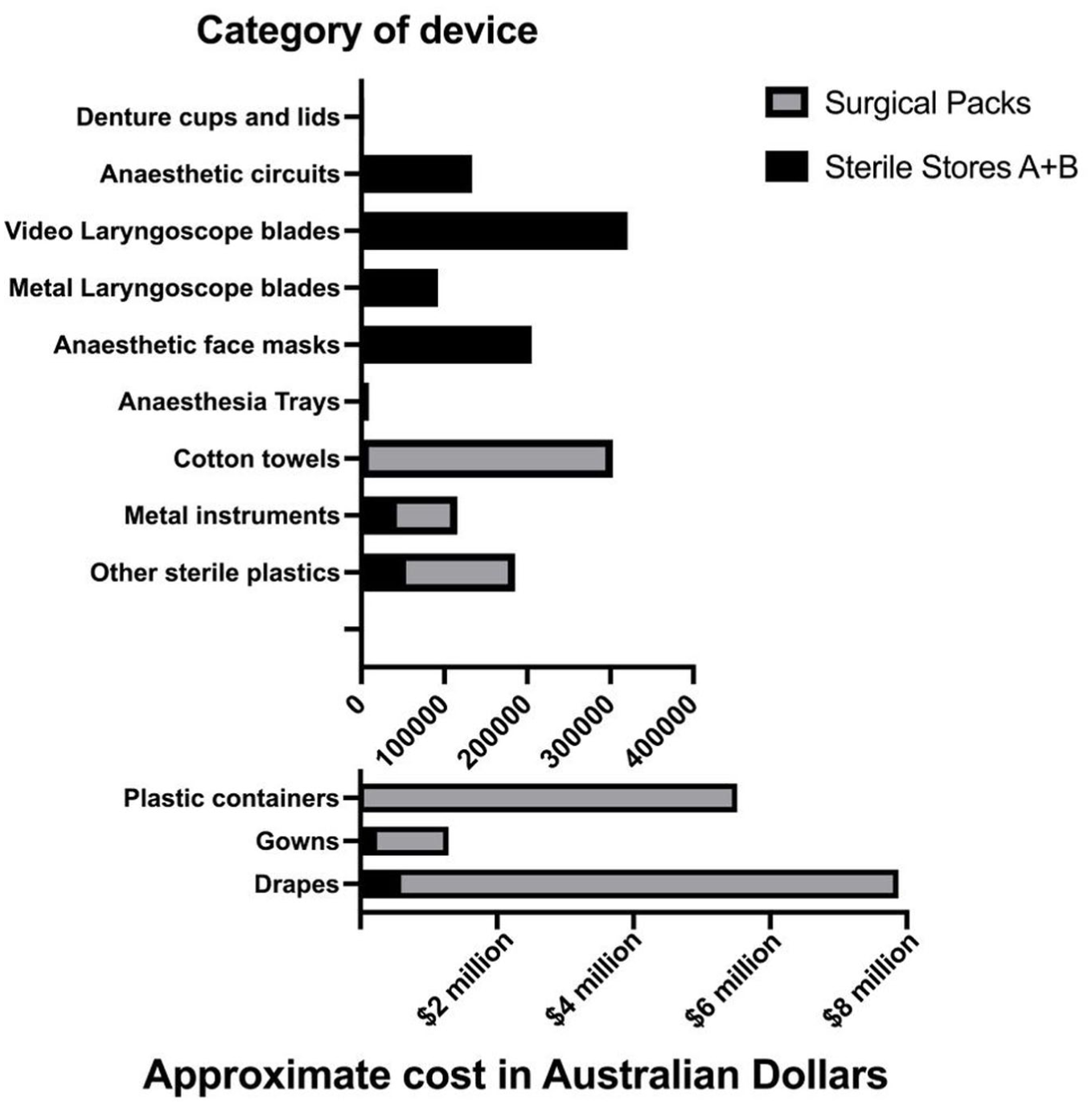
Approximated purchasing cost of single-use devices purchased by two operating theatre suites between July 2019 and June 2024, where the device had an available reusable alternative.
