Abstract
Patient–ventilator dyssynchrony or asynchrony occurs when, for any parameter of respiration, discordance exists between the patient’s spontaneous effort and the ventilator’s provided support. If not recognised, it may promote oversedation, prolong the duration of mechanical ventilation, create risk for lung injury, and generally confuse the clinical picture. Seven forms of dyssynchrony are common: (a) ineffective triggering; (b) autotriggering; (c) inadequate flow; (d) too much flow; (e) premature cycling; (f) delayed cycling; and (g) peak pressure apnoea. ‘Reverse triggering’ also occurs and may mimic premature cycling. Correct diagnosis of these phenomena often permits management by simple ventilator optimisation rather than by less desirable measures.
Background
Mechanical ventilation of intubated patients is controlled by a combination of patient and ventilator-determined variables. Except in the case of a completely passive (i.e. deeply sedated, paralysed, or comatose) patient, effective ventilation requires a close concordance between the patient’s effort and the ventilator’s contributions. Anomalies in this harmony—where variables such as the patient’s intrinsic flow of inspiration or desired inspiratory time do not match those offered by the ventilator—produce the undesirable situation of patient–ventilator dyssynchrony.
Does this phenomenon have a patient-relevant impact? Outcome data are limited. Significant dyssynchrony (or asynchrony, an equivalent term) results in ‘bucking’ of the ventilator and frank patient discomfort, but subtle dyssynchronies are often not obvious, requiring a well-trained eye or even specialised studies such as oesophageal manometry to detect. When present, patient distress and agitation may result in increased sedation requirements, which are associated with increased delirium and intensive care unit (ICU) length of stay. 1 However, observational studies suggest that even subtle dyssynchrony is associated with mortality and an increased duration of mechanical ventilation.2–4 This may be due to a variety of effects, as diaphragmatic effort that is dissociated from effective ventilator support can fatigue the respiratory muscles, confound ventilator weaning, and even predispose to lung injury by inducing regional alveolar stress. 5 More subjectively, inability by clinicians to understand unusual patterns during mechanical ventilation lends to it a general air of mystery, obscuring its fundamental nature as a physical, deterministic, and controllable process.
Basic concepts of mechanical ventilation
Failures of synchrony are most easily recognised and categorised in the context of a systematic approach towards ventilation as a whole.
Most mechanical ventilation in the modern ICU is performed using one of several basic modes. Although nomenclature in mechanical ventilation has become highly variable, several modes are both clinically commonplace and relatively standard in terminology. The first, used for full ventilatory support, is assist control ventilation with either a volume or pressure target (denoted respectively as volume control ventilation (VCV) or pressure control ventilation (PCV)). The second is pressure support ventilation (PSV) and is used when spontaneous patient effort is encouraged. A variety of other modes now exist, such as synchronised intermittent mandatory ventilation, pressure regulated volume control, volume support, and more, but for the purposes of this discussion are merely amalgams of existing principles. Several truly novel modalities have also been introduced, usually proprietary to specific ventilators (such as proportional assist ventilation and neurally adjusted ventilatory assist), but have not yet achieved widespread adoption. Many of the same principles of invasive mechanical ventilation are also applicable to non-invasive positive pressure ventilation, with appropriate modifications and some differences in nomenclature.
Disregarding idiosyncratic modes such as airway pressure release ventilation (APRV) or oscillatory ventilation, any mechanical breath is defined by three parameters:
The The The
Similarly, four variables are at work within the ventilator to fill these parameters: delivered inspiratory volume, inspiratory
Common ventilator modes and parameters.
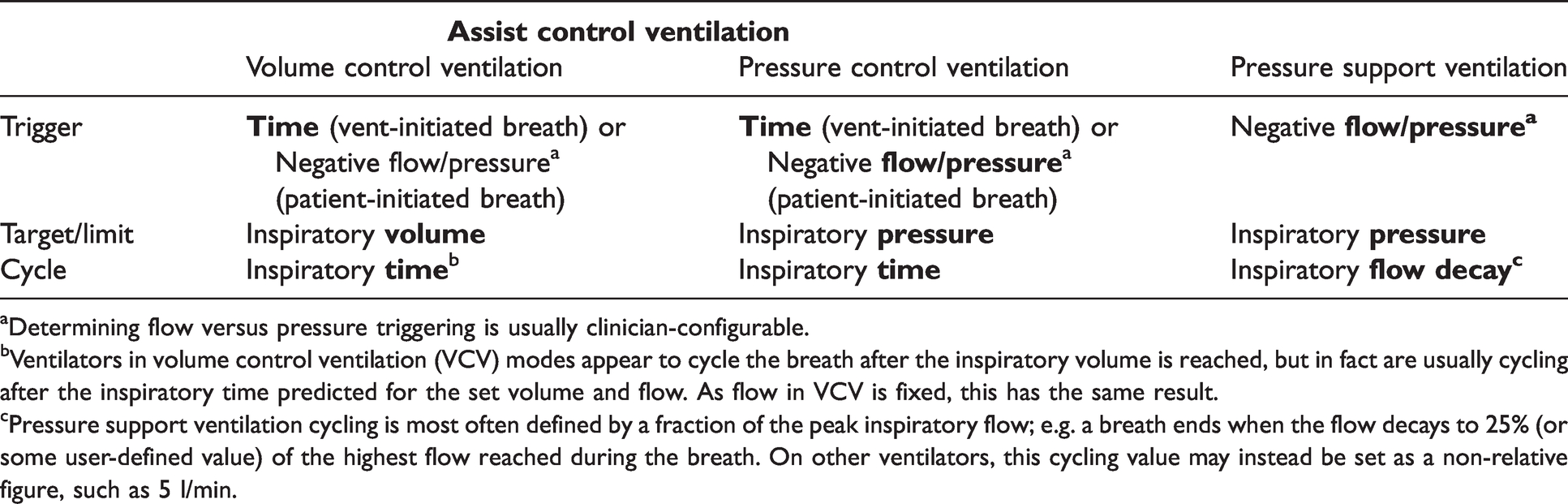
Determining flow versus pressure triggering is usually clinician-configurable.
Ventilators in volume control ventilation (VCV) modes appear to cycle the breath after the inspiratory volume is reached, but in fact are usually cycling after the inspiratory time predicted for the set volume and flow. As flow in VCV is fixed, this has the same result.
Pressure support ventilation cycling is most often defined by a fraction of the peak inspiratory flow; e.g. a breath ends when the flow decays to 25% (or some user-defined value) of the highest flow reached during the breath. On other ventilators, this cycling value may instead be set as a non-relative figure, such as 5 l/min.
With this framework, it becomes more straightforward to understand synchrony, using the following maxim: Ventilatory dyssynchrony occurs when, for any parameter of respiration, discordance exists between the patient’s spontaneous effort and the ventilator’s provided support.
An obvious corollary is that dyssynchrony occurs most often in patients with a spontaneous respiratory drive. Completely apnoeic patients, such as the deeply sedated, are usually straightforward to ventilate, because the ventilator can control the respiratory pattern with little interference from the patient. This maxim and its corollary hold true for nearly all dyssynchronies, with a few rare exceptions such as reverse triggering and ‘peak pressure apnoea’. Another consequence is that modes of ventilation which directly control more variables of ventilation are generally at higher risk of producing dyssynchrony than those which allow more control by the patient; for example, VCV is more likely to yield certain dyssynchronies (such as flow dyssynchrony) than PCV, and PCV is at higher risk than PSV.
The common types of dyssynchrony
Seven types of dyssynchrony are commonly encountered. As terminology is inconsistent, these are best recognised by their features rather than by a universal nomenclature.
Triggering dyssynchrony
These relate to breath initiation.
Flow dyssynchrony
These relate to the delivery of the breath, in which mismatch typically involves flow.
3. 4.
Cycling dyssynchrony
These relate to the ending of the breath. Also known as expiratory dyssynchrony.
5. 6.
Alarm dyssynchrony
A final idiosyncratic dyssynchrony exists, which we will denote:
7.
A clinical approach to patient–ventilator dyssynchrony
Let us consider these seven phenomena in turn. Most can be readily identified by a combination of clinical examination and inspection of the real-time ventilator waveforms, particularly the scalars depicting pressure and flow over time. All figures depicted here are derived from VCV with a decelerating ramp of flow (constant pressure), but in other modes the key features are similar.
In many cases, the most important information missing from the ventilator’s graphics is the patient’s contribution to the circuit; that is, the negative pressure introduced by spontaneous patient muscular effort (usually denoted Pmus). While this can be extrapolated to some extent, it cannot be directly measured without specialised methods, the most useful of which is oesophageal pressure monitoring. In this technique, a balloon is introduced into the oesophagus, permitting transduction via the compliant muscular walls of an oesophageal pressure that approximates the pleural pressure, which falls during spontaneous patient inspiration (Figure 1). Relatively few centres utilise this device in clinical practice, so its use is not discussed here, but its potential utility in diagnosing dyssynchrony is high, and other applications—such as titrating lung-protective ventilation in obese patients—may present as well.
Failure to trigger
(also known as ineffective triggering, wasted effort)
Causes
A majority of triggering failures occur in the setting of intrinsic positive end-expiratory pressure (‘autoPEEP’). Any combination of factors that prolong the inspiratory time constant (e.g. large tidal volumes), prolong the expiratory time constant (bronchoconstriction, fixed airway obstruction, high compliance), or shorten the time available for the latter (rapid respiratory rate) will tend to promote the presence of autoPEEP. Practically, the most common contributing factor is chronic obstructive pulmonary disease (COPD) and similar obstructive lung pathologies.3,4 The expiratory flow obstruction in these patients prolongs the expiratory time, causing mechanical breaths—whether patient or ventilator-initiated—to occur before complete exhalation of the prior breath. The result is air trapping and unintended autoPEEP.
