Abstract
Persistent tachycardia in patients with septic shock predicts poor outcome. This study sought to investigate the effect of the cardiac pacemaker current inhibitor ivabradine on heart rate and cardio-circulatory function in patients with septic shock. After informed consent, 60 patients with septic shock and persistent tachycardia (heart rate >95 /minute) were prospectively randomly assigned to receive either standard therapy for septic shock (group S) or standard therapy along with enteral ivabradine (group I) for the initial 96 hours after enrolment. Primary outcome was the difference in heart rate between the two groups during the first 96 hours. Secondary outcomes included the effect of ivabradine on haemodynamic, oxygenation, myocardial function and organ function parameters, incidence of adverse events and 30-day overall survival. Heart rate was lower in group I compared to group S (median difference in area under the curve –25.6 (95% confidence intervals –31.4 to –15.9) /minute; P <0.001). Vasopressor requirements, blood lactate levels, Sequential Organ Failure Assessment scores and E/e′ ratio were lower in group I compared to group S. Stroke volume index and ejection fraction were higher in group I while cardiac index and oxygen delivery parameters were maintained similar to group S. There was no difference in 30-day mortality or in the incidence of serious adverse events. Enteral ivabradine is effective in reducing heart rate, and improving haemodynamic parameters and cardiac function in patients with septic shock and persistent tachycardia, without increasing the incidence of adverse events.
Introduction
Tachycardia is a key compensatory mechanism in septic shock. Many patients, however, develop refractory tachycardia, which persists despite correction of hypovolaemia with fluids and vasopressors. This may be the result of excessive sympathetic stimulation from endogenous and exogenous sources, or due to the direct effect of bacterial endotoxins and inflammatory mediators on the sinoatrial node (SAN).1–5 Evidence suggests that persistent tachycardia may be harmful for patients with septic shock.6–9
Studies have shown that beta-blockers are effective in reducing heart rate in patients with septic shock and tachycardia, without negatively effecting haemodynamics.10–18 However, the concern that beta-blockers may negatively affect cardiac contractility, and may worsen myocardial dysfunction and shock, limits their widespread acceptability.
Ivabradine is a cardiac pacemaker current (If current) inhibitor that lowers heart rate without any negative effect on cardiac conduction or contractility. Ivabradine has been evaluated for heart rate control in patients with acute decompensated heart failure and cardiogenic shock, with promising results.19–25 Although some preliminary findings suggest that ivabradine may be useful in the management of patients with septic shock, 26 , 27 clinical evidence is limited.
This study was planned with the primary objective of assessing the effectiveness of enteral ivabradine as a heart rate–lowering agent in patients with septic shock and persistent tachycardia. The secondary objectives were to assess the effect of heart rate control with enteral ivabradine on haemodynamic, echocardiographic, physiological and biochemical parameters, and on intensive care unit (ICU) morbidity and mortality, in patients with septic shock.
Methods
This was a single-centre, prospective, open label, randomised controlled trial registered prospectively in the Clinical Trial Registry of India (CTRI/2017/09/009849). The study was conducted in the multidisciplinary ICU of the All India Institute of Medical Sciences, New Delhi, after approval by the institutional ethics committee (ref. no. IECPG-181/23.08.2017). Enrolment occurred between October 2017 and February 2019. Written informed consent was obtained from the next of kin of all individual participants included in the study.
Adult patients admitted to the ICU with septic shock were assessed for enrolment after at least 24 hours of resuscitation. Patients with heart rate more than 95 /minute and requiring noradrenaline support of 0.02–0.4 µg/kg/min to maintain mean arterial pressure (MAP) more than 65 mmHg in spite of adequate volume resuscitation (central venous pressure (CVP) more than 10 mmHg and resting inferior vena cava (IVC) diameter more than 1.5 cm) were included in the study.
Patients with possible safety concerns and confounding factors were excluded:
patients with pre-existing cardiovascular disease (coronary artery disease, congestive heart failure, cardiac rhythm abnormalities and conduction defects, congenital heart disease or pacemaker in situ); patients with a history of pre-existing chronic renal failure (glomerular filtration rate less than 15 ml/min/1.73 m2); patients with severe liver dysfunction (model for end-stage liver disease (MELD) score more than 30, serum bilirubin more than 2.5 times upper limit of normal (ULN) or serum transaminases more than five times ULN); patients with known seizure disorder; patients with severe impairment in oxygenation (ratio of partial pressure of oxygen in arterial blood to fraction of inspired oxygen (PaO2/FiO2) less than 150 mmHg or central venous oxygen saturation (ScvO2) less than 70%); patients requiring high dose noradrenaline (more than 0.4 µg/kg/min) or a second vasopressor to maintain MAP more than 65 mmHg; patients with any contraindication to enteral drug administration (including patients with gastrointestinal perforation, mechanical bowel obstruction or bowel ischaemia); pregnant or lactating patients; patients less than 18 years or more than 75 years of age; patients with active bleeding; post–cardiac arrest patients; patients with haemoglobin less than 70 g/l; patients with body mass index (BMI) more than 35 kg/m2; patients requiring the use of potent cytochrome P450 3A4 inhibitors such as antifungals of the azole-type (specifically ketoconazole and itraconazole), macrolide antibiotics (specifically clarithromycin and erythromycin) and HIV protease inhibitors (specifically nelfinavir and ritonavir).
Included patients were randomly allocated in a 1:1 ratio, based on computer-generated randomisation using sequentially numbered opaque envelopes, into the following two groups:
group S (standard treatment group)—standard treatment for septic shock; group I (ivabradine group)—standard treatment for septic shock plus enteral ivabradine.
An arterial line and central venous catheter were inserted in all patients for invasive blood pressure and CVP monitoring. The point of enrolment after initial haemodynamic stabilisation was taken as time 0. Patients in group I were administered 2.5 mg ivabradine (orally or crushed and injected through the enteral feeding tube, preferably with feed) every six hours, starting at enrolment. Heart rate was assessed prior to each dose. If the heart rate was less than 70 /minute, the dose was omitted. If the target heart rate (less than 95 /minute) was not achieved after 48 hours, the subsequent doses were increased to 3.25 mg. Once a heart rate of less than 95 /minute was achieved, the dose was reduced to 2.5 mg. Ivabradine was continued until 96 hours after initiation of therapy. Beyond this period, the decision to continue ivabradine was left to the discretion of the treating intensivist. In case a patient developed severe symptomatic bradycardia (heart rate less than 60 /minute and worsening hypotension), intravenous isoprenaline infusion (0.1–0.5 µg/kg/min) was started and ivabradine was discontinued. Isoprenaline was gradually tapered and stopped once a heart rate of more than 70 /minute was achieved. Ivabradine was also discontinued if noradrenaline requirement increased to more than 0.5 µg/kg/min, if noradrenaline requirement persistently increased by more than 0.1 µg/kg/min from baseline for more than 12 hours following initiation of ivabradine, if a patient developed potentially life-threatening hepatic impairment (serum bilirubin more than five times ULN or serum transaminases more than ten times ULN) or severe impairment in oxygenation (PaO2/FiO2 less than 100 mmHg or ScvO2 less than 65%), active haemorrhage, cardiac arrhythmias or conduction abnormalities, new-onset seizures, or if any medication having potentially harmful interaction with ivabradine needed to be administered to the patient. Apart from this, ivabradine could also be discontinued at any point of time by the treating intensivist if it was felt that ivabradine therapy was harming the patient. Patients in whom ivabradine had to be discontinued at any point of time due to the above reasons were still considered a part of group I, and data collection was continued. If consent was withdrawn after enrolment, any intervention related to the study was discontinued, and the particular patient was omitted from analysis.
