Abstract
Polymyxin B haemoperfusion is commonly used to adsorb endotoxins in septic shock caused by Gram-negative bacterial infections. Polymyxin B haemoperfusion has been reported to improve hypotension in Gram-positive bacterial infections; however, its efficacy and mechanism in treating such cases are unclear. We hypothesised that polymyxin B haemoperfusion would be equally effective in improving haemodynamics during Gram-positive bacterial infections as in Gram-negative bacterial infections. We conducted a retrospective study that included patients with septic shock admitted to the intensive care unit. The patients were divided into two groups according to bacterial culture results: Gram-negative rod (GNR) and Gram-positive coccus (GPC). We calculated the vasoactive inotropic score (VIS) before (0 h) and 2, 6, 12 and 24 h after polymyxin B haemoperfusion therapy. Data were analysed using two-way analysis of variance and post hoc tests for the associations between infection type and treatment time. Overall, 157 patients with septic shock were enrolled in the study: 81 and 76 patients were treated or not treated with extracorporeal haemoperfusion therapy, respectively. Although there was no significant difference in the VIS in polymyxin B haemoperfusion between patients with GNR and GPC infections, there was a significant decrease in the VIS over time, even when GPC was the causative organism. In addition, the degree of reduction in the VIS was significantly different in both the GNR and GPC groups compared with that in the non-extracorporeally treated group. Thus, polymyxin B haemoperfusion for septic shock caused by GNR reduced the VIS and could be effective even in cases of GPC infection.
Introduction
In septic shock, immune cells such as macrophages and dendritic cells are stimulated by pathogen-associated molecular patterns, such as the endotoxin lipopolysaccharide, an outer membrane component of Gram-negative bacteria. 1 This induces inflammatory cytokines, platelet-activating factors, and nitric oxide synthase activity.1,2 These changes often lead to fatal haemodynamic consequences and multi-organ failure. Endotoxin removal is achieved via direct haemoperfusion therapy that uses polymyxin B-immobilised fibres to adsorb endotoxins from the blood of patients with septic shock.3 –5 Large-scale randomised controlled trials (RCTs) of polymyxin B haemoperfusion revealed improvements in mean arterial pressure and PaO2/FIO2 (P/F), as well as significant improvements in clinical outcomes in patients with septic shock; however, these RCTs have not demonstrated clear improvements in mortality.6 –9
Polymyxin B haemoperfusion is used to treat septic shock caused by Gram-negative bacterial infections, such as intra-abdominal infections. In the hyperacute phase of septic shock, polymyxin B haemoperfusion could result in prolonged hypotension. In contrast, recent reports have shown that this therapy may also have an ameliorative effect on hypotension in patients infected with Gram-positive organisms. 10
No studies have directly compared the efficacy of polymyxin B haemoperfusion therapy between septic shock caused by Gram-negative- and that caused by Gram-positive organisms. If polymyxin B haemoperfusion could be performed independent of the causative bacteria, it might provide earlier intervention for patients with septic shock. Based on this, we hypothesised that polymyxin B haemoperfusion therapy would be equally effective in improving haemodynamics in septic shock patients with Gram-positive bacterial infection and those with Gram-negative bacterial infection. Therefore, we aimed to analyse the effect of polymyxin B haemoperfusion retrospectively. To this end, we divided septic shock patients into polymyxin B haemoperfusion intervention and non-intervention groups and measured the change in catecholamine dose with time during polymyxin B haemoperfusion according to Gram staining of the causative bacteria detected in each group.
Materials and methods
Design, setting and population
This single-centre retrospective observational study was conducted at the intensive care unit (ICU) of Sapporo Medical University Hospital in Sapporo, Japan. The Institutional Review Board of Sapporo Medical University approved the study design and protocol, approval number 322-179. We enrolled patients with unplanned ICU admissions between January 2016 and December 2021. Owing to the observational nature of the study, the results were released on an opt-out basis. We studied patients with septic shock to determine the effect of polymyxin B haemoperfusion on improving hypotension based on Gram stain results. We excluded patients who were under 15 years of age, were pregnant, had severe liver damage, and had mixed Gram-negative rod (GNR) and Gram-positive coccus (GPC) infections, Gram-negative coccus or Gram-positive rod infection, and fungal infections based on blood cultures. Patients received polymyxin B haemoperfusion therapy for less than 24 h. Patients who opted out of the study via a published opt-out document were also excluded from the study.
Data collection
Data on patient characteristics, such as age, sex, and the underlying disease, were collected. The Charlson Comorbidity Index (CCI) and the Acute Physiology and Chronic Health Evaluation II (APACHE II) score at ICU admission were calculated. The Sequential Organ Failure Assessment (SOFA) score was calculated during ICU admission and at 24, 48, and 72 h from the beginning of polymyxin B haemoperfusion therapy. Data for ventilator days, continuous renal replacement therapy, length of ICU stay, P/F, heart rate, and prognosis were extracted from the electronic medical record. Treatment with or no treatment with intravenous steroids, 24-h infusion volume and fluid balance during polymyxin B haemoperfusion therapy, and lactate levels were also recorded. Additionally, mean arterial blood pressure (MAP) was recorded during polymyxin B haemoperfusion therapy for up to 72 h.
When the causative organisms were identified using primary cultures before treatment, the patients were classified into two groups: GNR and GPC. We used the results of the first culture for antibiotic administration. If a single bacterium was identified on blood culture and was consistent with the clinical findings, we considered this bacterium to be the causative organism. Sputum, urine, and ascites cultures were included besides blood cultures. Among these cases, we considered the bacteria identified using the culture to be the causative organism if they were consistent with the clinical findings, including imaging and physical findings. In cases of intra-abdominal infection, if the blood culture showed positive results, the detected bacterium was considered to be the only causative organism. Negative blood culture results, clinical findings consistent with peritonitis, and detection of only GNR in the ascitic fluid were considered to indicate a single GNR infection. Cases in which more than one putative source was detected in each bacterial culture were excluded because they did not meet the objectives of the study.
