Abstract
Sugammadex has been used for more than ten years in Australia and New Zealand and has been implicated as an effective treatment, and in some cases a potential cause, of a critical incident. We aimed to identify and analyse critical incidents involving sugammadex reported to webAIRS, a de-identified voluntary online critical incident reporting system in Australia and New Zealand. We identified 116 incidents where the reporter implicated sugammadex as either a cause (23 cases) or a treatment (93 cases) during anaesthesia. There were 17 incidents suggestive of sugammadex anaphylaxis, although not all were confirmed by skin testing. There were six incidents when bradycardia was temporally related to sugammadex administration, although it was not possible to exclude other causes or contributory factors. There were nine incidents in which sugammadex was used to reverse aminosteroid-related neuromuscular blockade successfully in a ‘can’t intubate, can’t oxygenate’ (CICO) situation, and a further 67 incidents in which sugammadex was used to reverse aminosteroid neuromuscular blockade as part of the management of other critical incidents. While sugammadex was used during the management of 16 cases of anaphylaxis, there was no clear indication that this altered the course of the anaphylaxis in any of the cases. These reports indicate that sugammadex can be a potential trigger for anaphylaxis and that its use may be associated with the development of significant bradycardia. However, it is not possible to estimate or even speculate on the incidence of these sugammadex-related incidents on the basis of voluntary reporting to a database such as webAIRS. The reports also indicate that sugammadex has been used successfully to reverse residual or deep aminosteroid neuromuscular blockade in critical incident situations and to help rescue CICO scenarios. These findings provide further support for ensuring the ready availability of sugammadex wherever aminosteroid muscle relaxants are used.
Keywords
Introduction
Sugammadex was introduced as a neuromuscular reversal agent in Australia and New Zealand in 2009. Since the introduction of sugammadex into clinical practice, case reports have been published that have implicated it as both a cause and a treatment of critical events, most commonly as a treatment of a ‘can’t intubate, can’t oxygenate’ (CICO) event or rocuronium anaphylaxis, and as a cause of anaphylaxis in its own right. These reports have previously been limited to individual case reports and small case series.1 –19
Incident reporting has been an important learning tool in high-risk industries for many years. In medicine, feedback of incidents to clinicians is an important component of incident reporting, using both detailed information of the incident and analysis of trends and patterns in reporting. 20
The webAIRS incident reporting system was established in Australia and New Zealand in 2009 and is a multinational, voluntary, de-identified reporting system used to collect data on anaesthetic incidents related to safety and quality in anaesthesia. 21 There are currently 217 hospitals (175 in Australia and 42 in New Zealand) out of an estimated >1450 hospitals in Australia and New Zealand 22 registered to provide data to webAIRS. The majority of hospitals do not all provide their total number of anaesthetic cases (denominator data). However, nine hospitals in New Zealand that report to webAIRS also report denominator data, with an incident reporting rate of 1.5 incidents per 1000 anaesthetic episodes of care. At the time of this database study, there were 8905 incidents reported in the webAIRS database, which (assuming a reporting pattern consistent with these New Zealand hospitals) would reflect approximately 5.9 million anaesthetic episodes of care.
We aimed to examine the webAIRS database to identify critical incidents in which sugammadex had been used and to classify these according to whether sugammadex was a possible cause of the incident. We then analysed these incidents for potential learning points to help improve both the quality and safety of sugammadex use in anaesthesia.
Methods
The webAIRS database was queried for any incident where ‘sugammadex’ or the partial words ‘sugam’ or ‘suggam’ were mentioned. The case reports were then accessed electronically, and each event was reviewed individually by two researchers and categorised into events where the use of sugammadex was ‘critical’, ‘incidental’ or ‘not used’. Where there was disagreement as to the classification between the reviewers, the two researchers conducted a further review of the individual case together to reach a consensus. Data were then analysed to classify cases further where sugammadex was categorised as ‘critical’ or ‘other’ to be summarised as a narrative review.
