Abstract
Making a diagnosis of perioperative anaphylaxis and identifying culprit drugs are diagnostic challenges. The aim of this study is to describe the perioperative presentation of anaphylaxis and results of patients who underwent allergy evaluation. This is a retrospective review of perioperative anaphylaxis of severity Grade 2 and above based on the Australian and New Zealand Anaesthetic Allergy Group criteria from 2015 to 2019 in a tertiary paediatric hospital. Data collected were demographics, clinical features, investigations and management. Of the 35,361 cases of paediatric anaesthesia, there were 15 cases of perioperative anaphylaxis, giving an incidence of four in 10,000. The median age was seven years (interquartile range four–15 years) with a male predominance of 86.7% (13/15). The severity of anaphylaxis was Grade 2 in 33.3% (5/15) and Grade 3 in 66.7% (10/15). The commonest presenting feature was hypotension (13/15, 86.7%) while the earliest symptom was respiratory change (9/15, 60.0%). Dynamic tryptase was raised in 75% (6/8) of the patients with adequate tryptase samples. Eight patients (53.3%) completed allergy testing, of whom five patients (62.5%) had IgE-mediated anaphylaxis with skin test positive to cefazolin (
Keywords
Introduction
Anaphylaxis is a severe drug hypersensitivity reaction that can be caused by an underlying immunological IgE- or non–IgE-mediated mechanism. The incidence of perioperative drug-related hypersensitivity in the adult population varies from one in 353 to one in 18,600 procedures. 1 , 2 In 2018, the United Kingdom National Audit Project 6 (NAP6) review of perioperative life-threatening allergic reactions from all National Health Service (NHS) hospitals in a year found an overall incidence of one in 10,000. 3
However, there are limited published reports in the paediatric population and the estimated incidence of paediatric perioperative anaphylaxis ranges from one in 2100 in a single French paediatric centre 4 to one in 37,000 in the NAP6 subgroup of paediatric patients. 3
Appropriate management of perioperative anaphylaxis involves prompt recognition and resuscitation followed by investigation and identification of the causative agent. The guidelines from the British Society for Allergy and Clinical Immunology (BSACI), 5 European Academy of Allergy and Clinical Immunology (EAACI) 6 and the Australian and New Zealand Anaesthetic Allergy Group (ANZAAG) 7 include recommendations on serial tryptase readings, in vitro drug-specific IgE levels, skin prick testing (SPT), intradermal testing (IDT) and, in selected cases, drug provocation testing. However, the current guidelines are not specific to the paediatric population and there is great heterogeneity among centres on the management of perioperative anaphylaxis. The paediatric patient with perioperative anaphylaxis poses a challenge to anaesthetists and allergists due largely in part to the discomfort of intradermal skin tests resulting in high rates of non-compliance to allergy evaluation. Yet, unlike adults, children may be subjected more readily to general anaesthesia for diagnostic imaging and minor procedures. As such, it is important to identify culprit drugs for the safety of future anaesthesia as perioperative anaphylaxis is life-threatening, with a reported mortality rate of 9%. 8
Since 2015, we have introduced an integrated inpatient crisis management workflow between paediatric anaesthesia and allergy for investigation of perioperative anaphylaxis, including time-sensitive tryptase sampling, early inpatient allergy referral and comprehensive outpatient skin testing.
In this study, we aim to describe the clinical characteristics of perioperative anaphylaxis in our paediatric population. Our secondary aim is to describe the allergy evaluation, including dynamic tryptase calculation and skin test results.
Methods
After institutional ethical board review (SingHealth Centralised Institutional Review Board number 2015/3141), we conducted a retrospective review of perioperative anaphylaxis diagnosed between January 2015 and May 2019 in the KK Women’s and Children’s Hospital, an 830-bed children’s hospital with a throughput of 6000 to 8000 general anaesthesia cases per year.
Cases of perioperative anaphylaxis were identified using the paediatric anaesthesia quality assurance database, which captures perioperative critical adverse incidents that are documented by the anaesthesia team directly involved in the care of the patients. Critical incidents related to perioperative anaphylaxis were extracted from the quality assurance database using the keywords ‘drug reaction’, ‘drug allergy’, ‘allergy’ and ‘anaphylaxis’.