Fixed vasopressor and fluid protocols were followed in all patients (Appendix 1). Vasopressor requirement was expressed as a composite vasoactive–inotropic score (VIS) as described by Gaies et al. 28 (Appendix 2).
Heart rate and rhythm, peripheral oxygen saturation and invasive blood pressure were monitored continuously. Blood gas, haematological, and biochemical analyses were performed intermittently. Troponin I (TnI) and N-terminal pro-brain natriuretic peptide (NT-proBNP) levels were measured using Nano-Check (Nano-Ditech Corp., Cranbury, NJ, USA) quantitative rapid immune-chromatography assay. Myocardial function and haemodynamic parameters (including stroke volume, cardiac output and other derived parameters) were assessed using transthoracic echocardiography (TTE) by taking the average of the readings of two independent observers blinded to the group allocation. Left ventricular systolic and diastolic function was assessed by measuring left ventricular ejection fraction (LVEF) and E/e′ ratio, respectively. Assessment of right ventricular function was done by measuring tricuspid annular plane systolic excursion (TAPSE) (for details of TTE assessment protocol see Appendix 3; for details of the data collection protocol see Appendix 4).
Outcomes
Patients were followed up to 30 days after enrolment. The primary outcome was the difference in heart rate between the two groups during the first 96 hours after enrolment. Secondary outcomes included the effect of ivabradine on haemodynamic, oxygenation, myocardial function and organ function parameters, incidence of serious adverse events, need for organ support (defined as need for vasopressors, mechanical ventilation or renal replacement therapy), length of ICU stay, and 30-day overall survival.
Sample size calculation
Sample size was calculated based on a two-sided t-test, using the StatsToDo open access online calculator. Considering a 20% difference in heart rate as clinically significant, estimated standard deviation (SD) of heart rate in sample population at 25%, an alpha error of 5% and power of study at 80%, an estimated sample size of 26 per group was required. With an additional 15% safety margin for non-parametric analysis, a sample size of 30 patients in each group was chosen.
Statistical analysis
Statistical analyses were performed using GraphPad Prism v.7 and IBM SPSS v.23. The intention-to-treat principle was followed. Considering the relatively small sample size, continuous data were summarised as median with interquartile range (IQR) and difference between groups was expressed as median difference with 95% confidence intervals (CIs). To avoid multiple comparisons, the survival time adjusted area under the curve (AUC) relative to baseline was calculated for continuous variables with repeated measurements, as suggested by Matthews et al. 29 AUC was calculated relative to baseline to standardise for potential baseline imbalance and was divided by survival time to adjust for death within the initial 96 hours of the study period. AUC for each variable, and other quantitative data, were compared between the two groups using the Mann–Whitney U-test. Qualitative data were compared using chi-square tests. Thirty-day overall survival was compared by means of log-rank test and Kaplan–Meier plot. The hazard ratio for group I allocation was adjusted to age and disease severity (Sequential Organ Failure Assessment (SOFA) score at recruitment) using Cox’s regression analysis. A P-value of less than 0.05 was considered significant.
Results
Figure 1 shows the flow of participants through the various stages of the trial. Demographic characteristics of the study population have been described in Table 1. Both groups were similar with respect to baseline parameters (Table 2). The median administered dose of ivabradine in group I was 2.5 (IQR 2.5–2.5) mg every six hours.
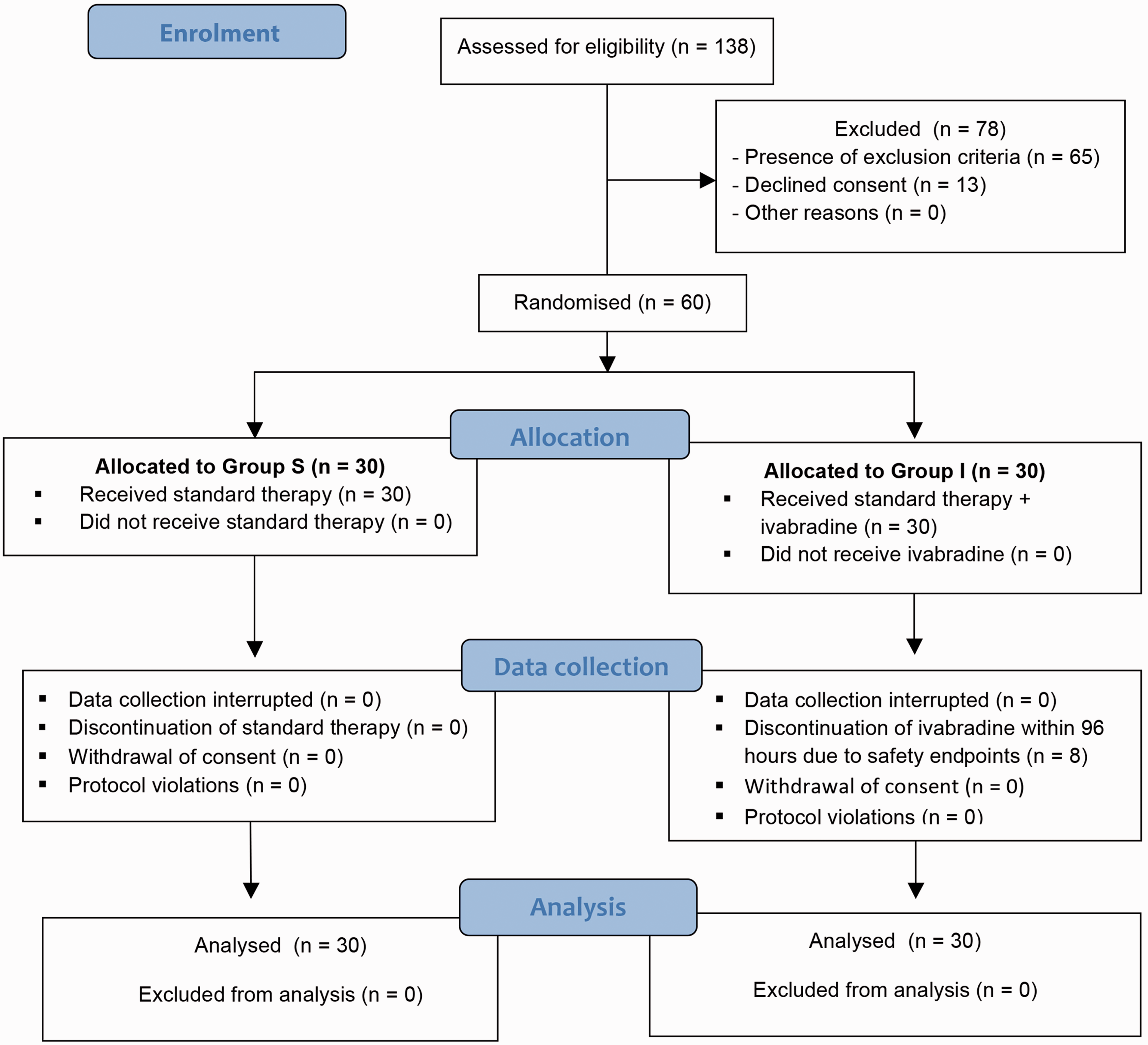
Consolidated Standards of Reporting Trials (CONSORT) diagram showing flow of patients through various stages of the trial.