Clinical data on circulatory dynamics and doses of vasoactive and inotropic agents were extracted as follows: blood pressure, and dopamine (DOA), dobutamine (DOB), norepinephrine (noradrenaline) (Nad), vasopressin, and epinephrine (adrenaline) (AD) doses were extracted before polymyxin B haemoperfusion induction and at 2, 6, 12 and 24 h (T0, T1, T2, T3, and T4, respectively) following polymyxin B haemoperfusion induction.
The vasoactive inotropic score (VIS) [VIS = DOA +DOB + (AD + Nad) × 100 µg/kg/min + vasopressin ×10,000 unit/kg/min + milrinone × 10 µg/kg/min] was calculated as the indicator of haemodynamics in each case. The VIS has been reported as an indicator of haemodynamic support requirements in patients with septic shock.11,12 Clinically, the doses of vasoactive and inotropic agents were titrated in response to a decrease in blood pressure, with a higher VIS indicating a greater need for vasoactive and inotropic agents.
Criteria and methods for initiation of polymyxin B haemoperfusion therapy
Sepsis was diagnosed following the Third International Consensus Definition for Sepsis and Septic Shock (Sepsis-3). 13 Target blood pressure, vasoactive medication utilisation, and infusion management were achieved in accordance with the Surviving Sepsis Campaign Guidelines 2021. 14 Targeting a mean blood pressure of 65 mmHg or higher, initial infusion with crystalloid fluid was started. Preload was assessed mainly by the respiratory variation of the inferior vena cava diameter based on ultrasound, left ventricular capacitance, and stroke volume variation without routine measurements of central venous pressure. Initial crystalloid infusion volume was approximately 30 mL/kg, and Nad was started if target blood pressure could not be achieved or if deemed necessary by the physician. If target blood pressure could not be achieved with Nad, vasopressin was started in combination. Polymyxin B haemoperfusion therapy was indicated for patients diagnosed with sepsis, with the initiation criterion being septic shock with a persistent serum lactate level more than 2 mmol/L despite adequate volume infusion and catecholamine administration [catecholamine index (CAI) more than 5; CAI =DOA + DOB + (AD + Nad) × 100 μg/kg/min)], with the final decision depending on the overall assessment of the effectiveness of polymyxin B haemoperfusion by the treating physician.
A double-lumen catheter was placed in a central vein for blood adsorption. The devices used in this study were the TR-2020 machine (Toray Medical Co, Ltd, Tokyo, Japan), the N-DHP-200A (Toray Medical) extracorporeal circuit, and the PMX-20R (Toray Medical) endotoxin removal column. Polymyxin B haemoperfusion was performed at a blood flow rate of 80 to 120 mL/min. Nafamostat mesylate was used as an anticoagulant at 30 mg/h, maintaining an activated coagulation time of approximately 150 s. Polymyxin B haemoperfusion therapy was initiated in the ICU immediately after the indications were met. If the circuit had thrombus occlusion, polymyxin B haemoperfusion therapy was continued with the exchange of circuits for up to two columns. In cases of hepatic or renal dysfunction, continuous renal replacement therapy was administered in a parallel circuit. Polymyxin B haemoperfusion therapy was performed for 24 h. The safety and efficacy of polymyxin B haemoperfusion therapy for at least 24 h until improvement have been reported in sepsis resulting from abdominal infection,15,16 and polymyxin B haemoperfusion therapy was managed 24 h per day by a specialised clinical engineer.
The non-extracorporeally treated group included patients with septic shock with a lactate level of more than 2 mmol/L who did not undergo polymyxin B haemoperfusion at the discretion of their physician. The T0 value for the non-extracorporeally treated group was defined as the time at which the patient was judged to be in septic shock with a lactate level of 2 mmol/L or more.
Statistical analysis
The data were evaluated for Gaussian distribution using the Shapiro–Wilk normality test. Normally distributed data are presented as mean (standard deviation (SD)), whereas non-normally distributed data are presented as median and interquartile range (IQR). Categorical data are presented as percentages (%). The Mann–Whitney U test was used to analyse continuous variables for patient characteristics, whereas Fisher’s exact test was used for categorical variables. Vasoactive inotrope score data were divided into polymyxin B haemoperfusion and non-extracorporeally treated groups in the GPC and GNR groups, and two-way analysis of variance was used for comparison between the groups over time. Bonferroni multiple comparisons were used for changes in VIS over time. The polymyxin B haemoperfusion and non-extracorporeally treated groups were stratified based on GPC and GNR, and repetitive measurements were conducted, considering temporal factors (within-subjects: pre-polymyxin B haemoperfusion therapy and 2, 6, 12 and 24 h post polymyxin B haemoperfusion). Statistical significance was set at P < 0.05. Statistical analyses were performed using the SPSS version 27 software (IBM Corp, Armonk, NY, USA).
Results
Patient characteristics
Figure 1 shows the flowchart of the patient enrolment process. Of the 157 patients admitted to our ICU during the study period, 81 were treated and 76 patients were not treated with extracorporeal haemoperfusion therapy, respectively; 19 patients in the polymyxin B haemoperfusion group and 23 in the non-extracorporeally treated group were excluded based on the exclusion criteria. Sixty-two patients in the polymyxin B haemoperfusion group and 53 in the non-extracorporeally treated group were included in the study. The number of patients in the GNR and GPC groups in each group were 46 versus 16 (polymyxin B haemoperfusion group) and 34 versus 19 (non-extracorporeally treated group).