These medical records and anaesthesia documents were then reviewed by the investigating team and categorised according to the ANZAAG grading for anaphylaxis
5
(Figure 1
Overview of recruited patients.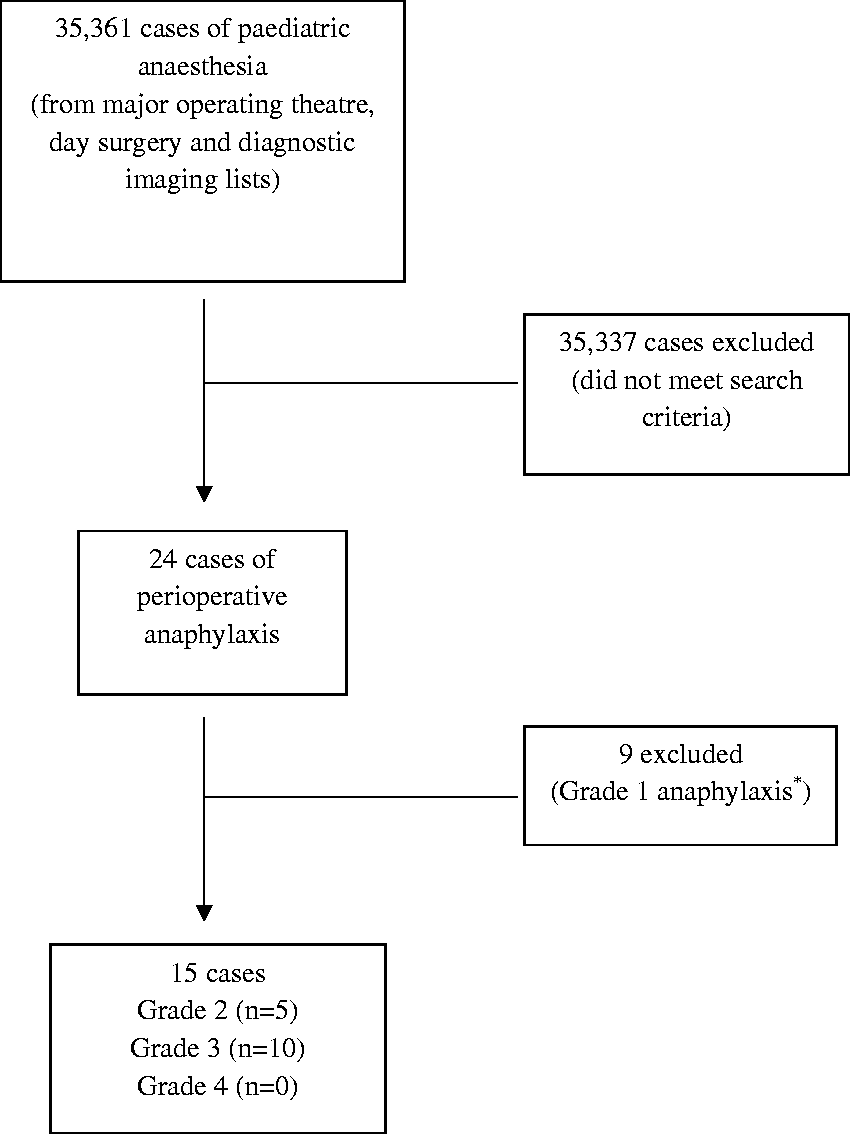
Each case of perioperative anaphylaxis followed an integrated crisis management workflow which included serial acute and baseline tryptase collection according to the BSACI guideline. 5 A raised dynamic tryptase was defined according to the 2019 EAACI position paper on perioperative anaphylaxis as acute tryptase taken within four hours of reaction greater than ((1.2 × baseline tryptase) + 2) ng/ml. 6 A referral to the paediatric allergy service was made, often within 24 hours of the event. SPT and IDT if indicated were performed four to six weeks after the event.
Skin testing was performed using histamine (0.01 mg/ml) as a positive control, 0.9% saline as a negative control and suspected culprit agents, including latex. Perioperative drugs were prepared according to non-irritant concentration guidelines stipulated in the 2013 EAACI Drug Allergy Interest Group position paper. 9 A positive SPT result was a mean wheal size of 3 mm or greater above the negative control. A positive IDT result was an increase of 3 mm or greater of the mean wheal size from 0 to 20 minutes or an absolute wheal size of 8 mm or greater. If a culprit agent was detected by positive skin tests, alternative drugs were tested; for example, alternative beta-lactam antibiotics or neuromuscular blocking agents.
