Abstract
Perioperative intravenous lidocaine administration by anaesthetists is purported to confer a variety of benefits across a range of surgical procedures. It remains unclear whether the available evidence regarding efficacy and safety is sufficient to influence Australasian practice broadly, and whether significant barriers to uptake exist. We therefore conducted a survey of Fellows of the Australian and New Zealand College of Anaesthetists to evaluate patterns of lidocaine use, and perceptions relating to benefit and safety. Of 979 survey invitations, 295 (30.1%) responded. Of these, 51.9% of anaesthetists incorporate lidocaine administration into their practice. Amongst users, the most common indication is open abdominal or pelvic surgery (88.9%), with the principal intent of reducing acute pain and opioid use (both 92.2%). Only 51% perceive lidocaine to have a role in the prevention of chronic post-surgical pain, and less than a third administer it for operations strongly linked to this condition. Nearly all (91%) users deliver the drug by intraoperative bolus and infusion, with the majority using doses between 1.0 and 1.5 mg/kg for both the bolus and the hourly infusion rate. When parallel local anaesthetic techniques are employed, 44.4% restrict the dose, 37.3% turn the lidocaine infusion off first and 15.7% make no modifications. Most respondents terminate infusions by the end of surgery (52.3%) or in the post-anaesthesia care unit (26.8%). Few deliver postoperative infusions without electrocardiographic monitoring (5.9%). There were no reports of life-threatening events. The dichotomy in Australasian use of perioperative lidocaine revealed by this survey confirms that large multicentre trials are now required to guide practice accurately.
Keywords
Introduction
The administration of perioperative lidocaine by anaesthetists is purported to confer a variety of benefits across a range of surgical procedures. These include opioid sparing and the reduction of acute postoperative pain 1 the reduction of gastrointestinal morbidity and nausea after abdominal surgery 2 and the reduction of chronic post-surgical pain (CPSP). 3 These effects are consistent with multiple mechanisms of action. 4 Whilst lidocaine is principally a voltage-gated sodium channel blocker, it is increasingly recognised to dampen systemic inflammation after tissue injury 5 and preserve some aspects of cell-mediated immunity when tested in vitro. 6 Lidocaine also confers concentration-dependent antagonism of N-methyl-D-aspartate (NMDA) receptors that play a key role in the transmission of pain. 7 Such NMDA antagonism occurs across a clinically relevant range. 8
Despite the promise of lidocaine as an intervention in surgical patients, the current evidence base comprises relatively small trials and heterogeneous patient groups. A Cochrane systematic review and meta-analysis updated in 2018 concluded that the efficacy of continuous lidocaine is uncertain for all early postoperative outcomes. 9 Our recent meta-analysis of six trials with 420 patients investigating lidocaine demonstrated a reduction in chronic pain at three- or six-month timepoints in all the studies (odds ratio=0.29; 95% confidence intervals 0.18–0.48). However, we also concluded that the quality of evidence was low, as trials consistently exhibited a high risk of bias, significant design limitations and inadequate safety surveillance. 3
The decision to administer an amide local anaesthetic systemically must also consider the possibility of causing harm. Although serious adverse events such as seizures or life-threatening arrhythmias have not been reported across substantial numbers of trial patients (n = 2802 in acute pain trials, n = 420 in CPSP trials 3 ), the full scale of adverse events will not be known until sufficiently large Phase III and IV trials are performed.
Given the limitations of the available evidence regarding the efficacy and safety of perioperative lidocaine, the extent to which Australasian anaesthetists have adopted this practice and what key translational barriers exist remain unclear. We therefore conducted an anonymous survey of a random selection of Fellows of the Australian and New Zealand College of Anaesthetists (ANZCA) in order to evaluate patterns of lidocaine use and perceptions relating to benefit and safety. No such multicentre surveys were found following an extensive literature search, making this study the first of its kind.