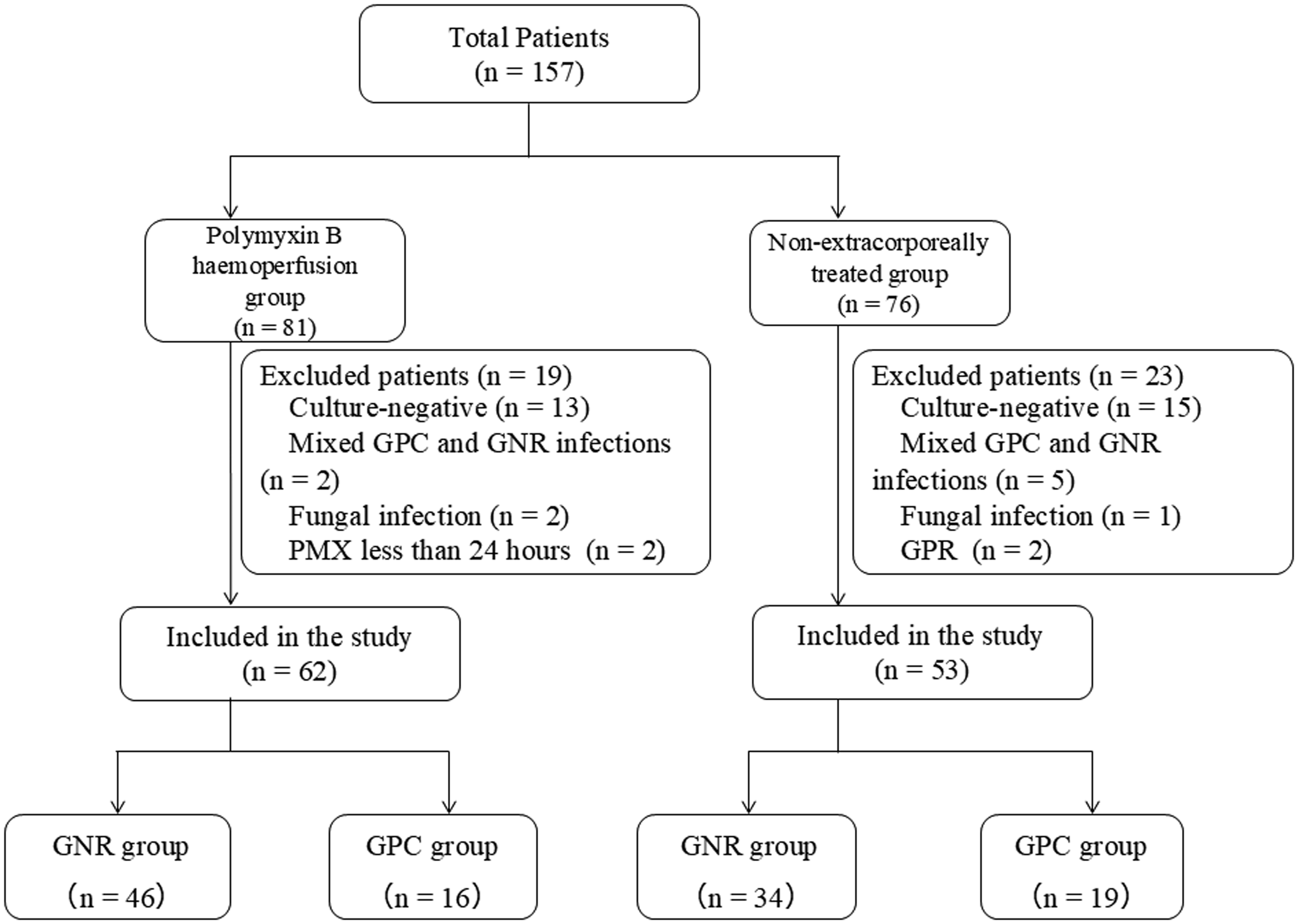
Patient flow.
Patient characteristics are presented in Tables 1 and 2. Based on the characteristics of patients infected with GPC, the CCI in the polymyxin B haemoperfusion group was significantly higher than that in the non-extracorporeally treated group. Based on the characteristics of patients with GNR infection, the CCI and ICU length of stay in the polymyxin B haemoperfusion group were significantly higher than those in the non-extracorporeally treated group. In patients with GPC and GNR infections, the VIS before perfusion therapy with polymyxin B in the polymyxin B haemoperfusion group was significantly higher than that in the non-extracorporeally treated group. The SOFA score decreased significantly for up to 72 h with polymyxin B haemoperfusion in the GNR group only, compared with that in the non-extracorporeally treated group. The P/F ratio did not differ significantly according to polymyxin B haemoperfusion in either the GNR or GPC group. Polymyxin B haemoperfusion was completed within 24 h in all patients, but the column was replaced in three cases in the GNR group and two cases in the GPC group because of intra-circuit thrombus formation.
Characteristics of patients in the Gram-positive coccus group.
