Abstract
Severely ill patients with COVID-19 are challenging to sedate and often require high-dose sedation and analgesic regimens. Ketamine can be an effective adjunct to facilitate sedation of critically ill patients but its effects on sedation level and inflammation in COVID-19 patients have not been studied. This retrospective, observational cohort study evaluated the effect of ketamine infusions on inflammatory biomarkers and clinical outcomes in mechanically ventilated patients with SARS-CoV-2 infection. A total of 186 patients were identified (47 received ketamine, 139 did not). Patients who received ketamine were significantly younger than those who did not (mean (standard deviation) 59.2 (14.2) years versus 66.3 (14.4) years; P = 0.004), but there was no statistically significant difference in body mass index (P = 0.25) or sex distribution (P = 0.91) between groups. Mechanically ventilated patients who received ketamine infusions had a statistically significant reduction in Richmond Agitation-Sedation Scale score (–3.0 versus –2.0, P < 0.001). Regarding inflammatory biomarkers, ketamine was associated with a reduction in ferritin (P = 0.02) and lactate (P = 0.01), but no such association was observed for C-reactive protein (P = 0.27), lactate dehydrogenase (P = 0.64) or interleukin-6 (P = 0.87). No significant association was observed between ketamine administration and mortality (odds ratio 0.971; 95% confidence interval 0.501 to 1.882; P = 0.93). Ketamine infusion was associated with improved sedation depth in mechanically ventilated COVID-19 patients and provided a modest anti-inflammatory benefit but did not confer benefit with respect to mortality or intensive care unit length of stay.
Introduction
Severe disease including fever and pneumonia has been reported in up to 20% of patients diagnosed with the novel coronavirus (SARS-CoV-2), 1 with some early reports of intensive care unit (ICU) admission being required in 9–21% of patients.2,3 A surge in the body’s inflammatory response accompanied by a cytokine surge or ‘storm’ has been linked to multi-organ dysfunction and death from COVID-19, the disease produced by SARS-CoV-2, and is often present in ICU patients.1,4 High sedation and analgesic requirements have been documented in these patients and have been reported to be associated with younger age, higher body mass index, greater degrees of hypoxia, and elevated inflammatory markers. 5 Maintenance of an adequate level of sedation and analgesia is critical in mechanically ventilated patients with COVID-19 to adequately oxygenate and perfuse vital organs.6,7 Ketamine is an appealing drug for this purpose because it lacks respiratory depression at routine clinical doses, 8 provides potent analgesia at subanaesthetic doses 9 and may reduce inflammation, in some studies.10 –12 Although several preclinical laboratory investigations demonstrated ketamine’s role in regulating inflammatory homeostasis in animals in stressful conditions, human data on critically ill COVID-19 patients are lacking.10,13 It was also once thought that ketamine’s clinical activity was solely from its interactions with N-methyl-D-aspartate receptors, but it is now known that ketamine has complex interactions with several other receptors that provide opportunity for new clinical targets, including treatment-resistant depression, descending modulation, and mixed models of pain.10,13 –15 Subanaesthetic infusions of (R,S)-ketamine, notated as ‘ketamine’ throughout this article, have been used successfully for challenging mechanically ventilated patients for almost 10 years at our institution and were used throughout the COVID-19 pandemic in critically ill patients. Ketamine infusions may help provide deeper sedation and spare the use of high-dose opioids and paralytic agents.
The purpose of this study was to examine the effect of ketamine infusions on sedation level, inflammatory markers and clinical outcomes in mechanically ventilated COVID-19 patients, compared with a similar patient cohort that did not receive ketamine. We hypothesised that ketamine would provide a deeper level of sedation, decrease inflammatory markers and improve patient-centred outcomes such as mortality and ICU length of stay in a critically ill patient population with SARS-CoV-2.
Materials and methods
This was a retrospective, observational cohort study of mechanically ventilated patients with a confirmed positive SARS-CoV-2 infection at one of five ICUs in the Jefferson Enterprise, which is a group of more than 10 hospitals located in and around Philadelphia, PA, USA. The Institutional Review Board of Thomas Jefferson University determined that the study was exempt from review on 29 April 2020 (Control #20E.432). The electronic medical record (EMR; Epic) was queried using the date range 1 April 2020 to 30 September 2020 for patients who were mechanically ventilated and had a positive laboratory test for SARS-CoV-2 during the admission. Patients were excluded when missing or incorrect information was identified in the EMR. These discrepancies were typically related to discharge dates, incomplete follow-up data following transfer to other facilities and incomplete weaning mechanical ventilation data. The study population consisted of two study groups: one group (Group K) received a ketamine infusion and the second group (Group C) did not receive ketamine. Ketamine infusions were administered at the discretion of the physicians in each ICU and the dosing was determined by the treating physicians. Enterprise general guidelines of 0.5 mg/kg per h as the starting rate with a one-time increase to 1 mg/kg per h if sedation remained inadequate were created and made available to hospitals in the study but these guidelines were not strictly enforced. Ketamine was added to the analgesic regimen when ventilator asynchrony or inadequate sedation were present despite high doses of opioids, benzodiazepines or other sedatives. Typically, when several sedatives were unable to achieve adequate sedation despite high doses, a ketamine infusion was initiated. In addition, intravenous sedation was titrated based on Richmond Agitation–Sedation Scale (RASS) values with a target of –2 to –3 for most patients.
