Abstract

Keywords
Massive transfusion (MT) is a resource intensive event, associated with significant morbidity and mortality. 1 Excessive transfusion of blood product is associated with harm and waste of a limited resource. Restrictive transfusion, when haemoglobin (Hb) is less than 70 g/l, has been demonstrated to reduce mortality and re-bleeding in patients with upper gastrointestinal (UGI) haemorrhage when compared with liberal transfusion (Hb <90 g/l). 2 In cardiac surgery, the TRICS III trial demonstrated non-inferiority of restrictive transfusion (Hb <75 g/l) compared with liberal transfusion (Hb <95 g/l). 3 At our institution, concern was raised regarding cases in which excessive blood product replacement was given as part of MT. To determine the veracity of this, we audited our incidence of over-transfusion.
Our audit was granted ethics approval as a quality assurance activity (reference 21059). Data were from a record of massive transfusion protocols (MTPs) from January 2020 to May 2021 and patient medical records. Our definition of MT required transfusion of blood products totalling 50% or greater of patient blood volume within four hours, or 100% or greater of blood volume within 24 hours. 4 Patient blood volume was calculated using a 70 ml/kg estimate up to 100 kg body weight. Cases of intraoperative mortality due to inability to control bleeding were not included. We categorised outcomes according to post-transfusion Hb: ‘mild over-transfusion’ (Hb 80–100 g/l) and ‘significant over-transfusion’ (Hb >100 g/l); ‘appropriate transfusion’ for post-transfusion Hb of 70–80 g/l and ‘under-transfusion’ for Hb less than 70 g/l. Pre- and post-transfusion Hb were defined, respectively, by the Hb concentration before the first unit of packed red blood cells (PRBCs), and two to four hours following MTP completion.
Of 83 cases of MTP activation, 30 episodes occurring in 29 patients met the criteria for inclusion (Table 1). Forty-seven excluded cases received less than the defined blood products for MT. Four were excluded due to intraoperative or preoperative mortality. Two duplicates were removed. The median transfused volume was 9 units PRBCs, 5.5 units fresh frozen plasma (FFP) and 8 units platelets. Twenty-seven of 30 episodes met the definition of over-transfusion. The median (interquartile range) Hb post-transfusion was 98 (91.3–106.3) g/l. At discharge, median Hb was 96.5 (89.8–106.5) g/l. No included patients met the criteria for under-transfusion. However, under-transfusion may have occurred in patients who did not survive.
Patient characteristics.
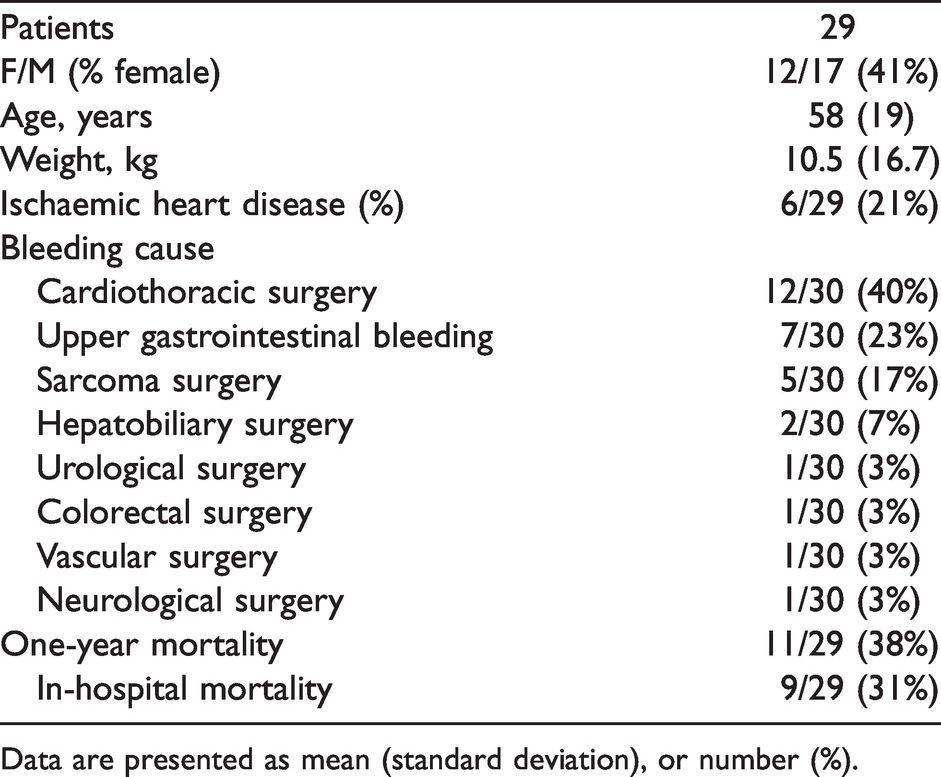
Data are presented as mean (standard deviation), or number (%).