Methods
This survey was approved by the East Metropolitan Health Research Ethics committee (RGS0000001320). A question set was developed and refined following informal discussions with colleagues and a trial of its interpretability and ease of use. As there were no validated surveys serving this purpose, a pragmatic design process was undertaken aimed at minimising differences in interpretation of the questions. The question set addressed the demographics of the respondents (questions 1–4; such that it could be compared to the ANZCA Fellowship demographics), the prevalence of lidocaine use (question 5) and the motivation for this use (questions 6–8). We enquired about the dosing strategy employed (questions 9–11), changes in practice when using lidocaine (questions 12 and 14) and which adverse effects the respondent had experienced when using lidocaine (questions 13 and 16). Those who reported not using lidocaine were asked their reasons for this (question 15), and all respondents were asked whether the drug should be given to unmonitored patients on the ward following surgery (question 17) in an attempt to gain an insight into whether anaesthetists’ current attitudes have the potential to limit the drug’s use on the ward. The question set was translated into an electronic survey, and data were collected and managed using REDCap electronic data-capture tools hosted at the University of Western Australia. 10 The survey itself contained smart logic steps such that the questions asked were dependent on prior answers, as outlined in Supplementary Figure 1. This avoided questions being answered by those to whom the questions did not apply. The survey could not be completed unless relevant questions were answered. ANZCA determined that the survey complied with the institutional policy, and the final survey was reviewed and approved by the ANZCA Clinical Trials Network group prior to circulation. 11 An email describing the project and an electronic link to the survey were sent to a randomly selected sample of 1000 Australasia-based Fellows by ANZCA administrators independent of the researchers. All of these Fellows were either practicing or previously practising anaesthetists, some of whom may also been pain medicine Fellows. A further reminder was sent two weeks later, and the survey was closed after four weeks. In total, 979 surveys successfully reached the intended recipient. The researchers had no involvement in this randomisation process, and they were blinded from the identity of the respondents whose answers were anonymous. No internet provider addresses were collected. The sample size was the maximum sample size permitted by ANZCA in an attempt to limit survey fatigue.
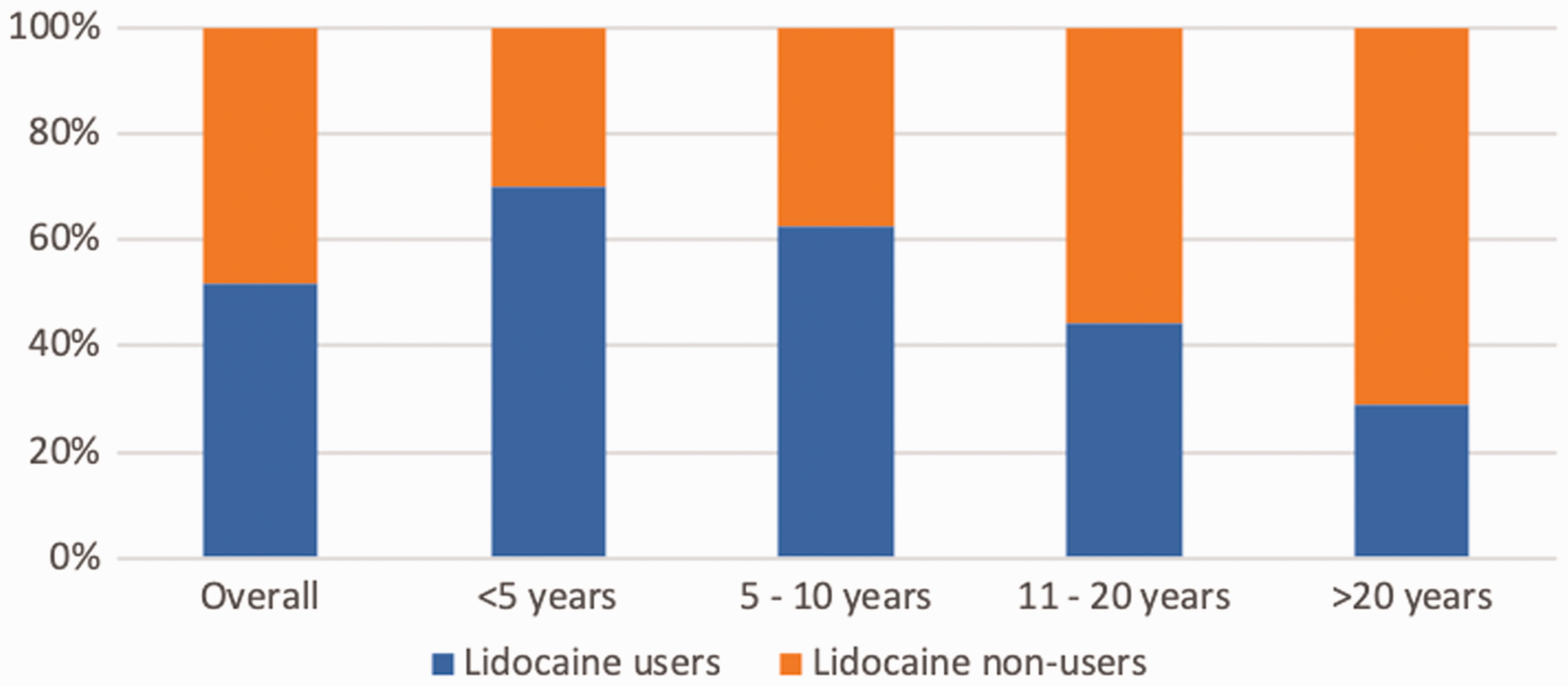
Users of intravenous perioperative lidocaine when categorised in terms of experience.