Data extracted from Epic (Verona, WI, USA) included: demographics; dates of admission; discharge disposition; medications given during admission; RASS scores; Acute Physiology and Chronic Health Evaluation II (APACHE II) scores; lactate dehydrogenase (LDH); lactate; ferritin; interleukin-6 (IL-6); and C-reactive protein (CRP). Serum biomarkers were routinely drawn per enterprise guidelines for all COVID-19 patients upon admission and then continued during hospitalisation for the purpose of trending inflammatory status and response to therapies.
Statistical methods
EMRs with demographic and clinical variables were provided in chronological order according to date of admission and patients’ medical record number. Tables were generated with descriptive statistics according to treatment group. Data are presented as means (standard deviation (SD)), medians with interquartile ranges (IQR), modes, frequencies and percentages. RASS median scores were calculated.
Univariate statistics (analysis of variance (ANOVA) and chi-square tests) were used to compare relationships between outcome variables and blood analytes, and for non-normally distributed laboratory data, the Mann–Whitney U test was used to determine between-group effects. Binary logistic regression models were constructed to examine associations between treatment and hospital length of stay, hospital mortality and other outcome variables. Hospital survival time was also examined using Kaplan–Meier survival analysis and Cox proportional hazard regression analysis using time of hospital admission until death or discharge (censoring). Univariate Cox regressions were performed for each covariate with a P-value <0.20 used for retention of variables in the multivariate model. Laboratory test results obtained during the patient’s hospital stay were examined using a one-way ANOVA to assess between group differences.
Ketamine dosing data were obtained from the Epic EMR. Complete drug administration records were provided for 41 patients who received ketamine sedation while mechanically ventilated in the ICU. Daily ketamine administration was calculated, and descriptive data figures are provided for total dose (mg) and duration (days) of drug administration. Pearson correlation and regression analysis were used to examine relationships between total ketamine dose and days of drug administration, and total ketamine dose versus age.
With respect to biomarker data, patient characteristics were summarised by counts with percentages or medians with IQR. Group differences in laboratory biomarker results were modelled and tested using generalised estimating equations to account for correlations among repeated assessments on the same patient. To evaluate the temporal relationships of ferritin and lactate between the two groups, we used daily ferritin and lactate concentrations according to date and time of blood draws. These data were analysed using Pearson correlation and regression analysis. The P-value was set at <0.05 for statistical significance. SAS version 9.4, SPSS version 28.0 and GraphPad Prism version 9.0 were used for statistical analysis.
Results
A total of 186 patients were identified from the Epic query: 47 who received ketamine and 139 patients who did not (Table 1). Group K patients were significantly younger than those in Group C (mean (SD) 59.184 (14.196) vs 66.292 (14.443) years, P = 0.004). There were no statistically significant differences in body mass index (BMI) or sex distribution between the groups. APACHE II scores were not significantly different between Groups K and C, respectively (30.00 (IQR 14.00) vs 27.00 (IQR 15.00), P = 0.293). Study patients’ characteristics are further detailed according to treatment, comparing those administered ketamine versus no ketamine in Figure 1. The median RASS scores were significantly lower for the Group K patients versus Group C patients (–3 (IQR 3.00) versus –2 (IQR 2.00), P < 0.001) (Figure 2). The modes for the two groups were –3 (Group K) and –2 (Group C). The Confusion Assessment Method for the ICU assessments were not significantly (P = 0.905) different among the two groups.
Demographic and sedation data for mechanically ventilated patients with SARS-CoV-2 who did and did not receive ketamine.
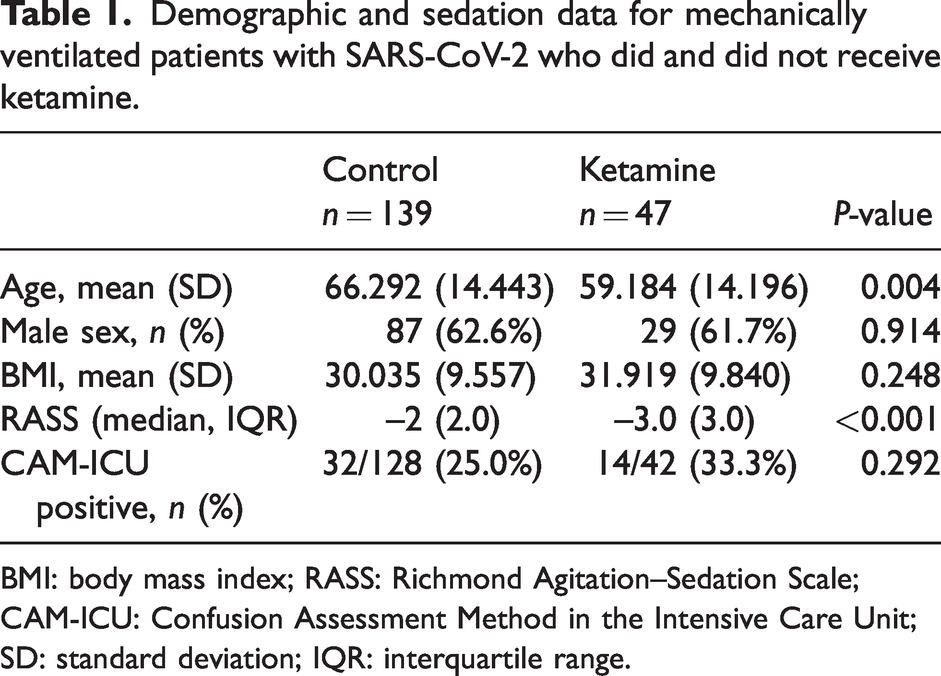
BMI: body mass index; RASS: Richmond Agitation–Sedation Scale; CAM-ICU: Confusion Assessment Method in the Intensive Care Unit; SD: standard deviation; IQR: interquartile range.
