Abstract
Summary
Perioperative neurocognitive disorders including postoperative delirium (POD) are common complications of anaesthesia and surgery, associated with morbidity, mortality and a large economic cost. Currently, limited data are available on the incidence of POD in the New Zealand population. The objective of this study was to utilise New Zealand national level datasets to identify the incidence of POD. Our primary outcome was defined as a diagnosis of delirium via ICD 9/10 coding within seven days of surgery. We also analysed demographic, anaesthetic and surgical characteristics. All adult patients undergoing any surgical intervention under sedation, regional, general or neuraxial anaesthesia were included, and patients who received surgical intervention under local anaesthetic infiltration alone were excluded. We reviewed ten years of patient admissions from 2007 to 2016. Our sample size was 2,249,910 patients. The incidence of POD was 1.9%, much lower than previously observed, potentially indicating significant under-reporting of POD in this national level database. With acknowledgement of the limitations of potential undercoding and under-reporting, we found that the incidence of POD was higher with increasing age, male sex, general anaesthesia, Māori ethnicity, increasing comorbidity, surgical severity and emergency surgery. A diagnosis of POD was associated with increased mortality and hospital length of stay. Our results highlight potential risk factors of POD and disparities in health outcomes in New Zealand. Additionally, these findings suggest systemic under-reporting of POD in national level datasets.
Introduction
Perioperative neurocognitive disorders following anaesthesia and surgery, including new onset postoperative delirium (POD) are common in older patients, and are associated with poor short- and long-term outcomes.1,2 Postoperative delirium is linked to cognitive decline, increased hospital length of stay (HLOS), discharge to long-term care, mortality, and higher healthcare costs and healthcare inefficiency. Postoperative delirium has been reported to contribute to about US$32.9 billion per year of health expenditure in the USA. 3 In Australia, the Australian Commission on Quality and Safety in Healthcare estimates that each episode of POD costs an additional A$27,791.1,2,4
The incidence of POD has been reported to be as high as 50%–70% in high-risk populations. 2 The disease incidence observed in clinical trials varies depending on the risk profile of the study population, the frequency and duration of delirium assessments and the surgical procedure.1,2 The reported incidence may vary due to the involvement of multidisciplinary management, intervention pathways for high-risk patients and variations in clinician recognition and coding.5,6 Perioperative delirium may present as either hyperactive or hypoactive subtypes, the latter being more difficult to detect. 7
Understanding the incidence of POD at a national level is crucial for establishing demographic and regional need for intervention. In New Zealand, studies have been limited to single centre observations of in-hospital POD. On medical wards, delirium incidence has been reported to be 11.2%–29%.8,9 Among surgical patients, Xiang et al. showed the incidence of POD to be as high as 39% in older surgical patients with neck of femur fractures. 10 These studies suggest that POD is a significant problem in New Zealand; however, there is a lack of national level data for the surgical population. Additionally, it is unclear whether patient factors, including ethnicity and surgical characteristics, have an impact on the incidence of POD in New Zealand.
Patient, anaesthetic and surgical characteristics are all recorded in New Zealand national level health databases. 11 These collections additionally contain International Classification of Diseases (ICD) codes, 12 which relate to the presence of a clinical diagnosis and provide insight into the incidence of disease at the population level. 11 Diagnostic coding is completed by clinical coders after reviewing documentation completed by medical staff. Clinical coding may not always correlate exactly to disease incidence/prevalence; however, national level databases remain valuable tools for providing population level data and have been used previously to identify health outcomes and disease incidence.13–17 To our knowledge, the incidence of POD has never been examined using national level datasets.
We hypothesised that there is a high burden of POD in the surgical population. The primary aim of this study was to utilise a national level dataset to define the incidence of POD in adult patients within seven days of surgery in New Zealand. Secondary aims were to identify high-risk population groups for POD and to compare the incidence of POD in the New Zealand surgical population with previously reported rates in the literature.