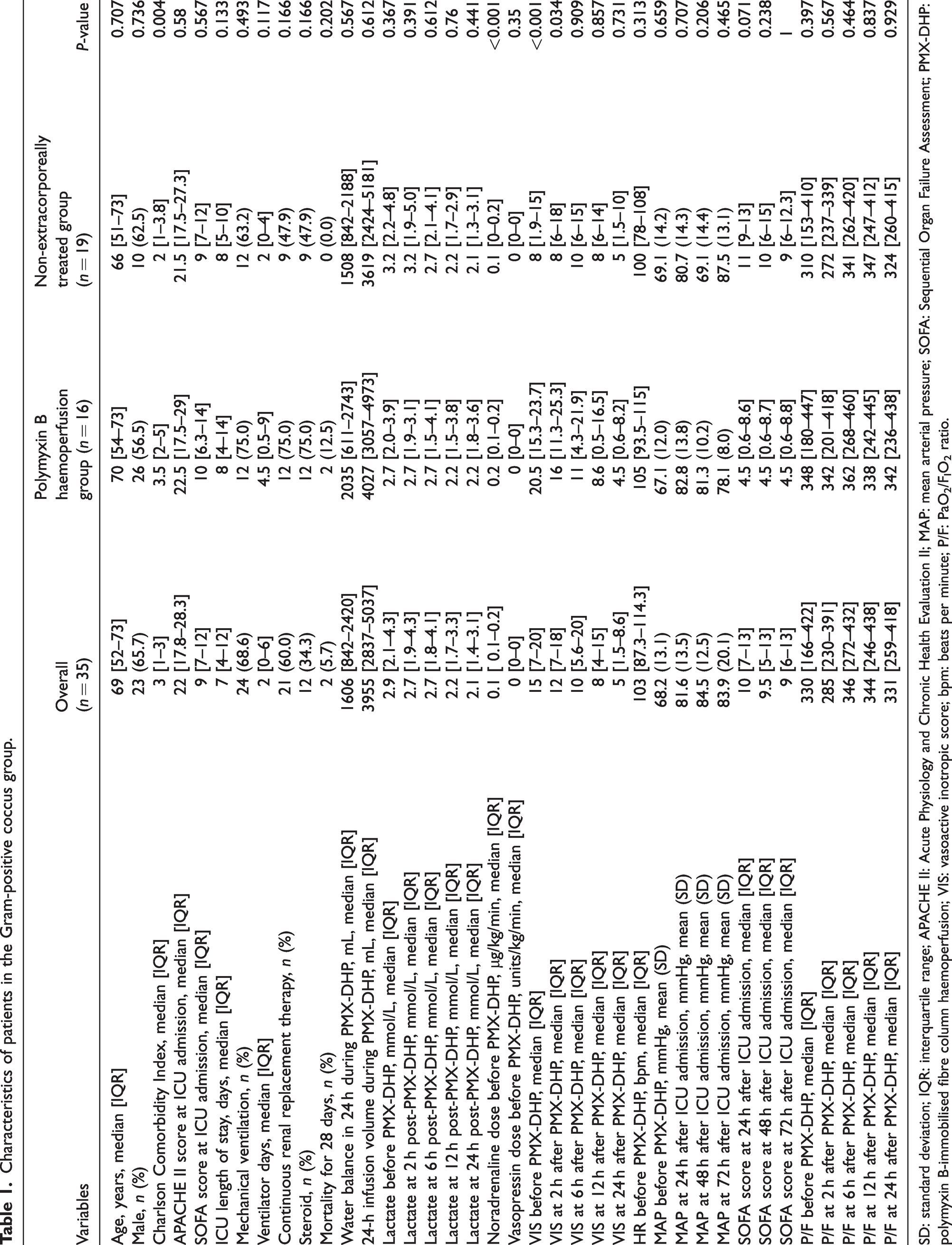
SD: standard deviation; IQR: interquartile range; APACHE II: Acute Physiology and Chronic Health Evaluation II; MAP: mean arterial pressure; SOFA: Sequential Organ Failure Assessment; PMX-DHP: polymyxin B-immobilised fibre column haemoperfusion; VIS: vasoactive inotropic score; bpm: beats per minute; P/F: PaO2/FIO2 ratio.
Characteristics of patients in the Gram-negative rod group.
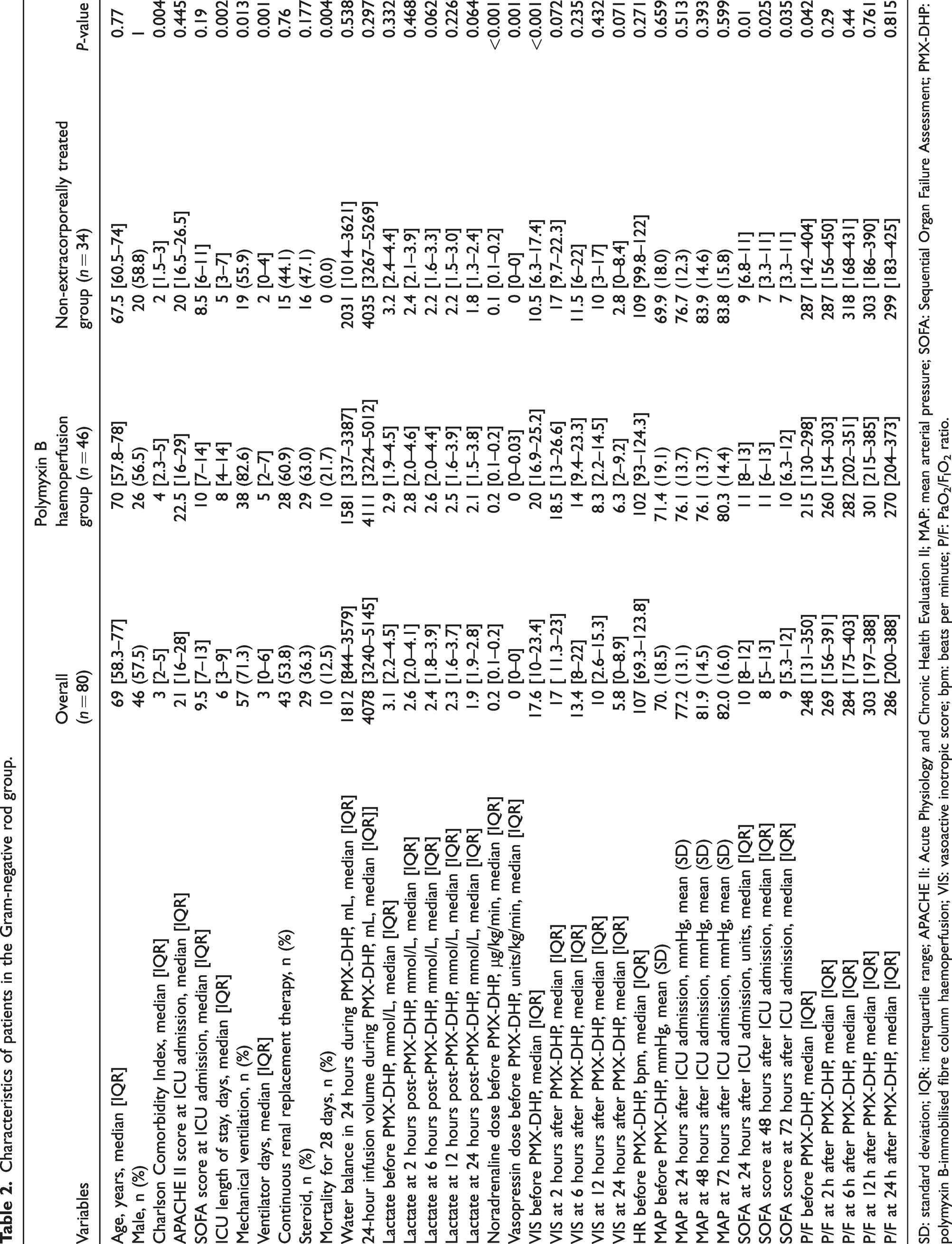
SD: standard deviation; IQR: interquartile range; APACHE II: Acute Physiology and Chronic Health Evaluation II; MAP: mean arterial pressure; SOFA: Sequential Organ Failure Assessment; PMX-DHP: polymyxin B-immobilised fibre column haemoperfusion; VIS: vasoactive inotropic score; bpm: beats per minute; P/F: PaO2/FIO2 ratio.
