Abstract
Cognitive impairment and older age are major risk factors for postoperative delirium. Professional societies have advocated preoperative screening to identify at-risk individuals for implementation of interventions, which have moderate effectiveness in preventing delirium. However, it remains unclear from the guidelines whether screening should be completed routinely for all older individuals or targeted, and also which specific screening tool is preferred. In addition, the responsibility for screening remains undesignated in the guidelines provided. We reviewed the benefits and risks of routine screening. We also performed a cost-benefit analysis of routine screening (versus no screening). Furthermore, we summarised the sensitivities and specificities of commonly used screening tools and reviewed evolving screening tools that may have an increasing role in future practice. We concluded that routine screening is useful and appears to be cost-effective for reducing postoperative delirium, with a cost-benefit ratio of 2.89 (adjusted ratio of 2.34), and has additional advantages over other approaches such as targeted screening or routine intervention.
Introduction
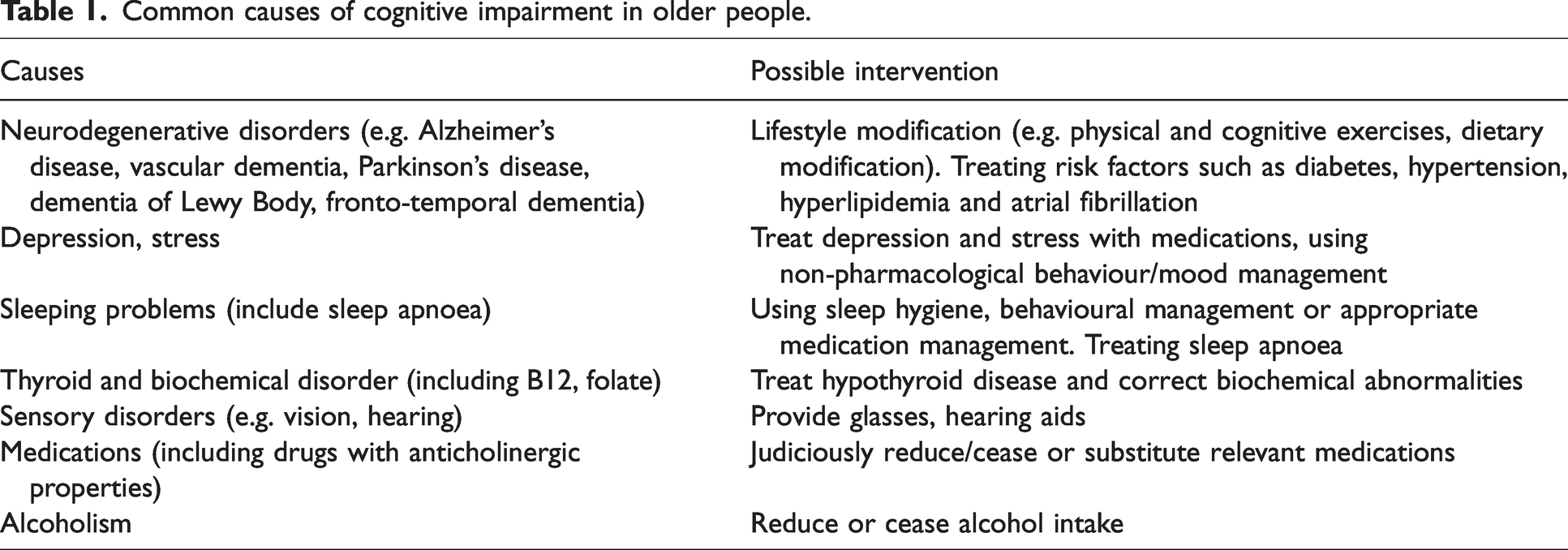
Older age and preoperative cognitive impairment are two major risk factors for postoperative delirium.1,2 Patients above the age of 70 undergoing major elective surgery and anaesthesia are at a significantly higher risk of postoperative delirium1,2 and the odds ratio doubles per 10-year increase in age. 3 A list of common causes of cognitive impairment in older people is presented in Table 1.