Methods
Ethical approval for this study was obtained from the New Zealand National Health and Disability Ethics Committees (HDEC 21/NTB/27, Ministry of Health, Wellington, New Zealand). The study design and manuscript preparation complied with Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational research. 18
We conducted a retrospective observational study with data obtained from the New Zealand Ministry of Health National Minimum Dataset (NMDS) and the National Mortality Registry. These are national level datasets collected by the New Zealand Ministry of Health.19,20 The NMDS captures over 99% of all operations in 71 private and public hospitals in New Zealand.15,19,20 The data captured include patient, surgical and anaesthetic characteristics as well as ICD-9 or ICD-10 codes pertaining to the diagnosis of delirium during a pre-defined time frame. 11 ICD-9 and ICD-10 codes were mapped, matched and assessed for appropriateness in classification of POD before application in the data extraction process.21–23 The New Zealand National Mortality Collection captures population mortality statistics, including cause of death, as compiled from multiple sources including the New Zealand Cancer Registry, the New Zealand Transport Agency, Water Safety New Zealand and from documentation provided by certifying doctors, coroners and medical records officers in public hospitals. 20 These data are codified utilising ICD-10-AM. 20 Clinical outcomes included in this study were coded in these databases by trained clinical coders at each individual hospital and reported to the New Zealand Ministry of Health as per standard practice. 11
All patients aged 18 years or over undergoing surgery in New Zealand from 1 January 2007 to 31 December 2016 under neuraxial, regional, sedation or general anaesthesia were included in the study. Patients who had multiple procedures during an episode of care had each procedure and relevant postoperative course analysed. Patients were excluded if the procedure was performed under local anaesthesia infiltration alone, as this group of patients are unlikely to require overnight hospital admission and be assessed for POD.
The primary outcome was the incidence of POD, defined as the presence of an ICD-9 or ICD-10 delirium code following surgery until discharge or seven days postoperatively, whichever occurred earlier.
Secondary outcomes included patient characteristics such as ethnicity, age, sex, American Society of Anesthesiologists Physical Status (ASA PS), procedural characteristics such as surgical specialty, surgical severity, duration, urgency and HLOS. Anaesthetic characteristics included anaesthetic type (general, regional or neuraxial anaesthesia).
The primary outcome of POD was reported as an incidence. A secondary analysis was performed for all patients with postoperative delirium using the following covariates: age (less than 55 years, 55–65 years, 65–75 years, 75–85 years and greater than 85 years), sex (male or female), ASA PS (as ordinal values: 1, 2, 3, 4 and 5) and ethnicity (New Zealand European, Māori, Pasifika, Asian or Other, as per NMDS coding).
Anaesthesia was coded as either general anaesthesia or non-general anaesthesia (i.e. sedation, neuraxial, regional anaesthesia). General anaesthesia was identified by the presence of an Australian Classification of Health Interventions ICD-10 code. Patients were separated into exposure groups based on the type of anaesthesia (general versus non-general) and patients with both general anaesthesia and non-general anaesthesia codes were assigned to the general anaesthesia group.
Surgical severity was documented by the Johns Hopkins five-category operative severity classification system as described by Pasternak. 24 Categorisation was allocated by three investigators and surgical severity was assigned based on surgical invasiveness and potential for blood loss. Severity 1 procedures are determined to carry minimal risk of intraoperative blood loss or low surgical invasiveness (e.g. breast biopsy or hysteroscopy). Severity 2 procedures have minimal to moderate invasiveness with expected blood loss of less than 500 ml (e.g. diagnostic laparoscopy, umbilical hernia repair). Severity 3 procedures have moderate invasiveness with potential blood loss of 500–1500 ml (e.g. hip or knee joint replacement). Severity 4 procedures are highly invasive procedures with expected blood loss greater than 1500 ml (e.g. spinal surgery, major genitourinary surgery). Lastly, severity 5 procedures were highly invasive operations with expected blood loss greater than 1500 ml and expected postoperative intensive care unit stay (e.g. cardiothoracic surgery or major vascular surgery). 24
Additional covariates included surgical urgency (elective or emergency/acute), cancer status (as either yes or no) and surgical specialty (cardiac, orthopaedic, urological, etc.) as per NMDS coding. Other outcomes included 30-day all-cause mortality and HLOS.