Discussion
This study describes some of the environmental and financial implications of procurement decisions that favour single-use medical devices in operating theatres where a reusable alternative device is known to be utilised in nearby health services. A large number of barriers and enablers to implementing reusable operating theatre devices have been identified elsewhere and are not re-explored here; however, the cost of using reusable devices is frequently perceived to be greater than the cost of single-use devices. 23 This study did not explore the cost of reusing devices but does demonstrate the funds that could potentially be leveraged towards reprocessing costs when introducing more reusable devices. A study of barriers and enablers for enhancing the uptake of reusable equipment identified perceptions about greater cost of reusable devices and this study estimates the outgoings for the purchase of single-use devices and for their disposal as well. 23 Additionally, a study of barriers to uptake of reusable devices proposed interventions to influence their uptake in theatres, of which one was the graded introduction of simple equipment and avoidance of complex reusable medical devices that are more challenging to reprocess. 23 Simple equipment included bowls, jugs, drapes and gowns while complex equipment included cannulated and interlocking devices. The findings of this study quantify this opportunity by excluding those complex devices. Therefore, the real opportunity for waste reduction is higher than the findings presented here because this study does not include every device that could be replaced with a reusable device. The implications for waste reduction across all hospital areas outside of operating theatres is even greater, but beyond this study’s scope. Health services with sustainability goals must explore their supply chains and address single-use medical device procurement as part of their net zero strategies and waste reduction plans. The results of this study are relevant to healthcare workers working in any area where simple single-use equipment is used despite a reusable alternative being available, including non-sterile personal protective equipment such as protective gowns. In 2023 this health service made a swap from single-use non-sterile kidney dishes to reusable kidney dishes and trays across all healthcare areas, with financial and waste savings seen within the first year of implementation.
Limitations
The findings are limited by being single service, although this health service can be utilised as a base for extrapolating waste reduction opportunities statewide. If all Victorian public hospitals purchased similar single-use devices, the approximate waste from these devices alone could be up to 5757 t of waste over five years across the state, with costs of approximately A$124m in purchasing and A$9m in disposal alone. Further, not all hospitals use the same single-use medical devices and in the same proportions, so applying these findings needs to take the local context into account. Of note, this study used a clinical waste rate of 57% based on a local study finding; however, this is higher than other studies from surrounding hospitals. 5 The purchasing cost of devices that were predominantly supplied through pre-packed surgical packs could not be directly measured and should be interpreted with caution; however, the figures present a guide and invite reflection on the speed with which capital investments in reusable equipment might be returned over time. The true cost of purchasing a device is not represented by the purchasing costs alone, and we have not reported on any costs of reprocessing (although these are reported elsewhere), which is only one of many barriers to more reusable devices in operating theatres.10,23,24 One financial barrier to reusable device use in theatres is that the financial structures in hospitals are complex, and purchasing costs of capital equipment such as a fleet of reusable devices and purchasing costs of disposable devices might come from separate cost-centres. 23 Not all drapes could be directly measured; however, the mass of drapes was estimated by methodology that is replicable and minimised further waste. Further, it does not include all potential devices because only simple high-use devices were included. Examining high-value single-use medical devices such as bronchoscopes and endoscopes might provide differing perspectives.
Conclusion
Although the figures presented provide context to the volume of waste experienced by health professionals working in high-waste environments such as operating theatres it does not translate that simply switching to reusable medical devices is easy and can be achieved rapidly. Not included in this study is the financial and environmental cost of decontaminating, cleaning and re-sterilising devices, although these have been studied elsewhere and favour reusable devices, particularly when renewable energy is utilised.10,14 Qualitative research shows that there are a large number of barriers to uptake of reusable devices in operating theatres across all levels, from state-level procurement down to individuals’ knowledge and trust of reprocessing of devices, despite this occurring for a wide range of already in-use reusable surgical instruments. 23 Coordinated eco-leadership with executive support is required in order to have a successful staged replacement of single-use medical devices with their reusable alternatives. Operational costs for single-use device purchasing should be redirected into investments in robust reprocessing and capital equipment investments in reusable devices. The benefits of replacing single-use devices include waste reduction and minimising environmental pollution, cost savings from purchasing and disposal plus the potential for higher quality products (particularly for devices such as trays and bowls). In a survey of National Health Service England staff, nearly all (98%) felt that it was important that the health service supports the environment and it is also possible, though unstudied, that reducing handled waste improves staff perceptions of their healthcare employer and encourages greater engagement with improving their workplace. 25
Footnotes
Acknowledgements
We would like to acknowledge Dana Peters for her assistance in data acquisition and Dr Scott McAlister for his assistance in greenhouse gas emissions estimation.
Author contributions
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