Demographic characteristic of the study population.
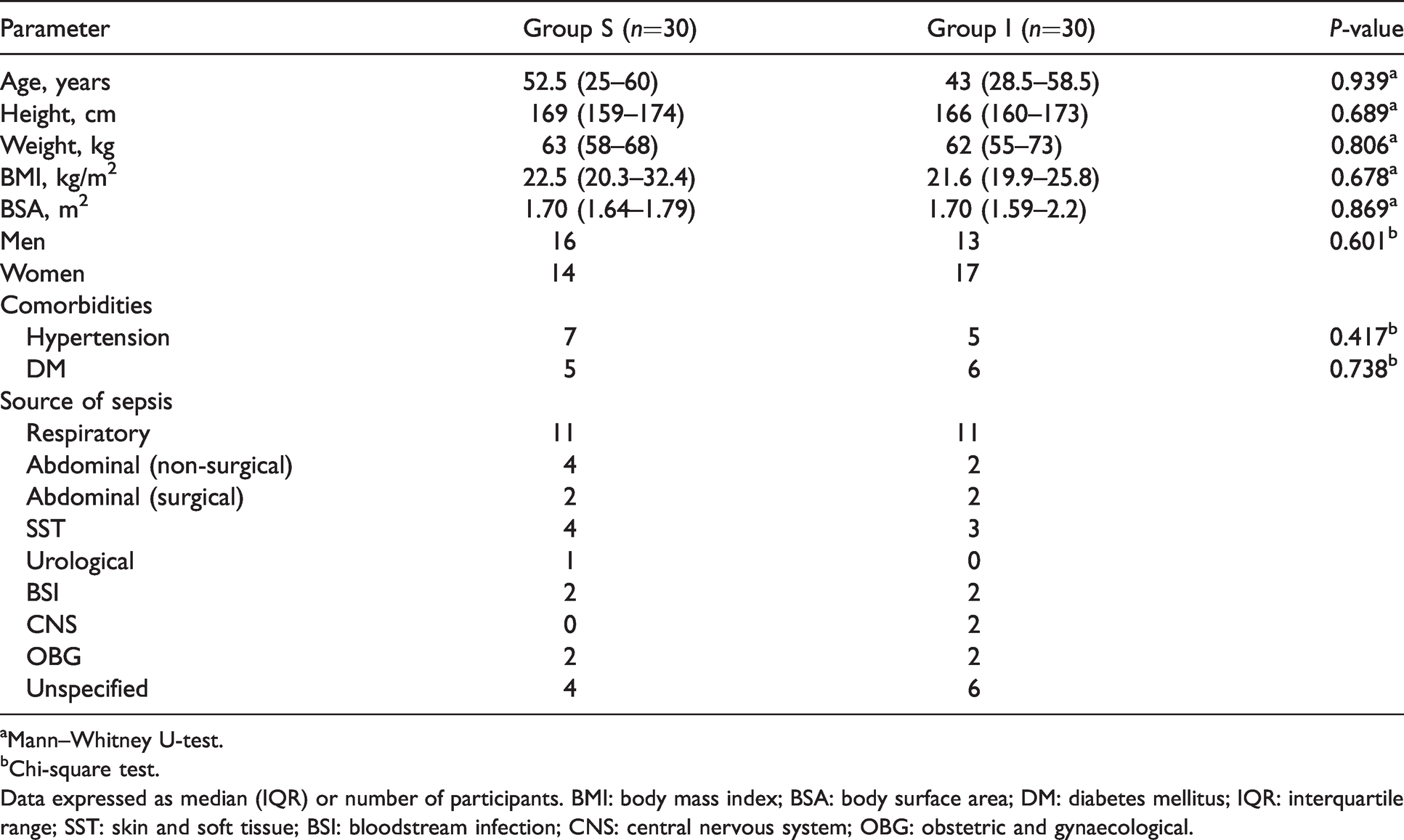
aMann–Whitney U-test.
bChi-square test.
Data expressed as median (IQR) or number of participants. BMI: body mass index; BSA: body surface area; DM: diabetes mellitus; IQR: interquartile range; SST: skin and soft tissue; BSI: bloodstream infection; CNS: central nervous system; OBG: obstetric and gynaecological.
Baseline characteristics of the study population.
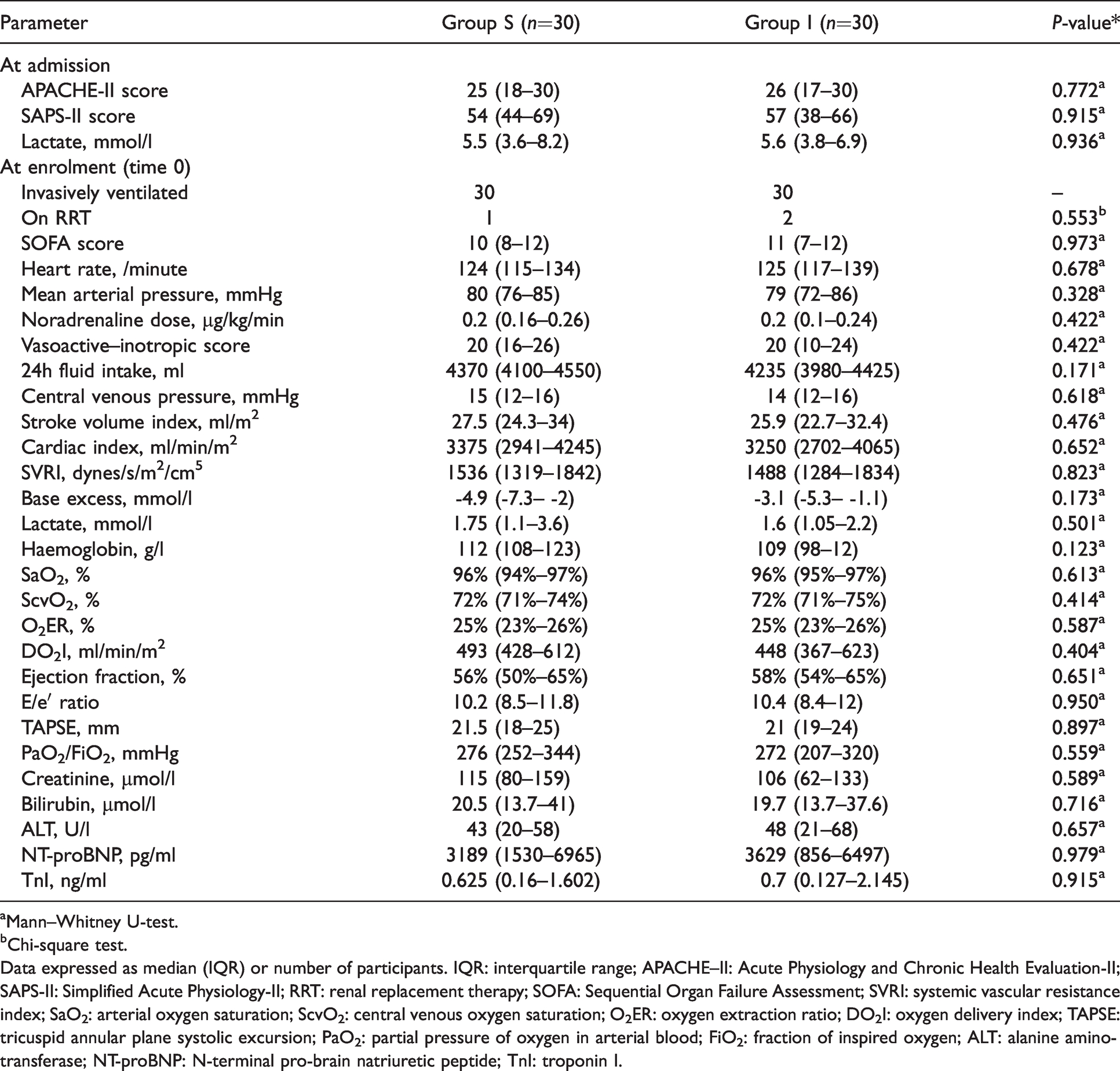
aMann–Whitney U-test.
bChi-square test.