Common causes of cognitive impairment in older people.
Postoperative delirium is common in older patients undergoing major surgical procedures, with an incidence rate of about 24%. 4 Its occurrence is associated with poorer prognoses, longer hospital stays (an additional 5 to 7.7 days with delirium versus no delirium) and higher mortality (9% to 17% with delirium versus 1% with no delirium). 3 Multiple predisposing and precipitating risk factors have been identified that contribute to both cerebral metabolic insufficiency and neuro-inflammation, postulated to be the pathological pathways for the postoperative delirium state, however, the exact pathogenesis is still not fully understood. Currently, the most effective intervention proven to reduce the likelihood of delirium onset is based on multicomponent environmental and supportive measures.5 –9 The multicomponent interventions in an early randomised controlled trial study 10 targeted six risk factors: orientation and therapeutic activities for cognitive impairment, early mobilisation to avert immobilisation, nonpharmacologic approaches to minimise the use of psychoactive drugs, interventions to prevent sleep deprivation, communication methods and adaptive equipment (particularly eyeglasses and hearing aids) for vision and hearing impairment, and early intervention for volume depletion. 5 In later studies, staff education and nutrition were included and orientation was adjusted to frequent reorientation. 9 Upon analysing individual components, it was concluded that the following are crucial components and should be included as a minimum: staff education, individualised care (person‐centred care), reorientation at frequent intervals, and early mobilisation. 9
In the USA, guidelines from the American College of Surgery recommend preoperative cognitive assessments for older adults (70 years and older), although there is no widely accepted standard approach. 11 For instance, there are a number of tools for cognitive screening, ranging from quick assessments including the Clock Drawing Test or the Mini-Cog©, to the more comprehensive tests such as the Mini-Mental State Examination (MMSE) or Montreal Cognitive Assessment (MoCA), the latter of which takes longer (6 to 10 min) to complete. 12 Cognitive screening can facilitate the implementation of interventions during the pre-, intra-, and postoperative periods, which can help preserve cognition and independence after surgery in those at risk of delirium. 13
In the UK, guidelines regarding the perioperative care of older people and those with neurocognitive disorders were published by the Association of Anaesthetists of Great Britain and Ireland in 2014. 14 This was in response to a national enquiry into older patient postoperative outcomes that identified that only 36% received ‘good' care, which contained medical, medication, cognitive and functional assessments as well as a risk score. 15 In a subsequent survey of Scottish hospitals (November to December 2020), of the respondents from anaesthetics departments, only 48% specifically screened for cognitive impairment when preassessing patients aged over 75. A specialist geriatric preassessment service that routinely screens for cognitive impairment was uncommon, operating in only 20% of centres. 16
In Australia, ‘A framework for perioperative care in Australia and New Zealand’ was published in 2021. 17 It is an initiative of the Australian and New Zealand College of Anaesthetists in collaboration with representatives from multiple colleges and learned societies in Australia and New Zealand, including geriatrics; internal, rural and general medicine; as well as physicians, surgeons, pain specialists and intensivists. This document suggested that high procedural-risk surgical patients should be referred to a perioperative medical team for further risk assessment using appropriate tools to assess cognitive, functional and frailty scores. However, the document did not recommend specific tools or assign responsibility for facilitating such screening. 17
In comparing the above guidelines, they all recommend some type of cognitive assessment, in particular for at-risk individuals. However, all have steered away from being too restrictive in recommending specific screening tests or stipulating who should be responsible for screening.
The aims of this article are therefore to examine whether cognitive screening should be applied to all older patients undergoing elective surgery versus no screening, specifically analysing the benefits versus risks, as well as the cost-effectiveness. Additionally, we explore which type of screening tools are recommended.
Methods
Rather than a systematic review, this ‘point of view’ article aims to provide a balanced approach to the study’s questions, based on our assessment and interpretation of current relevant literature. Our recommendations are also based on this assessment and interpretation.