Patient triggering on modern ventilators occurs by either a pressure or flow trigger. In the former, the patient inspires until a threshold of negative pressure is reached (e.g. –2 cmH2O), at which point mechanical inspiration is triggered. In the latter, the triggering threshold is instead set at a threshold of negative flow, such as –2 l/min.
Flow triggering is enabled by a continuous flow-by gas current (bias flow) through the ventilator circuit, usually between 2 and 20 l/min. As the patient inspires, they divert flow between the inspiratory and expiratory branches of the circuit, and this differential is noted by sensors. The relatively recent adoption of flow triggering—now the default on many ventilators—was hoped to decrease the problem of ineffective triggering, and in some cases it may do so, although the benefit seems greatest for pressure support breaths.6,7 However, even with flow triggering, the presence of intrinsic PEEP still hampers the patient’s ability to trigger, because flow can only occur once alveolar pressure becomes negative.
With a pressure trigger, on the other hand, the implications of autoPEEP are clear. If mechanical ventilation is triggered at –2 cmH2O, at rest, this requires the patient to produce a negative inspiratory force (NIF) of only 2 cmH2O. However, if 5 cmH2O of autoPEEP is present, that positive pressure must first be drawn down to zero; the total required pressure gradient to trigger a breath thus becomes –7 cmH2O. At higher levels of autoPEEP, pressure triggering becomes very difficult to achieve, particularly as a large end-expiratory volume also places the diaphragm in a flattened, less advantageous position for further inspiration. As exhalation continues, one or more breath attempts may fail to trigger until autoPEEP has dwindled enough to permit successful triggering. (Switching to PSV does not resolve this problem, because triggering in PSV is no different than patient-initiated triggering in assist control modes.) Attenuation of the maximal NIF by weakness of the respiratory muscles further exacerbates the problem, and in severe cases—such as neuromuscular disease—may cause inability to trigger even in the absence of autoPEEP.
Clinical implications
Ineffective triggering is a subtle and often unnoticed phenomenon, and is likely underappreciated. In most formal audits it is the most common type of dyssynchrony found,2,4 although it may be less ubiquitous in populations with a lower prevalence of COPD, such as surgical ICU patients. In mild cases (‘triggering delay’), a discomforting sensation of lag is created as patients exert effort on the circuit without immediately receiving a breath. In more severe cases, inspiratory effort fails to trigger a breath altogether, and dissociation is created between the patient’s intrinsic respiratory rate and the actual ventilatory rate. Either situation increases respiratory effort, induces unnecessary diaphragmatic fatigue, and may provoke patient distress leading to increased sedation; the falsely low apparent ventilatory rate also presents an inaccurate picture of the true rapid shallow breathing index (RSBI). Whatever the mechanisms, ineffective triggering is associated with a reduced chance of liberation from mechanical ventilation.3,4
Clinical signs
Patient examination (by inspection or palpation with a hand on the chest and abdomen) may reveal respiratory effort which is not followed by a ventilator breath. This may manifest as flexing of the abdominal muscles, retraction of the intercostal spaces, or paradoxical thoracoabdominal movement. Unfortunately, such clinical findings are usually subtle and in many cases may not be appreciable.
Waveform manifestations
Small perturbations are visible, appearing negative in the pressure tracing, but positive in the flow tracing (Figures 2 and 3). In minor cases, these are actually followed by a breath, but only after undesirable delay; in moderate cases, they intermittently fail to ‘‘trigger’’ a subsequent breath; in severe cases they rarely or never trigger a breath, and all breaths are triggered instead by a time trigger. They are therefore similar in significance to P waves in the setting of heart block.
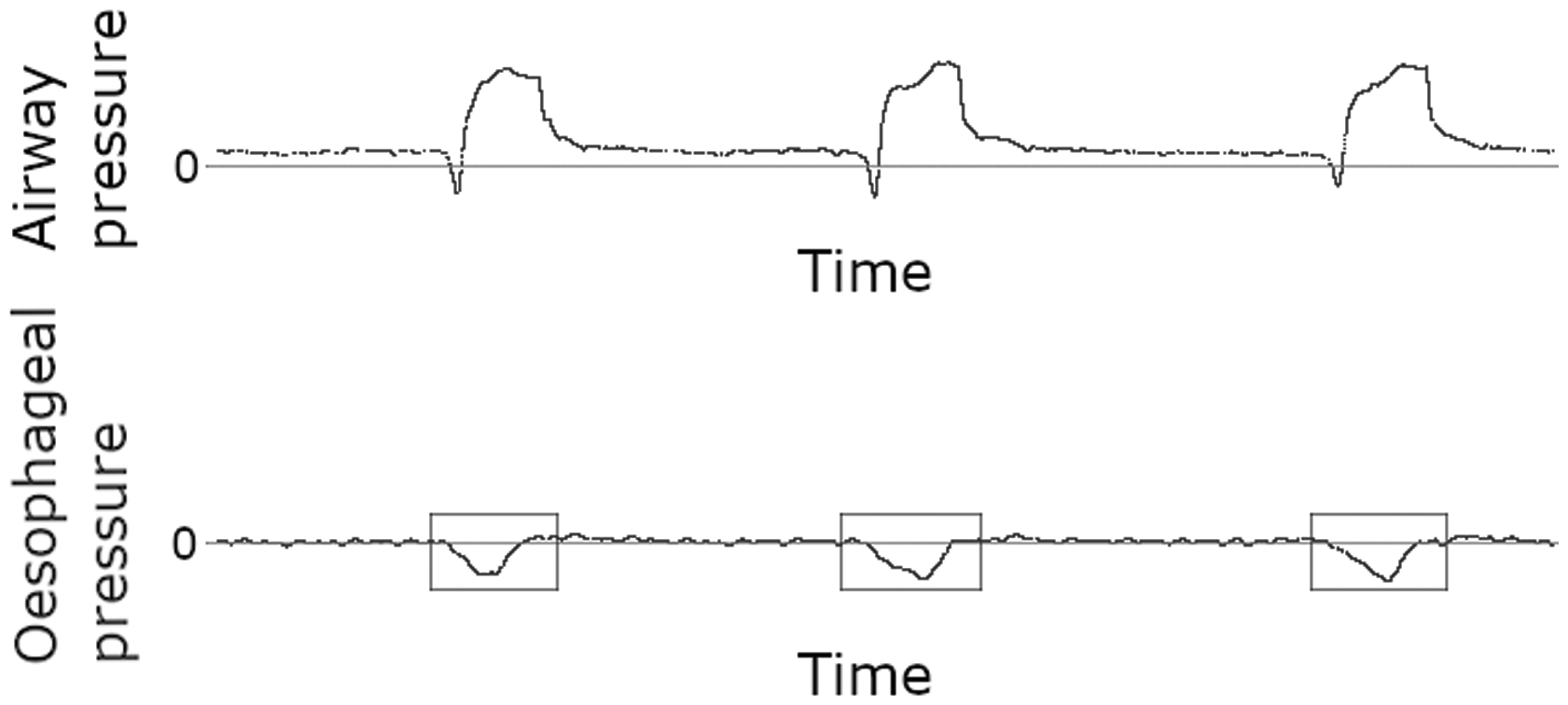
Simultaneous tracings of airway and oesophageal pressures. Negative deflections of the latter (boxes) prove the presence of muscular effort during each breath. (Source image courtesy of Elias Baedorf-Kassis, personal files)
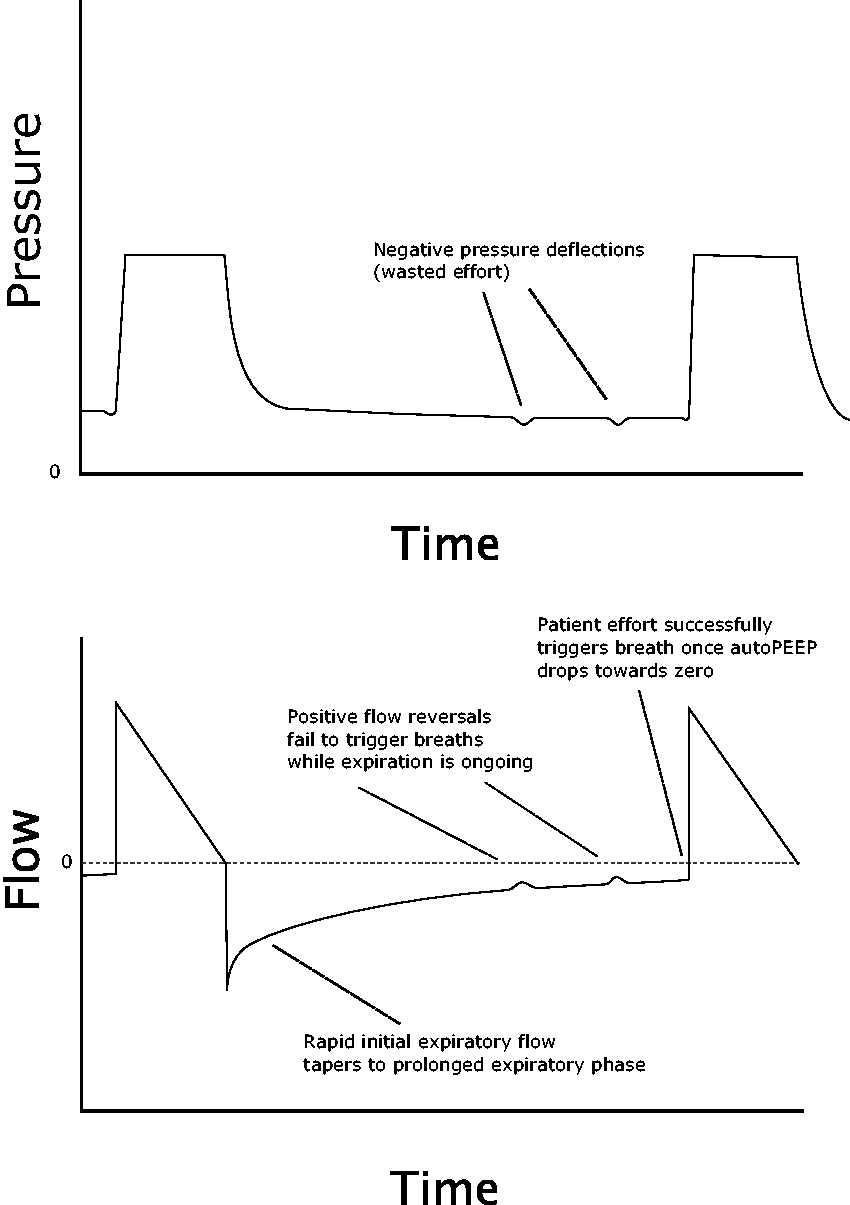
Multiple failed efforts before finally triggering a successful breath. Note the prolonged expiratory phase with resulting intrinsic positive end-expiratory pressure (autoPEEP).