Data expressed as median (IQR) or number of participants. IQR: interquartile range; APACHE–II: Acute Physiology and Chronic Health Evaluation-II; SAPS-II: Simplified Acute Physiology-II; RRT: renal replacement therapy; SOFA: Sequential Organ Failure Assessment; SVRI: systemic vascular resistance index; SaO2: arterial oxygen saturation; ScvO2: central venous oxygen saturation; O2ER: oxygen extraction ratio; DO2I: oxygen delivery index; TAPSE: tricuspid annular plane systolic excursion; PaO2: partial pressure of oxygen in arterial blood; FiO2: fraction of inspired oxygen; ALT: alanine aminotransferase; NT-proBNP: N-terminal pro-brain natriuretic peptide; TnI: troponin I.
Table 3 shows the trends in haemodynamic parameters during the initial 96 hours of intervention. Treatment with ivabradine resulted in lower heart rate in group I compared to group S (median difference in AUC –25.6 (95% CI –31.4 to –15.9) /minute; P <0.001; Figure 2(a)). Noradrenaline requirement was lower in group I, as was vasoactive–inotropic score (VIS) (Figure 2(b)). Stroke volume index (SVI) was higher in group I compared to group S (Figure 2(c)). There was no difference in MAP or cardiac index (Figure 2(d)) between the two groups.
Trends in haemodynamic parameters over initial 96 hours.
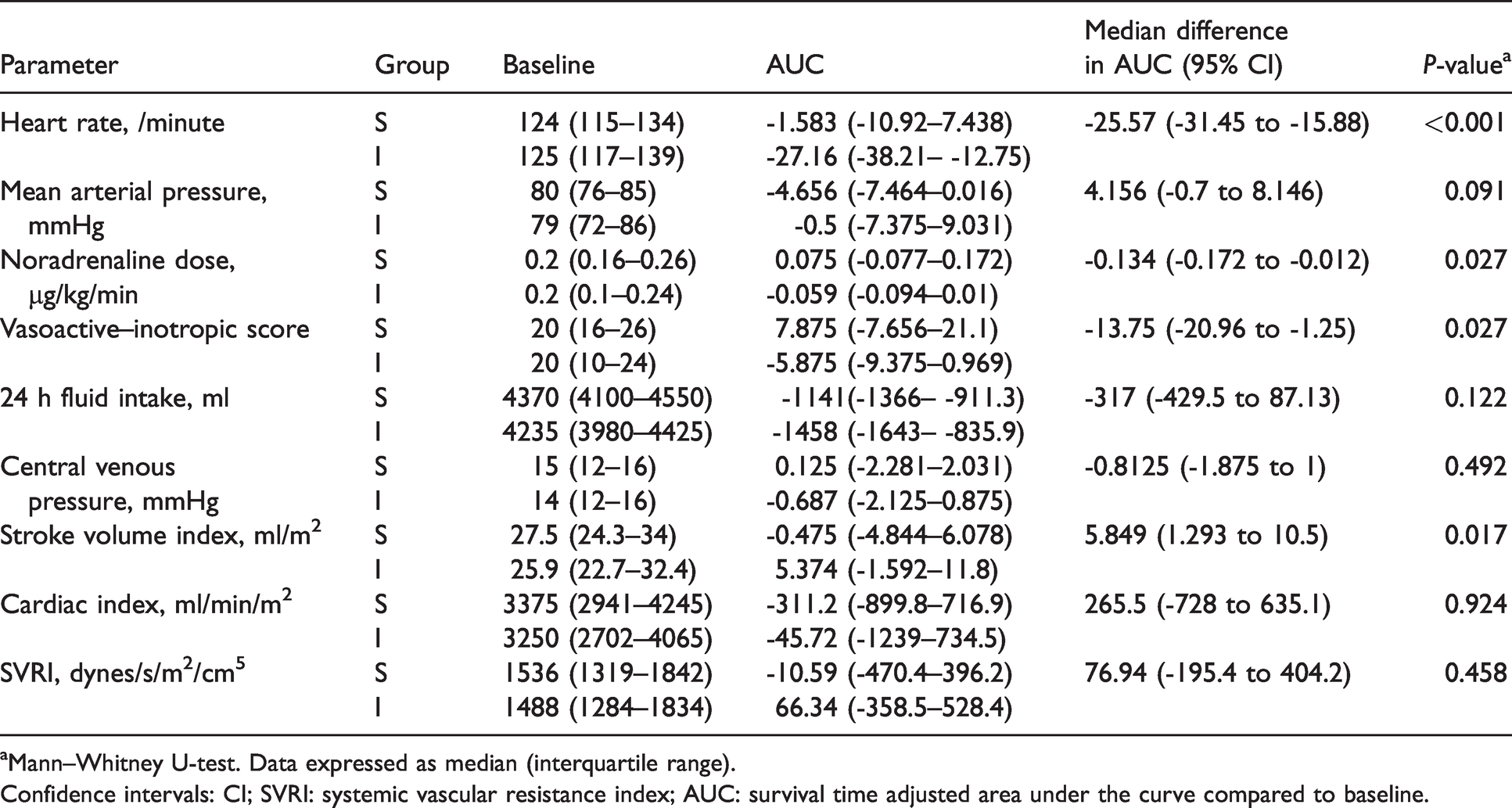
aMann–Whitney U-test. Data expressed as median (interquartile range).
Confidence intervals: CI; SVRI: systemic vascular resistance index; AUC: survival time adjusted area under the curve compared to baseline.
