Abstract
Summary
Iron-deficiency anaemia (IDA) is a global health problem. The impact of IDA on outcomes in obstetric patients who undergo caesarean section (CS) is unknown. We assessed the feasibility of conducting a large study to investigate perioperative anaemia and outcomes after CS. With ethics approval and trial registration, 60 obstetric patients (30 planned CS, 30 emergency CS) were included. Feasibility categories were willingness to participate in a study, to undergo additional blood tests (haemoglobin, ferritin), to have weight measured and to undertake quality of recovery (QoR) questionnaires. Of eligible people approached, 100% agreed to participate and 100% would participate in a future study. Ninety percent (95% confidence interval (CI) 82.4% to 97.6%) and 83% (95% CI 73.9% to 92.7%) agreed to additional tests in hospital and after discharge, respectively. Ninety-eight per cent (95% CI 95.1% to 100%) consented to being weighed, and 100% completed QoR questionnaires. Preoperatively, 8.5% (95% CI 1.4% to 15.6%) of participants were anaemic. Postoperative haemoglobin was measured in only 22 (36.7%, 95% CI 24.5% to 48.9%) participants, and 40.9% (95% CI 20.4% to 61.5%) were anaemic, suggesting at least a quadrupling of the proportion of anaemic patients from pre- to postoperatively. Ferritin was not measured postoperatively in any participant. The prevalence of participants discharged with IDA was unquantifiable. Postoperative IDA in obstetric patients is likely to be a serious problem that is currently unrecognised. Our data suggest that a definitive study to determine associations between maternal anaemia and perioperative outcomes after CS surgery is feasible.
Introduction
Anaemia, defined as a haemoglobin concentration of less than 110 g/l in pregnant women, is a serious and widespread global health issue affecting 25% of the obstetric population.1,2 Anaemia, specifically caused by iron deficiency (ferritin less than 30 µg/l), can lead to significant morbidity including fatigue, poorer cognition, infection-inhibited lactation and adverse mental health effects.3 –6 Iron-deficiency anaemia (IDA) is a major risk factor for postpartum haemorrhage contributing to maternal mortality. 7 Fetal and neonatal complications include premature birth, low birthweight and neurological impairment. 8
Traditionally, all pregnant women have been grouped together when examining relationships between IDA and outcomes after birth, with no dichotomisation into women who undergo major surgery (caesarean section (CS)) and those who do not (vaginal birth). Pregnant women are rarely considered perioperative patients despite nearly one in three undergoing major surgery (CS), 9 and even the word ‘surgery’ is seldom associated with birth. 10 This means that obstetric patients who undergo CS have not been sufficiently recruited into longitudinal research studies to determine the relationship between perioperative IDA and short- and long-term outcomes after surgery. It might be expected that, like non-pregnant surgical patients, perioperative IDA in the obstetric surgical population is associated with significant morbidity, mortality and cost to health services. 11 Given the haemoglobin threshold for the definition of anaemia in this population is still much lower than other surgical groups (less than 110 g/l compared with less than 130 g/l12,13), and the fact that CS is one of the most common surgical procedures performed globally, 14 the lack of critical investigation in this area is likely to be obscuring a serious and preventable global health issue.9,15 We think there is an urgent need to address this problem.
One of the first steps in investigating IDA and its relationship to outcomes in obstetric patients after CS is to determine whether a study is feasible, including assessing the willingness of these patients to be involved in this form of research, which includes additional tests, questionnaires, weight measurement and follow-up. Unlike other surgical populations, it cannot be assumed that obstetric patients will consent to participating in research, as there are many additional unique challenges faced by this group when recovering from surgery. The Pregnancy Risk Assessment Monitoring System identified major stressors in the postpartum period. These included the need for additional social support, breastfeeding problems, lack of education about the care of the newborn after hospital discharge, the need for assistance with postpartum mental health issues including postpartum depression, the need for an extended postpartum hospital stay and financial issues. 16 Taking on the additional load of being involved in a research study under these circumstances may not be attractive to potential participants no matter how important the research question is.
We aimed to determine the feasibility of undertaking a large study (Maternal anaemia and perioperative outcomes after CS surgery (MAPOC)) by assessing the willingness of postoperative obstetric patients to participate in research about IDA. We conducted a prospective observational study assessing participation rates in the feasibility study itself, willingness to participate in a future study, the participants’ likelihood to undergo serial procedures and assessments relevant to the physiology and pathophysiology of anaemia (such as blood tests and weight measurements) and serial quality of life (QoL) questionnaires, in hospital and after discharge. We collected data in these feasibility categories, data resulting from that participation (weight data, QoL questionnaire data), and routinely recorded data to assist with the design of a future study and future study questions in this area.