Current and future screening tools
Common brief screening tools
According to a recent systematic review and meta-analysis, the widely used screening tools – MMSE, Mini-Cog, and MoCA – have a combined sensitivity and specificity for the detection of major neurocognitive disorder of 0.81 (95% confidence interval (CI), 0.78 to 0.84) and 0.89 (95%CI, 0.87 to 0.91), respectively. 18 The approximate time required for carrying out the aforementioned screening tools range from 5 min or less to 10 min. 18 The time required, the sensitivity, specificity, domains tested and limitations of the commonly used brief screening tests are summarised in Table 2.
Comparisons of time required, domains tested and accuracy of some brief cognitive screening tools.
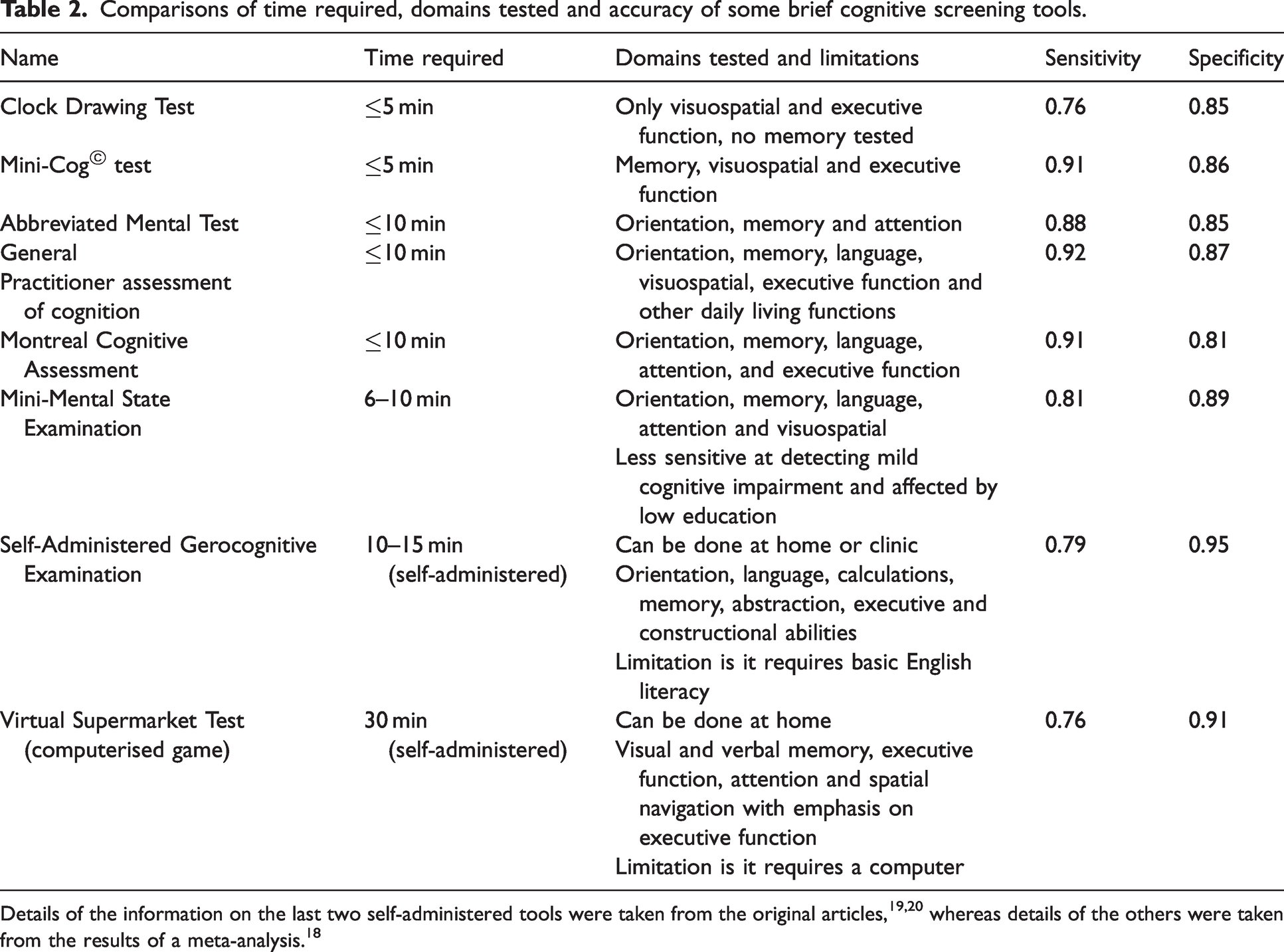
From the clinician’s perspective, an ideal tool for preoperative cognitive impairment screening would be one that is quick to perform and has high accuracy. For screening purposes, high sensitivity is vital even at the expense of specificity. The cause(s) of cognitive impairment may vary, and a positive screening result does not imply definitive diagnosis of neurodegenerative disease. Rather, the final diagnosis of the cause depends on clinical history, examination as well as other relevant investigations. Therefore, its specificity is less crucial as the test is not for diagnosis, but for screening of a number of causes of cognitive impairment.