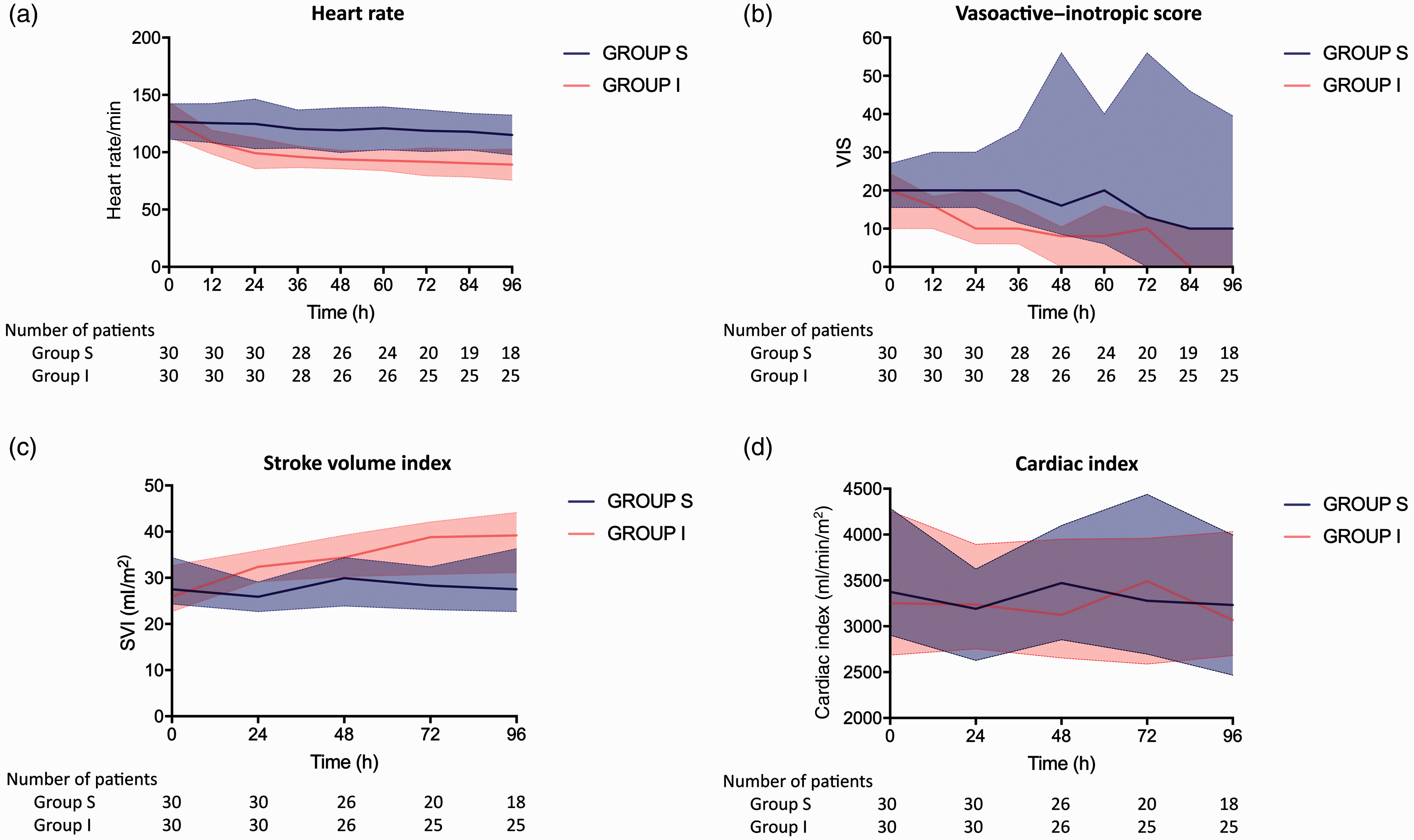
Trends in haemodynamic parameters over initial 96 hours. Solid lines represent median values. VIS: vasoactive–inotropic score; SVI: stroke volume index CI.
Table 4 shows the trends in oxygenation, microcirculatory function, myocardial function and organ function parameters. Arterial lactate levels were lower in group I. There was no difference in ScvO2 or oxygen extraction ratio between the two groups. LVEF was higher while E/e′ ratio was lower in group I compared to group S. SOFA score was lower in group I compared to group S.
Trends in oxygenation, microcirculatory function, myocardial function and organ function parameters over initial 96 hours.
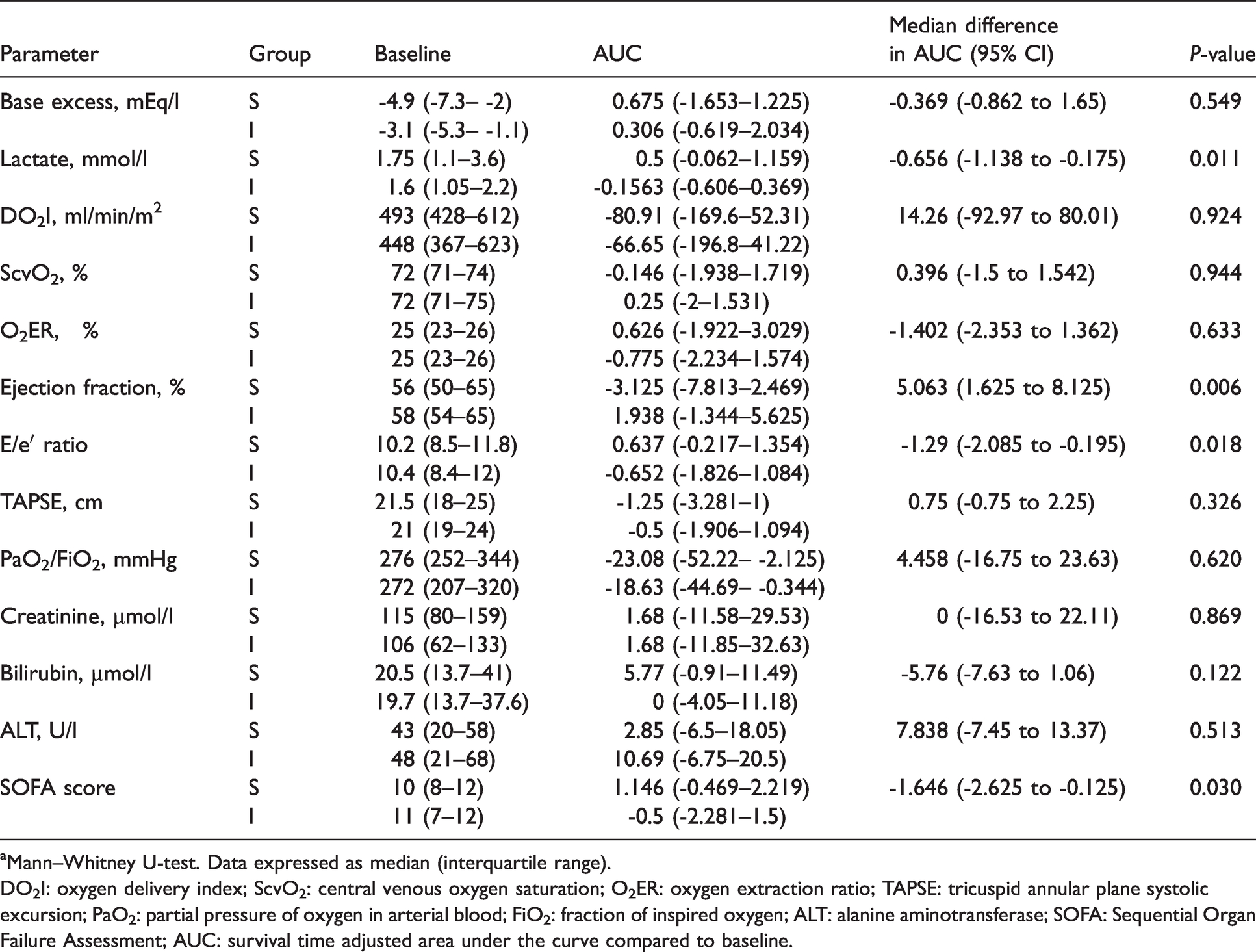
aMann–Whitney U-test. Data expressed as median (interquartile range).
DO2I: oxygen delivery index; ScvO2: central venous oxygen saturation; O2ER: oxygen extraction ratio; TAPSE: tricuspid annular plane systolic excursion; PaO2: partial pressure of oxygen in arterial blood; FiO2: fraction of inspired oxygen; ALT: alanine aminotransferase; SOFA: Sequential Organ Failure Assessment; AUC: survival time adjusted area under the curve compared to baseline.