Potential remedies
Reduce intrinsic PEEP: AutoPEEP is present in many cases of ineffective triggering, and should be managed by the usual methods: increase the expiratory time, decrease the inspiratory time, and decrease the respiratory rate. One method of decreasing inspiratory time (and hence increasing available expiratory time) in VCV modes is by increasing the inspiratory flow rate, which can be modestly effective; however, this should be done with caution, as higher flow rates may secondarily increase the spontaneous respiratory rate, for reasons that are not clear. 8 Improving triggering by reducing autoPEEP may also increase the effective respiratory rate due to the higher number of successful breaths, which may in turn tend to worsen autoPEEP. In such cases the goal should be effective triggering at a reasonable rate. Finally, the most definitive method of reducing autoPEEP is by addressing the underlying airway resistance, such as treatment of obstructive disease by bronchodilators and corticosteroids, or removal of an undersized, kinked, or obstructed endotracheal tube.
Reduce inspiratory pressure: Ineffective triggering is associated with higher levels of inspiratory pressure (in PCV/PSV modes), perhaps due to increased autoPEEP in the presence of larger tidal volumes. 4 Reducing excessive amounts of support may improve triggering. 9
Add extrinsic PEEP: If intrinsic PEEP is present and cannot be eliminated, increasing the ventilator PEEP can reduce the work of triggering assisted breaths, because the patient will only need to draw down inspiratory pressure to the level of extrinsic PEEP, not to zero. If autoPEEP can be measured accurately with an end-expiratory pause manoeuvre, machine PEEP can be set at approximately 75% of the measured autoPEEP, a figure which should limit the work of triggering without adding work of expiration and hence worsening autoPEEP (extrinsic PEEP will not alter expiratory flow until it exceeds intrinsic PEEP). 10
Reduce trigger thresholds: Shrinking the pressure or flow trigger towards zero will increase ease of triggering (although patients will still need to generate enough negative pressure to cross the baseline). However, very sensitive triggers may result in autotriggering, the inverse problem, which will paradoxically increase the respiratory rate and may worsen autoPEEP; titration should therefore be performed with caution.
Promote respiratory drive: Greater muscular strength and respiratory drive will increase the probability of successful triggering, even in the presence of obstacles such as autoPEEP. This can be promoted by reduced sedative and opioid use, avoidance of hyperventilation or overoxygenation, and scaled breathing exercise for patients with diaphragmatic weakness.
Switch triggering type: If a pressure trigger is in use, it can be switched to flow triggering (the trigger threshold should then be adjusted to maximise appropriate triggers and minimise autotriggering). Novel trigger types making use of neurally adjusted ventilatory assist or oesophageal pressure manometry may be particularly helpful, but are unavailable in most centres.
Autotriggering
(also known as accidental triggering)
Causes
As discussed above, autotriggering is exacerbated by sensitive trigger settings and is particularly common with flow triggering. In that context, and in the absence of any intrinsic PEEP, small artefacts in the circuit may easily stimulate breaths. The most common sources are:
Cardiac oscillations: Pressure waves transmitted from robust cardiac contractions.
11
Fluid in the circuit: Particularly common with actively humidified circuits, which experience condensation (‘rainout’); this collects at dependent points of the circuit (often near the wye), and its sloshing can produce pressure fluctuations. Air leaks: Active loss of gas from the circuit can generate a continuous negative flow, causing repeated autotriggers.
12
Clinical implications
Autotriggering can produce respiratory rates in excess of both patient comfort and respiratory physiology. Respiratory alkalosis may result. As tachypnoea can lead clinicians to infer patient distress, autotriggering may also indirectly cause increased sedation and failed breathing trials, all tending to prolong the duration of mechanical ventilation. The false appearance of spontaneous breathing may also confuse the picture of brain death. 13
Clinical signs
Autotriggering can be difficult to appreciate. The main clinical feature is an unusually rapid respiratory rate. Less specific although highly sensitive is a respiratory rate in excess of the set ventilator rate, despite the presence of neuromuscular blockade. Inspection of the ventilator circuit may reveal dependent fluid collections or an air leak.
Waveform manifestations
A jagged or irregular waveform may be noted, particularly on the pressure tracing; in the case of cardiac oscillations, this may correspond to the heart rate (Figures 4 and 5).
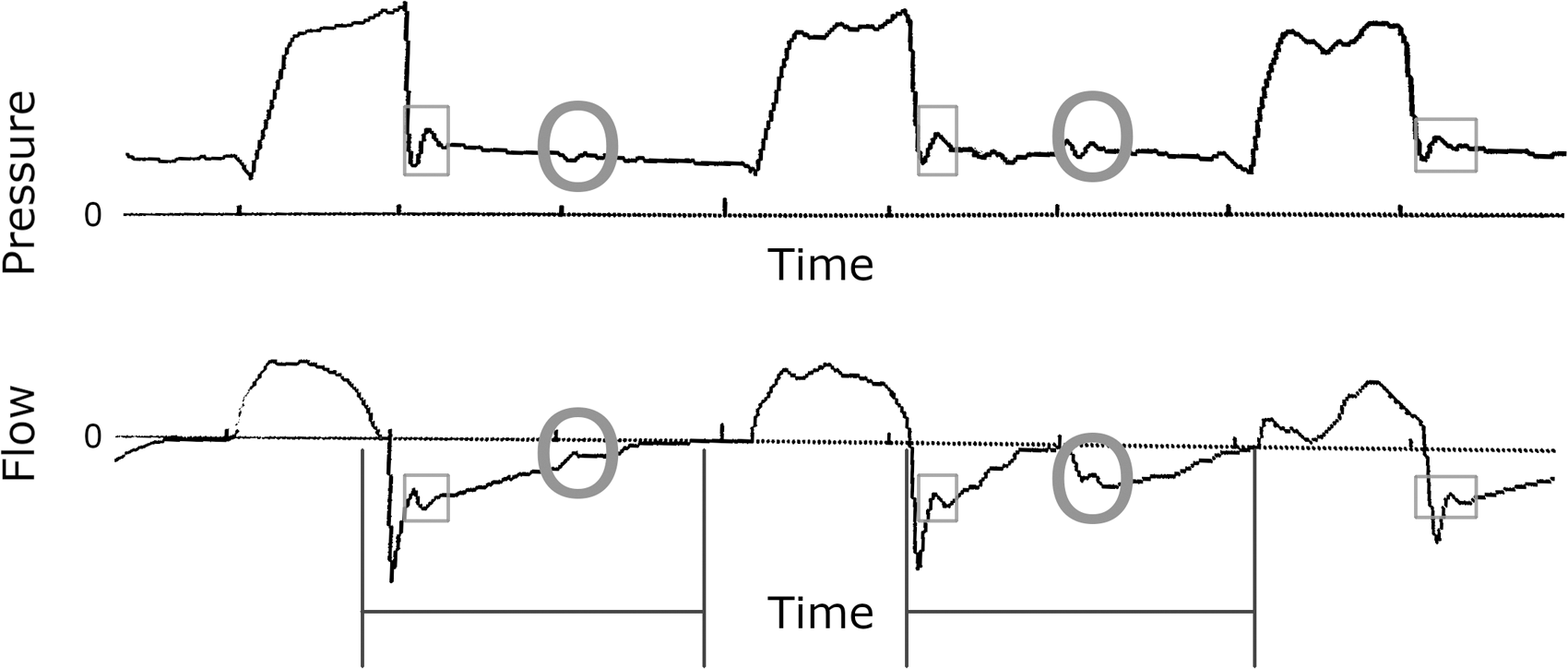
Pressure (top) and flow (bottom) scalars showing in vivo examples of ineffective triggering. Here the wasted efforts are mainly visible on the pressure tracing, and the effort that finally does trigger is strong enough to result in flow starvation for the ensuing breath (see Inadequate flow).