For the purposes of this study, presumed anaphylaxis was defined as a critical event with the reported presence of two or more of the following: high airway pressure, rash, tachycardia and hypotension; and a temporal relationship to administration of a medication. This definition is consistent with that of presumed anaphylaxis as defined by the Australian and New Zealand College of Anaesthetists and Australian and New Zealand Anaesthetic Allergy Group Perioperative Anaphylaxis Management Guidelines 23 , and the World Allergy Organization. 24 A CICO event was defined as a medication-induced inability to maintain a patent airway with reported consequent oxygen desaturation, despite the failure of at least two of the following techniques: manual bag–mask ventilation (with or without adjuncts), a supraglottic airway, and intubation with an endotracheal tube. Heart rate limits were not specified for the purpose of defining bradycardia, but rather the occurrence was recorded based on specific reports of bradycardia by the authors of each incident.
This study was approved by the Human Research Ethics Committee at the University of Sydney, Australia (2018/510).
Results
The webAIRS database of 8905 cases was examined, spanning the period from its creation in 2009 to 22 April 2021. A total of 345 reports mentioned sugammadex over this period. Figure 1 shows the classification of these cases. In total, there were 116 incidents in 115 cases where sugammadex either was considered to be the cause of the critical incident or was used specifically as part of the treatment of the critical incident.
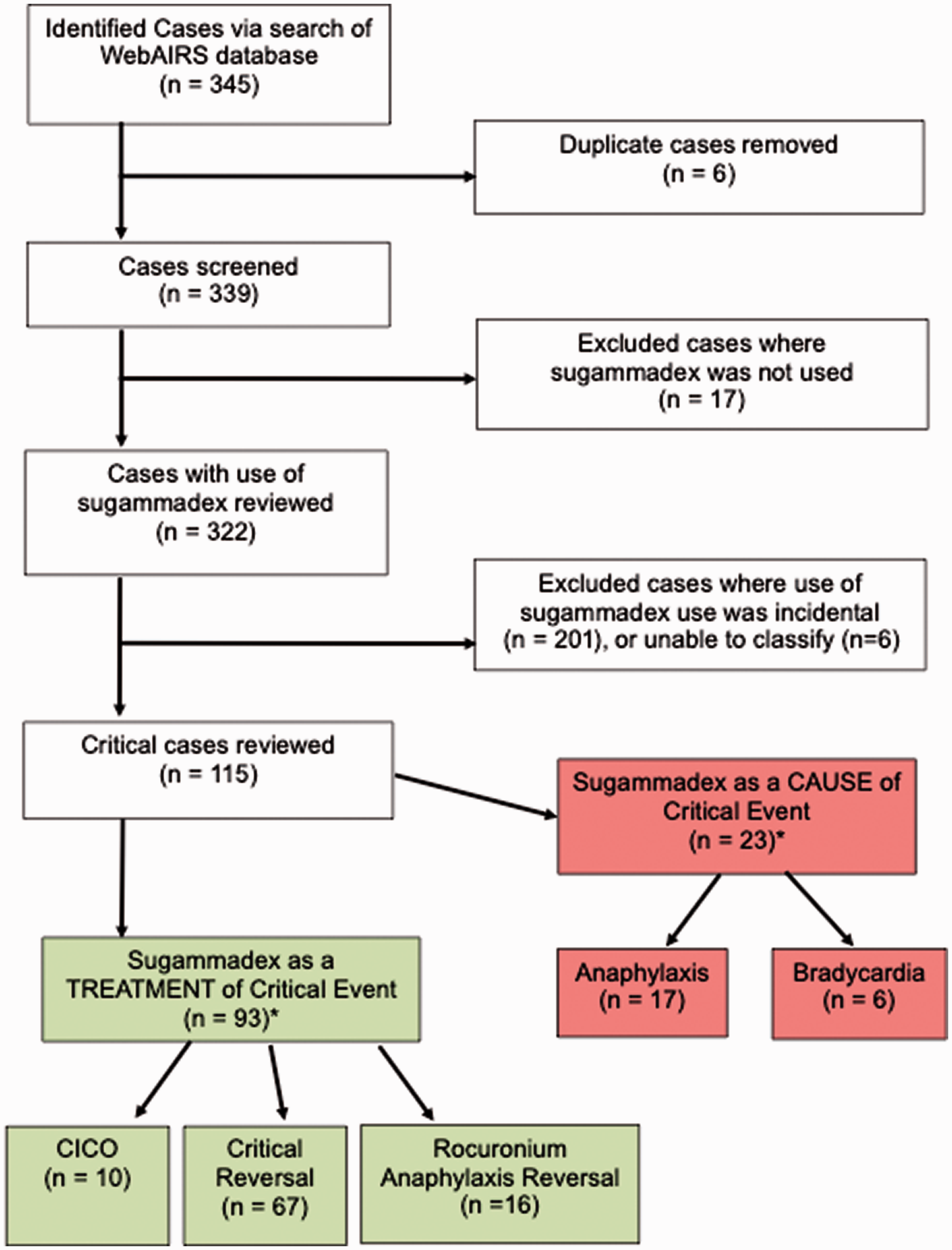
Schematic diagram of case classification. *One patient had sugammadex at the time of possible rocuronium anaphylaxis as a ‘treatment’, which also allowed reversal of deep block to wake the patient, leading to 116 events in 115 cases.