At 96 hours after enrolment, median NT-proBNP levels were lower in group I compared to group S (group S 7715 (IQR 1918–15,000) pg/ml versus group I 2815 (IQR 444.5–8251) pg/ml; median difference –4900 (95% CI –7016 to –332) pg/ml; P = 0.021]. There was no significant difference in median TnI levels between the groups at 96 hours (group S 0.31 (IQR 0.1–2.145) ng/ml versus group I 0.18 (IQR 0.1–1.465) ng/ml; P = 0.975).
Survival at 96 hours was higher in group I (25/30) compared to group S (18/30; P=0.045). Median heart rate at 96 hours after enrolment was 90 (IQR 82–100) /minute among 30-day survivors as compared to 103 (IQR 92–122) /minute among non-survivors (P=0.085). At the end of 30 days, there was no significant difference in the number of survivors between group I (12/30) and group S (8/30; P=0.273). There was no significant difference in overall 30-day survival between the groups (Figure 3). Multivariable Cox’s proportional regression analysis of the model including group allocation, age and SOFA score at enrolment revealed an adjusted hazard ratio for group I allocation of 0.628 (95% CI 0.333 to 1.182; P=0.149). Among the survivors, there was no significant difference between the groups with respect to the median length of stay in ICU (group S 12 (IQR 7.25–23.25) days versus group I 16.5 (IQR 6.5–27.5) days) or the median number of days free from organ support within 30 days from enrolment (group S 20.5 (IQR 10.5–24) days versus group I 17.5 (IQR 9.25–25.5) days). Renal replacement therapy was required in eight patients in group S and six patients in group I during the study duration.
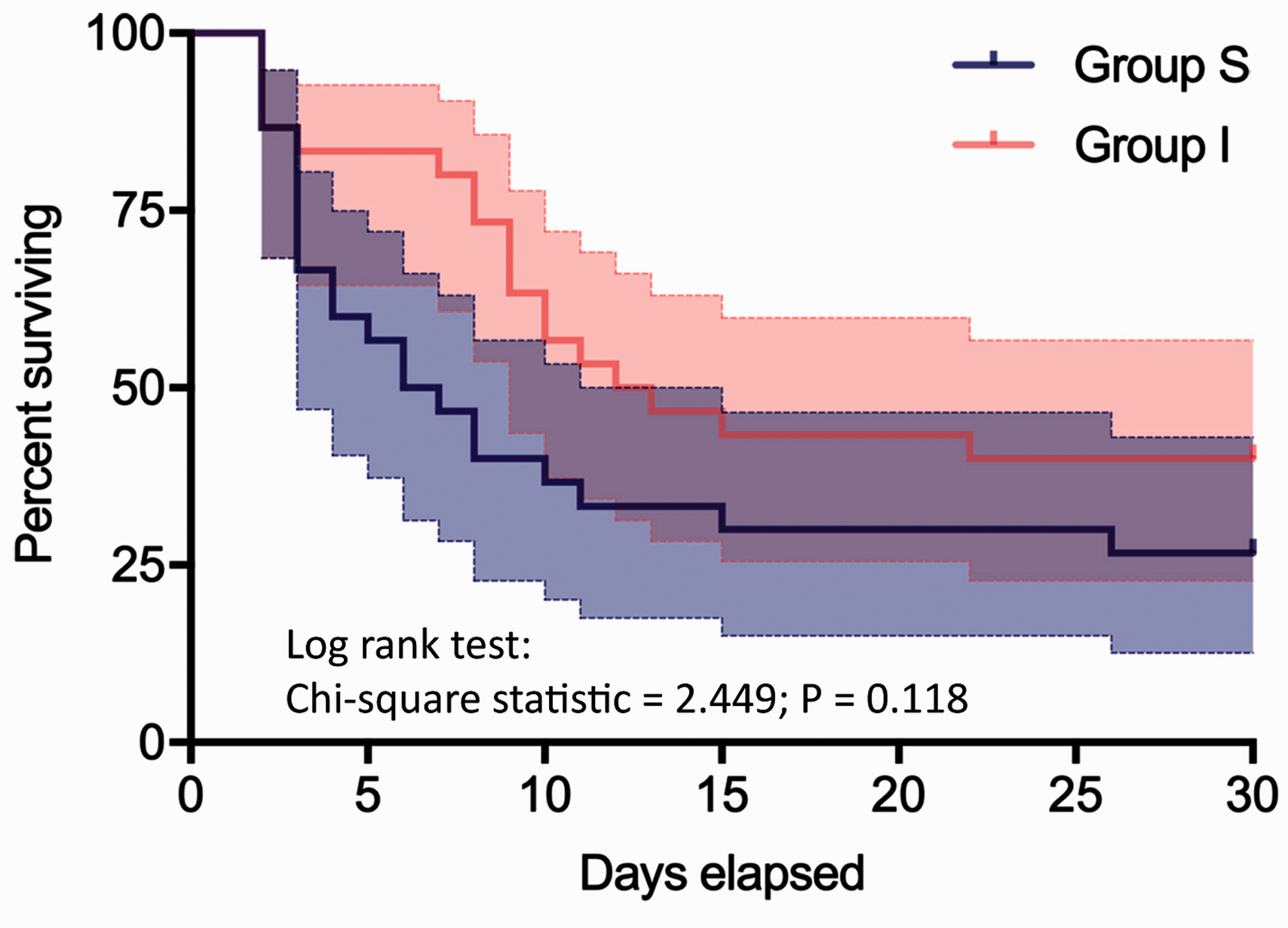
Kaplan–Meier survival analysis. Solid lines represent median values.
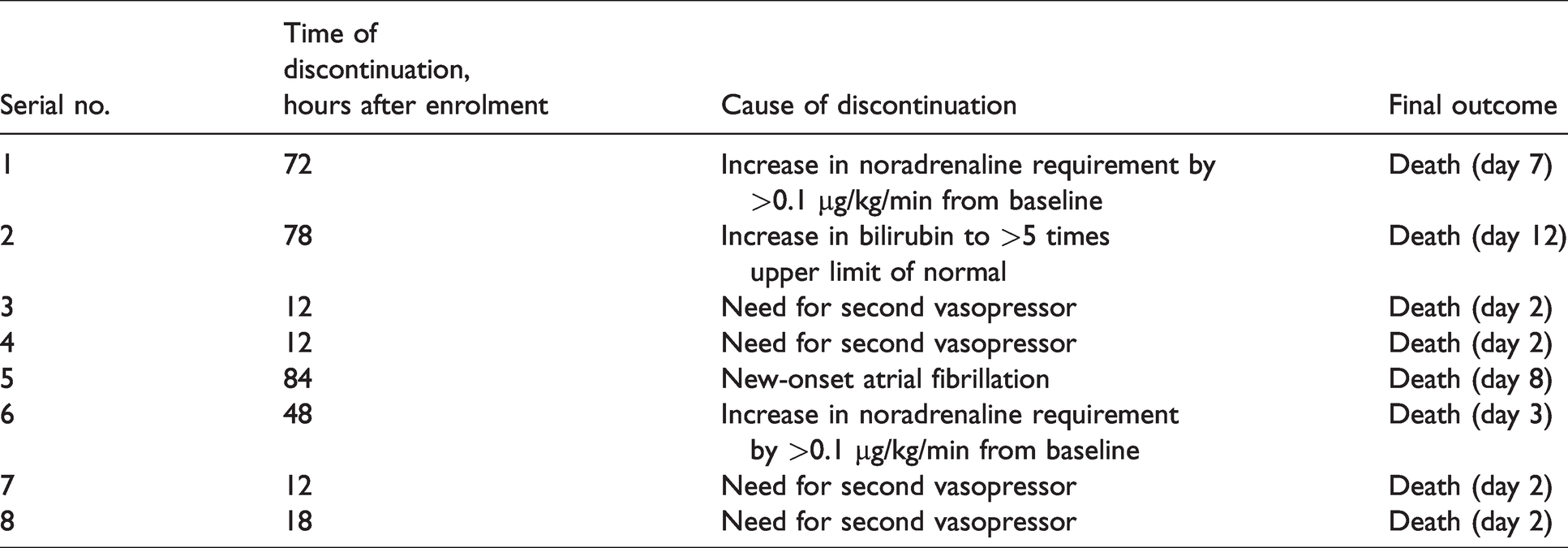
Ivabradine therapy had to be stopped within the first 96 hours of enrolment in eight patients [Table 5]. Scheduled ivabradine doses were temporarily withheld in three patients due to heart rate less than 70 /minute. None of the patients treated with ivabradine developed severe symptomatic bradycardia (heart rate less than 60 /minute with worsening hypotension) or conduction anomalies. During the study period, seven patients in group S and four patients in group I developed severe hepatic dysfunction. One patient in each group had new-onset seizures. There was no incidence of stroke in any of the patients.