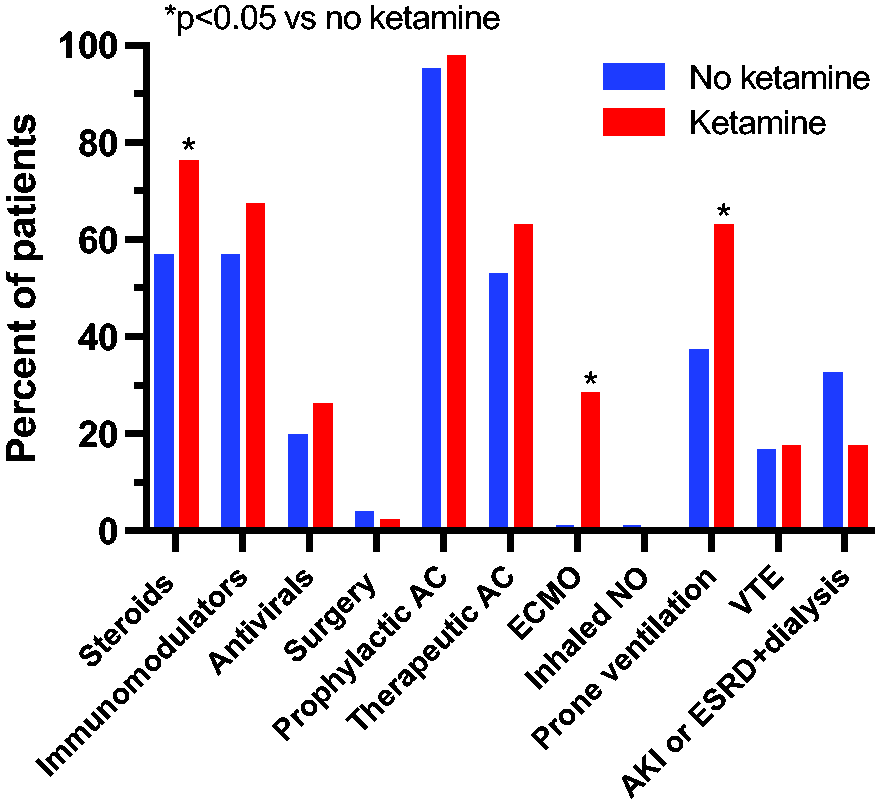
Study patient characteristics by ketamine status.
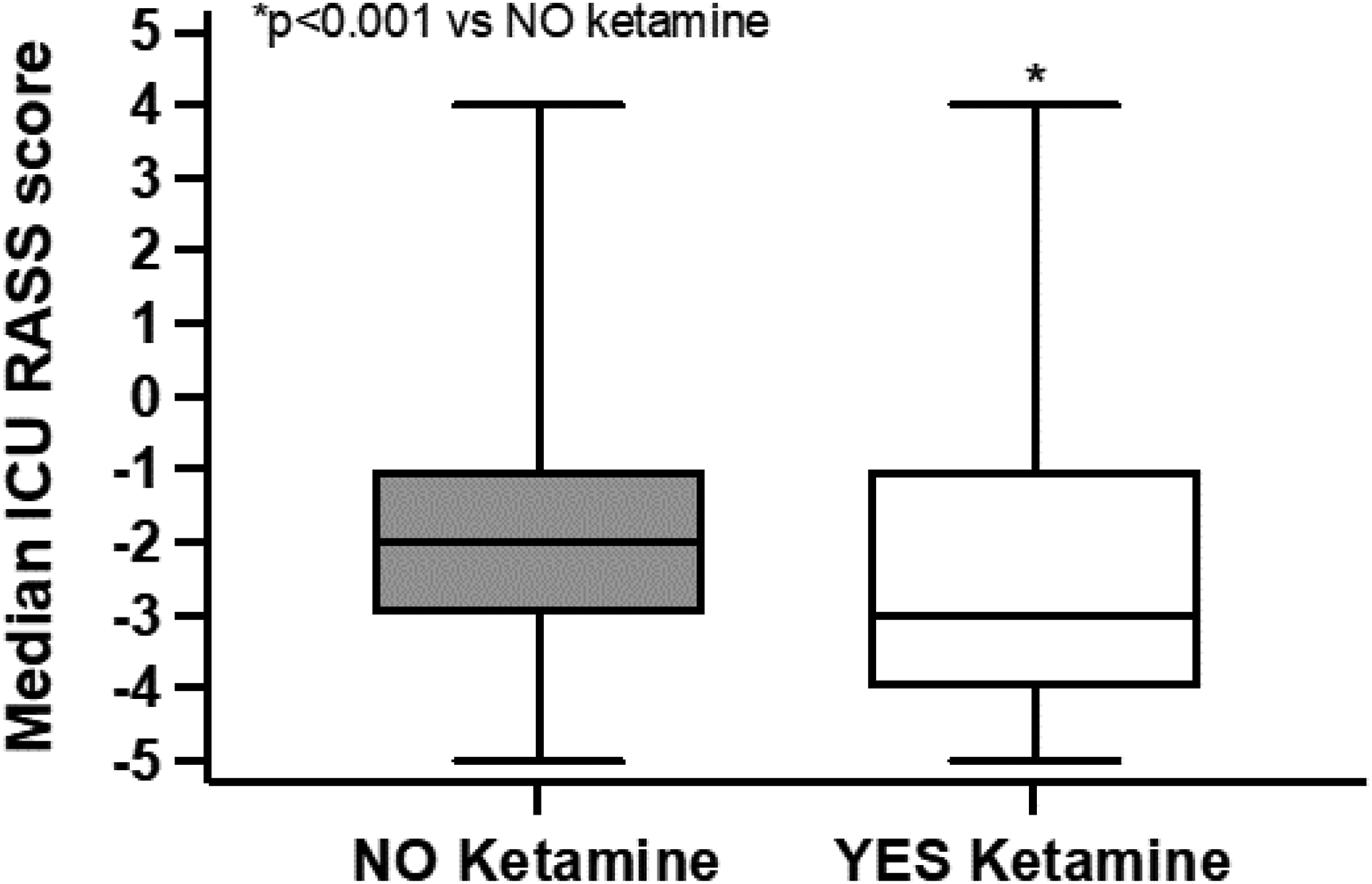
Median (interquartile range) Richmond Agitation–Sedation Scale (RASS) values.
Ketamine dosing
Total ketamine dose versus total days of drug administration is shown in Figure 3. The correlation R = 0.964 is statistically significant (P < 0.0001). Figure 4 shows a poor but significant