The primary and secondary outcomes were analysed using multivariable logistic regression. Analysis was completed in R (R Studio version 3.4.0, Boston, MA, USA) and statistical significance for univariate analyses was defined as a P value of <0.05. ASA PS was our only source of missing data, with 37% of patients missing an ASA PS code. As this comprised such a large percentage of the total data, we classified all those patients as ‘ASA PS missing’ and kept them in the analysis.
Summary statistics for baseline characteristics such as age, sex, ethnicity, ASA PS, cancer status, surgical urgency, surgical specialty, anaesthesia code and HLOS were reported using descriptive statistics such as frequencies and percentages for categorical data, and means, medians, ranges, standard deviations and interquartile ranges as appropriate for continuous data.
Results
We obtained data for all admissions via NMDS from January 2007 to December 2016, which included 6,360,618 patients. Figure 1 shows the study Consolidated Standards of Reporting Trials (CONSORT) diagram. 25 The final analysis dataset consisted of 2,249,910 admissions.
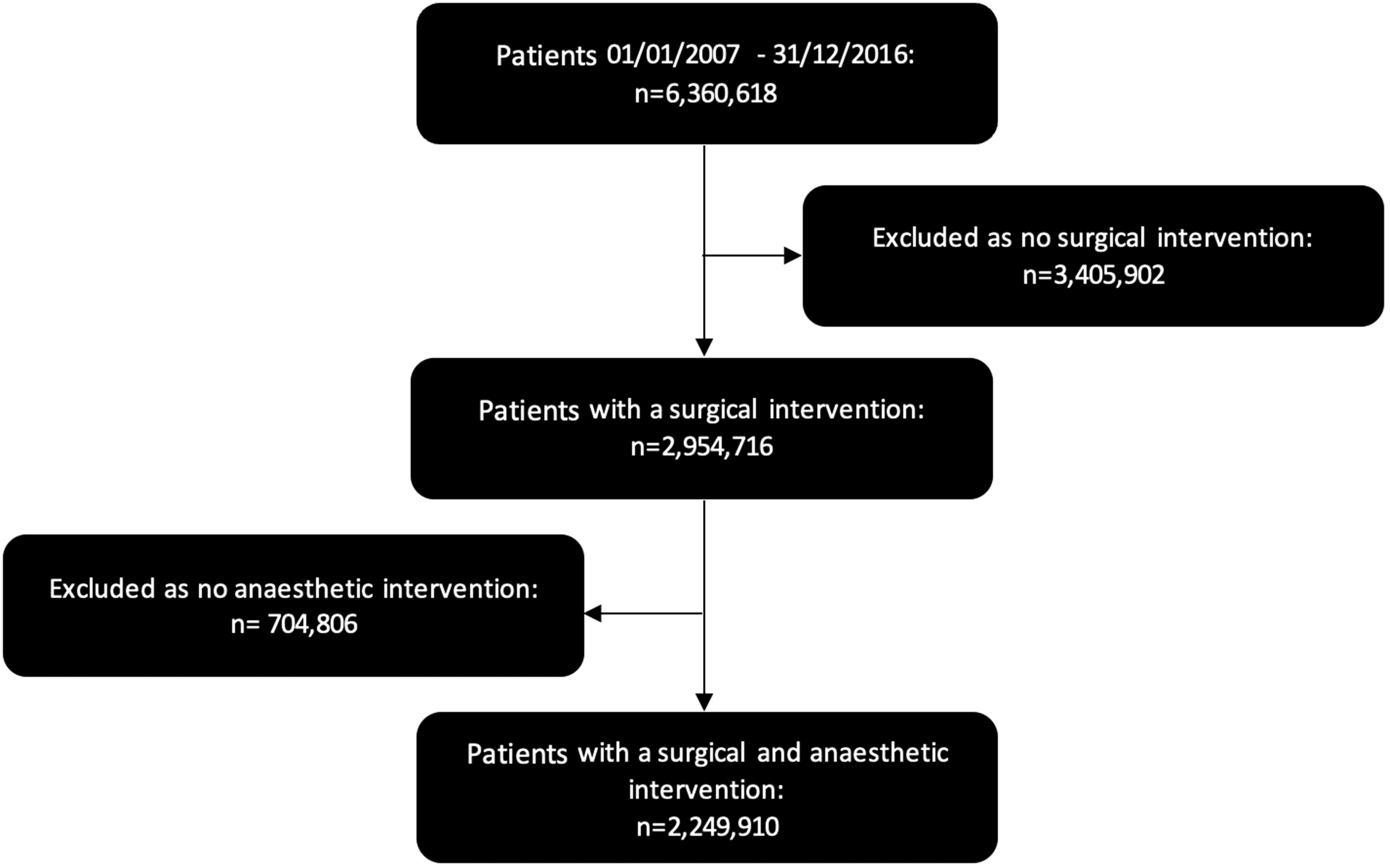
CONSORT (Consolidated Standards of Reporting Trials) diagram.
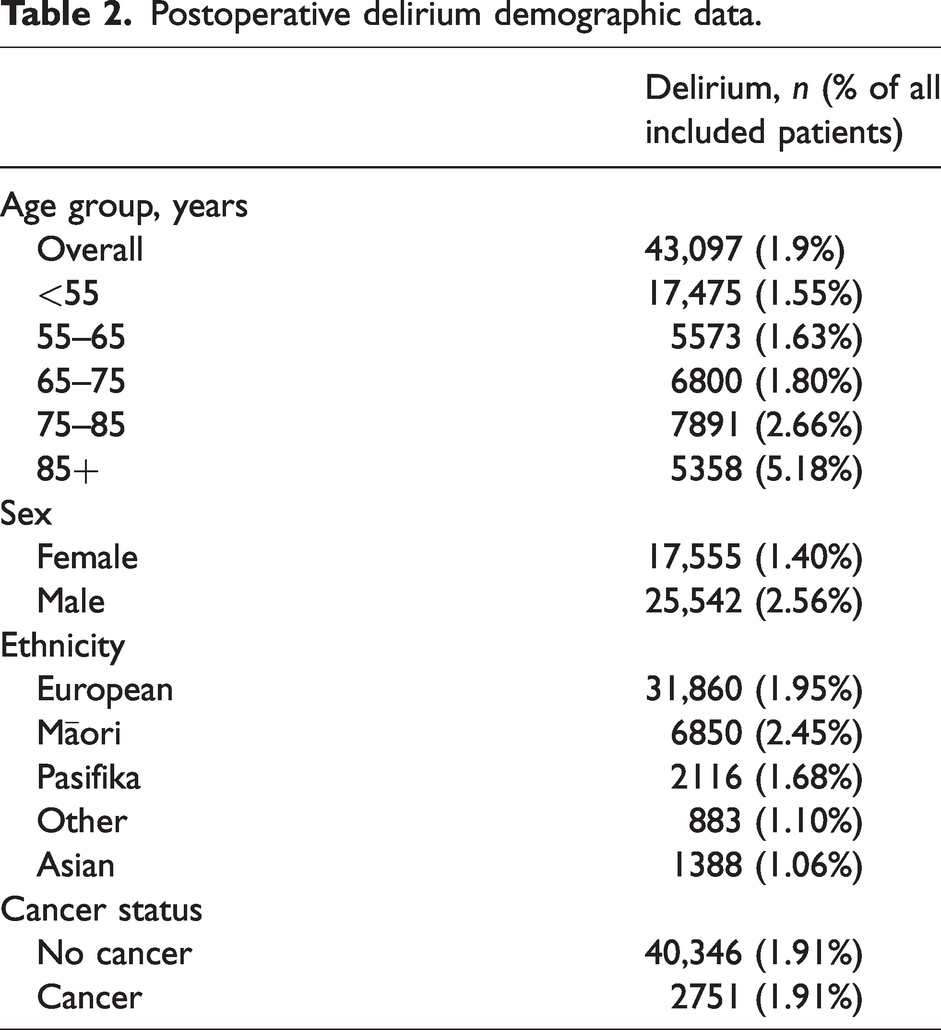
The overall incidence of POD was 1.9%. Table 1 shows the baseline characteristics of patients. Table 2 shows POD outcomes in relation to demographic data including age, sex and ethnicity. The incidence of POD was higher for older patients (POD incidence 5.2% for age >85 years versus 