Details of patients in whom ivabradine had to be discontinued within 96 hours of enrolment.
Discussion
This study shows that in a cohort of patients with septic shock and persistent tachycardia, enteral administration of ivabradine after initial haemodynamic stabilisation resulted in a significant reduction in heart rate. Ivabradine therapy was associated with lower vasopressor requirements, higher stroke volume index, similar cardiac index, improved left ventricular systolic and diastolic function, lower lactate levels, better SOFA scores and lower mortality in the first 96 hours after enrolment compared to standard treatment alone. There was no difference in 30-day survival or in the incidence of serious adverse events.
The reduction in heart rate observed in patients receiving ivabradine in the present study (median difference in AUC –26 /minute) was similar to those reported by previous investigators with intravenous esmolol: –22 /minute by Morelli et al., 14 –25 /minute by Yang et al., 15 –27 /minute by Xinqiang et al. 16 and –26 /minute by Wang et al. 17 Based on the findings of the present study, it appears that ivabradine is of similar efficacy as esmolol, with respect to heart rate reduction in patients with septic shock. A smaller, albeit statistically significant difference in heart rate (median difference –9 /minute) was reported by Nuding et al. in the MODIfY trial with the use of ivabradine. 30 The smaller effect size can probably be explained by the lower baseline heart rate (mean (SD) 106 (22) /minute) in their study population as compared to the present study (mean (SD) 128 (15) /minute).
Lower vasopressor requirement in group I compared to group S may have been the result of improved cardiac performance with heart rate control. SVI was higher in group I compared to group S (median difference in AUC 5.8 ml/m2). Morelli et al. and Xinqiang et al. have also reported higher SVI (by approximately 3 ml/m2) in patients receiving esmolol. 14 , 16 The increase in SVI may be explained by the reduction in heart rate which allows more time for diastolic filling and systolic ejection, and restores ventriculo-arterial coupling. 31 , 32 Reduction in heart rate was also associated with better left ventricular systolic and diastolic function. At the end of 96 hours, NT-proBNP levels were lower in group I compared to group S, implying less ventricular strain. The improvement in myocardial function was probably the result of better myocardial oxygen demand–supply matching. This may have helped restore the cardiac force–frequency relationship and frequency-dependent acceleration of myocardial relaxation, which have been found to be impaired in sepsis-related tachycardia. 33 , 34 Of note, there was no significant difference observed between the groups with respect to right ventricular contractile function. The right ventricle, with its smaller muscle mass and lower workload compared to the left ventricle, may be less prone to oxygen demand–supply derangements.
The net effect of lower heart rate and higher stroke volume in patients in group I was that cardiac output remained similar to patients in group S. Thus, systemic oxygen delivery remained unaffected. On the other hand a reduction in cardiac output was observed with the use of esmolol by Morelli and colleagues 13 , 14 and Yang et al. 15 It is possible that as compared to esmolol, the absence of negative inotropic effect with ivabradine helps maintain cardiac output more effectively at lower heart rates.
In the present study serum lactate levels were lower in the ivabradine group compared to the control group. A plausible explanation may be the vasopressor-sparing effect of heart rate control, resulting in decreased lactate production through aerobic glycolysis 35 and enhanced lactate clearance due to improvement in microcirculatory flow. 13 The lower SOFA scores in the ivabradine group can be primarily attributed to a lesser vasopressor requirement compared to the control group.
No significant difference in 30-day mortality was detected between the two groups. However, mortality early in the course of illness (with 96 hours of enrolment) was significantly lower in group I (16.6%) compared to group S (40%). Evidence suggests that the pathophysiology of multiple organ dysfunction in the early (within the first 4–5 days) and late phases of sepsis are different–early organ injury being mediated by unregulated inflammatory response and late insult the result of immune paralysis and increased microbial burden. 36 , 37 As tachycardia in sepsis is primarily caused by the systemic inflammatory response, it is appropriate that any intervention to control heart rate will have greater impact during the early phase of septic shock. The MODIfY trial found no significant difference in mortality at 96 hours, 28 days or 180 days with the use of ivabradine. 30 This lack of observed mortality benefit may have been because it included patients of both septic and cardiogenic shock. Overall 30-day mortality observed in the present study was 67% (40 out of 60 patients). This is similar to the 63%–68% mortality reported among patients with severe sepsis admitted in ICUs in India.38–40 The higher mortality observed in India compared to developed countries may be attributed to systemic delays in referral and admission to well-equipped centres for managing septic shock. In addition, patients with septic shock and persistent tachycardia form a subset with a higher chance of mortality.6–9
Stringent criteria for stopping the intervention in case of any clinical scenario remotely attributable to or worsened by ivabradine resulted in the discontinuation of ivabradine in eight out of 30 (27%) patients within the first 96 hours. No improvement was noted in any of these patients following withdrawal of ivabradine and all of them subsequently died. Although ivabradine was discontinued, these patients were still considered as part of group I and data collection continued as per intention-to-treat protocol. Discontinuation of the therapeutic intervention in 27% of patients in the intervention group may have diluted its net effect on the primary outcome. However, such a measure was essential from an ethical standpoint.
An important aspect of any new intervention is the additional cost associated with it. A 100 mg vial of esmolol costs approximately INR 220 (US$3.89) in India, whereas ivabradine costs approximately INR 110 (US$1.94) per strip of ten tablets, each containing 5 mg. Considering that the median dose of esmolol required, as reported by Morelli et al., was 100 mg/h, 14 the cumulative cost of 96 hours therapy with esmolol was approximately INR 21,100 (US$373) per patient. On the other hand, considering that the median dose of ivabradine required in the present study was 10 mg per day, the total cost of ivabradine therapy over four days was approximately INR 88 (US$1.56) per patient. Thus, heart rate control with ivabradine is potentially more economic than with esmolol.