negative correlation between total ketamine dose and age (R = –0.386, P = 0.013).
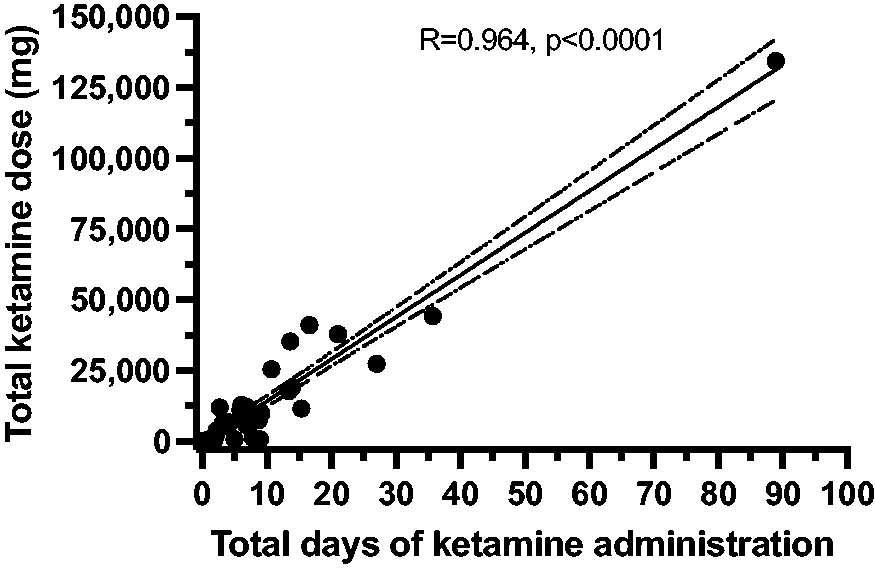
Illustration of total ketamine dose plotted against total days of drug administration. The correlation R = 0.964 is statistically significant.
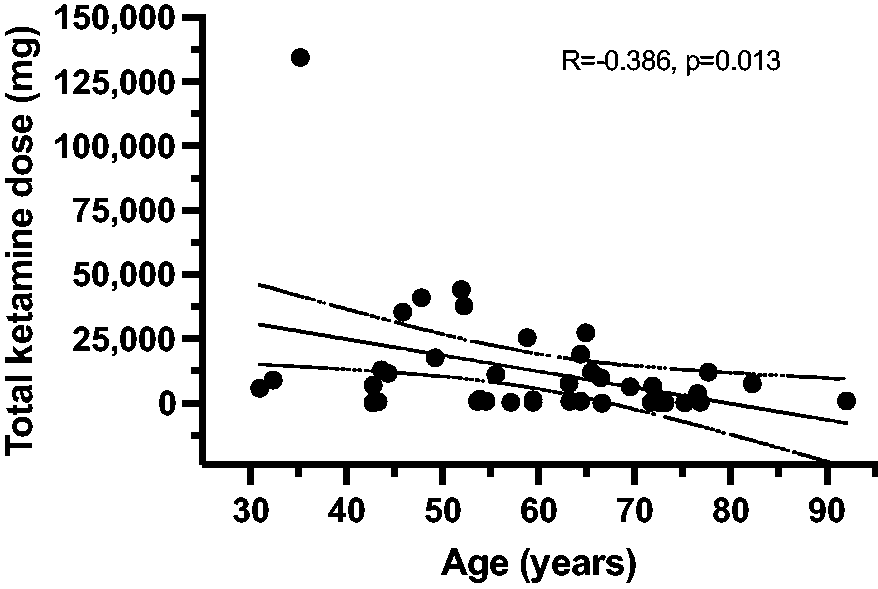
The figure shows a significant negative correlation between total ketamine dose and age (R = –0.38).