Our audit demonstrated a pattern of over-transfusion associated with MT, with no cases of under-transfusion. A similar incidence of over-transfusion has been identified in other institutions. 5 Our reported in-hospital mortality (Table 1), was slightly higher than in previous meta-analyses of MT (31% versus 24%). 6 Over-transfusion occurred at similar rates regardless of subgroup, although appropriate transfusion was most common in those with UGI haemorrhage (Figure 1). All MTs involved anaesthetists at some stage and took place, at least partially, in the operating theatre; demonstrating the importance of anaesthetists in MT management. With critical intraoperative bleeding, anaesthetists are required to rely on estimates of volume loss, ongoing bleeding and haemodynamic parameters to calculate blood product replacement. There may be a delay in acquiring frozen products due to thawing time. These factors may risk excessive administration of PRBCs for volume resuscitation prior to control of bleeding.
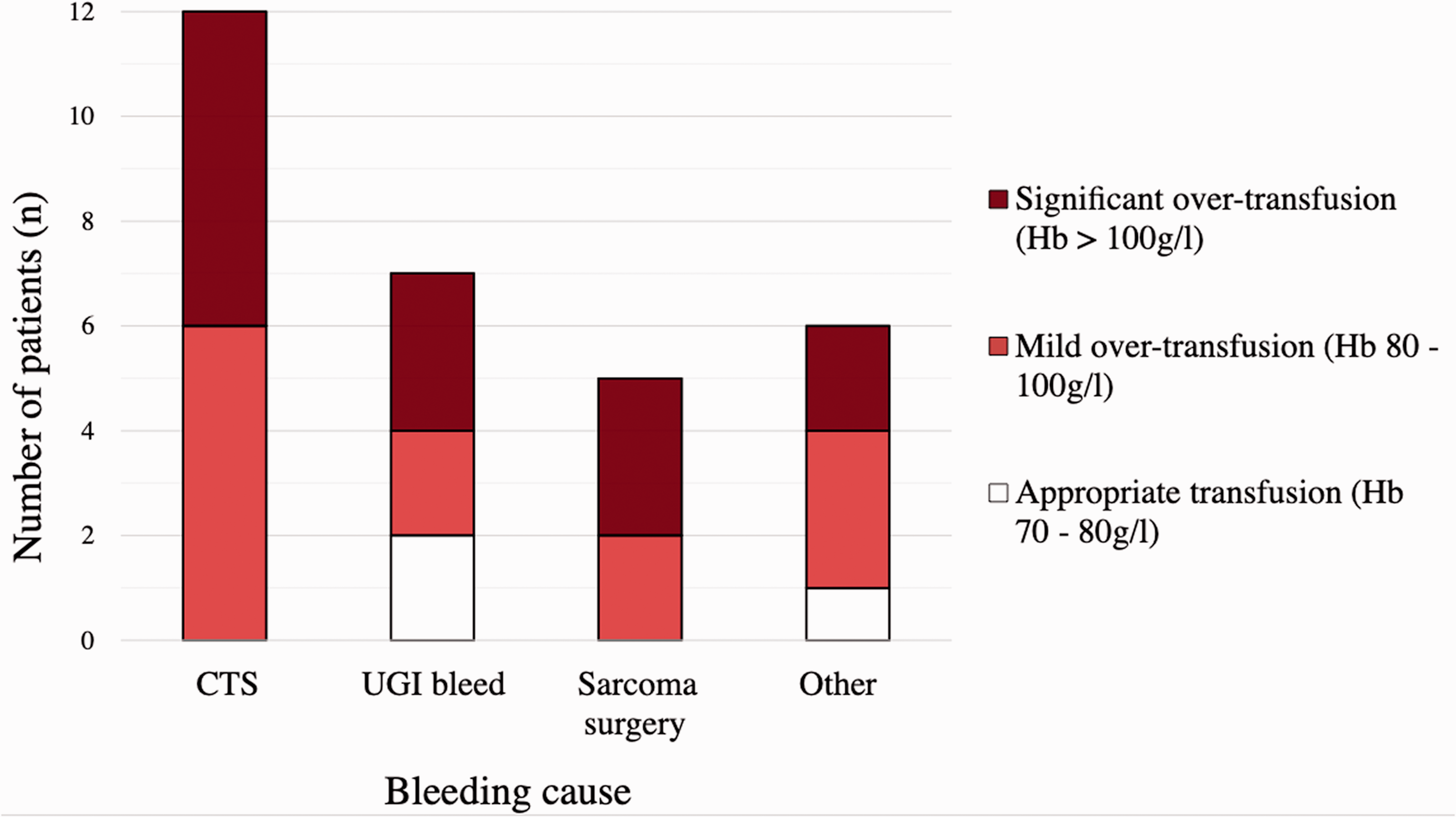
Proportion of patients receiving appropriate transfusion, and mild or significant over-transfusion. Hb: haemoglobin; CTS: cardiothoracic surgery; UGI: upper gastrointestinal.
Our institutional guideline for MT recommends giving PRBCs and FFP in a 2:1 ratio, considering platelet administration 1:1 with PRBCs. This is based on a protocol from the Australian National Blood Authority (NBA) guideline for MT. 4 The NBA does not specifically recommend any particular ratio of products, or a Hb target for transfusion. 4 European guidelines for MT target a Hb of 70–90 g/l, 7 and the Association of Anaesthetists of Great Britain and Ireland guideline suggests a 1:1 PRBCs to FFP ratio in early resuscitation. 