Abstract
Perioperative lidocaine (lignocaine) infusions are being employed with increasing frequency. The determinants of systemic lidocaine concentrations during prolonged administration are unclear. In the Long-term Outcomes after Lidocaine Infusions for PostOperative Pain (LOLIPOP) pilot trial, the impact of infusion duration and body size metrics on serum lidocaine concentrations was examined with regression models in 48 women undergoing breast cancer surgery. Lidocaine was delivered as an intravenous bolus (1.5 mg/kg) and infusion (2 mg/kg per h) intraoperatively, followed by a 12-h subcutaneous infusion (1.33 mg/kg per h) postoperatively. Dosing was based on total body weight. Wound infiltration with other long-acting local anaesthetics was permitted. Protein binding and pharmacogenomic data were also collected. Lidocaine concentrations (median (interquartile range) (range)) during prolonged administration were in the safe and potentially therapeutic range: post-anaesthesia care unit 2.16 (1.73–2.82) (1.12–6.06) µg/ml; ward 1.41 (1.22–1.75) (0.64–2.81) µg/ml. Concentrations increased non-linearly during the early intravenous phase of administration (mean rise 1.21 µg/ml per hour of infusion, P = 0.007) but reached a pseudo steady-state during the later subcutaneous phase. Higher dose rates received per kilogram of lean (P = 0.004), adjusted (P = 0.006) and ideal body weight (P = 0.009) were associated with higher steady-state concentrations. The lidocaine free fraction was unaffected by the presence of ropivacaine, and phenotypes linked to slow metabolism were infrequent. Serum lidocaine concentrations reached a pseudo steady-state during a 12-h postoperative infusion. Greater precision in steady-state concentrations can be achieved by dosing on lean body weight versus adjusted or ideal body weight (equivalent lean body weight doses: intravenous bolus 2.5 mg/kg; intravenous infusion 3.33 mg/kg per h; subcutaneous infusion 2.22 mg/kg per h.
Keywords
Introduction
Systemic lidocaine (lignocaine) infusions have been used for more than 50 years to treat or prevent a variety of conditions including cardiac arrhythmias, 1 chronic neuropathic pain, 2 cancer pain 3 and, more recently, acute 4 and chronic post-surgical pain. 5 For many of these clinical indications, prolonging the duration of lidocaine infusion offers a therapeutic advantage and minimisation of opioid-related side-effects. 6 However, maintaining safety reliably during prolonged intravenous infusion has proven problematic due to the risk of inadvertent intravenous bolus 7 and unrecognised systemic accumulation.8–11 As a consequence, many institutional guidelines advise that postoperative lidocaine infusions take place in critical care areas with continuous cardiac monitoring, and few clinicians are comfortable outside of these restrictions. 12
A recent development in perioperative trials is the use of the subcutaneous route to deliver a prolonged postoperative lidocaine infusion after intraoperative loading via the intravenous route.13,14 This offers a safety advantage by eliminating the risk of inadvertent intravenous bolus outside of the theatre environment. In addition, subcutaneous lidocaine has previously been shown to achieve appropriate systemic concentrations after a single bolus 15 and during titrated infusions lasting days to weeks in terminal cancer patients. 16 However, data in the surgical setting around the determinants of systemic lidocaine concentrations during prolonged subcutaneous infusion and the risk of unrecognised accumulation remain sparse. In particular, the influence of infusion duration, 17 obesity, 18 parallel local anaesthetic techniques 19 and inherited phenotypes linked to slow lidocaine metabolism 20 are under-explored. We therefore conducted this planned secondary analysis of our recently completed Long-term Outcomes after Lidocaine Infusions for PostOperative Pain (LOLIPOP) pilot trial 21 to evaluate the impact of these variables on systemic lidocaine concentrations during intravenous and prolonged subcutaneous administration.
Material and methods
This is a planned secondary analysis of the LOLIPOP pilot trial following on from the primary analysis published in 2021. 21 LOLIPOP was approved by the South Metropolitan Health Service Human Research Ethics Committee on 22 December 2017 and registered on 29 March 2018 (ACTRN12618000463279). 13 Briefly, women undergoing breast cancer surgery were randomised to lidocaine (n = 74) or placebo (n = 76) infusions starting intraoperatively and continuing for up to 12 h postoperatively. Intraoperative lidocaine 2% was delivered intravenously via a pre-programmed Alaris PK Pump (BD systems, New Jersey, USA), starting with a 1.5 mg/kg bolus over 5 min after induction of anaesthesia, followed immediately by an infusion of 2 mg/kg per h that continued into the post-anaesthesia care unit (PACU). In PACU, the intravenous infusion was replaced with a subcutaneous infusion of lidocaine 10%, delivered via a Niki T34 (BD systems, New Jersey, USA), a wearable syringe driver, at a rate of 1.33 mg/kg per h for up to 12 h, unless stopping or hospital discharge criteria were met at an earlier juncture. These doses and dosing rates represent 86.5% of lidocaine base and were calculated based on total (actual) body weight, capped at 100 kg. Doses were selected to target steady-state concentrations of 1.5 µg/ml based on results from equivalent perioperative trials14,22 and therapeutic thresholds identified in chronic pain populations. 23 Routine surgical practice with respect to long-acting local anaesthetics for wound infiltration or field blocks intraoperatively was permitted, with dose limits applied for ropivacaine (2 mg/kg) and bupivacaine (1 mg/kg).