Material and methods
After ethics approval (Royal Women’s Hospital, Parkville, Australia, HREC 33/21 1 September 2021) and prospective trial registration (ACTRN12621001066875), 60 obstetric patients (30 planned CS and 30 emergency CS) were studied at a single tertiary referral hospital with approximately 7500 births per year. 17
Inclusion criteria were age 18–40 years and gestational age 37 weeks or more having a CS. Exclusion criteria were planned combined procedure (such as tubal ligation or hysterectomy), pre-existing haemoglobinopathy, placental adhesive disorder, pre-eclampsia or immediate category 1 CS. 18
Eligible participants were asked if they would participate in a study about anaemia which involved understanding their views on participating in a longer-term research study about anaemia, and collecting sample data that would confirm their preferences and inform the design of a larger study. They would be interviewed, weighed and complete a questionnaire about their quality of recovery (QoR). Their medical record would be accessed to record their haemoglobin and ferritin measurements, and intraoperative outcomes, including intravenous fluid volume amount and type and blood loss. Prospective informed written consent before surgery was obtained from all participants. Participants were free to withdraw at any time.
Eligible participants were women who were undergoing CS and who were able to be contacted and reviewed by the study investigator (MO’D). Eligible participants scheduled for elective CS were contacted the day before surgery, and written consent was obtained on the day of surgery. Participants undergoing emergency CS were consented as soon as practical after being booked for surgery by the obstetric team.
Willingness to participate in research about anaemia was assessed by participation rates in this research study (the feasibility study itself), and once patients were participating in the feasibility study, willingness to participate in a future study about the same topic (the definitive (MAPOC) study). This was conducted at a postoperative visit by an investigator within the first 24–36 hours following CS. At the postoperative visit, the participants were also asked about the likelihood of agreeing to additional postoperative pathology testing (Likert scale: unlikely, likely, very likely and unsure). The participants did not undergo these additional blood tests. Participants were asked whether they were happy to have their weight measured. If they said yes, they had their weight measured. Participants were asked if they were happy to complete a questionnaire and if they said ‘yes’ they completed the questionnaire (the validated Obstetric QoR-10 QoR questionnaire (ObsQoR-10)), 19 (Online Supplementary Material) [permission to use sought and obtained].
Three sets of data were collected. The first set was data collected in the feasibility categories. The second set was data resulting from participating in the study (weight data, QoL data, duration of study time). The third set was data routinely recorded that may assist with the design of a future study and future study questions in this area. This included perioperative weight change, intraoperative blood loss and perioperative fluid administration, and serial haemoglobin and ferritin levels (Figure 1).
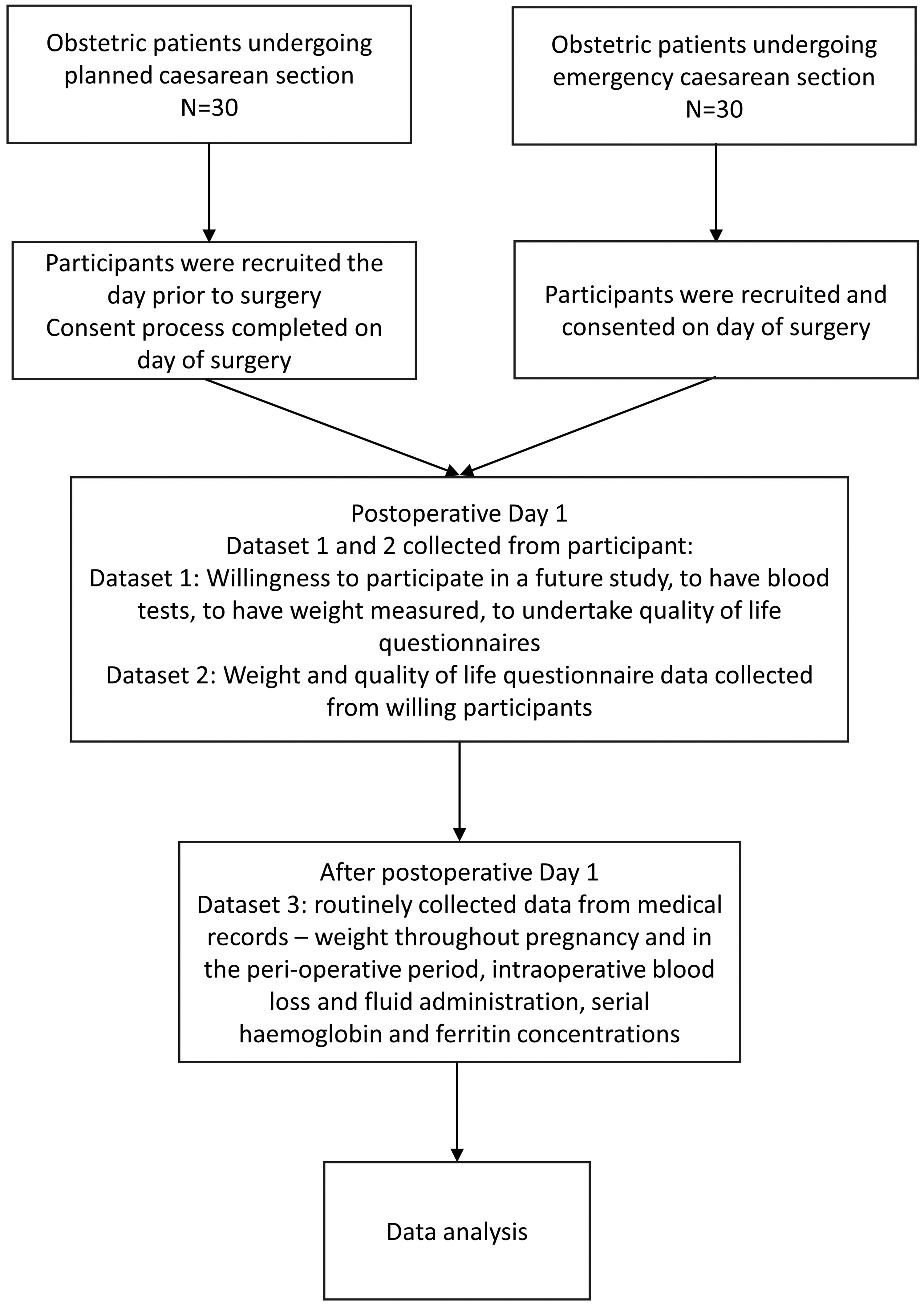
Study scheme. Flowchart of participant involvement showing primary outcomes and secondary outcomes data-collection process.