Statistical analysis
Data were analysed using descriptive methods. Percentages of responses to specific questions are presented as n (%). All statistical analyses were performed using STATA v15 (StataCorp LLC, College Station, TX, USA).
Results
In total, 979 email invitations to participate were successfully sent, and from this, 295 responses were received, giving a response rate of 30.1%. The characteristics of the survey respondents were similar to those registered as ANZCA Fellows (Table 1).
Demographic data on respondents and on the overall Fellowship in Australasia at the time of circulation.
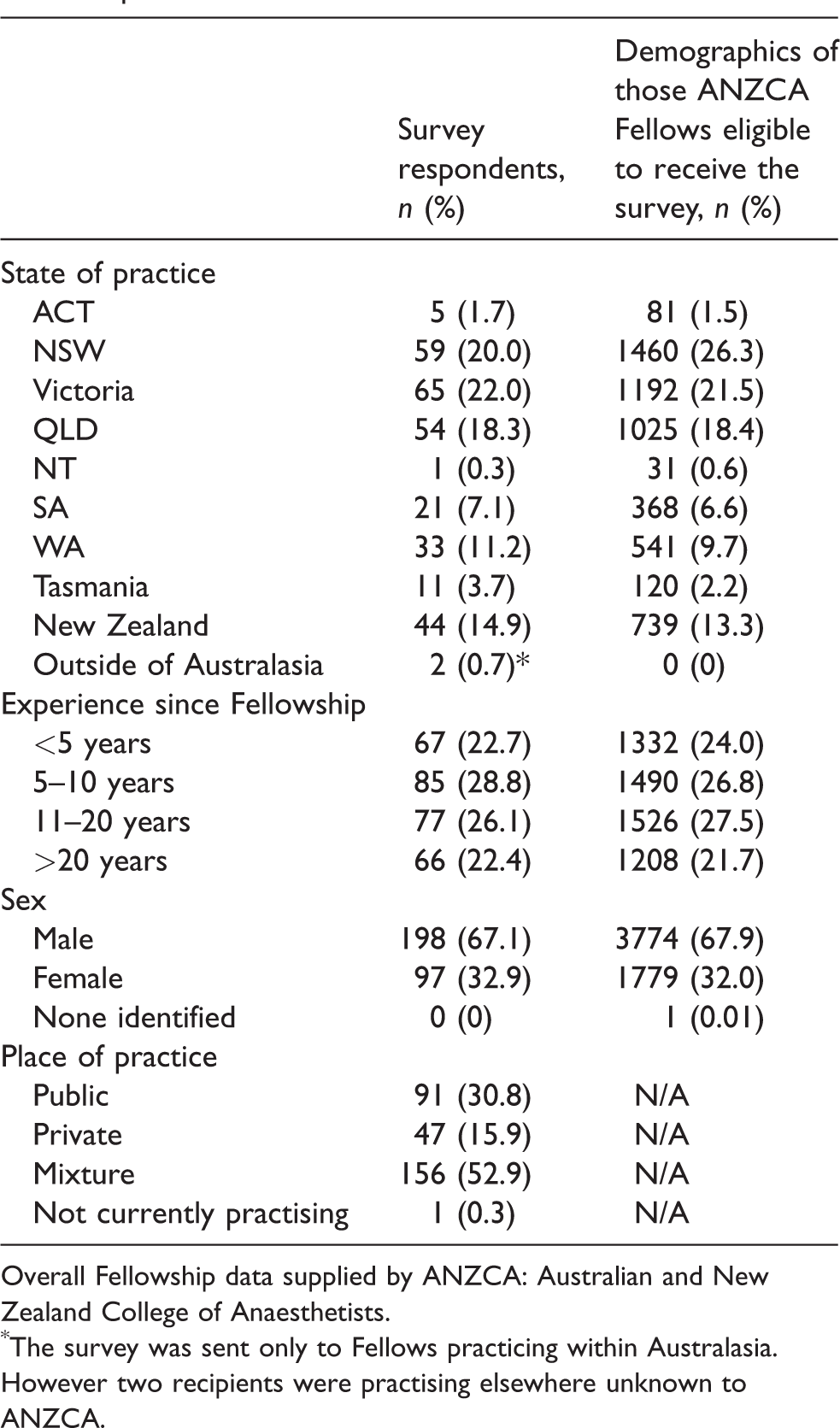
Overall Fellowship data supplied by ANZCA: Australian and New Zealand College of Anaesthetists.