Serum samples and assays
Blood samples were collected twice where possible; once in PACU during the intravenous infusion within 1 h of arrival (the PACU sample), and once on the ward during the subcutaneous infusion within a time-window of 4–12 h from the end of surgery (the ward sample). Serum was obtained then stored at –80°C in three aliquots (minimum 0.5 ml) and batch transferred to the laboratory. Samples were then assayed to quantify total serum lidocaine, the main metabolite monoethylglycinexylidide (MEGX) and the secondary metabolite glycinexylidide (GX), using a validated ultra performance liquid chromatography (UPLC) mass spectrometry method (coefficient of variation <10%). Total serum ropivacaine and bupivacaine were also quantified using UPLC mass spectrometry. Unbound local anaesthetics and lidocaine metabolites were extracted using temperature-controlled ultrafiltration and measured again with UPLC mass spectrometry. Finally, alpha-1 acid glycoprotein was quantified using an immunonephelometry assay on the IMMAGE system from Beckman Coulter.
This secondary analysis focuses on patients who were randomised to lidocaine and had at least one valid serum sample.
Dose rate calculations
In order to evaluate dosing strategies based on different body size metrics, the average dose rate received in mg/kg per h at each sampling point was calculated. For these calculations, the total lidocaine dose administered was divided by the infusion duration in hours, then divided again by one of four measures of body size commonly used in pharmacokinetic studies: total body weight, ideal body weight, adjusted body weight and lean body weight. Body size formulae evaluated by Green and Duffull were applied, 18 alongside a simpler formula for ideal body weight based on recent recommendations for easy bedside application (height in centimetres minus 105 for women). 8
Pharmacogenomics
At study conclusion, cheek swabs were mailed to participants allocated to the lidocaine arm. These were forwarded to myDNA (Melbourne, Australia) for pharmacogenomic single-nucleotide polymorphism analysis of CYP1A2 (rs76255, rs2069514, rs35694136), CYP3A4 (rs35599367) and CYP3A5 (rs776746, rs10264272, rs41303343). From the genotype results, individual phenotypes were summarised as follows: CYP1A2 normal, ultra rapid with inducer present, or unknown; CYP3A4 and CYP3A5 normal or intermediate. The dominant combined CYP1A2/CYP3A phenotype was ranked in order of the anticipated metabolism speed from slow to fast: Group 1, normal/intermediate; Group 2, normal/normal; Group 3, ultrarapid/intermediate; Group 4, ultrarapid/normal. If the CYP1A2 phenotype was unknown, no combined phenotype was assigned. The CYP3A4 and CYP3A5 categories were also combined to reflect an overall CYP3A phenotype 24 of poor, intermediate or extensive, and these in turn were explored in combination with CYP1A2 phenotype.
Statistical analysis
Continuous variables were tested for normality using D’Agostino’s omnibus test. All descriptive data for continuous variables are expressed as median (interquartile range (IQR)) (range) or number (percentage) because of skewed distributions.
To ensure adequate power, the analysis focused on regression models exploring covariates associated with lidocaine concentrations where there were at least 10 observations per covariate. Additional exploratory analyses were performed without a multiplicity adjustment of the statistical significance threshold.
Spearman’s rank correlation coefficient was used to identify covariates associated with the total serum lidocaine concentration in PACU during intravenous infusion, and the total serum lidocaine concentration on the ward during subcutaneous infusion. Backwards elimination linear regression models with natural log transformation were constructed to examine covariates in a multivariable context. Covariates entered into these models included age, infusion duration (from time of initial bolus) and average lidocaine dose administered per kilogram of body weight (mg/kg per h). These covariates were retained at a threshold of P < 0.1. Separate models were used for each body size metric.
The metabolic ratio for MEGX and GX was calculated by dividing the total lidocaine concentration by the metabolite concentrations. Changes in serum lidocaine and alpha-1 acid glycoprotein concentrations, metabolic ratios and protein binding in the early postoperative period were assessed in patients with both a PACU and a ward value, using the Mann–Whitney U test.
Serum lidocaine concentration-time profiles using the described drug delivery systems were evaluated with mixed-effects models incorporating non-linear fitting with fractional polynomials assessed using the χ2 test. All patients were assumed to have a lidocaine concentration of zero at time zero (the start of the lidocaine bolus).
To evaluate whether lidocaine is displaced from binding sites by the presence of other amide local anaesthetics, any correlation between the lidocaine free fraction and the serum concentration of ropivacaine and alpha-1 acid glycoprotein was assessed using linear regression.
The distribution of CYP phenotypes was summarised descriptively. Exploratory analyses of the impact of CYP phenotype on lidocaine concentration were conducted with the Mann–Whitney and Kruskal–Wallis tests as appropriate.
Analyses were completed in Stata (Stata Statistical Software: Release 16. 2019. StataCorp LLC, College Station, TX, USA) with a significance threshold of P < 0.05.