1.6% for age <55 years) and for males (POD incidence 2.6% versus 1.4% for females). Overall, the POD incidence was higher in Māori patients (2.45%,) compared with European patients (1.95%), and lower in Pasifika (1.68%), Asian (1.06%) and other ethnicity patients (1.10%).
Baseline demographic characteristics.

ASA PS, American Society of Anesthesiologists Physical Status.
aOne patient identified as neither male or female.
Postoperative delirium demographic data.
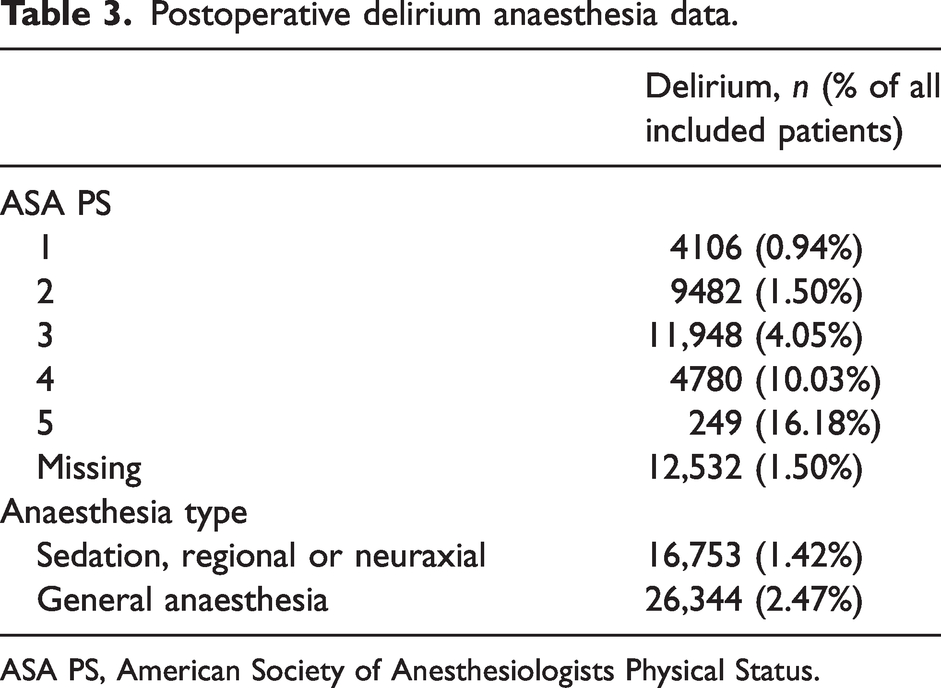
As shown in Table 3, increasing comorbidity as documented by ASA PS was also associated with an increase in POD (in those with ASA PS recorded): incidence for ASA 1 to ASA 5 patients was 0.9%, 1.5%, 4.1%, 10.0% and 16.2%, respectively.
Postoperative delirium anaesthesia data.