The survey was sent only to Fellows practicing within Australasia. However two recipients were practising elsewhere unknown to ANZCA.
Of the 295 respondents, 153 (51.9%) incorporate intravenous lidocaine administration into their practice (Table 2). The proportion of Fellows using perioperative lidocaine decreases as experience increases (Figure 1). A total of 142 respondents reported never using perioperative lidocaine for reasons including not knowing enough about the therapy (47.2%), being unsure of its effectiveness (44.4%) and being unsure of safety (19.0%).
Reasons for not using perioperative lidocaine amongst non-users.
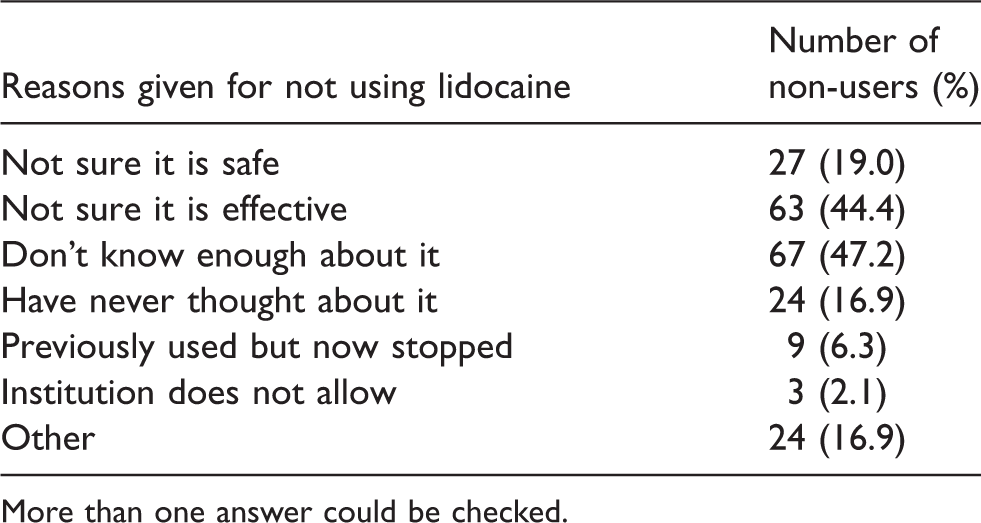
More than one answer could be checked.
Of the 153 lidocaine users, the most commonly reported surgical indications were open abdominal or pelvic surgery (88.9%) and laparoscopic surgery (58.8%; Table 3).