Cases were categorised as: (a) IgE-mediated anaphylaxis, with definite culprit agent proved by skin test; (b) non–IgE-mediated anaphylaxis, with negative skin tests; or (c) anaphylaxis—incomplete workup in cases who did not complete skin tests. For the category of non–IgE-mediated and anaphylaxis—incomplete workup, culprit drugs were identified by the anaesthetist and allergist after joint review of the anaesthetic charts taking into account factors including time interval to reaction and agents involved.
Statistics
The collected data were tabulated using the Statistical Package for Social Sciences (SPSS) and relevant descriptive frequencies were obtained and analysed.
Results
Baseline characteristics of study population.
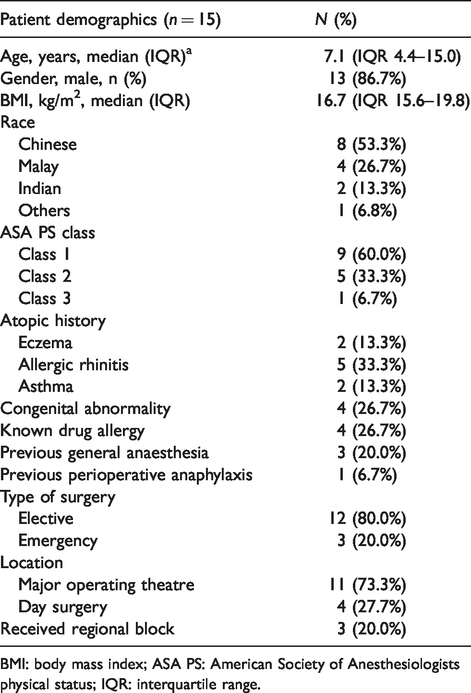
BMI: body mass index; ASA PS: American Society of Anesthesiologists physical status; IQR: interquartile range.
Clinical presentation and management of perioperative anaphylaxis.

Age-defined systolic blood pressure (SBP) 70 + (age (in years) × 2) mmHg.