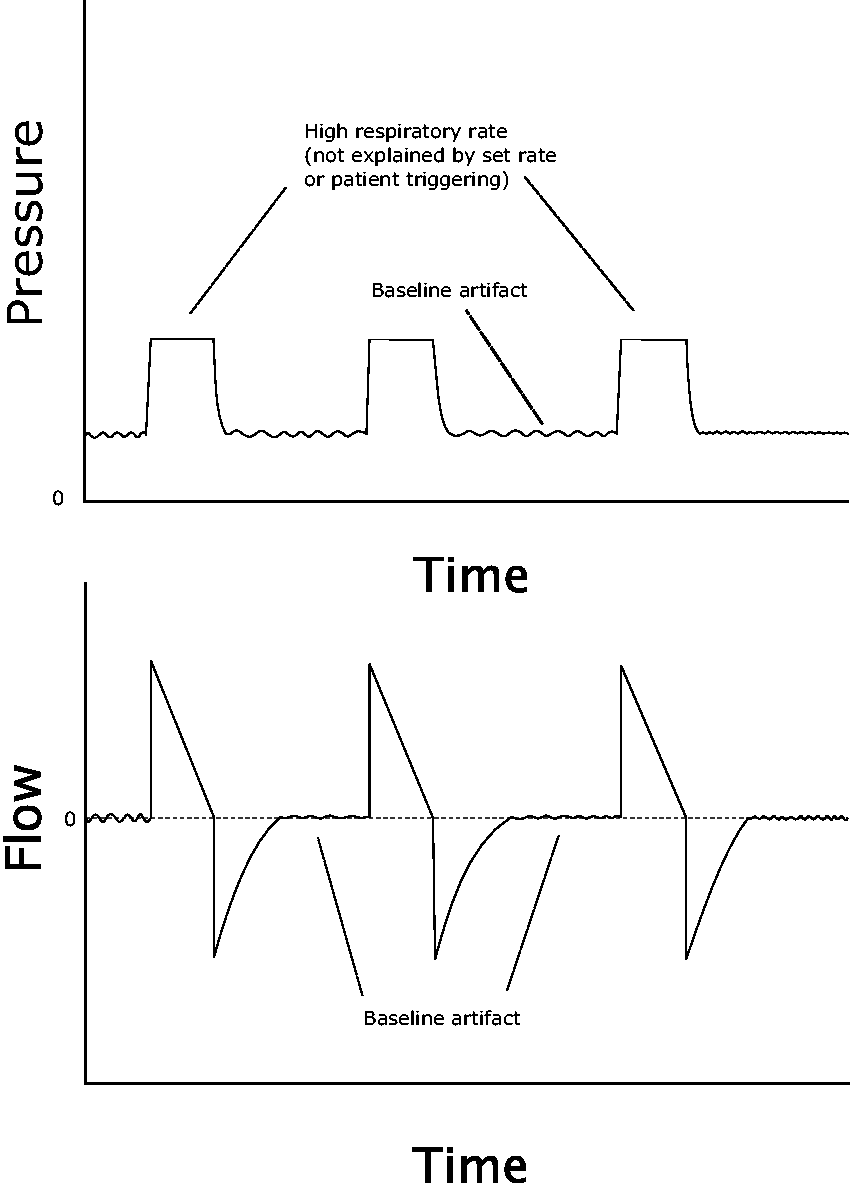
Autotriggering caused by a continuous artefact such as cardiac oscillations.
Potential remedies
Eliminate the source of artefact: Address leaks or drain excess fluids (cardiac oscillations usually cannot be eliminated). Reduce trigger sensitivity: Trigger threshold can be increased gradually until the contributory artefact no longer triggers breaths. Switch to a pressure trigger: If flow triggering is in use, switching to a pressure trigger may be attempted.
Inadequate flow
(also known as flow starvation, work shifting)
Causes
A spontaneous patient-initiated breath has a natural flow rate associated with it. The ventilator, however, will provide flow at a rate which is either fixed—as in VCV—or variable and patient determined, as in PCV or PSV. In the case of VCV, the set flow is often set lower than the patient’s desired rate, resulting in dyssynchrony.
In pressure modes, flow dyssynchrony is less common, because the ventilator adjusts flow dynamically to match patient effort, using flow as a free variable to achieve the manually set inspiratory pressure. However, a finite amount of time is required to build airway pressure, and initial flow can be increased faster or slower to reach the set pressure; because flow is itself a rate of change, this ‘rate of rate’ is known as the rise time (sometimes denoted ramp time, pressure slope, pressurisation rate, inspiratory percentage, or other names) and can be set too slow, creating flow dyssynchrony.14,15
Clinical implications
Flow starvation generally occurs in patients with a vigorous respiratory drive and strong muscles of inspiration. For many clinicians, the picture of a tachypnoeic patient with obvious distress and bizarre ventilator waveforms will prompt an increase in sedation, with all the associated harms. If such patients can be made comfortable by ventilator adjustments alone, they can be kept awake and transitioned more rapidly towards extubation.
Significant flow deficit also denotes a failure of the basic goals of mechanical ventilation (‘work shifting’), because it indicates a failure to unload and rest the patient’s respiratory muscles adequately, which may increase oxygen consumption, challenge efforts at liberation, and stimulate patient discomfort. 15 It may promote lung injury from barotrauma, as strong, unmatched negative pleural pressure establishes a high transpulmonary pressure gradient, even in the setting of a modest intra-alveolar pressure.
Clinical signs
In the setting of inadequate flow, patients will successfully trigger spontaneous breaths, yet can nevertheless be observed (on inspection and palpation) to exert continuous respiratory effort, actively pulling at the ventilator throughout inspiration. Paradoxical motion of the chest and abdomen are common, and patients are often tachypnoeic and uncomfortable in appearance.
Waveform manifestations
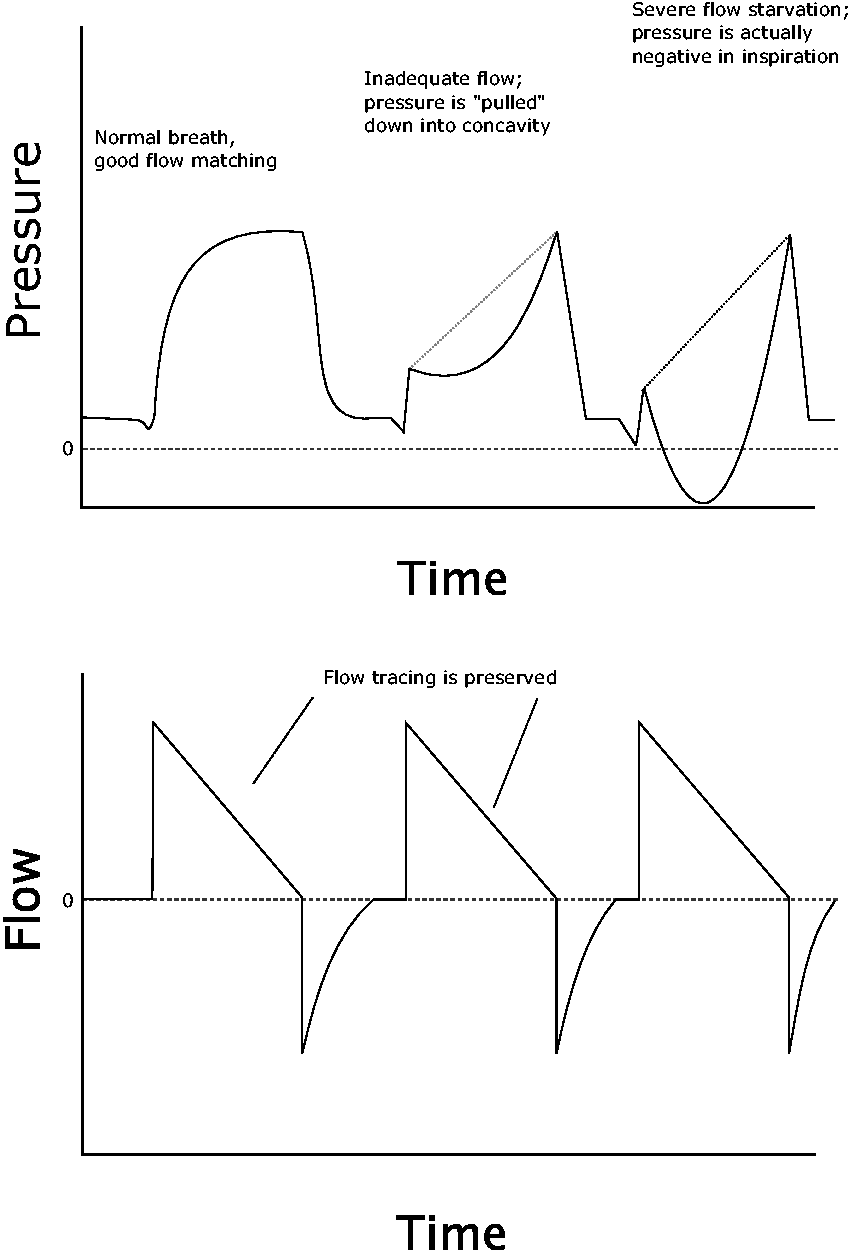
The pressure scalar should be carefully inspected (Figures 6 and 7). A normal waveform has the appearance of a flat or gently domed structure. In the case of flow starvation, the patient is attempting to inspire faster than the flow provided, pulling the airway pressure more negatively. In mild cases, the pressure curve becomes flattened and then upwardly concave. In severe cases, that concavity drops below the baseline, indicating a negative airway pressure and outright flow starvation.