Survey responses across the 153 respondents using perioperative intravenous lidocaine in their practice
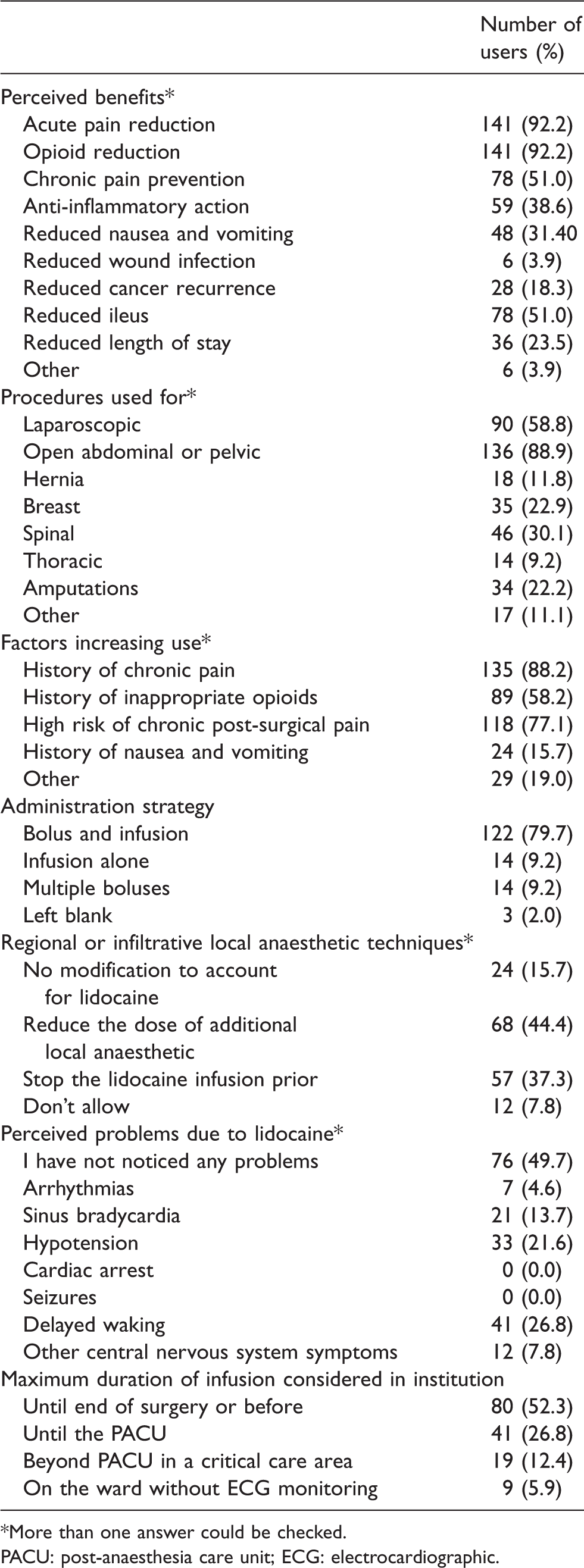
*More than one answer could be checked.
PACU: post-anaesthesia care unit; ECG: electrocardiographic.
Less than a third of users initiated perioperative lidocaine for other surgical procedures. The principal purpose reported was the reduction of acute pain and opioid use (each 92.2%). Other perceived benefits included the prevention of CPSP (51.0%), reduced ileus (51.0%), reduced nausea and vomiting (31.4%), reduced length of stay (23.5%) and reduced cancer recurrence (18.3%). Patient factors associated with high levels of both acute and chronic pain made most users more likely to administer perioperative lidocaine.
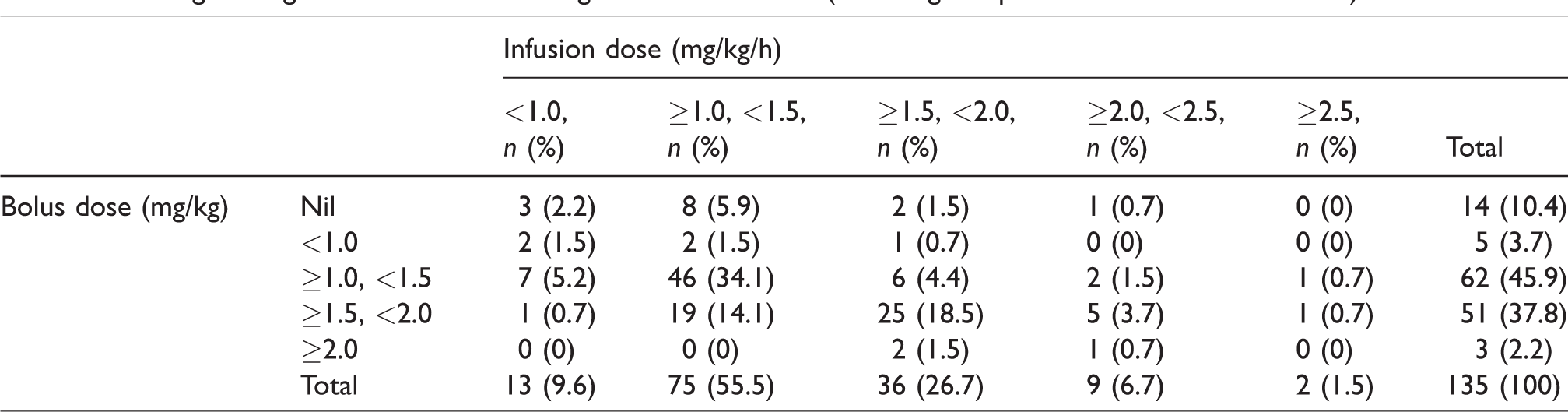
Lidocaine administration strategies included bolus and infusion (79.7%), infusion alone (9.2%) and multiple boluses (9.2%). Drug doses were reported by 135 respondents who use infusions as part of their practice (Table 4). One respondent stated their strategy was too complicated to be described. The most common bolus was ≥1.0 and <1.5 mg/kg (45.9%), and the most common infusion was ≥1.0 and <1.5 mg/kg/h (55.5%). The combination of these was the most common regimen overall (34.1%). When parallel local anaesthetic techniques were being used (by regional field block or wound infiltration), 15.7% of users reported unrestricted use alongside lidocaine, 37.3% turned the lidocaine infusion off first and 44.4% of users reduced the dose of additional local anaesthetic.