Heart rate as defined by age-defined range. Interquartile range: IQR.
Surgery was aborted in 40% of patients (six out of 15) as a result of anaphylaxis. Eight patients (53.3%) were transferred to intensive care; four patients required vasopressor support and two patients, mechanical ventilation. Three patients (20.0%) were transferred to high dependency and four patients (26.7%) were managed in the general ward. There was no mortality.
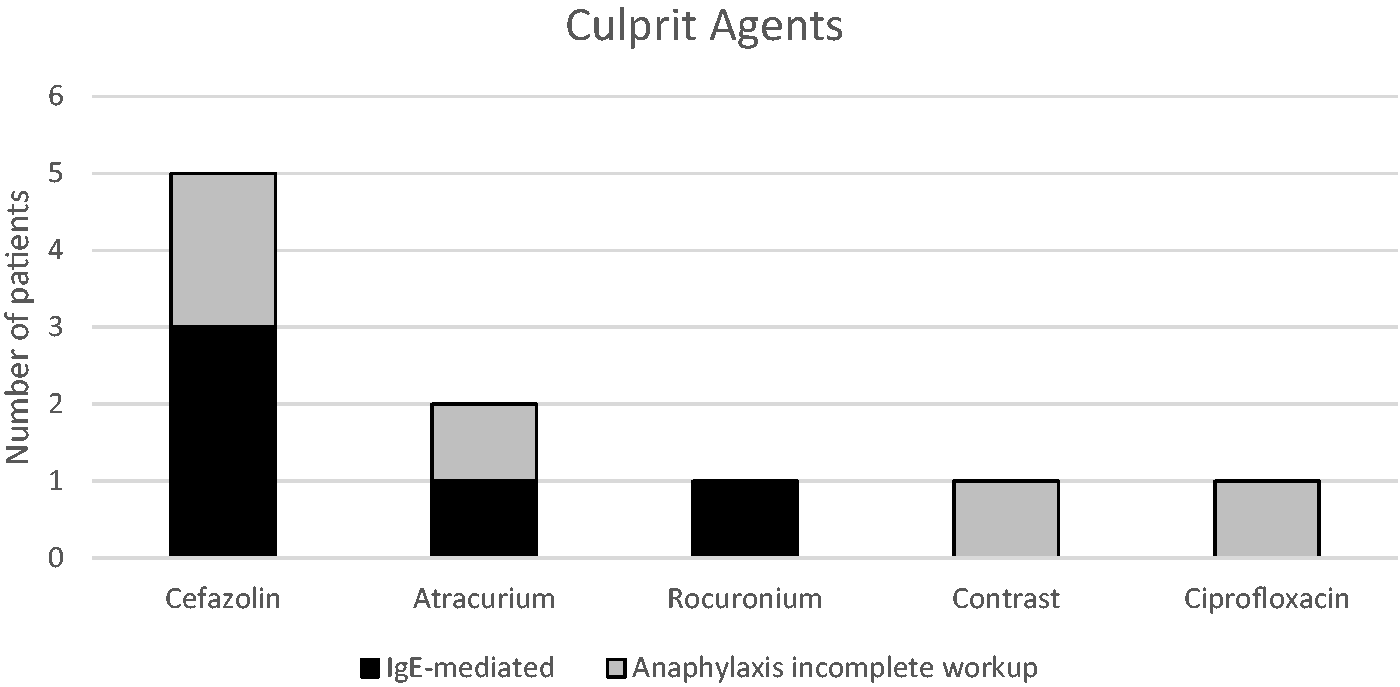
Patients were exposed to a median of six (IQR 3–7) agents prior to anaphylaxis. Drugs included induction and anaesthetic agents (propofol, thiopentone, sevoflurane, desflurane), opioids (fentanyl, morphine, remifentanil), benzodiazepines (midazolam), neuromuscular blocking agents (NMBAs) (atracurium, rocuronium), antibiotics (cefazolin, clindamycin, ciprofloxacin), local anaesthetics (bupivacaine, lidocaine), anti-emetics (ondansetron, dexamethasone) and others (contrast, paracetamol, tranexamic acid). Despite their common use, there were no reported perioperative anaphylactic events attributable to skin preparation agents including latex and chlorhexidine (proved by way of negative skin tests).