Results
Patient and procedural characteristics are summarised in Table 1. Lidocaine data were available for 48 patients and demonstrated a positively skewed distribution that normalised after natural log transformation.
Patient and treatment characteristics.
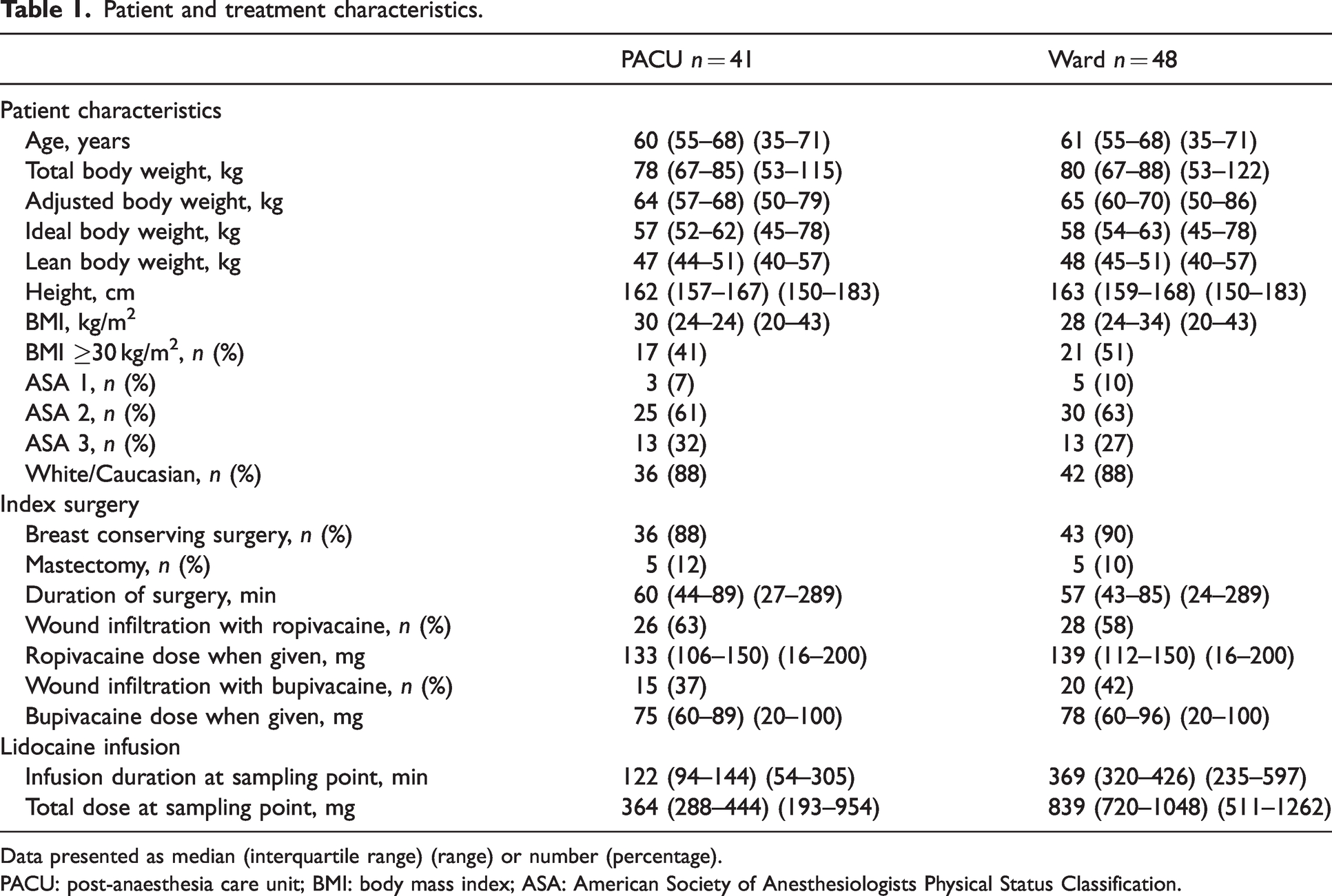
Data presented as median (interquartile range) (range) or number (percentage).
PACU: post-anaesthesia care unit; BMI: body mass index; ASA: American Society of Anesthesiologists Physical Status Classification.
Modelling of total serum lidocaine concentration
In the multivariable models at the PACU timepoint, infusion duration was the only variable associated with lidocaine concentration; coefficient = 1.21, R2 = 17.0%, P = 0.007 (Table 2). This relationship between intravenous infusion duration and lidocaine concentration was non-linear.
Univariable correlation and multivariable linear regression (backward elimination) for lidocaine concentrations in the post-anaesthesia care unit and on the ward.