Isolated microorganisms and infection sites (polymyxin B haemoperfusion group).

GNR: Gram-negative rod; GPC: Gram-positive coccus.
Isolated microorganisms and infection sites (non-extracorporeally treated group).

GNR: Gram-negative rod; GPC: Gram-positive coccus.
The highest proportion of intra-abdominal infections was observed in both the polymyxin B haemoperfusion and non-haemoperfusion treated groups. Escherichia coli and Klebsiella pneumoniae were the most commonly detected organisms in both groups; in GPC, Enterococcus faecalis was more commonly detected in the polymyxin B haemoperfusion group and methicillin-resistant Staphylococcus aureus in the non-haemoperfusion treated group (Tables 3 and 4).
Comparison of VIS time-course in the GPC group
Figure 2 shows the VIS trend from before treatment to 24 h after treatment for each treatment group of patients with GPC infection. The change in the VIS over time in GPC-infected patients was significantly different between the polymyxin B haemoperfusion and non-extracorporeally treated groups (F value =7.611, P ≤ 0.01). Although not significantly different, the VIS tended to be higher in the non-extracorporeally treated group towards the end of 6 h. In contrast, the VIS after 6, 12 and 24 h was significantly lower than that before polymyxin B haemoperfusion, respectively (P ≤ 0.001) in the polymyxin B haemoperfusion group.
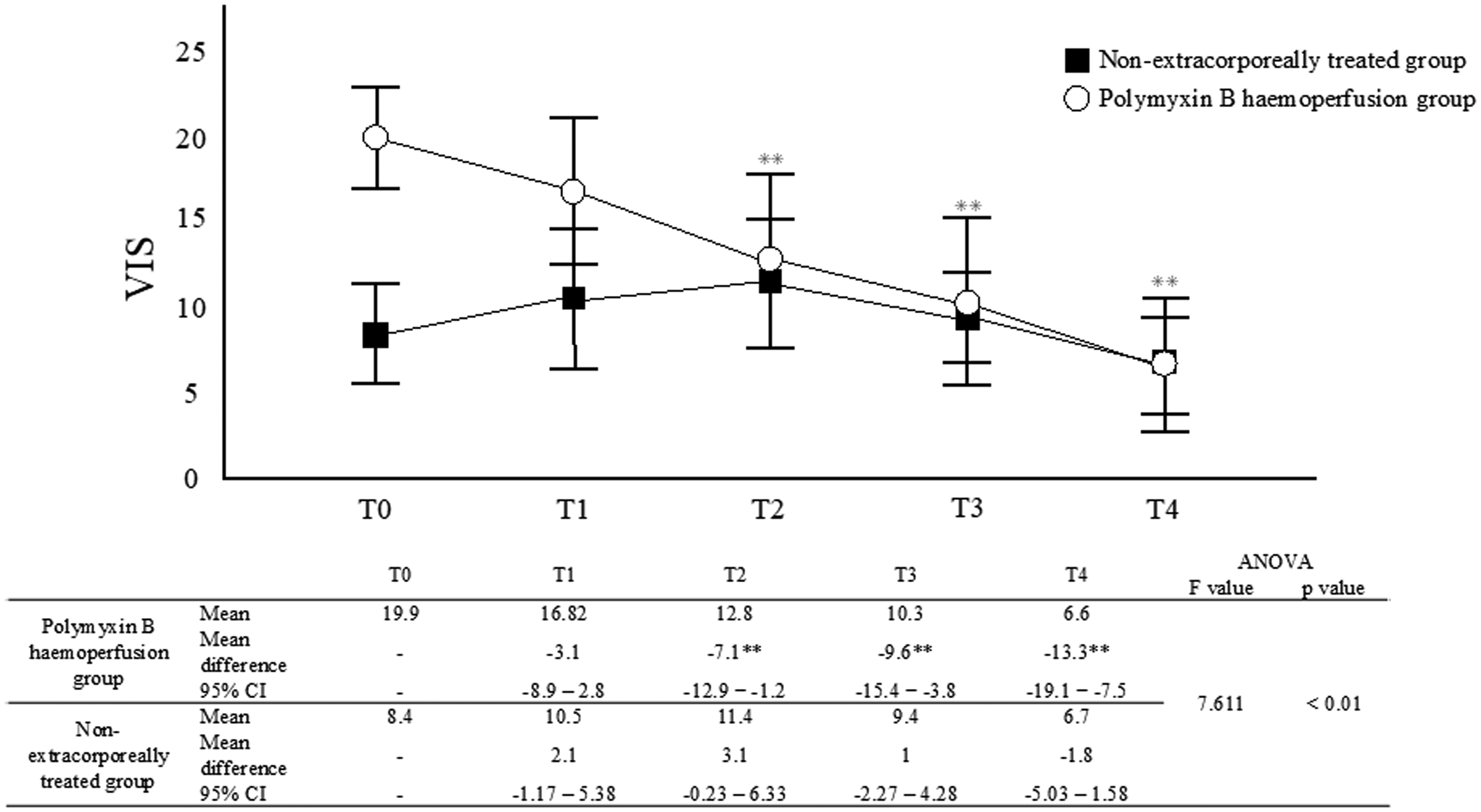
Trend of the vasoactive inotropic score over time in Gram-positive coccus-infected patients in the polymyxin B haemoperfusion group (n = 19) and non-extracorporeally treated group (n = 16): Two-way analysis of variance. T0 = baseline: T1 = 2 h: T2 = 6 h: T3 = 12 h: T4 = 24 h. **P < 0.001; all data represent mean ± SEM. The difference was statistically significant between T0 and T2, T3, and T4 in the polymyxin B haemoperfusion group. The non-extracorporeally treated group showed no statistically significant differences at any time. The table shows the time course of VIS in Gram-positive coccus-infected patients in the polymyxin B haemoperfusion group and the non-extracorporeally treated group: T0 = baseline: T1 = 2 h: T2 = 6 h: T3 = 12 h: and T4 = 24 h. To test the time course of both groups: two-way analysis of variance (ANOVA) was used, and the results are presented as the difference between means and 95% confidence intervals from T0 to T4. Differences from baseline over time are shown at **P < 0.01. The interaction between the two groups was calculated using two-way ANOVA, with F and P values calculated.