Data were analysed using SPSS Statistics Version 27 (IBM© SPSS© Statistics IBM Corporation 2020, Chicago, IL) using appropriate parametric or non-parametric tests according to data distribution. Mean, standard deviation (SD), 95% confidence interval (CI) of the proportions, median (quartiles) [lowest and highest value] or number and percentage are presented as appropriate. The 95% CI of the point estimate (p)—in our case proportion for the feasibility categories—was calculated using
Results
Sixty-one people were approached to be involved in the study and 61 (100%) agreed to participate. An additional participant was recruited in the planned CS group because one participant in that group was lost to follow-up due to being discharged from hospital before all data being collected. Sixty participants were analysed over a period of 8 weeks (Figure 2). Participant characteristics are listed in Table 1. Nearly one in eight participants did not have a higher educational level than secondary education, and one in seven participants did not speak English as their first language. Half the participants lived with children at home, and half were undergoing emergency surgery. One in three participants had a significant comorbidity.
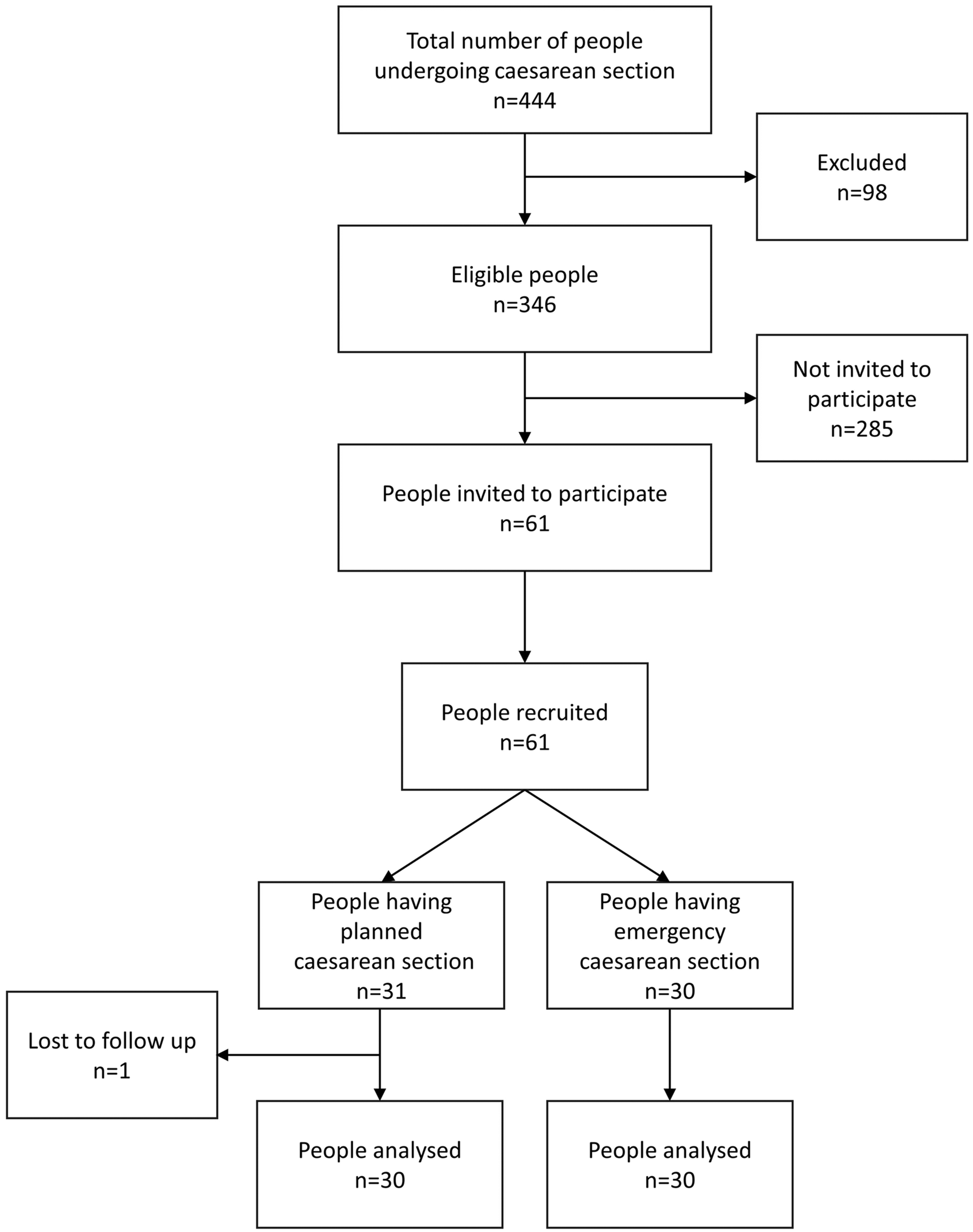
Participant flowchart. Flowchart according to Strengthening the Reporting of Observational Studies in Epidemiology guidelines for reporting observational studies. Some eligible participants were not invited to participate due to investigator availability. Participants were only recruited during the working hours of the investigator during the 8-week study period. Reasons for exclusion included: Category 1 caesarean section n = 29, pre-eclampsia n = 15, gestation <37 weeks n = 45, abnormal placentation n = 1, combined surgery n = 6, other n = 2.