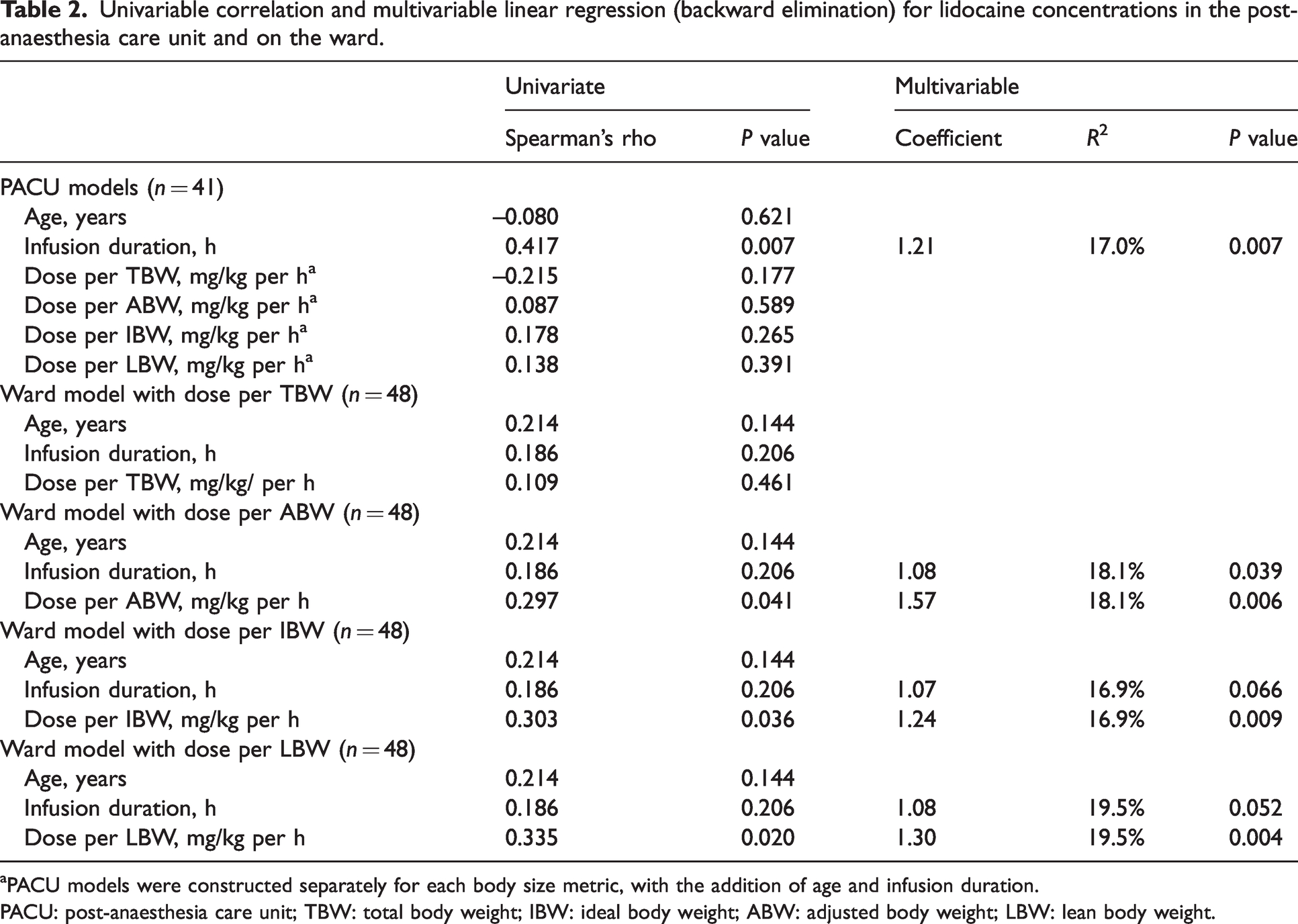
PACU models were constructed separately for each body size metric, with the addition of age and infusion duration.
PACU: post-anaesthesia care unit; TBW: total body weight; IBW: ideal body weight; ABW: adjusted body weight; LBW: lean body weight.
In the multivariable models of concentrations at the ward timepoint, the average lidocaine dose rate received per kilogram of adjusted body weight (ABW), ideal body weight (IBW) or lean body weight (LBW), but not total body weight, was associated with the serum lidocaine concentration (Table 2). The LBW model explained the most variance (R2 = 19.5%, P = 0.004); every 1 mg/kg per h increase in the average dose rate resulted in a 0.30 µg/ml increase in the lidocaine concentration. The relationships between doses received per body size metric and the resulting ward lidocaine concentrations are further described in Supplementary material Figure 1 online. Infusion duration was not a consistently significant covariate in the ward models.