Self-administered screening tools under development
In recent times, computerised cognitive assessments have been shown to be possible in older persons. For instance, a self-administered computerised game called Virtual Supermarket Test (VST) was used to detect mild cognitive impairment in a sample of persons with subjective memory complaints. The results were then compared to the MoCA and MMSE, against extensive neuropsychological batteries as reference. The results were promising: the VST had a sensitivity of 76.3% and a specificity of 91.4% versus the MoCA with sensitivity of 69.8% and specificity of 75%, and the MMSE with sensitivity of 63.6% and specificity of 66.7%. 19 However, the time required was 30 min and the sample sizes were small, with participant numbers in each group in the high forties.
There are other types of self-administered cognitive tests that do not require a computer. An example is the Self-Administered Gerocognitive Examination (SAGE) that only requires pen and paper to test different cognitive domains. 20 SAGE showed 79% sensitivity (compared with 71% for the MMSE) and 95% specificity (compared with 90% for the MMSE) in detecting those with cognitive impairment from normal subjects in a sample of 254 participants. The test can be completed in 10 to 15 min but does require a basic level of English literacy.
It is noteworthy that many of these self-administered screening tests have not been trialled in preoperative settings.
Benefits of routine screening
Benefit 1: Reducing occurrence of postoperative delirium
The incidence of postoperative delirium is high in older surgical patients. A 2021 systematic review, which included 19 studies with a total of 3533 postoperative older patients, demonstrated that the pooled incidence of postoperative delirium was 24%. 4 Preoperative cognitive impairment is strongly associated with postoperative delirium and highly prevalent in older patients, as illustrated in a recent large study of 1518 patients aged 70 years or more. 21 Postoperative delirium occurred in 30% of patients with baseline cognitive impairment compared with 11% of patients without it. There was also a sufficiently high prevalence of preoperative cognitive impairment to justify routine screening – 88% of the patients were screened for cognitive impairment before surgery, of whom 20% screened positive in this study. Furthermore, preoperative cognitive impairment had a higher incidence of a composite of postoperative complications (20% versus 12%) and a longer median length of stay in hospital (6 days versus 5 days). 21
Screening could facilitate the mitigation of these adverse outcomes through timely implementation of effective preventative measures against delirium. Most notable is the ‘Multicomponent Non-Pharmacologic Interventions for Prevention of Delirium,’ which is supported by a moderate level of evidence. 8
Recently, a Cochrane review 6 of interventions for preventing delirium in hospitalised non‐intensive care unit (ICU) patients concluded that they are probably effective in reducing the incidence of postoperative delirium by around 43% and may also reduce the duration of hospital stays by 1.3 days.
Benefit 2: Uncovering different causes of cognitive impairment
Although the most common cause of cognitive impairment is neurodegenerative disease (‘major neurocognitive disorder’ or in former terminology ‘dementia’), there are other significant causes including depression and certain medications.