Sugammadex as the cause of a critical event
Anaphylaxis
There were 17 cases where the use of sugammadex was followed by clinical signs of anaphylaxis. Of these cases, there were reports of hypotension (16 cases), tachycardia (seven cases), oxygen desaturation (eight cases), skin rash (seven cases), and raised airway pressures (six cases). Fifteen reports of anaphylaxis reported the need for adrenaline boluses, with five cases subsequently requiring an adrenaline infusion. In total, 11 cases were transferred ventilated to an intensive care unit (ICU). Two cases required reintubation in a post-anaesthesia care unit (PACU). Not all cases reported formal allergy testing as part of the follow-up to the presumed anaphylaxis incident (testing may have been performed, but it was not reported). Of the 12 cases that did report subsequent testing, nine cases reported elevated tryptase levels and four reported positive sugammadex skin testing; one case reported both an elevated tryptase and positive skin test. One case had inconclusive tryptase testing reported.
Bradycardia
In six cases, the incident reporters recorded a temporal association between the administration of sugammadex and an episode of bradycardia requiring treatment. All of these cases were administered atropine to treat the bradycardia, with one case needing cardiopulmonary resuscitation for asystole. A separate case was documented to need admission to the ICU for Takotsubo cardiomyopathy. Though the reporter of this incident indicated that this was believed to be precipitated by the administration of atropine, causation was not established or documented on the webAIRS report.
Sugammadex as treatment of a critical event
CICO event
In nine reports, a CICO event was successfully managed with adequate reversal of neuromuscular blockade using sugammadex. In all of these cases, the reporting anaesthetist documented a CICO event following either rocuronium (eight cases) or vecuronium (one case) administration, where subsequent use of sugammadex was rapidly followed by return of spontaneous ventilation and ‘rescue’ of the CICO event, with reversal of hypoxia and prevention of the need for front-of-neck access (FONA). One case reported a failure of sugammadex to rescue a CICO event. In this event, general anaesthesia was induced and both manual bag-mask ventilation and a planned supraglottic airway failed to produce a patent airway. Rocuronium (40 mg) was given, and the patient vomited before an endotracheal tube could be secured. On laryngoscopy, the cords could not be visualised with a subsequent drop in oxygen saturations. Sugammadex (200 mg) was given with no return of spontaneous respiration. Endotracheal intubation was then achieved following a second successful intubation attempt with resolution of the CICO event. This case did not exclude various other potential causes of failure to resume spontaneous ventilation. The patient had received midazolam, propofol, fentanyl, morphine and sevoflurane prior to the critical event. The patient’s weight was not documented in the webAIRS report, but the patient was classified as a male in his 50s in the obese category.
Management of rocuronium anaphylaxis
Sugammadex was reported as a component of the management of anaphylaxis on induction in 16 cases. These events all occurred immediately or shortly after induction of anaesthesia and administration of rocuronium. In these instances, sugammadex was administered as part of the resuscitation management (along with adrenaline, intravenous fluids and other supportive measures). In no cases did the administration of sugammadex lead to a rapid reversal of the signs of anaphylaxis or allow the rapid withdrawal of other supportive treatments, suggesting that sugammadex did not alter the course of the anaphylaxis. Of the 16 events, seven related to relaxant administration were confirmed as anaphylaxis to rocuronium in follow-up testing.