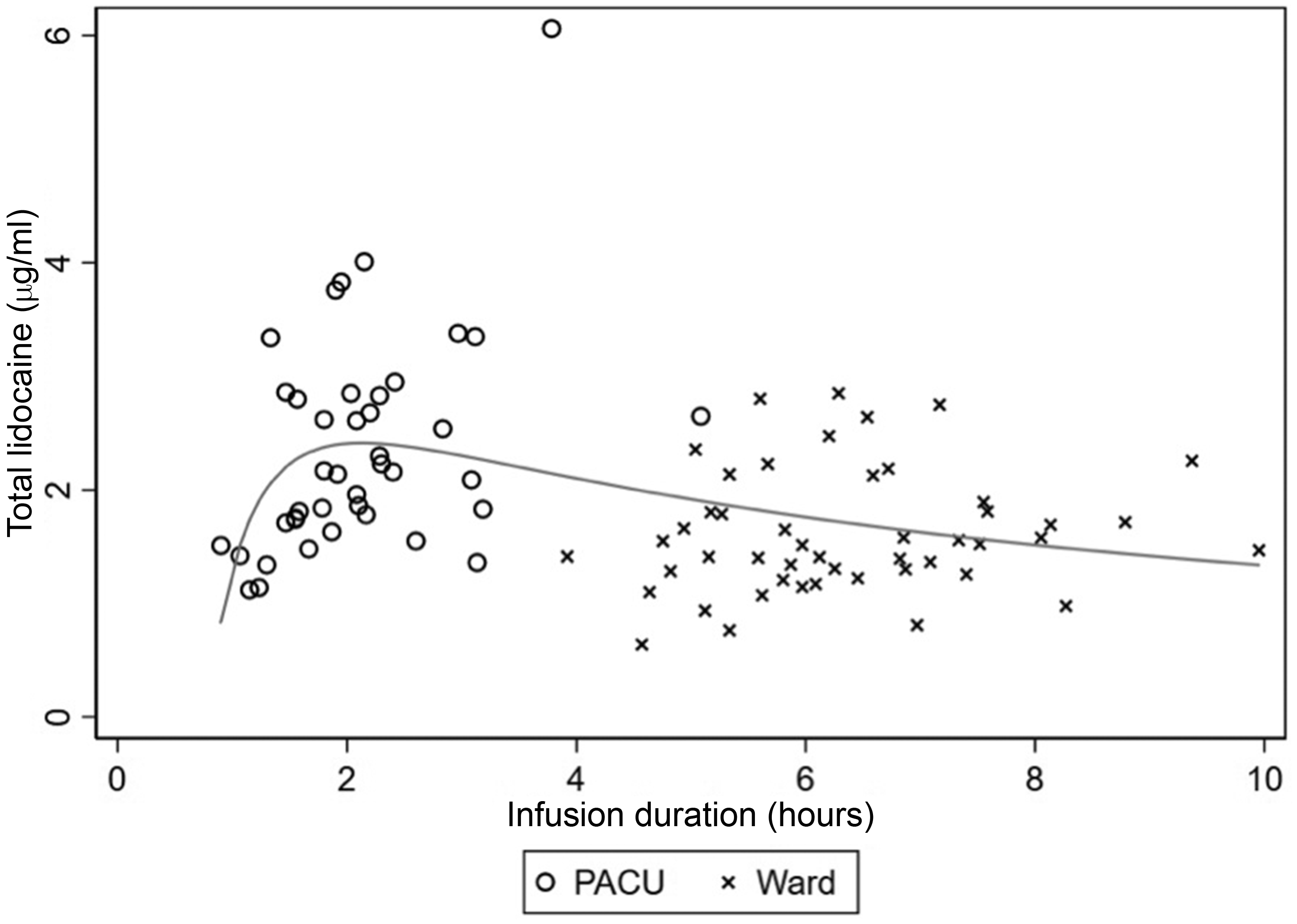
Non-linear fitted fractional polynomial curve of the relationship between infusion duration and total lidocaine concentration measured in the first 10 postoperative hours, with raw data included (mixed-effect model: R2 = 17.0%; P < 0.0001).
The exploratory mixed-effects model analysis of lidocaine concentrations across the perioperative infusion period incorporated two infusion duration covariates describing an early rise during the intravenous infusion followed by a gradual decline towards steady-state during the subcutaneous phase (Figure 1).
Paired samples
Data from participants with paired serum samples in PACU and on the ward (n = 39) are summarised in Table 3. Consistent with the higher doses during the intravenous phase, median (IQR) (range) total lidocaine concentrations were significantly higher in PACU than on the ward: 2.16 (1.73–2.82) (1.12–6.06) µg/ml versus 1.41 (1.22–1.75) (0.64–2.81) µg/ml, P < 0.0001. Only three patients had a higher lidocaine concentration at the later timepoint (+0.10, +0.44, +0.63 µg/ml).
Serum concentrations of lidocaine, monoethylglycinexylidide, glycinexylidide and alpha-1 acid glycoprotein in the post-anaesthesia care unit and on the ward (paired samples only).