Older persons may not volunteer a history of depression, and cognitive impairment may be the presenting symptom, often misdiagnosed as neurocognitive disorder or part of the ageing process. 22 Furthermore, depression may sometimes coexist with neurocognitive disorder. Unless deliberately screened for, the diagnosis may be missed. Treating depression successfully can improve the cognitive function of patients and reverse cognitive impairment.
Likewise, many medications can adversely affect cognitive function. Those commonly encountered are drugs with anticholinergic properties such as tricyclic antidepressants, antipsychotics, anticonvulsants, older antihistamines and dopaminergic drugs. Additionally, drugs used for sedation such as long-acting benzodiazepines and opioid analgesics can cause reversible cognitive impairment in older people. 23 Older people with underlying neurodegenerative diseases can also present with further decline in cognitive function after such drugs are used. Judiciously ceasing or reducing these medications can improve cognitive function. Therefore, preoperative cognitive screening can detect these potentially reversible, modifiable causes of cognitive impairment, which could subsequently be addressed to optimise the cognitive function of patients preoperatively.
Risks of routine screening
Risk 1: Cost
As resources are often limited in the healthcare sector, it is important to consider whether the cost of routine screening would be justified. Another consideration, if the healthcare sector is a fee-for-service model, is who should bear the cost.
To address this concern, a cost-benefit analysis of routine screening is demonstrated below. To our knowledge, this type of analysis has not been published before.
Estimated cost of routine screening in preoperative clinics
The approximate time required for carrying out one of the widely used dementia screening tools (MMSE) is about 10 min. 18 Hence, screening 100 older patients would equate to about 1000 min or 16.7 h. The average hourly salary of a perioperative nurse is about AUD$40/h. Therefore, the time taken to screen 100 older patients would incur a cost of about AUD$670.
A recent large study of patients aged 70 years or more demonstrated the prevalence of preoperative cognitive impairment is about 20%. 21 Assuming that all of these individuals proceed to have a non-contrast computed tomography head scan (cost in Australia about AUD$340) 24 as part of their workup, the total cost of imaging the 20 patients out of 100 who screened positive would be about AUD$6800 (20 × AUD$340).
Assuming that blood tests are routinely completed as part of the preoperative assessment for older people undergoing major surgery, and other cognitive impairment blood test screens such as B12, folate and thyroid function levels have minimal additional costs, the total cost for routine screening per 100 older persons would equate to about AUD$670 + AUD$6800 = AUD$7460.
Estimated cost-saving to the healthcare system of preventing delirium in hospital
Recently, a Cochrane review of interventions for preventing delirium in hospitalised non‐ICU patients concluded they may reduce the duration of hospital stay by 1.3 days. 6
Despite the effectiveness of implementing delirium prevention programmes and the reduction of postoperative delirium by 43%, it is noteworthy that the incidence of delirium was still about 10.5% in the intervention group, compared with 18.4% in the control group. 6
Therefore, out of those 100 older patients screened, delirium would be prevented in eight patients (18.4% minus 10.5%), which would reduce the total duration of hospital stay by 1.3 days × 8 = 10.4 days. The national average cost per admitted acute overnight stay is about AUD$2,074 for hospital-acquired delirium. 25 Therefore, this potential reduction in costs for these eight patients would be about AUD$2074 × 10.4 = AUD$21,570. This would represent a cost-benefit ratio of about AUD$21570 ÷AUD $7470 =2.89. However, this assumes a sensitivity of 100%. As the mean sensitivity of detecting delirium with screening tools has been estimated at 0.81, 18 the adjusted cost-benefit ratio would be about 2.89 × 0.81 = 2.34. That is, for every dollar spent in preoperative screening, AUD$2.34 could be saved by the prevention of delirium.