8 These recommendations are based on retrospective studies in a trauma population showing benefits associated with MT using higher PRBC:FFP ratios,9,10 which may not apply to our surgical patient population. A meta-analysis of trials comparing MT ratios found insufficient evidence to support using 1:1:1 (red blood cell:FFP:platelet) over 2:1:1 ratios in patients with critical bleeding. 6 Despite this, transfusing in a 1:1:1 ratio is unlikely to lead to over-transfusion of Hb, as on average one round of products (4 PRBCs, 4 FFP and 1 bag platelets) results in the transfusion of 192 g Hb in about 2411 ml, or 80 g/l Hb.
All patients requiring MT are at risk of being over-transfused. This may expose some to the direct risk of harm and risks inappropriate use of blood products. In our audit, we consider Hb as a surrogate for over-transfusion. We acknowledge that over-transfusion can result from excessive volume replacement using other products. However, high ratio (1:1:1) resuscitation may be an appropriate way to provide volume resuscitation while avoiding the over-transfusion of PRBCs in early bleeding. These data will guide our local guidelines and education for the management of critical bleeding. We will recommend initial ratio-based transfusion with a near one-to-one PRBC:FFP ratio and early transition to targeted transfusion using laboratory data or thrombelastography (if available) once bleeding is controlled. This represents a change from our current guideline, with more specific recommendations. In doing so, we aim to reduce the incidence of over-transfusion at our institution, and to optimise patient safety and our utilisation of blood products. We recommend that other institutions undertake similar audits of MT practice, and if required, implement interventions to reduce the incidence of over-transfusion.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial assistance for the research, authorship or publication of this work.