Hospital mortality
The overall hospital mortality for the study cohort was 51.60% and mortality according to treatment was 51.10% vs 51.80% for the K and C Groups, respectively (odds ratio (OR) 0.971; 95% confidence interval (CI) 0.501 to 1.882; P = 0.931). The log-rank Mantel–Cox test revealed no statistically significant difference in the survival distributions between Group K and Group C patients, respectively, with median survival times of 29 days (95% CI 19.234 to 38.766) and 19 days (95% CI 10.723 to 27.277); P = 0.297. The hazard function obtained from the Cox regression was 0.786 (95% CI 0.495 to 1.249; P = 0.308). Binary logistic regression showed overall mortality to be significantly associated with older age (OR 1.049; 95% CI 1.025 to 1.073, P < 0.001); however, there was no significant effect (P = 0.365) of ketamine treatment on hospital mortality after controlling for age. There was no statistically significant association between sex and mortality (P = 0.571) and ketamine treatment did not affect this (P = 0.927). There was no significant (P = 0.977) effect of BMI on mortality based on treatment received.
Length of stay and mechanical ventilation
Median hospital length of stay was 20 (IQR 24) days in Group K versus 12 (IQR 16) days in Group C (P = 0.033). The patients’ median ICU length of stay was 14.84 (IQR 19.95) days in Group K versus 6.47 (IQR 11.57) days in Group C (P < 0.001). Similarly, the time on mechanical ventilation was significantly greater for Group K patients compared with Group C patients, respectively (13.66 (IQR 20.28) days vs 4.22 (IQR 9.66) days; P < 0.001).
Biomarker analysis
With regard to biomarker analysis, in total, 175 patients had complete data and are described in Tables 2 and 3. There were 130 patients in Group C and 45 patients in Group K. Since the groups were imbalanced for race, age, steroid administration and immunomodulator administration, our biomarker models were adjusted for these variables. The immunomodulator group includes the following agents: tocilizumab, baricitinib, convalescent plasma; they are listed in approved immunomodulators on the Jefferson Health COVID-19 Inpatient Treatment guidelines.
Patient characteristics of the subset of patients with inflammatory data.
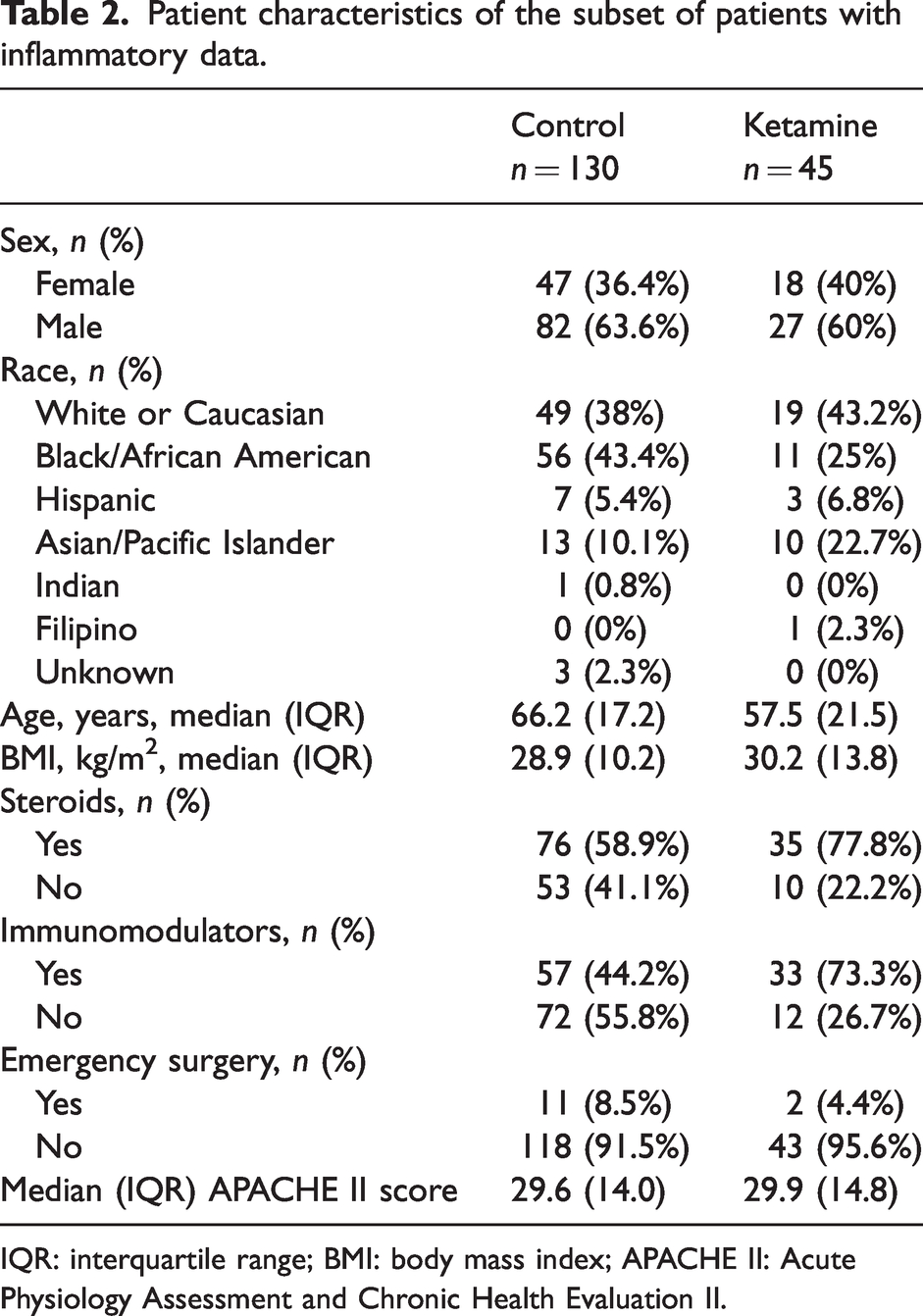
IQR: interquartile range; BMI: body mass index; APACHE II: Acute Physiology Assessment and Chronic Health Evaluation II.