Figure 2
Outcome of allergy testing and dynamic tryptase calculation. Description of culprit agents.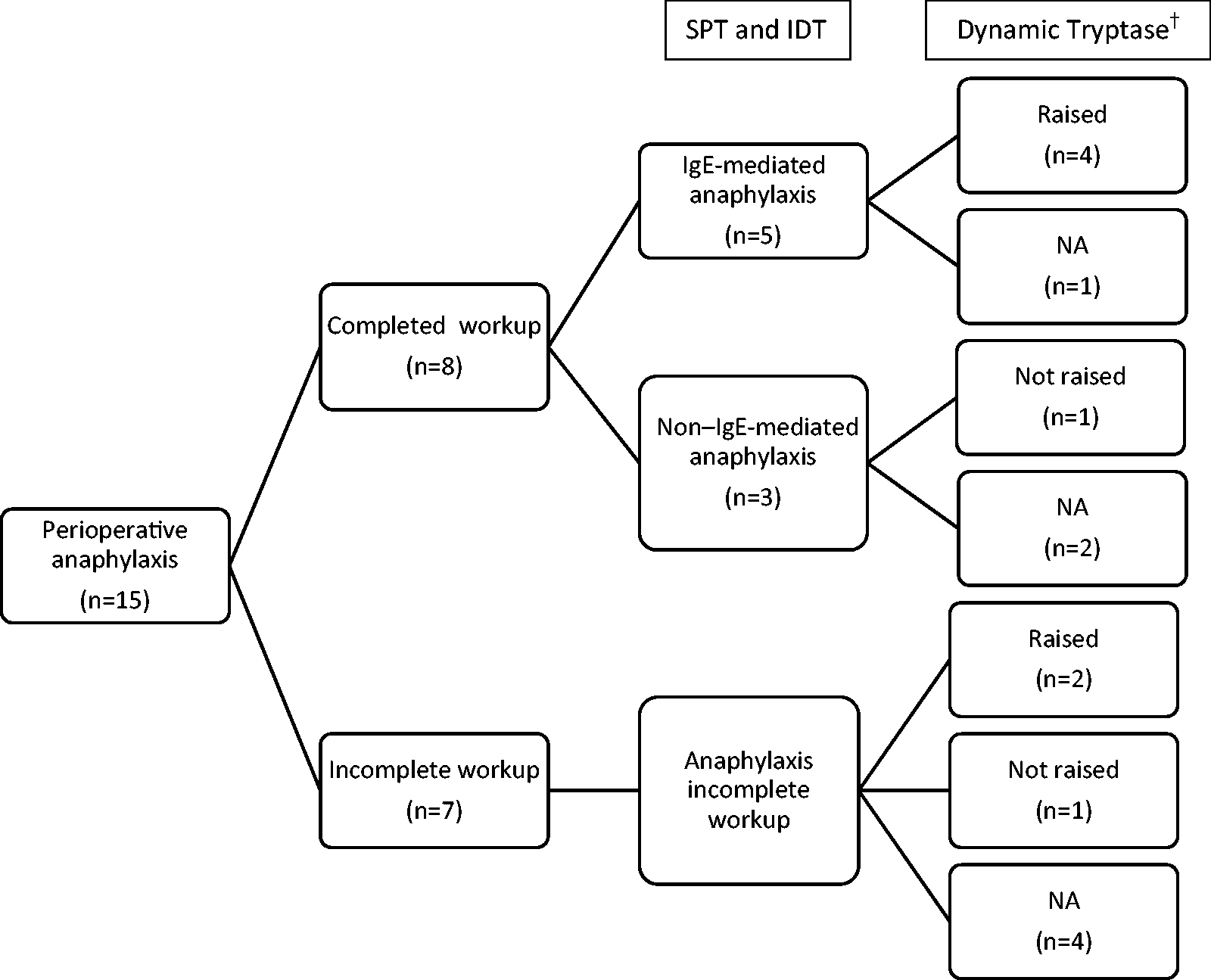
Only 53.3% of patients (eight out of 15) had an acute tryptase blood sample taken within four hours of reaction paired with a baseline sample taken more than 24 hours after reaction. Dynamic tryptase was raised in six patients (four IgE-mediated anaphylaxis, two anaphylaxis—incomplete workup) of whom two patients had acute levels that were greater than 11.4 ng/ml. A raised dynamic tryptase occurred in 80% of IgE-mediated anaphylaxis compared to 0% in non–IgE-mediated anaphylaxis.
Details of subsequent anaesthetics.
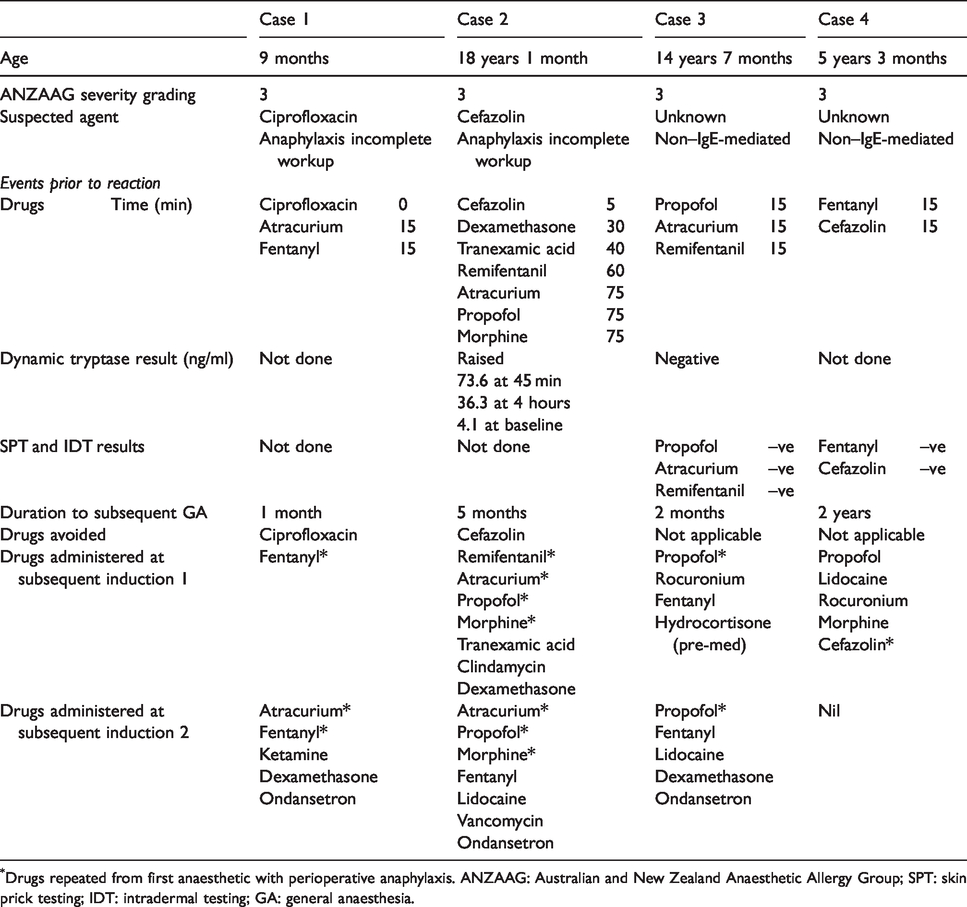
Drugs repeated from first anaesthetic with perioperative anaphylaxis. ANZAAG: Australian and New Zealand Anaesthetic Allergy Group; SPT: skin prick testing; IDT: intradermal testing; GA: general anaesthesia.