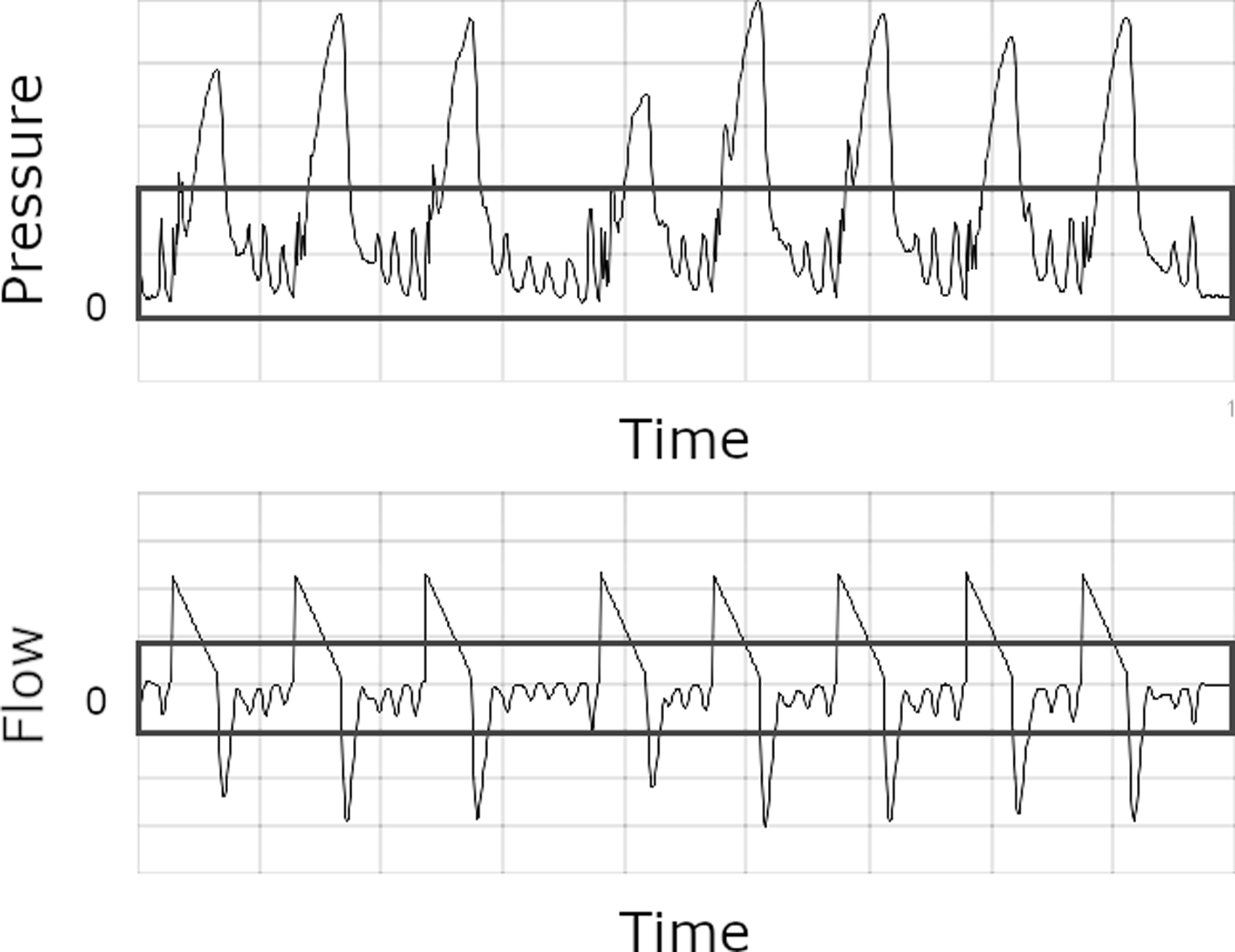
In vivo example of autotriggering from a continuous artefact such as cardiac oscillations (boxes).
Three breaths with increasing flow mismatch. Note these are all patient-triggered breaths (proved by the negative deflection at the start of each inspiration), and hence this is not an example of poor compliance (positive ‘stress index’). The flow curve is unchanged, because inspiratory flow is fixed in volume control ventilation (VCV), regardless of patient factors.
This concave waveform may easily be mistaken for the scooped-out appearance that develops in passive breaths when tidal volume exceeds lung compliance; this has been described as a ‘stress index greater than 1’ and may be associated with volutrauma. 16 However, that phenomenon occurs during passive breaths, whereas flow starvation occurs during active patient effort. Although the concave appearance may be virtually identical in both, the distinction can be easily made by either observing the patient for effort, or by noting on the ventilator whether the breath was triggered by the patient or by the passive time trigger.
Potential remedies
In volume control: Increase set inspiratory flow until the pressure waveform flattens out. If unable to match patient flow successfully (e.g. if effort is variable or very brisk), consider switching to a pressure mode.
17
In pressure control or pressure support: Shorten rise time.
14
If patient effort is pathological (i.e. resulting in unnecessarily large tidal volumes and minute ventilation): Attempt to resolve the cause of increased respiratory drive, such as managing pain and distress, controlling fever, etc. If this cannot be achieved, increase sedation.
Too much flow
(also known as pressure overshoot, flow overshoot)
Causes
This phenomenon is relatively unusual, as many clinicians are more likely to set flow rates too low than too high. However, if flow on VCV has been titrated up to prevent flow starvation, overcompensation may sometimes occur. In pressure modes, the same phenomenon can occur with excessively quick rise times, particularly when inspiratory pressure is also set very high.
Clinical implications
Excessive flow is not always pathological, but several undesired consequences are possible. First, it may result in patient discomfort. Second, it can affect cycling in PSV, in which the cycle-off parameter is typically defined by flow decay from the peak inspiratory flow (i.e. the breath ends when flow drops to a certain percentage of the peak flow, such as 25%). If that peak flow is artificially high—even for a brief ‘spike’, as in the case of flow overshoot—it will configure the cycle-off flow threshold too high as well. The breath will therefore be artificially shortened, resulting in premature cycling (see below) and inadequate tidal volumes.
Finally, a high flow rate has an independent in vivo effect of reducing neural inspiratory time and hence increasing respiratory rates. 8 This may result in inappropriate tachypnoea.
Clinical signs
Excessive flow may present with patient discomfort or the appearance of gagging.
Waveform manifestations
An early ‘spike’ or peak may be seen on the pressure scalar, as initial flow is delivered faster than respiratory compliance can accommodate it. The inspiratory duration may also be unusually short in VCV or PSV modes (Figures 8 and 9).
In vivo example of inadequate flow; note concave pressure curves (boxes).
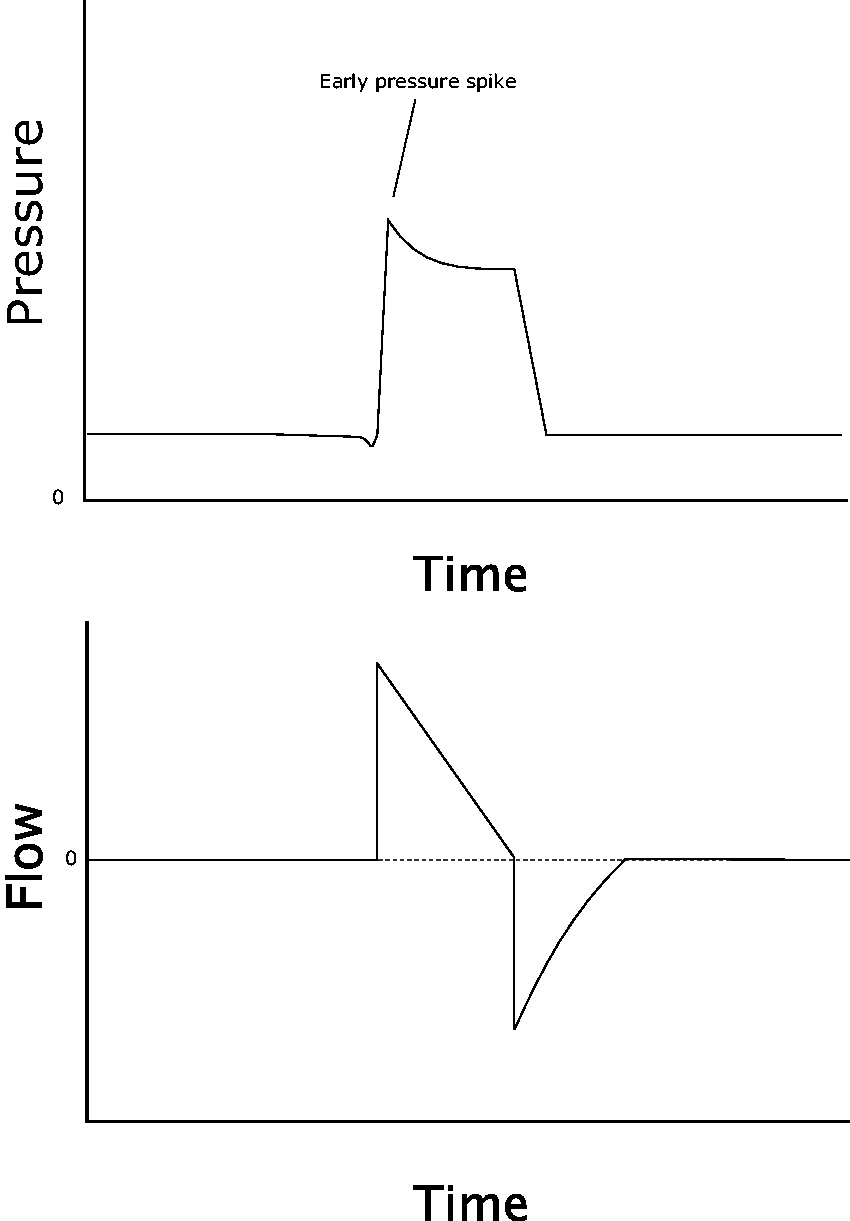
Initial flow overshoot creates an early spike or peak at the start of inspiratory pressure.
Potential remedies
Volume control: Decrease set flow. Pressure control/Pressure support: Lengthen rise time.
Premature cycling
(also known as short cycling, double triggering, breath stacking)
Causes
Premature cycling occurs when the neural (patient) I-time exceeds the machine I-time. In this phenomenon, inspiratory time, volume, and flow interact in important ways. In VCV, for instance, premature cycling can be correctly conceived as an inappropriately short I-time, but it may be easier to understand as an inappropriately small tidal volume; as machine I-time in VCV is determined by the volume and set flow, these amount to the same thing. Whatever the case, premature cycling in VCV is usually due to a patient seeking larger tidal volumes, and often occurs when small volumes are intentionally used for lung protection.