Generalised estimating equation modelling results for group differences in biomarkers among mechanically ventilated patients with SARS-CoV-2 who did and did not receive ketamine.
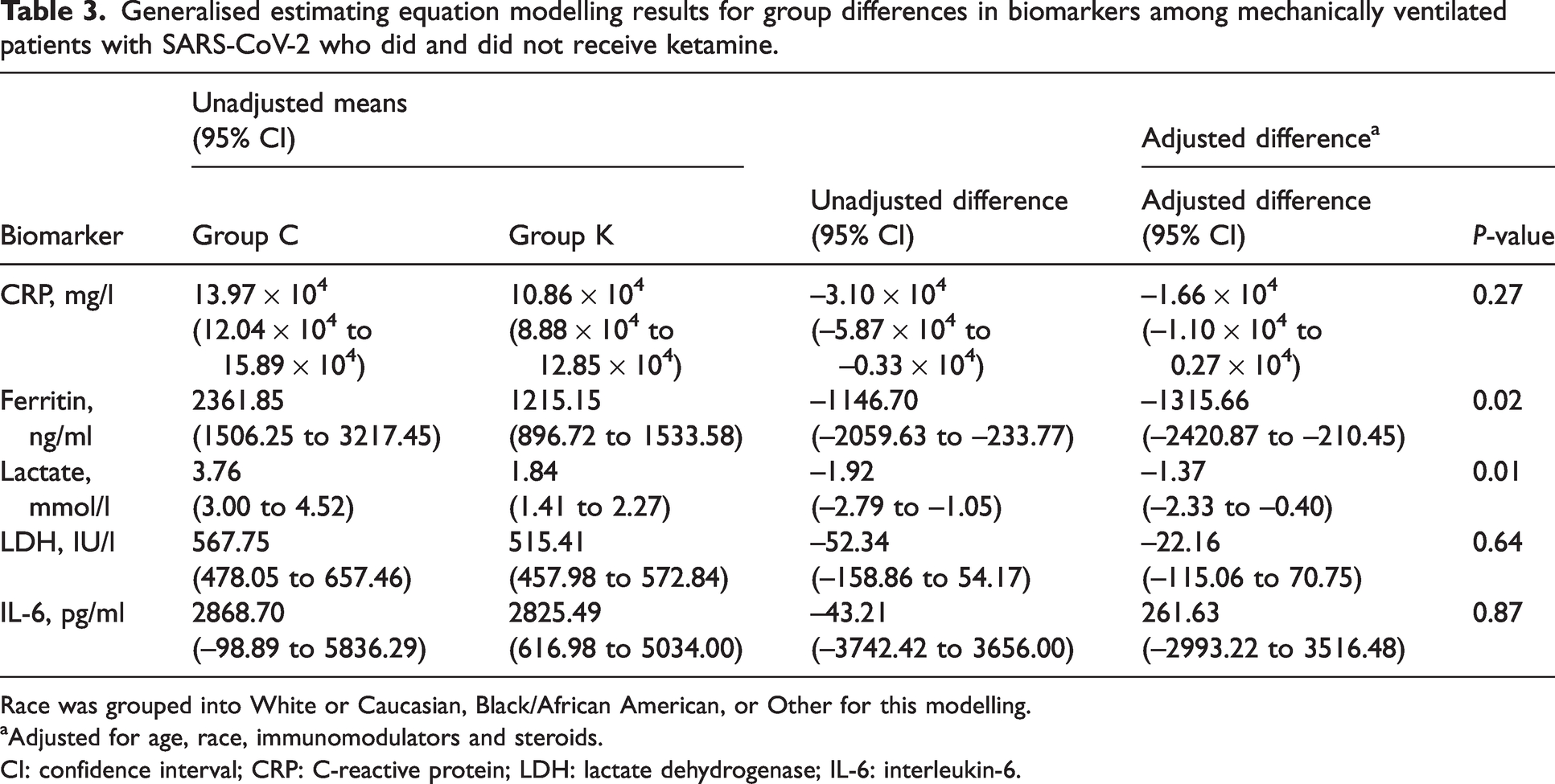
Race was grouped into White or Caucasian, Black/African American, or Other for this modelling.