Discussion
The incidence of perioperative anaphylaxis in our tertiary paediatric hospital is four in 10,000. Dynamic tryptase was raised in 80% of patients (four out of five) who had IgE-mediated anaphylaxis; however, we recognise that almost half of our patients (seven out of 15) did not complete allergy testing. Among the eight patients who completed allergy evaluation, five patients (62.5%) had IgE-mediated anaphylaxis with skin test confirming the culprit drug. Antibiotics, in particular cefazolin, were the most common cause.
Our study incidence is similar to the 2005 report by Karila et al. 4 showing an incidence of one in 2100 in a 12-year review of paediatric perioperative anaphylaxis in a French paediatric centre. However, it is much higher compared to data from NAP6. 10 The difference in incidence is likely to be related to study methodology, inclusion criteria and the patient population. 11
Recognition of paediatric perioperative anaphylaxis hinges on a high index of suspicion by the attending anaesthetist. Bronchospasm may be the first sign of anaphylaxis, or due to non-anaphylactic causes such as hyperactive airway disease or direct histamine release secondary to drugs such as atracurium. In our cohort, a child developed bronchospasm and significant hypotension during his first surgical procedure. Given his history of asthma, he was deemed to have an asthma exacerbation by the attending anaesthetic team. The child subsequently had a similar presentation on induction for another surgery. Anaphylaxis was recognised and the child was referred for allergy testing revealing an IgE-mediated anaphylaxis secondary to the rocuronium administered on both occasions. While there was no long-term morbidity, the critical event could have been prevented by a high index of suspicion and evaluation after the first event.
In our study, the first sign of anaphylaxis was a change in respiratory status in 60.0% of patients while hypotension was the commonest sign, occurring in 86.7%. This is similar to the NAP6 paediatric cohort of 11 patients in which bronchospasm and/or high airway pressures were the first clinical feature of anaphylaxis in 64% of cases and hypotension was the commonest sign, occurring in 81.8%. 12 In 2019, Khaleva et al. performed a ten-year multicentre retrospective study identifying 29 cases of paediatric perioperative anaphylaxis. 13 In the study by Khaleva et al., hypotension was present early and throughout anaphylaxis, accounting for 58.6% of the initial presentation and 89.7% at any time. We postulate that the difference in initial presentation is due to the patient population as the study by Khaleva et al. included only patients with severe anaphylaxis of the Ring and Messmer classification Grade 3 (life-threatening) and Grade 4 (arrest), while our study included patients of ANZAAG anaphylaxis severity Grade 2 (moderate, multi-organ manifestation) and above.