Caveat for the above calculation
The above calculation assumes that the intervention programme has minimal or no additional costs and can be embedded as part of the routine ward-based care of older patients. This raises the question that if the cost of intervention is inexpensive, why not implement such intervention for all older patients undergoing major surgeries and avoid screening altogether. This proposal would seem reasonable. However, screening confers the additional advantage of uncovering at-risk patients, which may prompt an increased level of attention to their care rather than an unfocused approach that could dilute such attention required by the at-risk individuals. In addition, it could also uncover and address reversible causes of cognitive impairment as described previously. Furthermore, although logical to provide the standard delirium prevention intervention care to all older people, it is not yet widely implemented in the real world.9,26
Risk 2: Accuracy of commonly used screening tools
Another concern around routine screening is the accuracy of currently available tools. According to a recent meta-analysis, the sensitivity of screening tools for the diagnosis of dementia is about 80% and specificity 90%. 18 Assuming that the sensitivity and specificity are equivalent for cognitive impairment, and the prevalence of cognitive impairment is 20% in older patients, 21 the positive predictive value (PPV) would be 67% with a false positive rate of 33% (see Table 3). This finding would discourage routine screening. However, the PPV could be higher in the selective screening of high-risk individuals where the prevalence of cognitive impairment is higher, as PPV increases with a higher prevalence.
Calculation of predictive values based on typical estimated sensitivities and specificities.
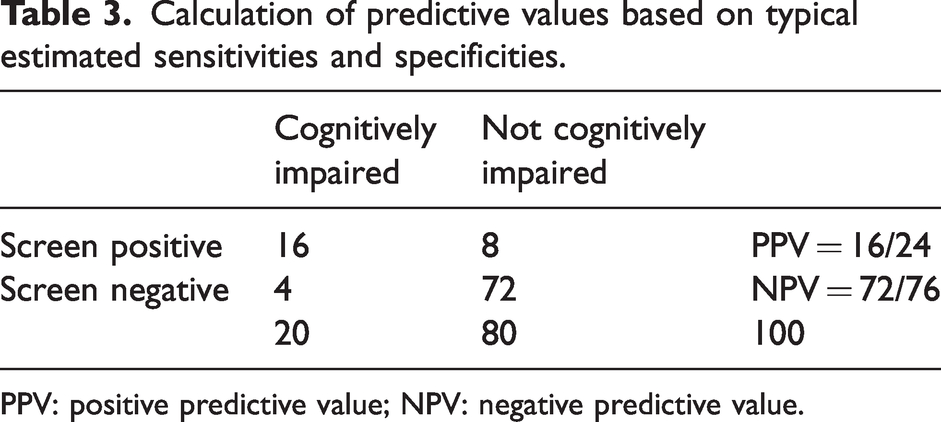
PPV: positive predictive value; NPV: negative predictive value.
Risk 3: Potential harm from inappropriate application of screening tools
The lack of high accuracy of current ‘screening’ tools means that it is inappropriate to use them as ‘diagnostic’ tools. Based on the previous calculation of PPV, approximately 33% of persons would be wrongly ‘diagnosed’ as having cognitive impairment. The current clinical method to diagnosing cognitive impairment is a holistic approach that integrates history, cognitive screening tool results and investigations targeting any reversible cause(s) as well as brain imaging. Detailed neuropsychological tests are reserved for difficult cases. If false positive screening results are incorrectly communicated as a diagnosis to screened older persons, it could cause distress, and some may even need counselling. To counter this, we have made suggestions below under ‘Recommendation 3: Addressing concerns about uncertain diagnosis’.
Whether to do routine cognitive screening: our recommendations
Postoperative delirium is associated with poorer patient outcomes and has a high incidence in older surgical patients, which may rise given the increasing number of older patients undergoing surgery. Concurrently, cognitive impairment is a strong risk factor for postoperative delirium and has a high prevalence in older persons. Thus, as there are several effective measures for reducing postoperative delirium, preoperative screening will be advantageous. Moreover, even if the patients have other reversible or modifiable causes of cognitive impairment, such as depression or medication use rather than neurodegenerative diseases, appropriate treatments could lead to improved outcomes for these patients as well.