Need for reversal of paralysis as a critical event
There were 67 cases of sugammadex being given to reverse paralysis as a critical event. In 20 of these cases, sugammadex was given in the PACU to treat poor respiratory effort secondary to apparent residual paralysis. Three of these reports documented that the reporter felt the administration of sugammadex prevented the need for reintubation of the patient. In 29 incidents, sugammadex was given in the operating theatre as treatment of residual paralysis following extubation (12 of these documented as a train-of-four count of less than two, with all reporting clinical sequelae of weakness, dyspnoea or desaturation). In 18 of these cases, neostigmine had already been administered. In two cases, a reversal agent had not been administered. In four cases, a repeat dose of sugammadex was required for inadequate reversal following a single dose. In each of these incidents, sugammadex was successful in reversing the residual paralysis. In four of these cases, the reporter specifically reported that the administration of sugammadex prevented the need for reintubation of the patient.
Five cases were abandoned following induction due to various concerns (including rapid atrial fibrillation on induction, deranged pathology results or oxygen desaturation). In these cases, sugammadex was administered with successful reversal of aminosteroid muscle relaxant deep neuromuscular blockade, facilitating timely extubation of the patient.
An additional four reports concerned incidents where a large dose of rocuronium had been given towards the end of a procedure to facilitate reintubation (for inadvertent extubation intraoperatively in two cases, one case of laryngospasm on extubation and another loss of airway due to an endotracheal cuff leak). In these cases, sugammadex was needed to reverse deep paralysis at the end of the case and allow timely extubation.
In seven cases, sugammadex was given to reverse neuromuscular blockade following drug errors. In one case, rocuronium remained in the intravenous fluid line and was inadvertently administered at the end of the case. In the other six reports, rocuronium was mistaken for other drugs (neostigmine and midazolam twice each, suxamethonium and parecoxib). In each of these cases, sugammadex was successful in reversing the subsequent neuromuscular blockade.
There were two additional cases of the critical use of sugammadex to treat paralysis: one where the patient was accidentally extubated on transfer to their bed at the end of the case, and a second where the anaesthesia team were unable to intubate the patient. In both cases, manual ventilation was possible using bag-mask ventilation, and sugammadex was given, allowing the return of spontaneous ventilation.
Discussion
By analysing a large incident reporting database, we identified several cases in which sugammadex was a confirmed or likely cause of a critical incident. This included the development of anaphylaxis, which has been previously reported. 25 However, we also identified several cases of severe bradycardia following sugammadex administration. While it was not possible to confirm that sugammadex was the cause of the bradycardia, it was also not possible to exclude sugammadex being the cause. Unfortunately, using a voluntary database, it is not possible to determine the incidence of an adverse or critical incident such as anaphylaxis or bradycardia. We also found a large number of incidents in which sugammadex has been reported to help rescue a CICO event successfully and to treat residual paralysis or deep neuromuscular block to either facilitate timely extubation or prevent the need for reintubation. In contrast, we found no reports that suggested that sugammadex altered the course of anaphylaxis caused by aminosteroid muscle relaxants.
Our findings add to the existing published experience with sugammadex and critical incidents. Prior reports outlining the role of sugammadex in individual critical anaphylaxis incidents have been limited to 16 published individual case reports1 –5,7 –12,14,16,18,19,26 and four case series; three series outlined three cases,6,15,17 and one series outlined two cases. 13 The 17 cases identified in our analysis may constitute one of the largest case series of sugammadex-induced or potentially sugammadex-induced anaphylaxis. Similarly, the identification of 93 incidents in which sugammadex was used to help manage a critical incident constitutes one of the largest reports of the use of sugammadex in critical incident scenarios.
Eleven previous individual case reports have described the use of sugammadex to rescue anaphylaxis caused by rocuronium,27 –37 with two case reports showing that sugammadex failed to reverse signs of rocuronium anaphylaxis.38,39 Another case series reported on 13 cases of suspected rocuronium allergy where sugammadex was given. 40 They concluded that sugammadex does not modify the clinical course of a suspected rocuronium hypersensitivity reaction. Our findings add further support to the conclusion that sugammadex does not influence the course of anaphylaxis due to aminosteroid agents.