Dosing strategies of anaesthetists using lidocaine infusions (assuming the patient has no co-morbidities).
Regarding maximum infusion durations, most anaesthetists terminate administration by the end of surgery (52.3%) or in the post-anaesthesia care unit (PACU; 26.8%). Very few consider continuation in a critical care area (12.4%) or on a ward without electrocardiographic (ECG) monitoring (5.9%). Across all 295 respondents, only 33.3% believed that hospital protocols should allow for ward-based infusions up to 24 hours without ECG monitoring, assuming a fit and healthy patient and a dose of 1.33 mg/kg/h.
With respect to safety, 49.7% of users had not noticed any problems attributable to lidocaine, and no users had encountered cardiac arrest or seizures. Problems perceived to be at least in part due to lidocaine included delayed waking (26.8%), hypotension (21.6%), sinus bradycardia (13.7%), other central nervous system symptoms (7.8%) and arrhythmias (4.6%). Nine respondents stated they no longer used lidocaine for a variety of reasons, including a change in caseload, but none cited major adverse events.
Discussion
This survey provides a timely snapshot of attitudes and practice relating to the use of perioperative lidocaine by anaesthetists in Australia and New Zealand, given the emerging popularity of this adjunctive anaesthesia technique. The main finding is that approximately half of respondents use perioperative lidocaine, principally for the purpose of reducing acute postoperative pain and opioid consumption, and the vast majority of users administer lidocaine intravenously by bolus and infusion. Despite the perception of many that lidocaine is indicated in certain circumstances, safety issues appear to be a significant concern amongst both users and non-users. Australasian dosing regimens are more conservative than the majority of international trials, and continuation of these infusions into the postoperative period on the ward, particularly without cardiac monitoring, remains very uncommon. These findings warrant examination in the context of current practice, the current evidence base and future trials.
Almost all anaesthetists using perioperative lidocaine do so with the intention of reducing acute pain and opioid consumption. A Cochrane review updated by Weibel et al. in 2018 reported potential reductions in postoperative pain, opioid consumption, ileus and nausea and vomiting. The authors concluded that the quality of evidence was very low, and they remain uncertain whether intravenous lidocaine has a beneficial impact on any of these outcomes. 9 The dichotomy of practice revealed by this survey is therefore consistent with the uncertainty in the current evidence base.
Only 51% of anaesthetists using perioperative lidocaine do so with the intention of reducing CPSP. Yet, paradoxically, anaesthetists administer lidocaine much less frequently during surgical procedures with a relatively high incidence of CPSP (spinal, breast, amputation, hernia and thoracic 12 ). Our recent meta-analysis of six trials with 420 patients demonstrated efficacy but with considerable uncertainty. 3 Again, the responses in this survey mirror the uncertainty in the literature.