Participant characteristics.
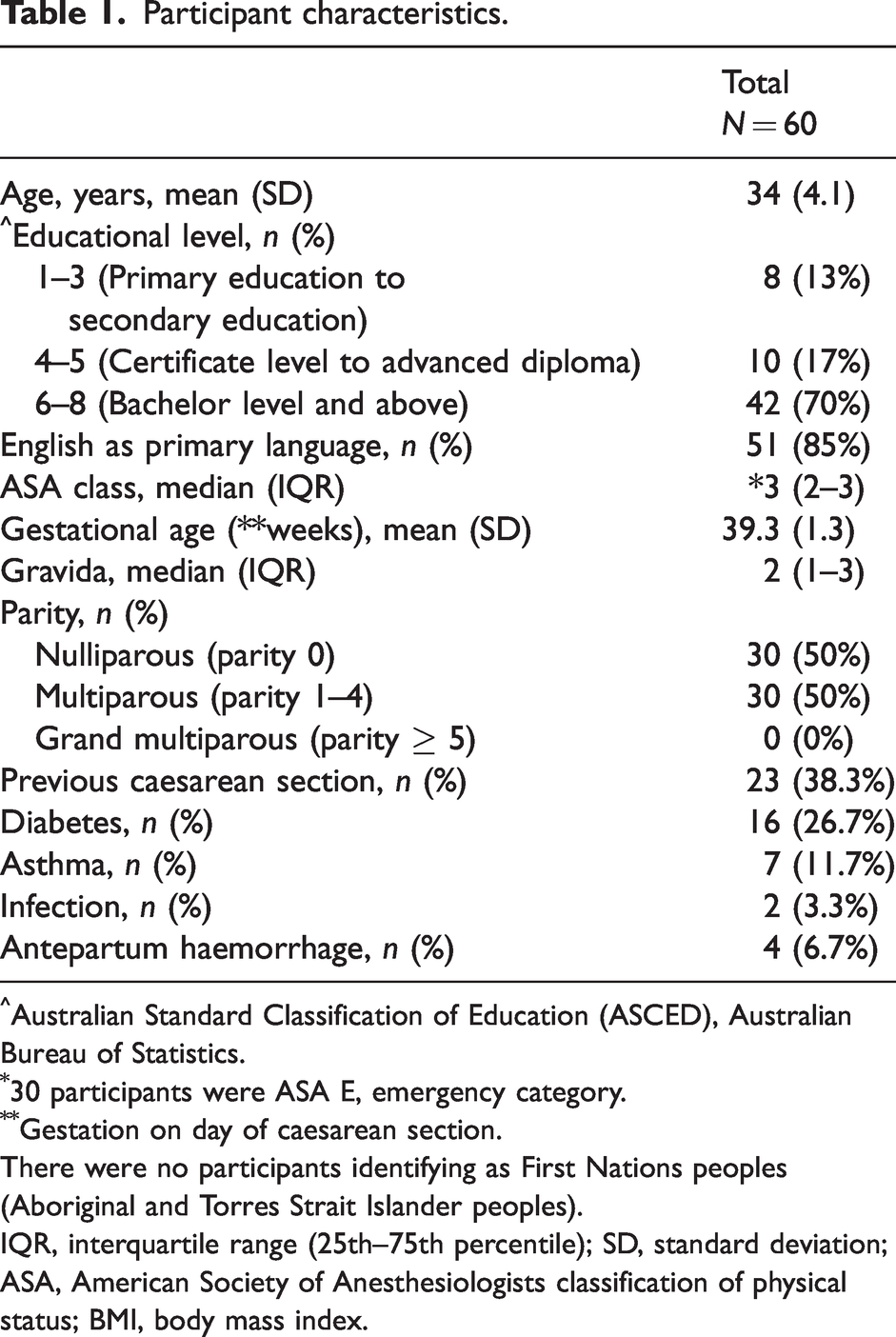
Australian Standard Classification of Education (ASCED), Australian Bureau of Statistics.
30 participants were ASA E, emergency category.
Gestation on day of caesarean section.
There were no participants identifying as First Nations peoples (Aboriginal and Torres Strait Islander peoples).
IQR, interquartile range (25th–75th percentile); SD, standard deviation; ASA, American Society of Anesthesiologists classification of physical status; BMI, body mass index.