The present study had some limitations. This was a single-centre study in a highly select group of patients. The study was open labelled as opposed to being blinded. Extensive exclusion criteria led to a greater number of patients being excluded than included in the study. The exclusion criteria, however, related to potential adverse effects of ivabradine and were deemed necessary to ensure patient safety. The median age of patients in group I was nine years less than group S—although this difference was not statistically significant, it may have skewed results in favour of group I to an extent. Haemodynamic parameters such as SVI and cardiac index were measured using TTE and not by indicator dilution techniques. Although the results of TTE assessment are prone to inter-observer variations, this method was chosen as it is non-invasive, inexpensive and safe. The predefined heart rate threshold of 95 /minute was chosen based on previous studies, and not individualised to the haemodynamic response of each patient. The dose range of ivabradine was limited to a maximum of 15 mg per day based on recommendations for conventional indications. Higher doses may have been effective in patients with inadequate heart rate response as enteral drug absorption is often impaired in shock. Patients with contraindications to enteral drug administration were omitted from the study. Serum levels of ivabradine could have been measured to provide a better understanding of its pharmacokinetics in patients with septic shock. However, the means thereof were unavailable. The Nano-Check (Nano-Ditech Corp., Cranbury, NJ, USA) quantitative rapid immune-chromatography assay kits used for measuring cardiac biomarkers had a measuring range of 0.1–30 ng/ml for TnI and 30–15,000 pg/ml for NT-proBNP. Values beyond this range had to be approximated to the upper or lower limits. Finally, this study was not adequately powered to detect differences in mortality or the incidence of adverse events.
In conclusion, the administration of enteral ivabradine to patients with septic shock and persistent tachycardia resulted in a significant reduction in heart rate and vasopressor requirements, and an improvement in cardiac and microcirculatory function parameters, without an increase in adverse events. Ivabradine appears to be a safe, effective and cost-effective agent for heart rate control in septic shock. Larger multicentre trials are warranted to evaluate the effect of heart control with ivabradine on clinical outcomes in critically ill patients.
Footnotes
Data Sharing Statement
Author Contribution(s)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has received financial support from the Indian Council of Medical Research, New Delhi. Grant No. 3/2/June-2017/PG-Thesis-HRD(7).
Appendix 1: Vasopressor and fluid protocol
The initial vasopressor was noradrenaline. If noradrenaline requirement increased to more than 0.5 µg/kg/min, vasopressin was started at 0.0006 units/kg/min. Vasopressin was reduced to 0.0004 units/kg/min once noradrenaline requirement decreased to less than 0.3 µg/kg/min and to 0.0002 units/kg/min once noradrenaline requirement decreased to less than 0.2 µg/kg/min. Vasopressin was stopped once noradrenaline requirement was less than 0.1 µg/kg/min. In case re-escalation of noradrenaline was required, vasopressin was increased in a similar manner. Adrenaline was started if noradrenaline requirement increased to more than 1 µg/kg/min and was titrated as per requirement and response. Hydrocortisone infusion (200 mg over 24 hours) was started if noradrenaline requirement increased to more than 0.5 µg/kg/min, and was continued until 24 hours after the patient had been weaned off vasopressors. During the first 96 hours of the intervention period, fluid challenges of 10 ml/kg were given in case vasopressor requirement escalation was accompanied with inferior vena cava (IVC) distensibility of more than 18% in paralysed or deeply sedated patients, or IVC collapsibility of more than 40% in spontaneously breathing patients, or if deemed appropriate by the treating intensivist. Packed red blood cells were transfused when haemoglobin concentration decreased to less than 70 g/l.
Appendix 2: Vasoactive–inotropic score
Vasoactive–inotropic score (VIS): dopamine dose (µg/kg/min) + dobutamine dose (µg/kg/min) + (100 × adrenaline (epinephrine) dose (µg/kg/min)) + (10 × milrinone dose (µg/kg/min)) + (10,000 × vasopressin dose (units/kg/min)) + (100 × noradrenaline (norepinephrine) dose (µg/kg/min)).
Appendix 3: Echocardiographic assessment protocol
Myocardial performance and haemodynamic parameters were assessed by transthoracic echocardiography using a 1–5 MHz phased array probe (Fujifilm Sonosite Inc., Bothell, WA, USA), with in-built software capable of automatic calculation of cardiac parameters. Left ventricular systolic function was assessed by measuring left ventricular ejection fraction (LVEF), using the modified Simpson’s method (bi-planar method of discs), by averaging the readings obtained in the apical four-chamber (A4C) and apical two-chamber views. In case one of these views could not be achieved to satisfaction, LVEF was calculated solely from the other view. In case neither of the two views was obtained to satisfaction, LVEF was calculated from fractional shortening of the left ventricular internal diameter measured using M-mode in the parasternal long axis (PLAX) view, just apical to the mitral leaflets. Left ventricular diastolic function was assessed by the measuring the E/e′ ratio. Early mitral inflow velocity (E) was measured using pulse-wave Doppler (PWD) at the opening of the mitral cusps, in the A4C view. Early diastolic tissue Doppler velocity of the mitral valve annulus (e′) was measured using tissue Doppler imaging (TDI), by taking the average of both septal and lateral mitral leaflets, in the A4C view. Right ventricular function was assessed by measuring tricuspid annular plane systolic excursion, using M-mode at the lateral tricuspid annulus in the A4C view. Stroke volume was calculating by multiplying left ventricular outflow tract (LVOT) velocity-time integral (VTI) with the LVOT cross-sectional area (CSA). LVOT-VTI was measured by taking the average of three consecutive readings during expiration, using PWD at LVOT midstream, just proximal to the aortic valve, in the apical five-chamber view. LVOT-CSA was calculated from the LVOT diameter measured just proximal to the aortic valve at mid-systole, in the PLAX view.
Appendix 4: Data collection protocol
The point of enrolment after initial haemodynamic stabilisation was taken as time = 0. The following data sets were recorded at the specified time intervals:
Enrolment data: Demographic and anthropometric data, ICU severity of illness scores at admission, probable source of sepsis and volume of fluid received in the preceding 24 hours were recorded. For intubated patients, Glasgow Coma Scale score was calculated using derived verbal score (Meredith W, Rutledge R, Fakhry SM, et al. The conundrum of the Glasgow Coma Scale in intubated patients: a linear regression prediction of the Glasgow verbal score from the Glasgow eye and motor scores. J Trauma 1998; 44: 839–844). Haemodynamic data: Heart rate, mean arterial pressure and vasopressor requirements were recorded every 12 hours from enrolment up to 96 hours. Central venous pressure, stroke volume index, cardiac index, systemic vascular resistance index and fluid intake (sum of enteral and intravenous fluid intake) were recorded every 24 hours from enrolment up to 96 hours. These measurements were recorded after ensuring patients were calm (Richmond Agitation Sedation Scale score 0 or less). Oxygenation and microcirculatory function data: Haemoglobin, arterial oxygen saturation (SaO2), delivery of oxygen index, ScvO2, peripheral oxygen extraction ratio (O2ER), base excess and arterial lactate levels were recorded every 24 hours from enrolment up to 96 hours. O2ER was calculated as (SaO2 – ScvO2)/SaO2. Myocardial function data: Left ventricular ejection fraction, E/e′ ratio and tricuspid annular plane systolic excursion were recorded every 24 hours from enrolment up to 96 hours. Troponin I and N-terminal pro-brain natriuretic peptide were measured at enrolment and at 96 hours. Organ function data: Ratio of partial pressure of oxygen in arterial blood to fraction of inspired oxygen, serum creatinine, bilirubin, alanine aminotransferase and Sequential Organ Function Assessment score were recorded every 24 hours from enrolment up to 96 hours.