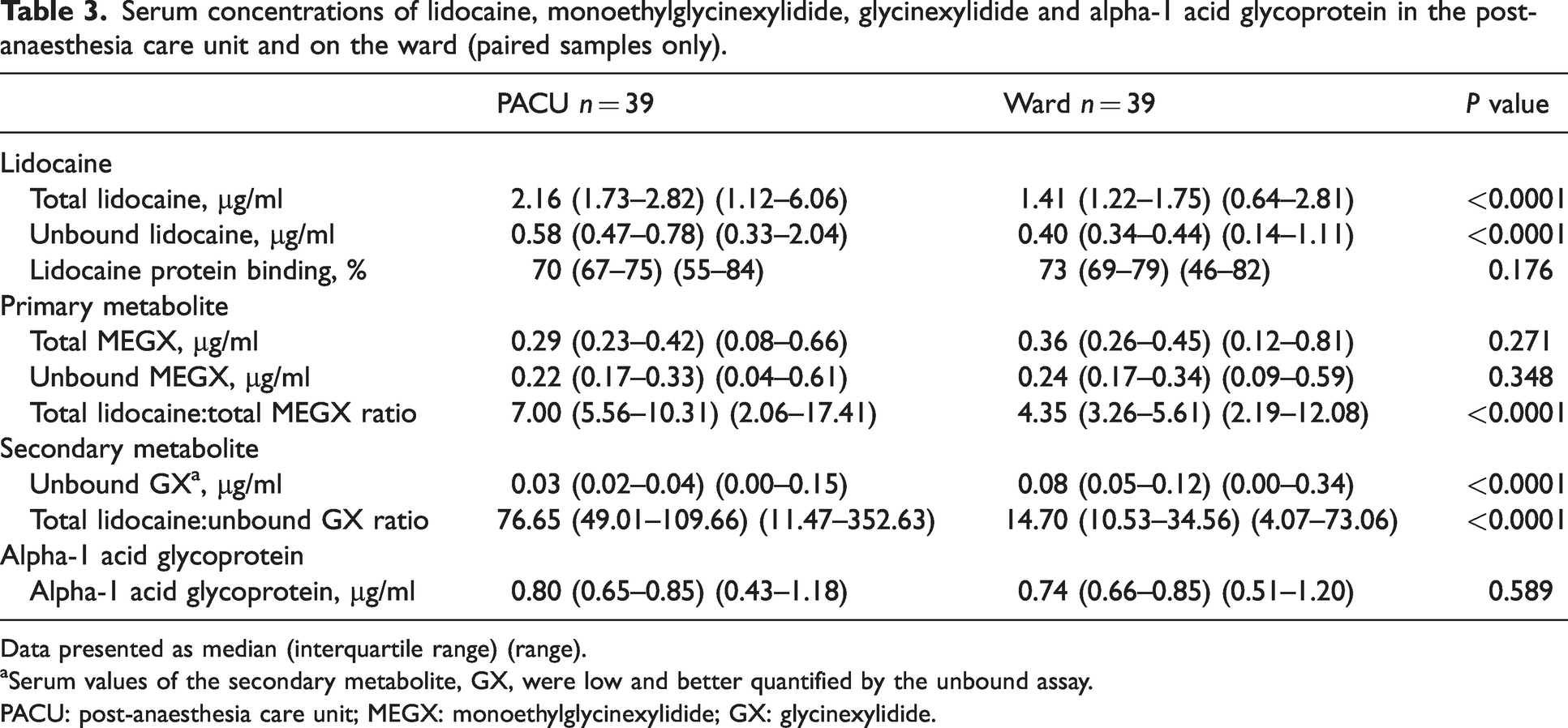
Data presented as median (interquartile range) (range).
aSerum values of the secondary metabolite, GX, were low and better quantified by the unbound assay.
PACU: post-anaesthesia care unit; MEGX: monoethylglycinexylidide; GX: glycinexylidide.
Neither alpha-1 acid glycoprotein concentration nor lidocaine protein binding changed significantly in the early postoperative period (Table 3). First-step metabolism to MEGX increased between PACU and the ward, reflected in a decreased metabolic ratio. Similarly, second-step metabolism to GX also increased between PACU and the ward, reflected in a decreased metabolic ratio.
Other amide local anaesthetics and protein binding
Serum ropivacaine and bupivacaine were detectable in less than 65% patients in PACU and on the ward (Supplementary Table 1). Total concentrations were low and did not exceed 0.39 and 0.14 µg/ml respectively. Protein binding (%) for ropivacaine and bupivacaine was considerably higher than for lidocaine. The lidocaine free fraction was not correlated with total ropivacaine or bupivacaine concentrations but was correlated with the alpha-1 acid glycoprotein concentration (Figure 2).