ASA PS, American Society of Anesthesiologists Physical Status.
Surgical severity was associated with increasing POD: the incidence was 2.5%, 4.4% and 8.0% for surgical severity groups 3, 4 and 5 respectively. This association was also reflected by increasing incidence of POD in surgical specialties with more invasive procedures (Table 4). The highest incidence of POD was observed in the thoracic (7.5%), neurosurgical (6.5%) and vascular (7.7%) surgical specialty groups. The reference group for surgical specialty is labelled ‘other’ surgical procedures and includes nine subspecialties, which had an incidence rate of less than 1% (eye and adnexa, ear and mastoid, nose, mouth and pharynx, blood and blood forming organs, urinary system, male genital organs, gynaecological procedures, obstetric procedures, breast procedures).
Postoperative delirium by surgical factors.
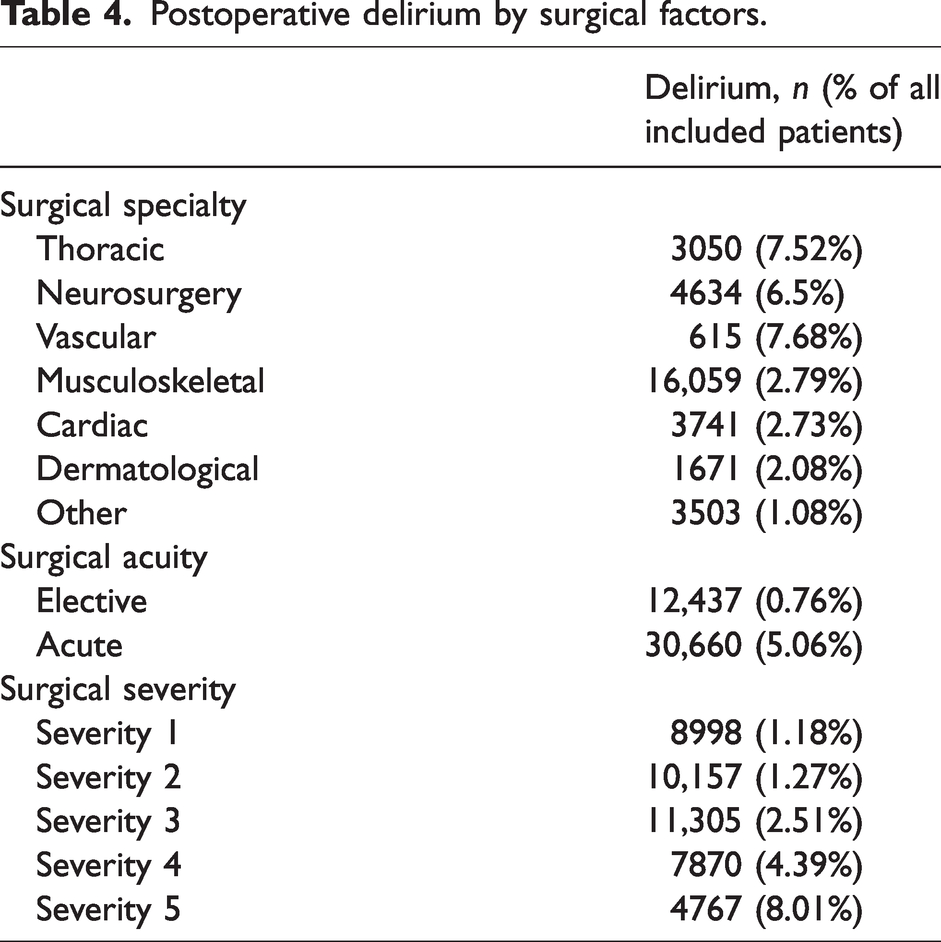
Patients undergoing emergency surgery had higher incidence of POD compared with those undergoing elective surgery (5.1% versus 0.8%). Patients who received general anaesthesia had a higher incidence of POD compared with patients who did not (2.5% versus 1.4%). Perioperative cancer status did not appear to affect the incidence of POD (1.9% both groups).