Comparison of VIS time-course in the GNR group
Figure 3 shows the VIS trend from before treatment to 24 h after treatment for each treatment group of patients with GNR infection. The change in VIS over time in GNR-infected patients was significantly different between the polymyxin B haemoperfusion and non-extracorporeally treated groups (F value = 7.760, P ≤ 0.01). The VIS in the polymyxin B haemoperfusion group was significantly lower at 6, 12 and 24 h than before polymyxin B haemoperfusion, respectively (P ≤ 0.001). The VIS was significantly lower at 24 h than before polymyxin B haemoperfusion therapy (P < 0.001). There were no significant differences in MAP between the non-extracorporeally treated and polymyxin B haemoperfusion groups at all time points.
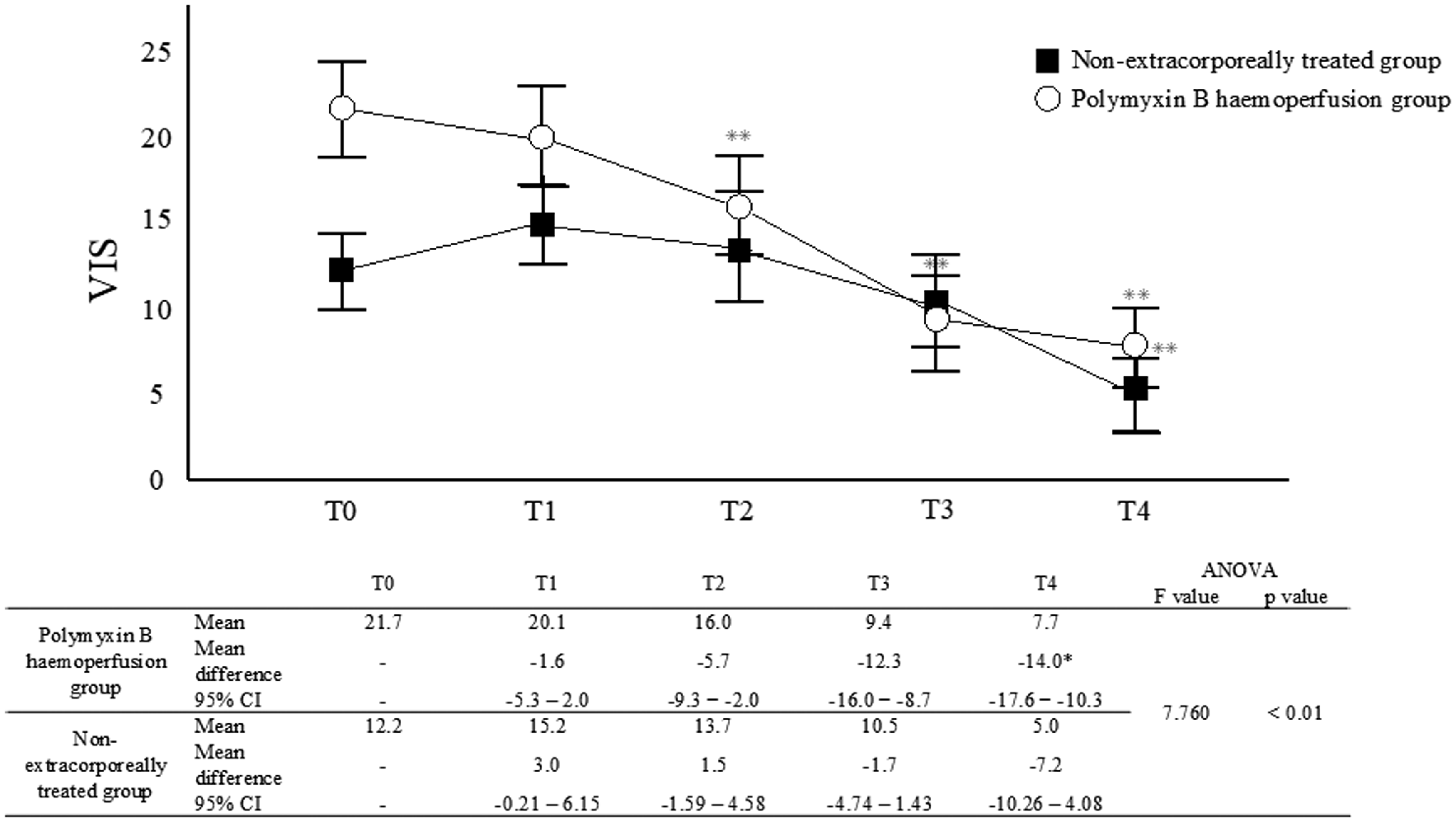
Trend of the vasoactive inotropic score over time in Gram-negative rod-infected patients in the polymyxin B haemoperfusion group (n = 46) and the non-extracorporeally treated group (n = 34). Two-way analysis of variance. T0 = baseline, T1 = 2 h, T2 = 6 h, T3 = 12 h, T4 = 24 h. **P < 0.001; all data represent mean ± SEM. This difference was statistically significant between T0 and T2, T3, and T4 in the polymyxin B haemoperfusion group. The nonextracorporeally treated group showed a statistically significant difference between T0 and T4. The bottom row shows the time course of VIS in Gram-negative rod-infected patients in the polymyxin B haemoperfusion and non-extracorporeally treated groups: T0 = baseline, T1 = 2 h, T2 = 6 h, T3 = 12 h, and T4 = 24 h. To test the time course of both groups, two-way analysis of variance (ANOVA) was used and the results are presented as the difference between means and 95% confidence intervals from T0 to T4. Differences from baseline over time are shown at **P < 0.01. The interaction between the two groups was calculated using two-way ANOVA, with F and P values calculated.