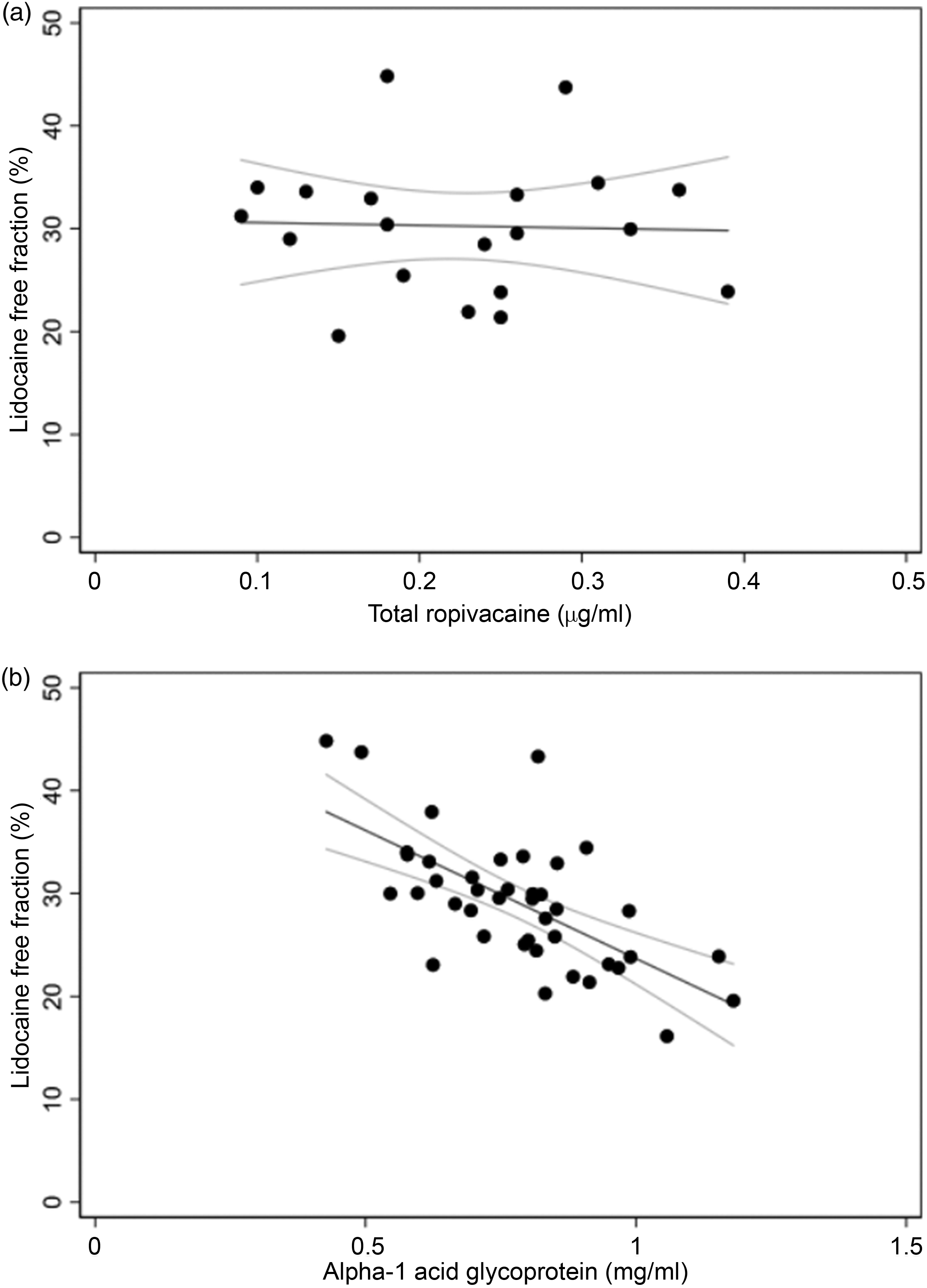
Scatterplots with linear regression for: (a) lidocaine free fraction versus total ropivacaine in post-anaesthesia care unit (PACU) (coefficient = –2.65, R2 = 0.12%, P = 0.886, n = 20) and (b) lidocaine free fraction versus alpha-1 acid glycoprotein in PACU (coefficient = –24.9, R2 = 43.4%, P < 0.001, n = 41).
Pharmacogenomics
Total lidocaine concentrations at the PACU and ward sampling points are summarised according to cytochrome P450 phenotypes in Supplementary Table 2. Phenotypes linked to slow metabolism were uncommon (≤12%). No statistically significant differences in lidocaine concentrations were apparent between phenotype groups at either timepoint.
Discussion
This secondary analysis of the LOLIPOP pilot study demonstrates that intraoperative intravenous lidocaine infusion, followed by a reduced postoperative subcutaneous infusion, results in relatively stable systemic drug concentrations 4–10 h after the initial drug bolus. Lidocaine exposure with our dosing strategy was highest and most variable in PACU, during a period of careful monitoring and one-to-one nursing. Thereafter, serum concentrations dropped slowly towards a pseudo steady-state. No ward lidocaine concentrations exceeded values purportedly linked to systemic toxicity (5 µg/ml). 8
The parameters we identified as significant determinants of systemic lidocaine concentrations at the early and later measurement points were consistent with pharmacokinetic studies in non-surgical settings. First, Bassan and colleagues have shown that it takes 6 h of lidocaine administration to approximate steady-state when a bolus and infusion combination is employed. 25 It is therefore unsurprising that infusion duration was the main determinant of lidocaine concentration in the PACU models, with sampling times ranging between one and five hours from the initial drug bolus. Clinicians using intravenous lidocaine intraoperatively with equivalent dosing schedules should be aware that longer operations will result in higher systemic concentrations.
Second, lidocaine metabolism observes first-order kinetics, 26 whereby the steady-state concentration (µg/ml) is determined by the fixed infusion rate (µg/kg per h) divided by the clearance rate (ml/kg per h). In surgical populations with lidocaine dosing per kilogram of total body weight, obese patients will receive a higher fixed infusion rate. They will not, however, exhibit a proportionally higher clearance rate, as this is largely determined by liver blood flow 27 that changes little with increased adiposity. Lidocaine clearance for a given individual is better reflected by alternative body size metrics such as LBW, which is well-established as a means of chronic drug dosing. 18 Indeed, obese patients in our study were observed to receive a higher fixed infusion rate per kilogram of lean, adjusted and ideal body weight and consequently exhibited higher steady-state concentrations (Supplementary Figure 1). Future studies should consider dosing lidocaine according to LBW to reduce some of the inter-individual variability in steady-state concentrations and avoid overshoot in obese patients (equivalent LBW doses: intravenous bolus 2.5 mg/kg; intravenous infusion 3.33 mg/kg per h; subcutaneous infusion 2.22 mg/kg per h).