Surgical patients with POD had worse outcomes compared with those without POD. The 30-day all-cause mortality was 7.5% in patients diagnosed with postoperative delirium and 1.1% in patients without a POD diagnosis (P < 0.001). HLOS was significantly higher in patients with POD, with a mean difference of nine days between patients with and without POD (mean HLOS 12 days versus three days, P < 0.001).
Discussion
We found that the incidence of POD reported in the NMDS was much lower than previously reported in single-centre observational studies. Xiang et al. found a postoperative delirium incidence of 34.8% in 184 patients over the age of 65 years with neck of femur fractures. 10 Similarly, Lin et al. found a postoperative delirium incidence of 13.5% in orthopaedic patients across two wards. 26 Tan and Scott studied both orthopaedic and medical patients and across their sample size of 250 patients and noted an incidence of delirium of 11.2%. 9 This large discrepancy suggests systemic under-diagnosis or under-coding or both. Our experiences mirror that of other researchers who similarly reported lower than expected diagnostic coding of delirium. In a population of over 900 patients in the USA, Inouye et al. found that only 2.6% of patients had a delirium code, despite 12.5% being diagnosed with delirium. 16 Travers et al. noted a 17.3% incidence of delirium in a population of 493 medical and surgical patients across four Australian hospitals. 17 However, this was associated with appropriate coding of only 2.4% on discharge. 17 Looking at New Zealand specific studies, we re-examined the data from Xiang et al. and noticed that, out of the 34.8% of postoperative patients diagnosed with delirium, only 3.2% had an appropriate coding of delirium on discharge. 10
Our findings of a low incidence of POD diagnosis or coding may reflect the complexity of this disorder as well as factors that could lead to its systemic under-recognition and prevention. We recognise that the lower incidence of POD in our study may be caused by the difficulty in detecting hypoactive delirium, which accounts for over 50% of cases of POD. 7 Without specialist diagnostic input, a large population of patients with delirium may not be recognised and treated appropriately. Another obstacle affecting data quality includes how medical data are coded in the New Zealand context. A medical diagnosis must be documented within patient notes and discharge summaries, which are prepared by junior medical staff who may have limited clinical experience, time, resources or supervision. Documented diagnoses are then coded into large national datasets such as the NMDS by clinical coders' review of patients' notes and discharge summaries. 13 Thus, there is a large reliance on accurate documentation and coding processes to ensure data quality.11,13 Going forward, it may be prudent to establish more appropriate diagnostic and coding guidelines, as well as greater utilisation of electronic record-keeping for the translation of clinical diagnoses into appropriate coding.
The ability to draw definite conclusions from our study is limited by the potential inaccuracy in clinical coding for POD. However, our previous experience with using NMDS data is that data completeness, accuracy and integrity for demographic and process datapoints is greater than 99%.15,27–29 Our results were consistent with previously identified risk factors for POD, including increasing age, acute surgery, comorbidity and association with invasive surgery. 1 Perioperative outcomes were also worse in patients diagnosed with POD as previously observed.1,2 Our study also identified ethnic disparities in the incidence of POD with higher incidences in Māori patients. This finding is historically consistent, and perioperative outcomes in Māori patients have been shown to be worse compared with other ethnicities in New Zealand.29–32 In our study, Māori patients had a higher risk of experiencing POD and experienced POD at a much younger age than New Zealand European (median age 41 years old in Māori patients, compared with 64 years old in New Zealand European patients). The Māori surgical population also had a younger age structure (median age, 41 years versus non-Māori, 56 years old). The potential disparity may be related to higher risk factors for POD in the Māori population (increased metabolic and cardiovascular disease burden) as well as institutionalised racism leading to structural and/or geographic disparities in access to culturally responsive healthcare.29–33 Gurney et al. discussed factors such as historical and structural institutionalised racism leading to socioeconomic disadvantage, higher comorbidity and disease burden and poor access to culturally safe healthcare. 29 These features are risk factors for POD.29,31,32 The potential higher incidence of POD among the Māori surgical population is an important issue for health equity in New Zealand and could contribute to increasing health inequities through the need for long-term care, whānau involvement and escalating healthcare needs.1,2 This potential disparity in POD reinforces the ongoing need to improve Māori health.