Feasibility categories comprising the first set of collected data are presented in Table 2. All recruited participants (60, 100%) were willing to be involved in a future study. Fifty-four (90%, 95% CI 82.4–97.6%) were likely to undergo additional blood tests in hospital, and 50 (83%, 95% CI 73.9–92.7%) to undergo additional blood tests after discharge from hospital to test for anaemia. All but one participant agreed to having their weight measured postoperatively (95%, 95% CI 95.1–100%). QoR questionnaires were completed by all participants (60, 100%).
Feasibility categories.
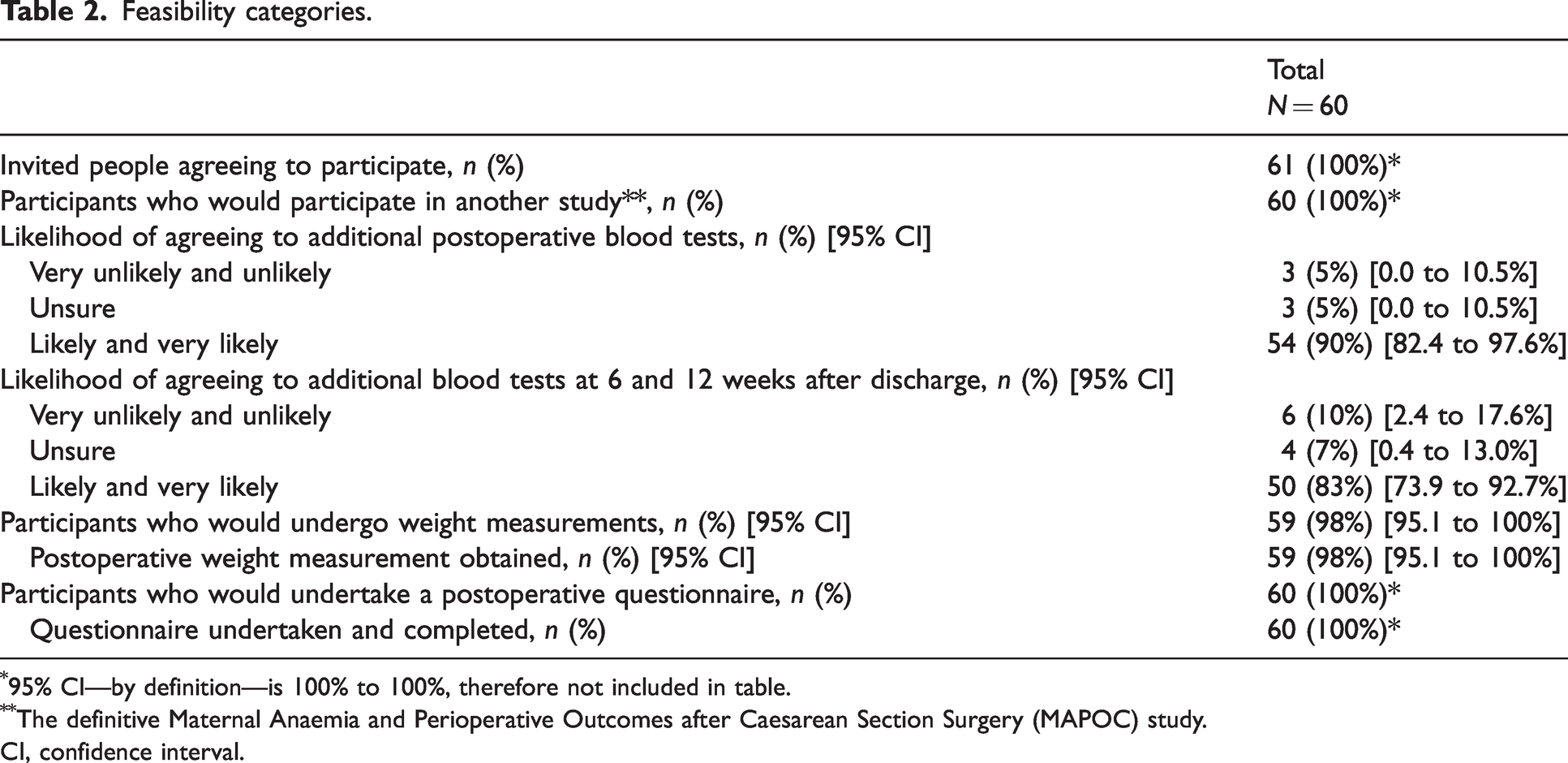
95% CI—by definition—is 100% to 100%, therefore not included in table.
The definitive Maternal Anaemia and Perioperative Outcomes after Caesarean Section Surgery (MAPOC) study.
CI, confidence interval.
The second set of data resulted from participating in the study and were weight data and QoL data. Postoperative weight, measured in 59 out of the 60 participants, was mean (SD) 82 (19.7) kg. All participants completed the ObsQOR-10 questionnaire. The overall median score was 72 (interquartile range (IQR) 54.75–85.88). When groups were compared, participants undergoing planned CS and participants undergoing emergency CS experienced a different QoR score (median difference 5; median score 73.5 (IQR 52.25–87.00) versus 68.5 (IQR 58.25–84.25)); however, there was a low likelihood of there being any actual true difference (P = 0.636), [Mann–Whitney test, exact P value reported taking into account tied values]. The time spent with participants to complete data collection was a mean (SD) of 9.0 (4.1) minutes.