Our study identified a causative drug in 62.5% of patients who completed evaluation, congruent to the 50%–60% estimate in the review by Mertes et al. 1 on the epidemiology of perioperative hypersensitivity. In our study, cefazolin was the top cause of anaphylaxis, followed by NMBA atracurium. This is in contrast with the data of Karila et al. showing that the most common cause was NMBA in 60.8% followed by latex in 27%, and none had antibiotic hypersensitivity. 4 The absence of latex anaphylaxis in our cohort is reflected in adult studies from the UK and Germany and is likely to be related to the improvement in modern equipment and the reduction of latex exposure in all patients. 6 , 14 The absence of chlorhexidine anaphylaxis was an interesting observation. Chlorhexidine was the third most common trigger of perioperative anaphylaxis in the NAP6 study, resulting in 18 cases of adult perioperative anaphylaxis. 3 The prevalence of chlorhexidine anaphylaxis in the paediatric population is still unreported and it was not featured in the multicentre study of Khaleva et al. 13 Its absence in our study was likely to be related to the small number of patients who completed allergy evaluation. Our study is in agreement with recent trends in which antibiotics are the commonest cause in predominantly adult studies from the United Kingdom, 10 United States and Spain. 15 , 16 In 2016, Kuhlen et al. showed that in a study of 25 patients with perioperative hypersensitivity reactions, a culprit drug was identified by way of a positive skin test in 36% and all were due to antibiotics. 15
Our study also showed that multiple intravenous medications were administered in quick succession. In our institute, cefazolin is commonly used by surgeons as empiric antibiotic cover. Given our findings that the median time to reaction was six minutes and antibiotics were the commonest culprit agents, it may be prudent for anaesthetists to space out the administration of antibiotics from induction agents and judiciously monitor clinical parameters after drug exposure. The benefits of the early recognition of perioperative anaphylaxis and identification of culprit agents should be weighed against the feasibility of spacing out the timing of antibiotic prophylaxis and induction drugs with its consequent potential time delays.
Tryptase is a specific marker of mast cell activation and is highly suggestive of anaphylaxis; however, levels are highly time sensitive and baseline levels differ between patients. The conventional cut-off for positive tryptase was greater than 11.4 ng/ml 17 based on the 95th percentile level in healthy individuals. In a retrospective study of 102 adult patients, Vitte et al. 18 proved that the dynamic tryptase algorithm from the international consensus recommendation of 2012 1 9 is more effective in diagnosing perioperative anaphylaxis, achieving a high positive predictive value and specificity, albeit a lower negative predictive value and sensitivity. In 2019, the EAACI position paper on the investigation of perioperative hypersensitivity recommended the use of the dynamic tryptase algorithm instead of the 11.4 ng/ml cut-off. 6 This algorithm was recommended in the update on anaphylaxis in paediatric anaesthesia of Stepanovic et al., 20 and applied in studies of perioperative anaphylaxis specific to childhood 13 and mixed adult and child populations. 10 In our cohort of paediatric patients, only 25% had acute tryptase above the conventional tryptase cut-off of 11.4 ng/ml, while the usage of the dynamic tryptase algorithm picked up a threefold increase of 75%.
It was disappointing that only 53.3% of our patients had time-appropriate blood samples for calculation of dynamic tryptase. We did not study the reasons behind poor compliance; however, we aim to increase the awareness of critical time-sensitive tryptase samples among the different disciplines and ranks of doctors and nurses caring for the child with perioperative anaphylaxis, including the operating theatre, post-anaesthesia care unit, paediatric intensive care and surgical ward.
In patients with skin test–negative non–IgE-mediated anaphylaxis, particularly when dynamic tryptase was not raised, certain drugs may be administered again with close supervision. In our cohort, two cases of non–IgE-mediated anaphylaxis safely received selected skin test–negative drugs during the subsequent anaesthesia after combined discussion between the allergy and anaesthesia teams. In patients with IgE-mediated anaphylaxis, the proved culprit agent must be avoided strictly and suitable alternative agents should be identified. For example, in patients with proven IgE-mediated NMBA anaphylaxis, alternative NMBAs are skin tested.
Our study had several limitations. First is the inherent limitation of a retrospective analysis. Secondly, the high rate of refusal to complete investigations. This may be because skin prick and intradermal testing are uncomfortable. Nearly half of our patients did not complete skin test evaluation, limiting the recommendations and choice of drugs for subsequent surgeries and increasing the risk of repeat perioperative anaphylaxis.
Conclusion
The incidence of paediatric perioperative anaphylaxis is four in 10,000 in our tertiary paediatric hospital. Although only half the patients completed allergy evaluation, a causative drug could be identified in 62.5%, with cefazolin being the most common culprit drug. It is important to evaluate all cases of perioperative anaphylaxis with a multidisciplinary approach including serial tryptase sampling and allergy skin tests.
Footnotes
Acknowledgements
Anaesthesia secretary: Hui Lian
Allergy nurses: Lim Hwee Hoon and Ding Xiao Mei
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