Adjusted for age, race, immunomodulators and steroids.
CI: confidence interval; CRP: C-reactive protein; LDH: lactate dehydrogenase; IL-6: interleukin-6.
Table 3 shows modelling results on unadjusted means and differences, as well as adjusted differences for each biomarker. Exposure to ketamine infusion was associated with having statistically significantly lower adjusted group means for ferritin (–1315.66 ng/ml; 95% CI –2420.87 to –210.45, P = 0.02) and lactate (–1.37 mmol/l; 95% CI –2.33 to –0.40, P = 0.01). Figure 5 shows the temporal changes of ferritin and lactate for the control and ketamine groups.
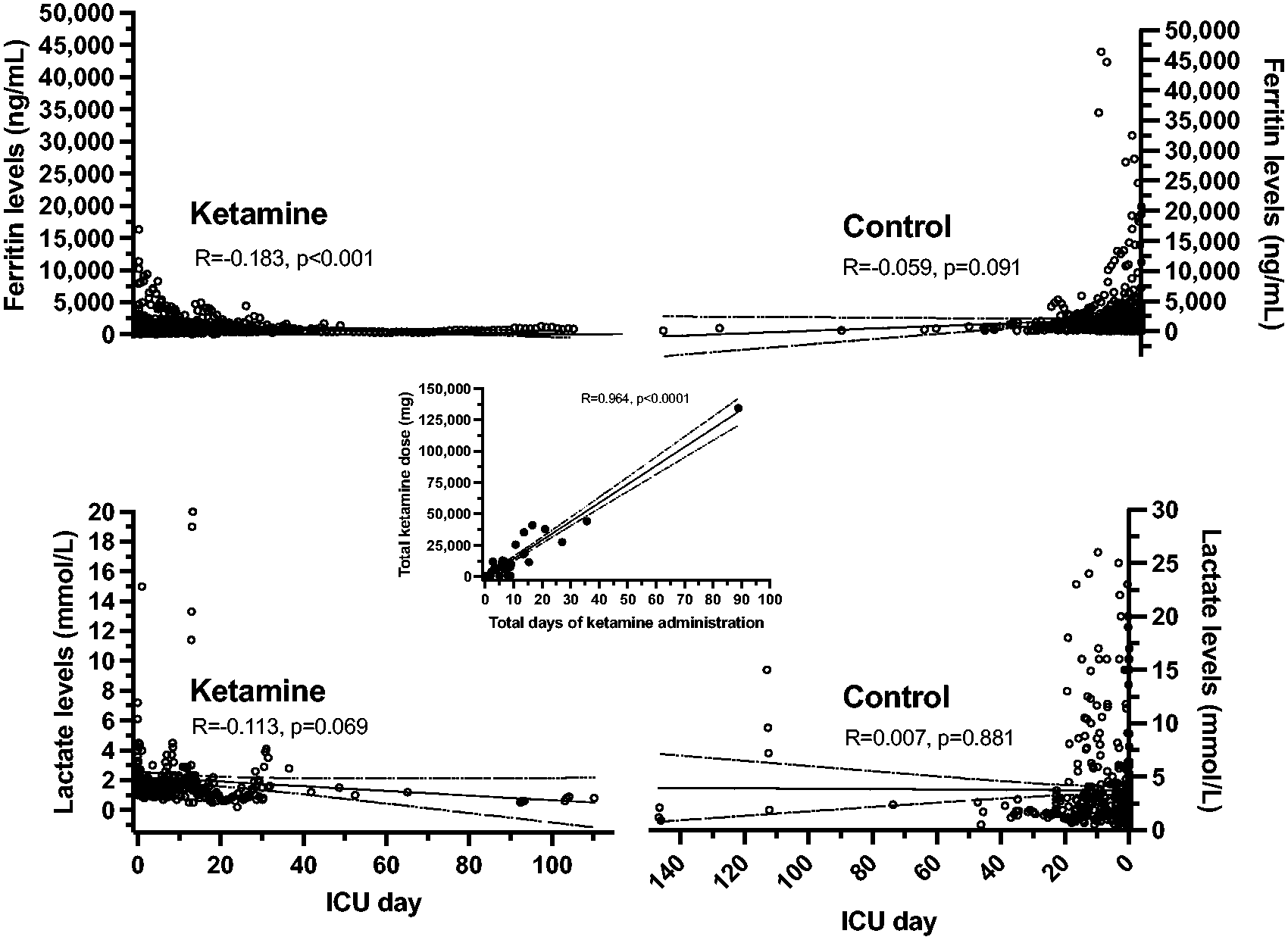
Illustration of the temporal relationships for ferritin and lactate concentrations for the ketamine (left figures) and control (right figures) groups. A low but significant correlation was observed for ferritin levels plotted for each intensive care unit (ICU) day measurement while on a ketamine infusion. The centre figure shows the high correlation of total ketamine dose administered plotted against the patients’ total days of drug infusion while on mechanical ventilation in the ICU.