The SOFA scores and MAP for each treatment group of the GPC- and GNR-infected patients over time are shown in Figure 4. In patients with GPC infection, there were no significant differences in SOFA scores in the polymyxin B haemoperfusion group, but there was a significant difference in SOFA scores in the non-extracorporeally treated group at 48 and 72 h compared with baseline (P = 0.003, P < 0.001, respectively). In patients infected with GNR, there were significant differences in SOFA scores in the polymyxin B haemoperfusion group at 48 and 72 h compared with baseline (P = 0.038 and P = 0.033, respectively). The SOFA scores in the non-extracorporeally treated group were not significantly different.
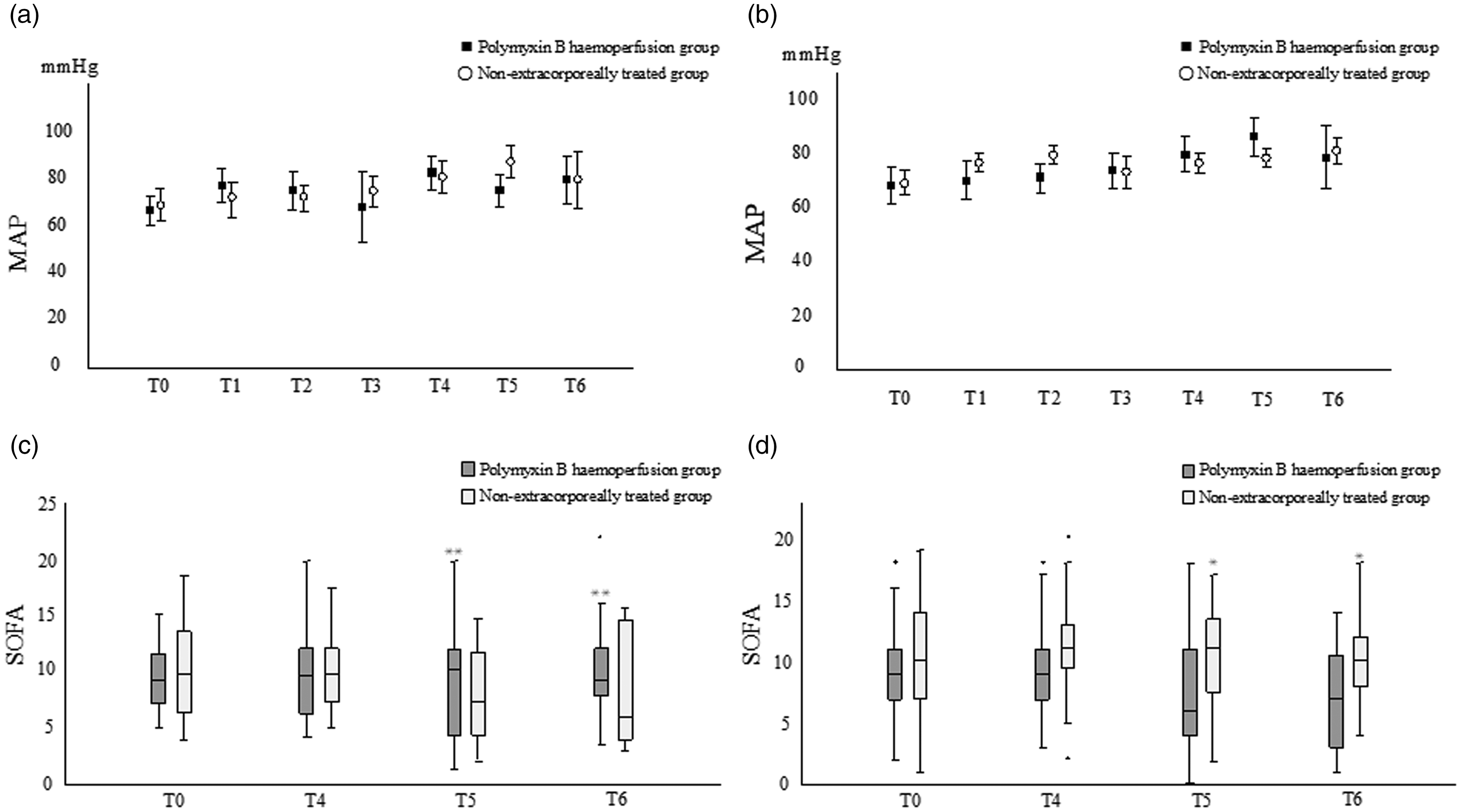
MAP and SOFA score for each treatment in the GPC and GNR groups: (a) MAP for the GPC group, (b) MAP for the GNR group, (c) SOFA score for the GPC group, and (d) SOFA score for the GNR group. Two-way analysis of variance. *P < 0.05, **P < 0.01. T0 = baseline, T1 = 2 h, T2 = 6 h, T3 = 12 h, T4 = 24 h, T5 = 48 h, T6 = 72 h. Circle markers in (a) and (b) indicate mean and standard deviation for the non-extracorporeally treated group, and square markers indicate those for the polymyxin B haemoperfusion group. Horizontal lines in (c) and (d) indicate median values; no statisticallv significant differences were observed in the polymyxin B haemoperfusion group in (c) at any time point. In the non-extracorporeally treated group, there was a statistically significant difference between T0 and T2 and T3: in the polymyxin B haemoperfusion group in (d), there was a statistically significant difference between T0 and T2 and T3. In the non-extracorporeally treated group, there was no statistically significant difference at any time point.