We observed lower incidences of POD in patients of Pasifika, Asian and other ethnicities relative to New Zealand European patients. We interpret this finding with caution due to concerns regarding data accuracy. This finding was surprising, especially given previously identified health outcome disparities in New Zealand for patients from minority ethnic backgrounds.34–36 There are many potential unmeasured factors at play. However, we postulate that a potential cause for these findings may relate to cultural differences in communication and potential language barriers,34–36 which may affect the accuracy of a clinical diagnosis of POD in these population groups. We believe that the incidence of POD in Pasifika, Asian and other ethnicities may be underestimated in our dataset, but further studies are required to clarify this.
It is important to acknowledge the limitations of our study. Firstly, our primary outcome relies on accurate diagnostic, documentation and coding processes in New Zealand. There is a suggestion of poor data coding and accuracy based on the lower than expected incidence of POD in our study. Variations in patients’ perioperative journeys could affect the accuracy of our results. We did not look at regional incidences of POD and acknowledge that some hospitals may have established pathways for screening and management of POD, which may not be reflected by the overall incidence rates of POD in our study. Secondarily, our data are retrospective and do not reflect the efforts made by health institutions over the last decade to improve the global recognition and management of this disorder. Examples of this includes the American Society of Anesthesiologists – ‘Brain Health Initiative’ and the Australasian ‘Caring for cognitive impairment’ campaign.37,38 Thirdly, although the majority of New Zealand data have been captured by the NMDS, this statistical dataset does not receive data from 10% of private hospitals. 15 This is unlikely to be of significance given the lower risk demographic of this patient group. Lastly, 37.1% of our study patients had missing ASA PS data. Although this subset was similar when compared with patients with full data, this still represents a large missing amount of data for this variable. Lastly, a conceivable source of error is the potential effect of cumulative exposure to surgical and anaesthetic events. We included patients in our analysis who had multiple procedures during an episode of care, with each procedure and relevant postoperative course analysed separately. We acknowledge that there is a likelihood that each exposure may potentially increase the subsequent incidence of POD. Nevertheless, it was felt that excluding multiexposure patients would be inappropriate.
Conclusion
The incidence of POD as recorded in the NMDS by ICD coding in adult patients undergoing surgery in New Zealand over the last ten years was 1.9%. This is much lower than expected compared with previously published studies. The true POD incidence is likely to be much higher in New Zealand and this outcome highlights concerns of substantial under-diagnosis and under-reporting of this clinically important disorder. This discrepancy could be attributable to a number of confounders and unmeasured factors that we could not determine from the NMDS data collection. We found that male sex, Māori ethnicity, increasing ASA PS grade, general anaesthesia, emergency surgery and increasing surgical severity were associated with a higher incidence of POD. However, we recommend interpreting this finding with caution, given the potential under-reporting for the incidence of POD.
This study highlights the need to improve diagnosis and coding of POD. Understanding disease prevalence and incidence is crucial in workforce planning and health resource allocation. Further investment in screening and care pathways are needed, alongside a greater focus on accurate documentation of POD.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X231156459 - Supplemental material for Incidence of postoperative delirium in surgical patients: An observational retrospective cohort study
Supplemental material, sj-pdf-1-aic-10.1177_0310057X231156459 for Incidence of postoperative delirium in surgical patients: An observational retrospective cohort study by Peter Y Xiang, Douglas Campbell in Anaesthesia and Intensive Care
Footnotes
Author Contributions
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The authors have no sources of funding to declare for this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