Four previous published case reports have outlined the use of sugammadex in a CICO event. Two of these outlined the successful use of sugammadex in an anticipated difficult airway.41,42 A third report was of the failure of sugammadex in an anticipated difficult airway, resulting in the need for FONA, 43 while a fourth report outlined only the planned use of sugammadex, with the case proceeding without sugammadex being used. 44 Two trials in critical event simulations investigated the use of sugammadex in an unanticipated CICO event.45,46 Both concluded that sugammadex could not be relied upon in these situations to prevent patient morbidity. We found nine cases where the use of sugammadex was involved in successfully rescuing a CICO event, providing low-level evidence to support the inclusion of sugammadex in failed airway algorithms. This is important, given that previous publications have questioned the usefulness of sugammadex to rescue a CICO event.45,47 This is also important across critical care specialties, as at least 20% of airway complications occur outside of the operating theatre environment. 48
Two previous case reports have outlined the successful emergency use of sugammadex to reverse residual paralysis in the PACU or ICU.49,50 Our findings provide additional data on 67 cases of residual paralysis, as well as 16 other cases where sugammadex was used in the treatment of paralysis following drug errors, or when reversal was required shortly after the administration of full doses of an aminosteroid muscle relaxant. The use of sugammadex in such incidents could save considerable time and resources.
Five previously published case reports described an event where reversal with sugammadex was considered as a possible cause of significant bradycardia,51 –53 conduction block 54 or Takotsubo cardiomyopathy, 55 and a further two cases where sugammadex administration was temporally linked to the development of bradycardia. We identified six cases where sugammadex administration was temporally linked with bradycardia and one where there was a subsequent development of Takotsubo cardiomyopathy after the use of atropine to treat the bradycardia. While not confirming causation, this raises the awareness of a potential important association being identified in multiple case reports and warranting further investigation.
As with any incident reporting study, a limitation of this work is that a lack of denominator data, which combined with the voluntary nature of reporting precludes any estimate of the rates at which particular incidents occur. Our data are from Australia and New Zealand and may not apply elsewhere. As has been reported previously in the first cross-sectional overview of the webAIRS database:
22
The value of webAIRS, as with all incident reporting systems, lies mostly in the narratives associated with each incident. It is not possible to calculate accurate incidences from data of this type. This is because reporting bias cannot be excluded. For example, it is possible that only ‘more serious’ incidents were reported to webAIRS, resulting in a skew to more patient harm and death as outcomes. Alternatively, there may have been under-reporting of the more serious outcomes, due to medicolegal concerns, despite the anonymity. The incidents reported by the currently registered sites may also not be representative of incidents in the broader anaesthesia community. Therefore, the summary data must be interpreted with caution. Nevertheless, the reports were received from a very wide range of sites across Australia and New Zealand and the confidential and de-identified nature of the reporting mechanism would have encouraged frank and unbiased reporting.
An additional limitation in relation to sugammadex anaphylaxis is the reliance on reports of clinical signs to diagnose anaphylaxis. While many of the webAIRS reports indicated that tryptase levels and further investigations would be performed, many of the cases did not include documentation of follow-up allergy testing. Therefore, the agent causing the anaphylaxis in many cases was not confirmed in the reports. The value of this report therefore lies not in incident rates but lessons to be learned in the evolution of an incident with a clinical diagnosis of anaphylaxis soon after giving a reversal drug. Whilst anaphylaxis during emergence from anaesthesia has been previously described, these reports highlight the clinical pattern observed and the actions of the reporters in managing these crises rather than the subsequent diagnosis and testing to confirm the agent. This would be best performed in a separate future study ideally performed in allergy clinics.
In conclusion, our findings from this large critical incident database indicate that sugammadex can be a potential trigger for anaphylaxis and that its use may be associated with the development of significant bradycardia. The reports also indicate that sugammadex has been used successfully to reverse residual or deep aminosteroid neuromuscular blockade in critical incident situations and to help rescue CICO scenarios. These findings provide further support for ensuring the ready availability of sugammadex wherever aminosteroid muscle relaxants are used.
Footnotes
Author Contribution(s)
Acknowledgements
We would like to acknowledge Professor Alan Merry for his manuscript review and suggestions and to acknowledge the contribution of all members of ANZTADC, past and present, and the large number of anaesthetists reporting incidents to webAIRS in the interests of greater patient safety.
Declaration of conflicting interests
MC has previously received travel grants to attend and present at meetings sponsored by Merck Sharp & Dohme.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