In PCV, premature cycling may have the same cause; however, as machine I-time is manually set, adjusting it is particularly straightforward.
In PSV, premature cycling may occur due to flow overshoot (as noted above) or an inappropriately set flow-cycle threshold.
Clinical implications
As with most dyssynchrony, premature cycling can result in patient discomfort and agitation. However, a more important consequence is seen when the patient’s effort to prolong the breath results in ‘double triggering’, or stacking of mechanical breaths. This creates an effective tidal volume and inspiratory pressure far in excess of the set parameters—as much as double the set volume—which may predispose to lung injury.
Clinical signs
Premature cycling is usually associated with strong patient effort, so patients will often be visibly roused and tachypnoeic.
Waveform manifestations
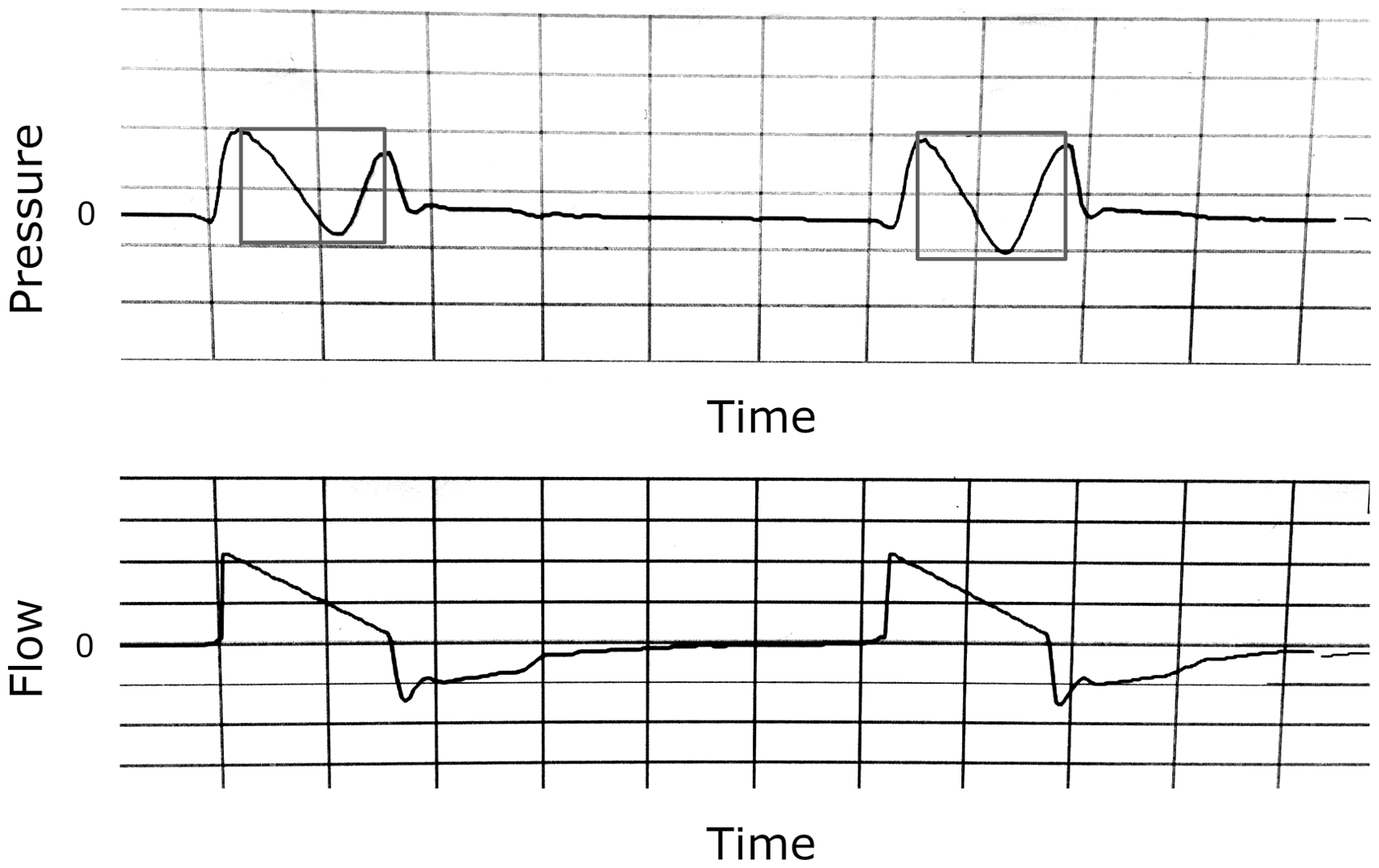
In minor cases, the patient’s effort continues beyond the ventilator’s, but does not trigger an additional breath (Figures 10 and 11). This can be seen as a positive deflection on the flow scalar at the start of expiration, as the patient continues to inspire after the ventilator has initiated exhalation. More subtly, the corresponding pressure scalar may show a sudden, near-vertical drop-off in pressure, rather than the more gentle downslope seen on regular inspiratory termination.
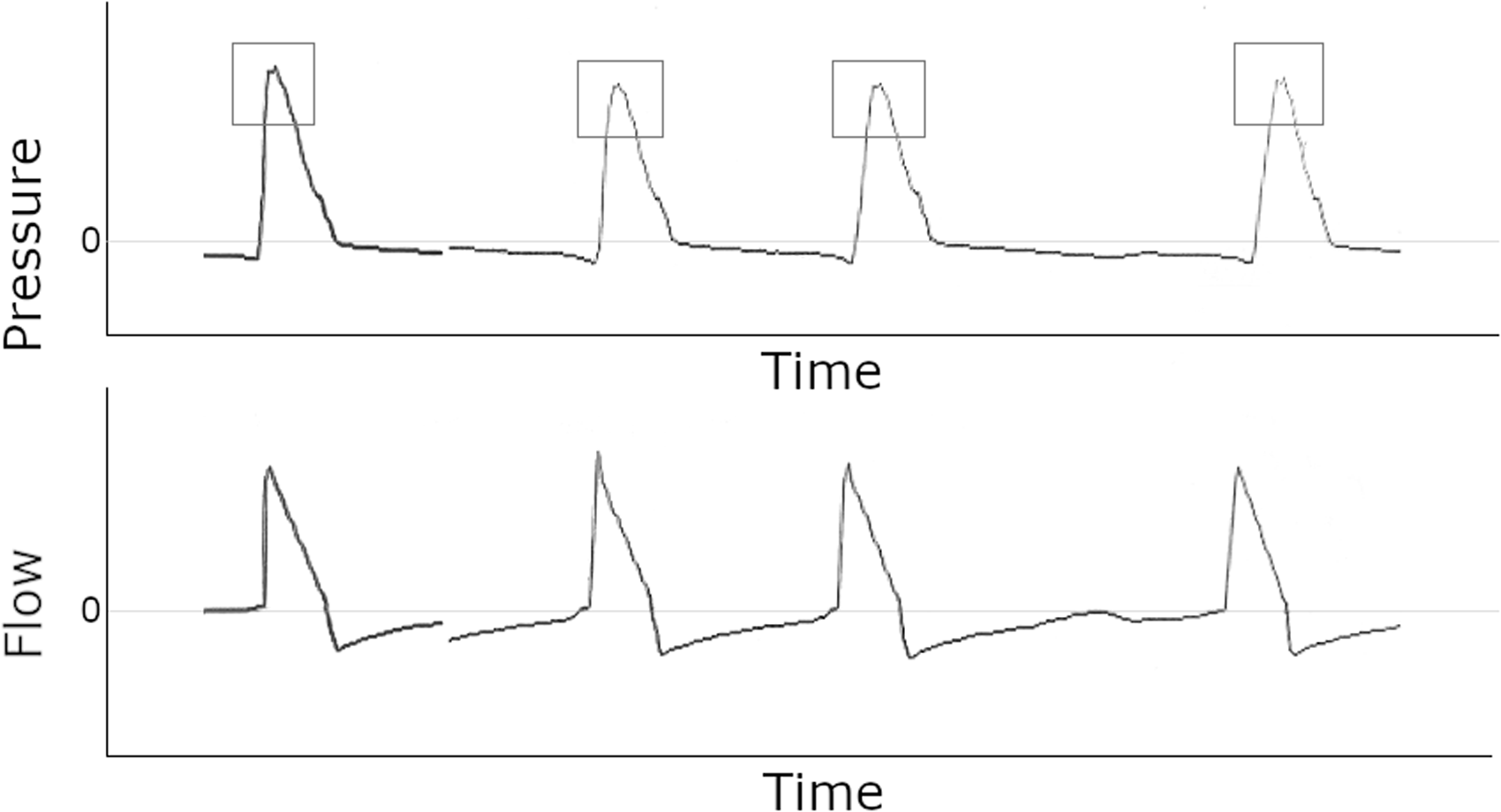
In vivo example of severe flow overshoot. Although the pressure curve would typically be square-waved in volume control ventilation with a decelerating flow ramp, here the dramatic pressure spikes (boxes) create a sloped waveform instead.