Safety concerns were a significant feature amongst respondents. Of those who do not use lidocaine infusions, 19% cited safety reasons. Amongst users, 79.1% currently terminate infusions in the PACU or earlier, only 33.3% believe hospital protocols should allow for postoperative ward infusions without ECG monitoring in healthy patients and only 5.9% actually use lidocaine in this way. The review by Weibel et al. included 69 trials and 4525 randomised patients, within which 50 collected data on adverse events. No studies reported any local anaesthetic toxicity resulting in seizures or cardiovascular collapse, and only two studies demonstrated a clear difference in minor adverse effects. 9 These findings are consistent with the survey results showing that no respondents had experienced life-threatening events, but a number of respondents had witnessed non–life -threatening arrhythmias, bradycardia, hypotension and delayed waking that they perceived to be at least in part related to lidocaine. However, such events commonly occur during the perioperative period in circumstances where lidocaine is not used, and it is therefore difficult to confirm a causal relationship. A large, double-blinded, randomised trial that systematically collects these as data points is required to quantify the true contribution of lidocaine to these events accurately.
The most popular dosing strategy from our survey was less than but not including 1.5 mg/kg followed by less than but not including 1.5 mg/kg/h. In a review of both high- and low-dose lidocaine infusions, a statistically significant reduction in acute pain in the high-dose group was observed, but there was no reduction in those receiving the low dose. 13 Studies in established chronic pain have suggested a certain threshold of plasma levels needs to be met before clinical effect is evident. 14 Kaba et al. suggest that a bolus (1.5 mg/kg) followed by an infusion of 2 mg/kg/h would achieve this plasma concentration in some but not all. 15 Of the 69 studies in the most recent Cochrane review, 1 only one had a dosing strategy that would fit into the survey’s most popular dosing response option. 16 The other 68 studies used doses higher than this. This phenomenon may reflect that many lidocaine trials exclude parallel local anaesthetic techniques (by field or wound infiltration), thus potentially allowing for higher doses than anaesthetists will accept when balancing risks in the real world. However, given that studies employing higher doses were only able to provide very low-quality evidence for lidocaine infusions and a very small clinical effect, it seems unlikely that lower doses would provide any significant clinical benefit.
Given the variation in practice revealed by this survey and the increasingly acknowledged limitations of meta-analyses, 17 large definitive trials of perioperative lidocaine infusions are now warranted. This survey highlights that the findings of such trials would hold considerable translational potential, regardless of the outcomes. For example, if lidocaine infusions were found to be not superior to placebo in preventing acute or chronic pain, half of the respondents would be expected to reconsider their practice. Conversely, if pain outcomes were improved and safety concerns were simultaneously addressed, many would adopt this intervention. One barrier to widespread implementation is safety and the related issue of how to merge lidocaine infusions with routine parallel local anaesthetic techniques. In particular, current attitudes amongst anaesthetists could feasibly contribute to postoperative infusions not being used in patients returning to surgical wards. Compelling data that prolonged drug exposure into the postoperative period is safe and adds benefit would be required to overturn such attitudes. Finally, it is clear that Fellows in the early stages of their careers are showing early adoption of perioperative lidocaine. Whether this reflects a readiness to embrace change or a less critical appraisal of the existing literature, high-quality evidence is urgently required to guide these cohorts better.
This survey is vulnerable to the weaknesses common to all surveys of this nature. The response rate was low but consistent with other ANZCA-endorsed surveys of bi-national practice. 18 Despite demographic consistencies, the relatively small survey sample may not fully reflect the overall ANZCA Fellow population being investigated, especially as anaesthetists with strong opinions on the safety or efficacy of the therapy may have been more likely to respond. We were also unable to quantify the frequency of perceived adverse events with lidocaine or the frequency with which lidocaine was being used, as efforts were made to limit the number of survey questions used.
Conclusion
Approximately half of our survey respondents incorporate perioperative lidocaine infusions into their practice with the principal aim of reducing acute surgical pain and opioid use. The majority of users employ conservative dosing strategies, restrict exposure to the intraoperative period, modify their practice when parallel local anaesthetic techniques are used and report no life-threatening adverse events. Non-users report inadequate knowledge and limited evidence as the primary reasons for avoidance. Large pragmatic multicentre clinical trials are now required to address the efficacy, safety and feasibility of perioperative lidocaine infusions definitively.
Supplemental Material
AIC889367 Supplemental Material - Supplemental material for A survey of perioperative intravenous lidocaine use by anaesthetists in Australia and New Zealand
Supplemental material, AIC889367 Supplemental Material for A survey of perioperative intravenous lidocaine use by anaesthetists in Australia and New Zealand by Martin A Bailey, Andrew J Toner and Tomas B Corcoran in Anaesthesia and Intensive Care
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Royal Perth Hospital Department of Anaesthesia and Pain Medicine funded the investigator time required to conduct the survey.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