The third set of data represented routinely recorded data that may assist with the design of a future study and future study questions in this area. Perioperative weight data, intraoperative blood loss and intravenous fluid therapy data are shown in Supplementary Tables 1 and 2, respectively. Weight gain from first hospital visit to the day of delivery (CS) was mean (SD) 12.2 (6.1) kg. The mean (SD) weight decrease after CS for the whole group was 2.1 (2.3) kg, with an estimated overall positive mean (SD) fluid balance on postoperative day 1 of 2286 (1047) ml. There was no evidence of a difference in weight change between the planned and emergency CS groups (mean (SD) difference 0.90 (0.64) kg, 95% CI –0.385 kg to 2.185 kg, P = 0.166). There was evidence of a more positive postoperative fluid balance in the emergency group compared with the planned CS group (mean (SD) difference 1374 (245) ml, 95% CI –879 ml to 1870 ml P <0.0001), [unpaired t-test with Welsh correction].
Haemoglobin and ferritin data are given in Table 3. Fifty-six (93%, 95% CI 87.0–99.7%) participants had haemoglobin measured at their first hospital appointment.
Haemoglobin and ferritin data.

For each n (%), the % given uses the denominator of the number of participants who had the measurement done at that time, not of the whole group (n = 60).
World Health Organization definition of postpartum anaemia at 1 week.
Not measured in any of the cohort participants.
Iron-deficiency anaemia defined as haemoglobin <110 g/l with ferritin <30 µg/l. Iron deficiency and iron-deficiency anaemia could not be quantified, as ferritin levels were not measured in this cohort as part of clinical care in the preoperative or postoperative period.
CI, confidence interval; SD, standard deviation.
Preoperative haemoglobin was measured in 59 (98%, 95% CI 95.1–100%) participants. Using a standard threshold value for anaemia (haemoglobin <110 g/l), 8.5% (95% CI 1.4–15.6%) of the tested participants were anaemic, and when using a contemporary threshold value of 130 g/l, 52.5% (95% CI 39.8–65.3%) were anaemic.
Postoperative haemoglobin was measured in only 22 (36.7%, 95% CI 24.5–48.9%) participants. Using a standard threshold value for anaemia (haemoglobin <110 g/l), 40.9% (95% CI 20.4–61.5%) of the tested participants were anaemic, and when using a contemporary threshold value of 130 g/l, 90.9% (95% CI 78.9–100%) of the tested participants were anaemic. There was at least a quadrupling of the proportion of anaemic patients from the pre- to postoperative period using the traditional definition of anaemia.
Most participants had ferritin measured at their first hospital appointment (90%). Over one in three participants were iron deficient at this time, and over one in two participants were iron deficient in the third trimester. Forty (67%) participants were prescribed oral iron, and four participants received an iron infusion in the preoperative period. In the postoperative period 17 participants were prescribed oral iron, and one participant received an iron infusion. The incidence of IDA was 3.6% (95% CI 0.0–8.7%) at first hospital appointment and 4.3% (95% CI 0.0–16.1%) in the third trimester. The incidence of IDA could not be determined postoperatively, as no participant had ferritin levels measured during this time.
Sample size ranges are shown in Supplementary Table 3. From these calculations it is likely that the sample size ranges from 550 participants required to investigate the relationship between anaemia at hospital discharge and outcomes in the postpartum period after hospitalisation, to 5900 participants to investigate the relationship between anaemia in the immediate preoperative period and outcomes after CS.
Discussion
We conducted the first study in postoperative obstetric patients to determine their willingness to participate in anaemia research. There are four key findings.
The first is that despite many obstetric patients having children at home, having undergone emergency surgery, having significant comorbidities to manage, and all having a newborn baby, 100% of obstetric patients were willing to participate in our study (and did participate). All our participants were also willing to participate in a future study. There was also willingness to undertake additional non-routine blood tests both in hospital and after discharge. Participants were willing to have a non-routine postoperative weight measurement, and almost all did have this measurement taken, and all were willing to complete a QoR questionnaire and did complete this questionnaire. The group included participants who did not have an education beyond secondary school, and who did not speak English as their first language. These participant characteristics and willingness data, supported by actual data from undergoing the weight and QoL elements of our study, suggest that a larger study is feasible. While over 80% of the cohort agreed to additional blood tests, we cannot be sure that they would also actually undertake these tests. However, based on our weight and QoL data where there was 100% concordance with willingness to undertake the assessment and actually undertaking the assessment, we think this supports the feasibility of much larger studies in this area.