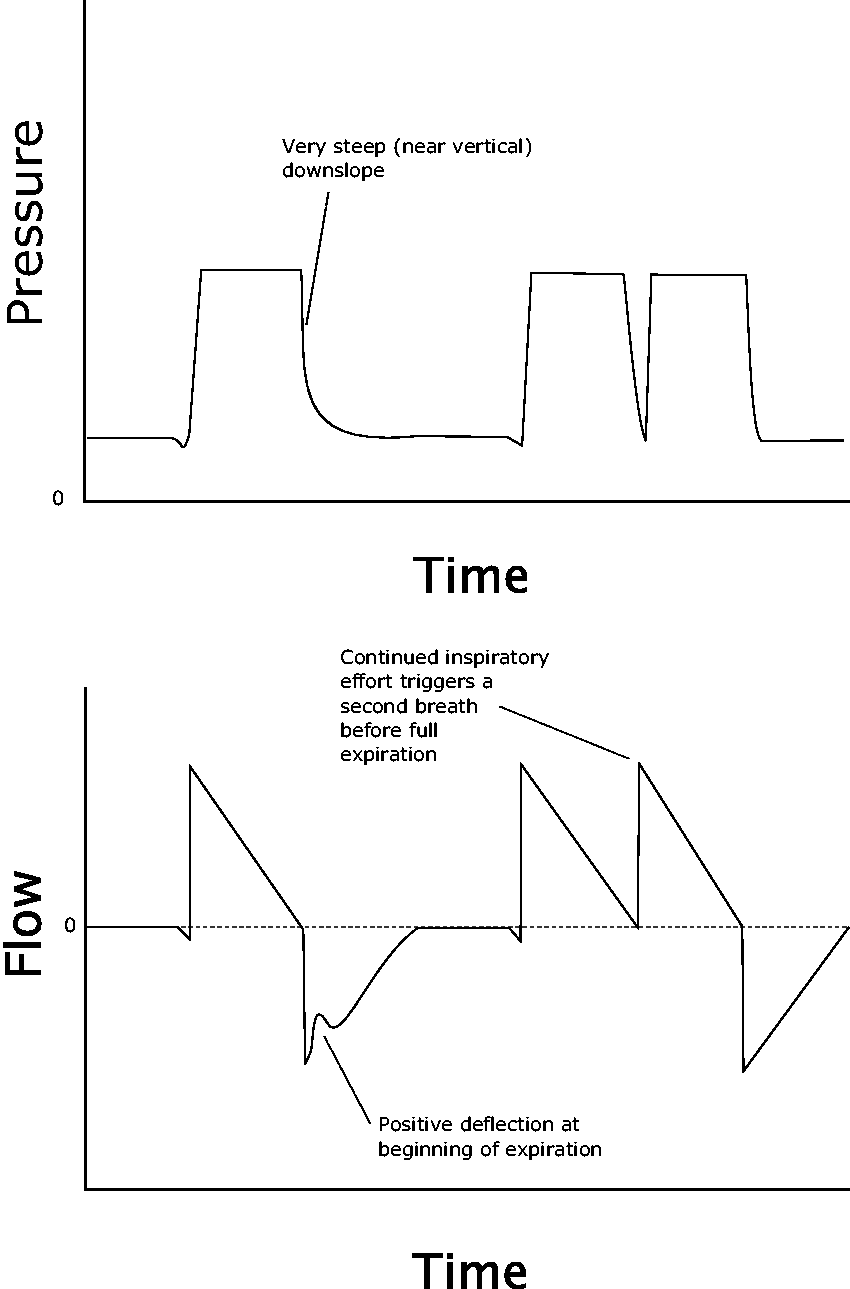
In the first breath, an upward flow deflection early in expiration reveals that neural I-time (inspiratory time) exceeds machine I-time. In the second breath, this continued effort is substantial enough to trigger another full ventilator breath, which stacks onto the first. Note that these are patient-triggered breaths, and hence this is not an example of ‘reverse triggering’.
In more significant cases during assist control ventilation, the prolonged patient effort actually triggers an additional ventilator breath, occurring shortly after the last breath and long before full expiration has completed. This results in ‘breath stacking’ or ‘double triggering’, with a characteristic appearance on the pressure tracing. PSV is less susceptible to this, because a second triggered breath is usually small.
Potential remedies
Volume control: Increase machine I-time. Most often, this requires increasing the tidal volume. In principle, it can also be achieved by reducing inspiratory flow; however, as premature cycling tends to occur in patients with a strong respiratory drive, this often results in flow starvation. Pressure control: Increase the set machine I-time. Pressure support: Decrease cycle threshold (e.g. 30% to 20%), thus prolonging the period of inspiratory support.
Reverse triggering: a special case of double triggering
Although most instances of double triggering are the result of premature cycling, one important subset has a distinctly different cause. This recently recognised phenomenon, dubbed ‘reverse triggering’ or ‘entrainment’, occurs when an initial ventilator breath triggers a subsequent patient-initiated breath. 18 This occurs for unclear reasons, but may be the result of reflex stimulation of the diaphragm. Unlike premature cycling, and indeed most other asynchronies—which tend to occur in the setting of a potent respiratory drive—reverse cycling occurs more often in deeply sedated or comatose patients; it has even been described in brain-dead individuals, in whom it can potentially confuse the neurological exam. 19 The stimulated diaphragmatic effort may be minor (a small waveform perturbation only), or may be sufficient to trigger a breath, in which case double triggering will occur.
Reverse triggering can most easily be identified by noting whether the initial breath was patient triggered (indicating that the stacked breath is the result of premature cycling) or ventilator triggered (suggesting reverse triggering). In cases in which the nature of the second breath remains unclear, a lengthy manual expiratory hold manoeuvre may be revealing, as withholding additional ventilator breaths may make the presence or absence of spontaneous patient effort (via negative pressure deflections) obvious. 20 The most effective strategy for managing it seems to be one that encourages patient-triggered respirations, such as transitioning to PSV, adjusting the set respiratory rate, or perhaps most effectively, reducing the level of sedation.18,21 Neuromuscular blockade is effective for refractory cases that are resulting in harmful sequelae.
Delayed cycling
Causes
Delayed cycling occurs when machine I-time exceeds neural I-time. Similar to premature cycling, it may therefore be caused by some combination of excessive tidal volume (in VCV), overly long I-time (in PCV), or an incorrect cycle parameter (in PSV). It is particularly common when PSV (a flow-cycled mode) is used in COPD, in which prolonged expiration causes late attainment of the flow decay threshold, and breath termination is consequently delayed.
Clinical implications
In addition to patient discomfort, delayed cycling often results in patients actively exhaling during ventilator insufflation, creating elevated peak pressures. It also may predispose to autoPEEP, as an unnecessarily long I-time shortens the effective time for expiration.
Clinical signs
Patients may be seen actively to expire via activation of the abdominal muscles.
Waveform manifestations
On the pressure curve, a positive inflection is seen at the end of inspiration; this reflects the pressure spike caused by the patient actively exhaling to resist the machine breath 22 (Figures 12 and 13).
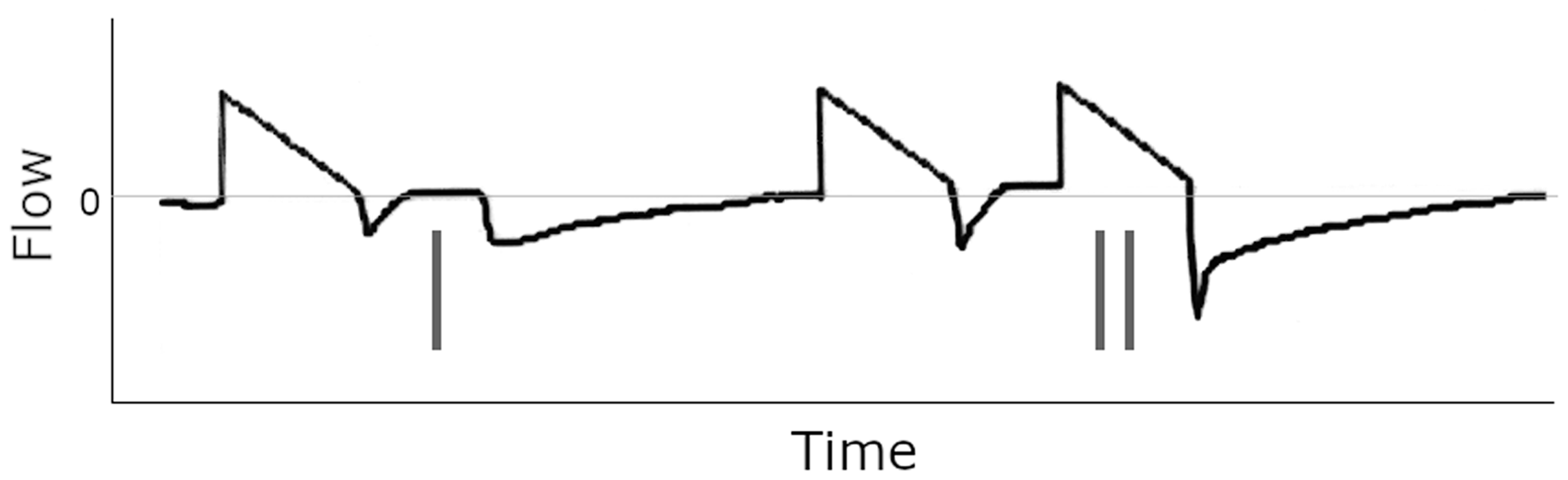
In vivo examples of premature cycling. In the first breath, continued inspiratory effort is visible at the start of expiration (single line). In the second, this effort is sufficient to trigger a stacked breath (double line). Note the prolonged expiratory time that results due to the additional tidal volume.