Therefore, cognitive screening, at a minimum, should be completed in higher-risk preoperative older patients. However, whether it should be carried out routinely is less clear, given the concerns about screening accuracy, and whether it would be more cost-effective to skip screening altogether and implement a standardised intervention for every older patient postoperatively. In our opinion, a practical approach would be to choose a screening tool that is brief and useful for the older age group.
Recommendation 1: Selection of brief screening tool(s)
In our opinion, as in older people (aged 70 or more), the prevalence of neurodegenerative disease is higher and they often have multiple comorbidities and risk factors for developing delirium, using a more comprehensive method like the MMSE or MoCA would be preferred as more cases could be detected (time: 6 to 10 min, sensitivity 0.81, and time: 10 min or less, sensitivity 0.91, respectively 18 ). We additionally believe that a screening tool that assesses the domain of attention, for example, ‘serial sevens’ or spelling ‘world’ backwards, would be advantageous, as impaired attention is a required diagnostic criterion for delirium (MMSE, Abbreviated Mental Test, and MoCA have this advantage). Reassessing a patient’s attention in the postoperative period may help clinicians identify delirium. Any deterioration from baseline may indicate the development of delirium and allow for the patient’s progress to be carefully monitored.
The above strategy would improve the value and yield of preoperative cognitive screening. Another feasible strategy concerning efficiency would be to utilise self-administered tools such as VST or SAGE. Novel tests are regularly being developed that could prove plausible time-saving options. However, further evaluation in the preoperative setting is required before such tests could be adopted routinely in preoperative screening for cognitive impairment.
Recommendation 2: Delegation of screening duty
Regarding whom should be responsible for performing the cognitive screening of at-risk patients, in our opinion, possible options include the anaesthetist or nurse in the preoperative anaesthetic clinic who could carry out the cognitive screening as part of their risk assessment. Alternatively, as there are disparities in the resources available in different hospitals and settings, cognitive screening in surgical consultation clinics remains an option. We recommend that healthcare facilities should specify the clinician responsible for screening to avoid miscommunication, which could result in screening not being done. The role could be allocated based on the available resources and expertise in each facility.
Recommendation 3: Addressing concerns about uncertain diagnosis
Despite good specificity, none of the screening tests are infallible. False positive screening errors can be navigated effectively if patients with a dubious positive result are subsequently referred to specialists such as geriatricians for confirmation perioperatively. Therefore, reversible, modifiable or treatable cognitive impairment could also be delineated and managed with this strategy. Even if a definitive diagnosis cannot be formed, patients could be advised to consult their general practitioner in the future if they remain concerned about the possibility of having underlying neurocognitive disorder.
Perioperative medicine could be enriched by a multidisciplinary approach, as older patients often require the involvement of multiple specialties postoperatively (for instance, if delirium develops). Seeking the input of the geriatrics team promptly, or preoperatively for older patients at risk of delirium or complications, may facilitate early discharge planning and help the management of possible postoperative complications.
Summary
We have argued that routine screening for cognitive impairment in older patients undergoing major elective surgery and anaesthesia is useful and appears to be cost-effective for reducing the occurrence of postoperative delirium. Moreover, we feel that it has additional advantages over other approaches such as targeted screening or routine intervention. The value of preoperative screening extends from reducing the occurrence of postoperative delirium to detecting patients with reversible cognitive impairment. Current guidelines do not specify who should bear the responsibility for screening. In our opinion, hospitals should delegate a specified clinician to avoid miscommunication, although the type of clinician may vary according to the setting, available resources and expertise. Several suitable screening tools exist, but a time-efficient approach could be to use Mini-Cog or the Clock Drawing Test for the younger old population with a lower prevalence of impairment (aged 60 to 70 years) and to use a more comprehensive tool that incorporates assessment of the attention domain in the older age groups (aged 70 years or more) with a higher prevalence of impairment. Newer initiatives such as self-administered cognitive tests have been developed and may aid in reducing the time and cost associated with administering these tests. We hope our emphasis on screening will help optimise the perioperative management of older patients undergoing elective surgery and anaesthesia.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