The second finding, leading on from willingness to undertake a non-routine postoperative weight measurement, is that we have quantified for the first time, the weight change from the pre- to postoperative period in obstetric patients undergoing CS, with the mean decrease in weight being approximately 2 kg. This result is not overly surprising given the blood loss, equivalent to 0.46 kg, the average newborn birthweight of 3.4 kg, the average placental weight of 0.6 kg (not measured), 20 the amniotic fluid weight, equivalent to 0.3 kg (not measured) and the perioperative period fluid input of approximately 2.8 kg. However, it is somewhat surprising—and contrary to traditional clinical opinion—that there was no significant difference in postoperative bodyweight change between the planned CS group and the emergency CS group. The larger overall positive fluid balance in the emergency cases, in the absence of change in weight differences compared with the planned CS group, suggests that perhaps pre-surgery losses (in labour) are not sufficiently replaced – or that blood loss is underestimated. There is a need to better understand fluid shifts and weight changes in the perioperative period in obstetric patients, and how these may impact recovery from surgery.
The third key finding relates to haemoglobin and ferritin and the issue of missing measurements. Measurement of haemoglobin and ferritin at the first hospital appointment was not routine practice. This is surprising given the background prevalence of IDA in reproductive-aged women, and the opportunity at the first hospital appointment to diagnose and treat IDA. When ferritin was measured at the first hospital appointment, 30% of participants were iron deficient. This is very similar to previous data reporting a 33% rate of iron deficiency at first hospital appointment. 21 Despite obstetric patients undergoing major surgery associated with significant blood loss, less than half of the participants had their haemoglobin measured postoperatively. When they did, nearly nine out of ten participants had a haemoglobin of less than 130 g/l, and 41% had levels less than 110 g/l. This is also consistent with previous work from the same centre reporting less than 50% of patients having a haemoglobin measured, and if they did, over 90% had had postoperative levels less than 130 g/l, with 55% having levels less than 110 g/l. 21
The incidence of preoperative iron deficiency and IDA was unable to be determined in the preoperative period, as no participant had ferritin levels measured preoperatively. Ferritin was measured in 48% of the group in early third trimester (approximately 28 weeks). Traditionally, normal ferritin levels in early third trimester have been used to reassure maternity providers that ferritin levels at term are also within normal limits. However, without measuring ferritin preoperatively, this assumption cannot be made due to the ongoing utilisation and depletion of iron stores—often without replacement—in the last three months of pregnancy. 15 Based on the findings of the third trimester ferritin measurements, it could be assumed that ongoing iron replacement and measurement of ferritin into the preoperative period is prudent in a significant proportion of the population to maximise preoperative preparation and minimise the impact of IDA.
Looking at this issue from another vantage point, however, while it is likely that most anaemia in patients in the preoperative period is IDA, without measuring ferritin we cannot be sure. Other less prevalent causes of anaemia must be diagnosed accurately and treated accordingly, which further highlights the need for timely measurement of ferritin and follow-up with perioperative physicians where appropriate. Undiagnosed causes of anaemia in the preoperative period is an unrecognised problem. Assuming that all anaemia is IDA may lead to incorrect treatment of anaemia, and unpredictable responses to routine fluid and blood resuscitation; for example, in the case of anaemia being due to haemolysis or haemoglobinopathies and blood transfusion in patients who experience postpartum haemorrhage. There is a need to raise awareness of this issue and consider measuring ferritin as well as haemoglobin in the preoperative period so that appropriate individualised care of obstetric patients can be given.
The rate of postoperative iron deficiency and IDA was also unable to be determined, as no participant had ferritin levels measured postoperatively. This is of concern and indicates that the potentially widespread and serious problem of postoperative IDA in obstetric patients is under-recognised. Based on our data, using the lower 95% CI value for the proportion of participants discharged home with a haemoglobin less than 110 g/l, it is likely that at least 20% of obstetric patients at our institution are discharged home after CS with anaemia. The number of participants—and patients in general—being discharged home with iron deficiency is unquantifiable but given the nature of the surgery performed and the incidence of preoperative iron deficiency, it is expected to be significant. As perioperative physicians, we should be ensuring that postoperative obstetric patients have routine haemoglobin and ferritin levels measured and should begin immediate treatment—including with iron infusions—if required. There are, however, no official perioperative recommendations in this area.
Regarding QoR, the participants generally reported high scores suggesting good quality of recovery. The mean ObsQoR-10 score in our cohort was similar to values gained during the validation of this score. 19 While the global scores (calculated out of 100 across the 10 domains) were not significantly different across the two groups of participants in our study, it may have been useful to see if any of the specific areas within the questionnaire such as pain level and overall feeling of control demonstrated differences between the two groups. Pleasingly, the questionnaire tool was simple and efficient to use in the postoperative period, and all participants were able to complete this with the assistance of the investigator, regardless of their primary language.
The sample size for the future MAPOC study will be dependent on which timepoint(s) are examined to determine the relationship between haemoglobin (and possibly ferritin) concentration and outcomes (first hospital appointment, third trimester, preoperative, postoperative (on hospital discharge) [the measurement that is rarely taken]). Our data have, however, enabled an estimate that 550 participants are required to examine the association between postoperative anaemia and outcomes after CS. This relatively low number is due to the high prevalence of anaemia in our sample. This high prevalence is consistent with our other work in this area when the prevalence of postoperative anaemia was also high. 21 If associations of anaemia at other timepoints and outcomes are examined, the sample size increases to approximately 5900 participants if the immediate preoperative time is used. Depending on the exact future study question and the outcomes of interest, either of these sample sizes or any in between are likely to be feasible. At hospitals with 7000 births per year and a 30% CS rate, and a study that had eligibility criteria widened to include all women undergoing CS, a single centre could recruit the postoperative period number of 550 participants in approximately 6 months. A multicentre collaboration—as planned, with local, statewide, national or international participation, including low- and middle-income countries—may achieve the much larger sample size within a relatively short period.