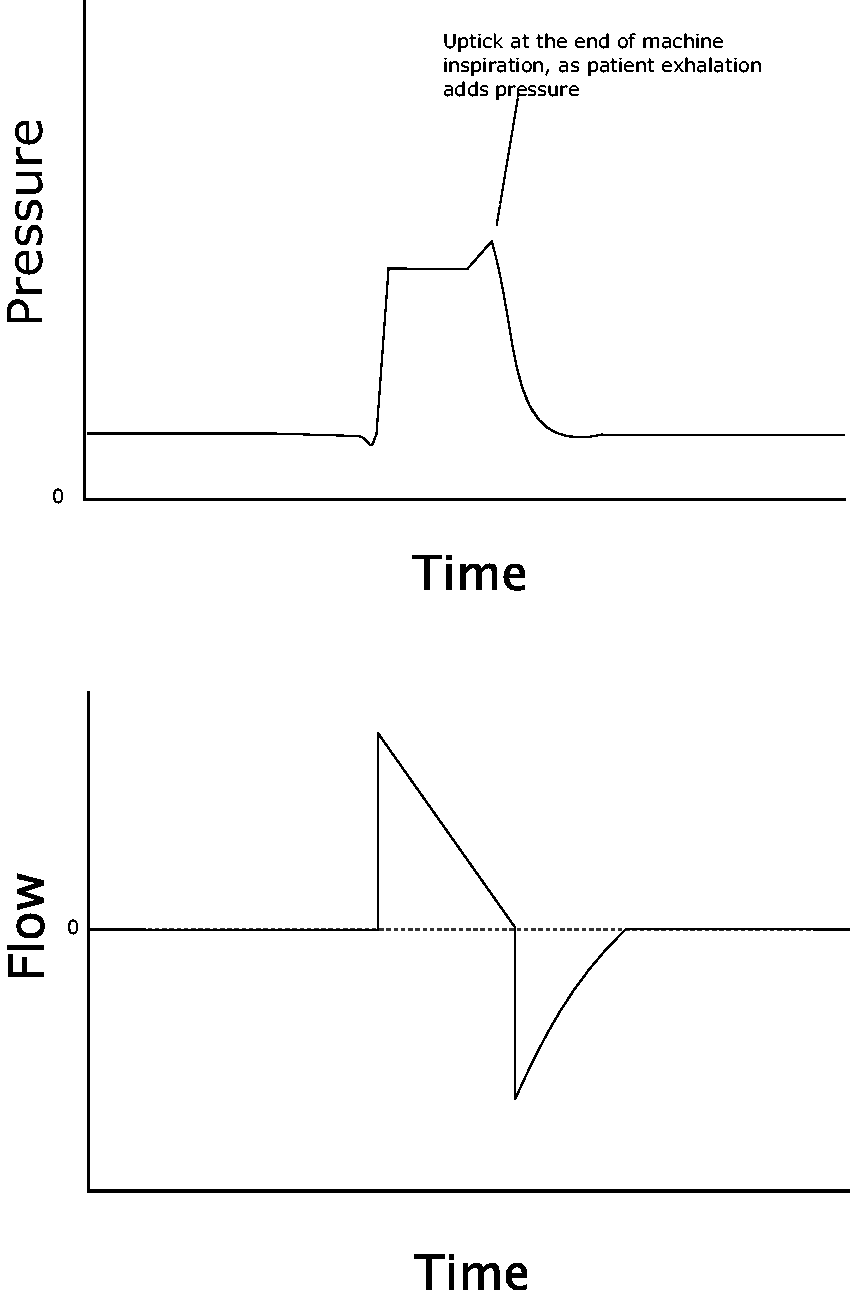
A small ‘horn’ is created at the end of the pressure curve as the patient attempts to stop the breath by exhaling against it. Flow remains fixed due to the volume control ventilation (VCV) mode.
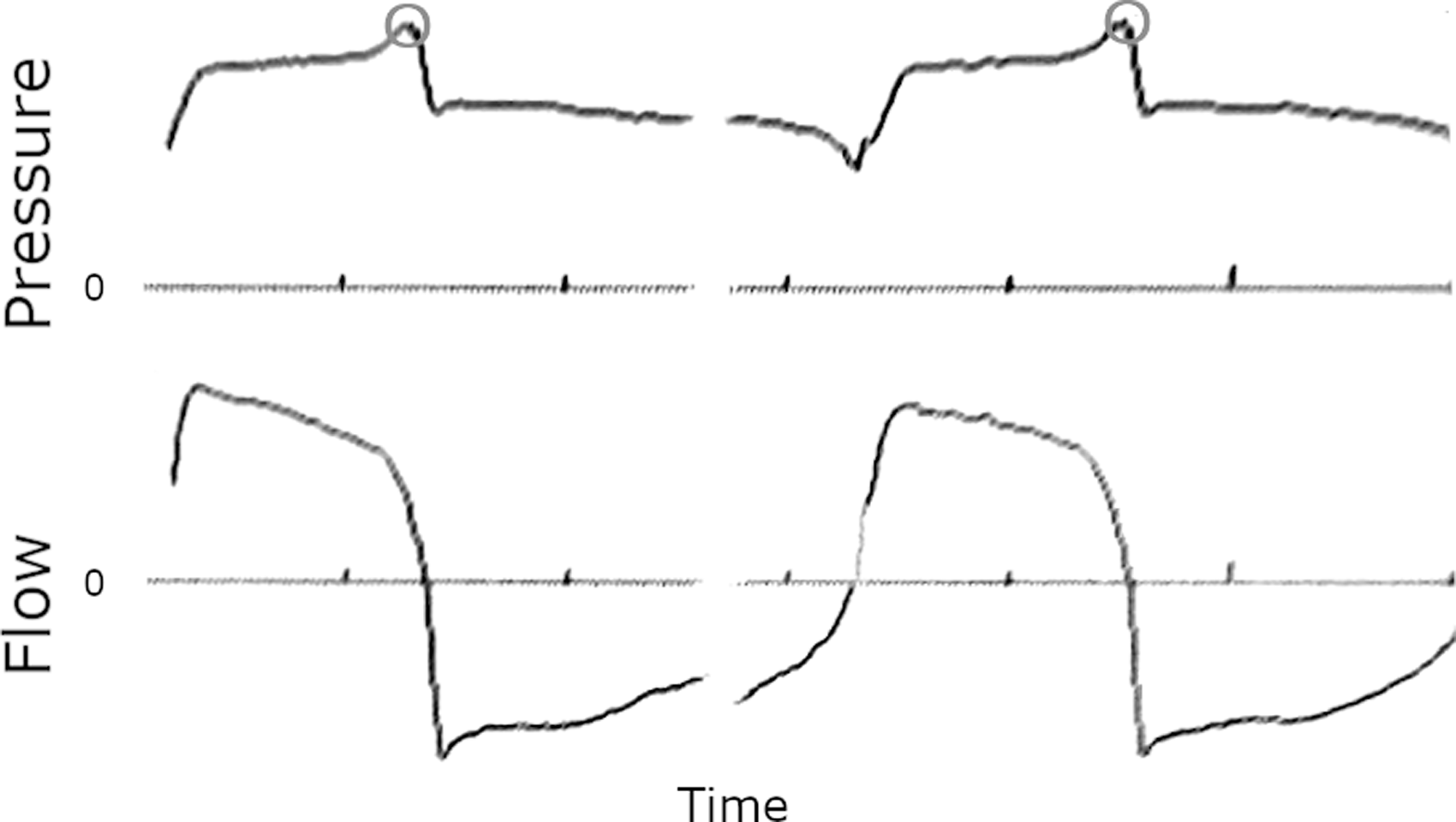
An in vivo example of delayed cycling. Circles denote the end-inspiratory pressure spike. Flow curve is unchanged. (Image courtesy of Alex Yartsev, personal files.)
Potential remedies
Volume control: Shorten machine I-time. This can be achieved by either increasing the flow rate or decreasing the tidal volume. Pressure control: Shorten set machine I-time. Pressure support: Increase the flow threshold for cycling (e.g. 30% to 40%), thus shortening the breath.
23
Peak pressure apnoea
A seventh type of dyssynchrony does not fit into our standard rubric, but has great potential to cause harm if not immediately recognised. 24 All modern ICU ventilators have the ability to set a variety of alarms, one of which is typically a ‘peak pressure alarm’ that sounds when the airway pressure at the ventilator outlet exceeds a set limit. This alerts staff to circuit occlusions, mucus plugging, and other troublesome issues (although also alarming from time to time due to coughing and similar transient events).
However, on the majority of ventilators, this parameter functions not only as an alert, but also as a maximum. When the pressure reaches the set limit or a figure close to it, inspiratory flow is automatically terminated. The result is a curtailed breath: in mild cases, the inspired volume is reduced, and in severe cases the volume may become zero or nearly zero, resulting in functional apnoea.
This phenomenon is often unrecognised and can be extremely hazardous; although high pressure alarms require troubleshooting, apnoea rarely improves the situation. Some patients, such as those with bronchoconstriction, have perpetually elevated peak pressures but a normal plateau pressure; others, such as the obese, have elevated plateau pressures but a normal transpulmonary pressure. In either case, elevated peak pressures must be tolerated to permit adequate ventilation.
‘Peak pressure apnoea’ is primarily a phenomenon of volume-targeted modes, as in pressure modes the inspiratory pressure is set directly by the clinician. Dual-control modes such as pressure regulated volume control, in which pressure is titrated automatically by the ventilator to achieve a set volume goal, are also susceptible.
Diagnosis is straightforward: the combination of a peak pressure that is repeatedly fixed at the set limit (often defaulted to 40 cmH2O), along with consistently low tidal volumes, is nearly pathognomonic for this phenomenon. The diagnosis can be confirmed and resolution immediately achieved simply by increasing the pressure limit, which will promptly result in an increase of both inspiratory pressure and tidal volume. The patient can then be evaluated to determine whether the elevated peak pressure is clinically acceptable or requires further manoeuvres to reduce it.
Conclusions
Patient–ventilator dyssynchrony is a common phenomenon which is nevertheless poorly understood. In many cases, subtle failures of synchrony are not noted, but may result in diaphragmatic fatigue or occult lung injury; in more frank cases, the standard clinician response may be to increase sedation, which is a blunt response and rarely the preferred solution. A keen understanding of the causes of dyssynchrony, and a practised eye at examining ventilator waveforms, is often sufficient to diagnose these problems and recommend a response which focuses on simple ventilator optimisation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