Discussion
The principal finding of this study was that patients receiving ketamine infusions experienced statistically significant reductions in RASS scores, suggesting that ketamine allowed for deeper sedation in a challenging population of mechanically ventilated patients. Moreover, ketamine infusions were also associated with a reduction in two inflammatory markers: ferritin and lactate. There was insufficient evidence for similar associations with CRP, LDH and IL-6. Several known inflammatory biomarkers typify the inflammatory surge in COVID-19.4,16 Mahat et al reviewed 83 studies and determined the following biomarker elevations to be related to severity and mortality: CRP, ferritin, IL-6, and neutrophil to lymphocyte ratio (NLR). In our study, we did not evaluate NLR. 16 Ketamine was not associated with a reduction in mortality or ICU length of stay.
The lack of association with reduced mortality is not surprising since ketamine in most cases was added to the sedation and analgesia regimen after other treatments were deemed inadequate and the disease burden was high. Evidence has suggested that once severe disease and the ‘cytokine storm’ occur in patients with COVID-19, multi-organ failure and death often materialise. 17 It is likely in the case of the patients in this study that despite possible anti-inflammatory effects, ketamine was added too late to the treatment regimen for any possible outcome benefit to be observed.
In our study, we utilised the RASS for ongoing assessments of depth of sedation since RASS was our institution’s standard method of sedation assessment. Processed electroencephalography monitoring was not routinely used and therefore those values were not available for our study. Patients receiving ketamine demonstrated significantly lower RASS scores, indicating a more deeply sedated state. Given analgesic properties of ketamine, the agent may function well as an adjunct in an analgo-sedation protocol, serve in an opioid- or benzodiazepine-sparing fashion, or deepen the sedation to reduce ventilator–patient dyssynchrony (VPD). 18 At our institution, ketamine is often added to the sedative regimen to reduce VPD and prevent ventilator-induced lung injury. Since ventilator asynchrony often challenges severe adult respiratory distress syndrome (ARDS) and patients with poor lung compliance, deeper levels of sedation and adjuvant neuromuscular blockade are often required.6,19,20 Similar reports observed that younger COVID-19 patients displayed higher disease burden and elevated inflammatory markers that required high sedation dosages. 5 Therefore, it is not surprising that patients in our study who received ketamine were younger (Figure 4). Given that the ketamine patients were younger and likely had higher disease burden, greater levels of inflammatory markers would have been expected but this was not the case, possibly as a result of treatment with ketamine.
Evidence suggests that severe ARDS from COVID-19 leads to a uniquely challenging form of VPD, requiring multiple sedative agents at higher doses than typically required in traditional acute respiratory failure seen prior to the pandemic.21 –23 It can be presumed that this phenomenon, now well known to the critical care community, resulted from the unique and devastating nature of the cytokine surge. Pata et al, in their retrospective cohort study, recognised that these sedation challenges exist and demonstrated safety and reduced mortality with ketamine in severe ARDS COVID patients. 24 Our study cohort represents a historic population of COVID-19 critically ill patients with data obtained during the early period of the pandemic, prior to immunisation and established therapies being validated; thus, the severity of respiratory failure and ARDS along with the disease burden was high. In our enterprise, ketamine was added when the sedation and analgesia requirements exceeded what was possible with maximal doses of other agents. Although a mortality benefit was not associated with ketamine, improved sedation depth was observed. This finding may be useful to critical care physicians who desire a deeper level of sedation in COVID-19 patients or similar severely critically ill populations.
Limitations
In addition to the retrospective study design, there are several other limitations to this study. There may have been differences in treatment approaches during this early part of the COVID-19 pandemic, particularly since this patient population was treated before the RECOVERY trial demonstrated the mortality benefit of corticosteroids. 21 Ketamine may not have been initiated for the same indications at different Enterprise hospitals. This may have resulted in inconsistencies between the study hospitals. Clinicians were not blinded and therefore their inherent biases may have affected which patients received ketamine. Laboratory and RASS assessments were largely driven by clinical factors and inconsistencies between clinicians and hospitals may have existed. We were also unable to account for heterogeneity in the COVID-19 disease itself, namely, the presence of ARDS. 4 ARDS takes many different forms and can be classified by clinical severity, aetiology and pathological phase.25,26 Complex inflammatory changes occur in the lung and are classified into an early, exudative phase followed by proliferative and fibrotic phases.26,27 Another key limitation of the study is that the plasma concentrations of ketamine and its metabolites were not assessed. Ketamine is rapidly and extensively metabolised by microsomal enzymes, raising the potential for multiple metabolic drug–drug interactions and pharmacogenetic effects, which would produce changes in the drug’s observed efficacy and toxicity. 28 In addition, we have recently demonstrated that a five-day continuous infusion of subanaesthetic doses of ketamine induces the drug’s metabolism, which could also alter the observed effects. 29 Thus, therapeutic drug monitoring will be an important addition to future studies.
Conclusion
Ketamine infusions provided improved sedation depth in a challenging mechanically ventilated COVID-19 cohort and a modest anti-inflammatory benefit. Mortality or ICU length of stay were not reduced in patients given ketamine.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Wyler, Leong, Baram, Denk, Long, Gawel and Schwenk have no conflicts of interest. Torjman and Wainer filed a patent application on the use of ketamine metabolites. Viscusi was a consultant or received funding from Heron, Esteve, Innacoll, and Salix. He is a member of the board of directors of the American Society of Regional Anesthesia and Pain Medicine, which endorsed the acute and chronic ketamine guidelines in 2018.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