Our study has provided important information about costs related to pathology from a single centre to help inform budgetary calculations for the future study and also for routine implementation in clinical practice. Taking a conservative approach and assuming the lower proportional limit of participants who have haemoglobin measured is 25%, then 75% of participants (or current hospital patients) will require an additional haemoglobin test. As ferritin was not measured in any postoperative patient, pathology costs will need to incorporate the need for 100% of study participants requiring this (or 100% of current hospital patients). With a study of 550 participants this equates to 413 tests to measure haemoglobin and 550 tests to measure ferritin.
The first limitation of this study is that it was a single-centre study, so may not represent the wider practice of diagnosis and management of IDA after CS. As this was a single-centre study it is important to note that the feasibility assessment only applies to our centre. It could be that patients from other institutions may have very different participation preferences or anaemia prevalence, therefore limiting feasibility at other centres. The second limitation is that we did not test the feasibility of undertaking a multicentre study. Ideally, feasibility would also test the ability to engage with multiple centres to undertake a future multicentre study. We hope, however, that the results of our study will encourage other centres to participate in a larger study with us. Furthermore, we did not define a priori thresholds for the categories for determining feasibility; that is, we did not define feasibility endpoints. We found what we considered very acceptable feasibility levels (more than 80% willingness to participate in all areas assessed) for a future study, based on expert opinion, but this can only be confirmed in a multicentre study across different sites. Thirdly, participant selection for recruitment was dependent on investigator availability. This may have led to a bias in time of the day and days that participants were recruited. We also chose a convenience sample of 30 participants in each group, and this has limited the power of this study. However, we were able to demonstrate that despite the small sample size of 60 participants, the lower limits of the 95% CI for the feasibility categories would likely be considered above an acceptable threshold for defining feasibility. The study did not recruit any First Nations peoples. When designing a large trial, better engagement with First Nations peoples through liaison officers and consumer advocates will be sought. We also did not collect data on the person’s sex recorded at birth (or other characteristics such as gender, variations of sex characteristics, sexual orientation, and cisgender and trans and gender diversity), which in the case of sex recorded at birth, may have an impact on the prevalence of IDA. Importantly, willingness does not necessarily translate into actual participation in a research study, especially after discharge from hospital. Modest dropout rates would need to be factored into larger studies and perhaps incentives used to encourage participation. Engaging with consumers to determine key outcomes of importance is key and would be undertaken in the definitive MAPOC study, and biostatisticians would be involved as part of the team to determine methodology and assist with definitive sample-size calculations and planned analysis. We did not ask the participants whether they would be interested in engaging with the research rather than being participants. Having consumers—in this case pregnant and recently pregnant women—working with the study team to prepare for research, conduct the research and use the research is becoming more important in all areas of clinical research and would be valuable. 22
The problem of perioperative IDA in obstetric patients is significant and likely under-recognised and under-treated. We have undertaken a study to determine the feasibility of conducting a larger study in this area and have demonstrated encouraging results suggesting that the feasibility of a larger study is favourable. We have also generated data which can be used to base budgetary calculations for pathology tests regarding the number of non-routine blood tests needed in the postoperative period to diagnose IDA (one ferritin measurement per participant, and two out of three participants requiring a full blood examination). We have highlighted the serious lack of data, and likely inadequate treatment of iron deficiency with and without anaemia in postoperative obstetric patients. While there are recommendations about the treatment of IDA in pregnancy, there is an urgent need for peak global bodies to update recommendations to include the diagnosis and treatment of iron deficiency, specifically in women after CS.23,24 If recommendations are not forthcoming, then conducting our proposed large multicentre trial to diagnose iron deficiency and IDA—and offer treatment and record outcomes in the short and long term—should be undertaken. A future study may enable analysis of participant characteristics on the perioperative outcomes measured, particularly parity, age, English as a primary language and educational background, to see whether there are any correlations between these variables and perioperative iron deficiency and anaemia outcomes. Outcomes in participants who decline treatment and remained iron deficient could be compared with outcomes in those people who had iron levels corrected to normal values. A study of this kind would definitively address this question and hopefully lead to global changes in how we can better care for obstetric patients after major surgery.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X241275127 - Supplemental material for A feasibility study of measuring maternal anaemia and postoperative outcomes after caesarean section
Supplemental material, sj-pdf-1-aic-10.1177_0310057X241275127 for A feasibility study of measuring maternal anaemia and postoperative outcomes after caesarean section by Mark O’Donnell and Safiya Virji, Hannah Hill, Elizabeth Potter, Claire Grimstvedt and Fiona McHugh in Anaesthesia and Intensive Care
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from the Australian and New Zealand College of Anaesthetists Academic Enhancement Grant 001/18. Professor Alicia Dennis is supported by the Fulbright Commission Australia as a 2023 Fulbright Scholar. Dr Mark O’Donnell was awarded the 2021 Kevin McCaul Prize from the Australian Society of Anaesthetists for this study protocol.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