Discussion
We compared the efficacy of polymyxin B haemoperfusion therapy based on the Gram staining classification of the causative bacteria. In the polymyxin B haemoperfusion group, the VIS improved in both the GNR and GPC groups from 6 h after induction of polymyxin B haemoperfusion. To the best of our knowledge, this is the first study to compare the effects of polymyxin B haemoperfusion therapy on different types of infectious organisms. Mortality, ventilator days, and 24-h fluid balance during polymyxin B haemoperfusion therapy were not significantly different between the groups. The mortality rate in the GNR and GPC groups was approximately 20%. There was no significant deviation from the findings of previous studies, which reported a mortality rate ranging from 16% to 32% for septic shock patients who underwent polymyxin B haemoperfusion therapy.6,17,18 In the present study, ICU length of stay was significantly shorter in patients with GNR infection without haemoperfusion than in those with haemoperfusion (Table 2). The possibility of haemoperfusion-associated adverse effect could be considered. However, the proportion of patients on ventilators and with a longer duration on ventilators was significantly higher in the GNR-infected group that underwent haemoperfusion (Table 1). Conversely, ICU length of stay, mechanical ventilation rate or ventilator days for patients infected with GPC did not significantly differ between the groups with and without haemoperfusion (Table 1). The longer ICU length of stay in GNR-infected patients with haemoperfusion may be due to the presence or absence of ventilator management.
The efficacy and safety of polymyxin B haemoperfusion therapy for sepsis and septic shock have been discussed. Several multicentre randomised trials of polymyxin B haemoperfusion therapy for 2 h, including the EUPHRATES study, have been conducted, but the observed clinical efficacy has not been consistent.6 –9,17 Moreover, most focused on GNR cases, such as intra-abdominal infections, and examined the effects of polymyxin B haemoperfusion, focusing on endotoxin adsorption and endotoxin levels. Two review articles suggested that polymyxin B haemoperfusion decreased endotoxin levels, improved MAP, and reduced organ dysfunction.19,20 Based on a subgroup analysis of the EUPHRATES trial, the most recent double-blinded RCT of polymyxin B haemoperfusion, it was suggested that a survival benefit existed in patients with a high level of endotoxin activity (EA), and endotoxin levels are likely to be important in polymyxin B haemoperfusion treatment against sepsis. 18 The present study suggests that polymyxin B haemoperfusion could be effective against septic shock caused by GPC as a result of an unknown mechanism. Previous studies reported elevated EA in infections and sepsis caused by GPC, but the mechanism is unclear.21 –23 Several studies have demonstrated the efficacy of polymyxin B haemoperfusion therapy against non-Gram-negative organisms.2,10 Based on these findings, it was hypothesised that polymyxin B haemoperfusion would be effective if the causative organisms were GNR or GPC, and our results confirmed this. In the present study, haemodynamics improved after polymyxin B haemoperfusion even in septic shock caused by GPC. The reason for this may include elevated endotoxin levels in the GPC group being adsorbed through polymyxin B haemoperfusion therapy. Polymyxin B haemoperfusion significantly reduced the VIS in the GNR group compared with the non-extracorporeally treated group, whereas the level of reduction in the VIS in the GPC group did not differ. It has been reported that EA in patients with infections and sepsis caused by GPC is lower than that in those caused by GNR.21,22 This may be responsible for the delayed haemodynamic improvement of patients with sepsis from GPC, in contrast to that in sepsis from GNR, in this study.
This retrospective study has a few limitations. First, confounding factors were not excluded, although patient characteristics, including water balance for the 24 h during polymyxin B haemoperfusion, were not significantly different between the groups. Second, the aim of this study was to investigate the effect of polymyxin B haemoperfusion by distinguishing groups according to the Gram staining of the initiating organisms. Therefore, if the bacteria detected in culture tests were a mix of GNR and GPC, they were excluded from the study. This situation is quite possible in clinical practice and may not accurately reflect the clinical effect of polymyxin B haemoperfusion. However, this method may be suitable for determining the effect of polymyxin B haemoperfusion by Gram staining and should be considered in large RCTs that have not determined the effect of polymyxin B haemoperfusion on the causative organism. Third, the nonsurgical treatment group included patients with septic shock with lactate levels of 2 mmol/L or more who did not undergo polymyxin B haemoperfusion, which may have introduced a selection bias. This is because polymyxin B haemoperfusion is performed when infection by Gram-negative bacteria is strongly suspected, and the definitive organism is often unknown at the time the procedure is initiated. In addition, lactate can behave erratically around 2 mmol/L during the administration of initial resuscitative infusions, and the decision to perform polymyxin B haemoperfusion or not is at the discretion of the treating physician. Therefore, even if the lactate level is above 2 mmol/L, there may or may not be cases where polymyxin B haemoperfusion is performed in actual clinical practice.
Finally, EA was not measured in this study, therefore, it may be necessary to check for differences in endotoxin concentrations between the GPC and GNR groups.
Conclusion
In conclusion, polymyxin B haemoperfusion for septic shock caused by GNR reduces the VIS and could be effective even in cases of GPC infection. This study suggests that polymyxin B haemoperfusion may improve inotropic scores in patients with septic shock, regardless of the organism or site of infection, enabling early intervention without waiting for blood culture results.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