In our models of lidocaine concentrations measured on the ward between 4 h and 10 h from the initial bolus, infusion duration was not a consistently significant covariate (ABW model, coefficient = 1.08, P = 0.039; IBW model, coefficient = 1.07, P = 0.066; LBW model, coefficient = 1.08, P = 0.052). However, these data remain consistent with small concentration increases over time, representing either the final approach towards steady-state, or autoinhibition of lidocaine metabolism by its own metabolites during prolonged exposure. 28 Consequently, gradual rises beyond 10 h cannot be ruled out. Indeed, Bauer and colleagues demonstrated falling lidocaine clearance rates in the first 30 h of drug infusion in healthy volunteers, 17 and many clinical studies have also reported lidocaine concentration rises during fixed rate infusions, well beyond the time required to reach steady-state.29,30 Importantly, the rises observed in these studies are gradual and unlikely to encroach on minor toxicity thresholds (5 µg/ml) when doses similar to those employed in our study are used for up to 24 h. 14 As longer-duration infusions may ultimately be necessary to maximise the antinociceptive, 4 antihyperalgesic 31 and anti-inflammatory 32 lidocaine effects in surgical patients, future work should evaluate whether autoinhibition is a robust and quantifiable phenomenon in the perioperative period.
The use of additional long-acting local anaesthetic agents was permitted in the pilot study in order to assess the added value of lidocaine over and above routine practice. Serum concentrations of ropivacaine and bupivacaine were low in PACU and on the ward, reflecting limited systemic absorption, and no major safety events were detected. 13 The absence of a correlation between the lidocaine free fraction and total ropivacaine suggests that significant competition for protein binding sites was not taking place, and the potential for synergistic toxicity demonstrated in vitro 19 is not a concern at these low concentrations. In this light, our data support the concept that recommendations to universally avoid other local anaesthetic techniques alongside lidocaine infusions 8 appear unnecessarily restrictive.33,34 Consistent with other breast surgery studies, 35 we observed no significant changes in concentrations of alpha-1 acid glycoprotein or lidocaine protein binding over time. Thus, total and unbound lidocaine concentrations are highly correlated in the very early postoperative period, and either can be used to probe efficacy and safety in breast cancer surgery patients.
In our pharmacogenomic analysis, phenotypes potentially linked to slow lidocaine metabolism via CYP systems were infrequent. The CYP3A4*22 variant, present in about 10% of European populations, has gained prominence due to its association with considerably reduced CYP3A4 protein expression and function. 36 The CYP1A2*1F variant is also common (over 50% in Europeans) and leads to rapid metabolism in the presence of inducers such as smoking. 37 The frequencies of these polymorphisms in the pilot study reflected the general population and were not associated with markedly different lidocaine concentrations. Although these data are only preliminary and based on a limited sample size, it remains reassuring that large fluctuations in lidocaine concentrations were not observed. Definitive pharmacogenomic studies can now be embedded within clinical trials based on these frequencies in a predominantly European-heritage cohort.
Finally, our data confirm that there is considerable inter-individual variation in lidocaine concentrations during perioperative administration, up to 80% of which remains unexplained. This variability is likely to pertain to inter-individual differences in liver blood flow and CYP function that are dynamic due to the impact of anaesthesia and surgery,38,39 and difficult to measure with conventional approaches. Even on the postoperative ward where haemodynamics are very stable in patients after breast surgery, we observed a positively skewed range of lidocaine concentrations spanning 2.17 µg/ml. At present, ensuring that the individuals creating this positive skew do not exceed toxic thresholds requires restraint in dosing regimens applied to whole populations. Ultimately, more population-specific data on lidocaine concentrations and safety are required to guide better precision.
This secondary analysis of the LOLIPOP pilot study has several limitations. First, we did not employ any population pharmacokinetic analyses to formally derive or test pharmacokinetic parameters. Second, the sample size was limited and no a priori power calculations were performed. Consequently, many of our analyses remain exploratory in the absence of formal multiplicity adjustments. Finally, our results cannot be generalised to other surgical populations, where postoperative liver blood flow and lidocaine metabolism are less predictable. 40
In conclusion, in patients undergoing routine care for breast cancer surgery we have shown that infusion of intravenous lidocaine intraoperatively followed by a lower dose subcutaneous infusion postoperatively resulted in pseudo steady-state concentrations 4–10 h after the initial bolus. Such steady-state concentrations were determined in part by the dose rate administered per kilogram of lean, adjusted or ideal body weight. Perioperative lidocaine concentrations exceeded or approximated therapeutic thresholds previously described in chronic pain cohorts 23 and those achieved in equivalent randomised trials. 8 Conversely, all ward concentrations were below the threshold traditionally linked to minor toxicity (5 µg/ml). Thus, in combination with reassuring safety data from the pilot study, 13 this dosing approach appears to balance efficacy and safety concerns. The definitive LOLIPOP trial (NCT05072314) is therefore employing a similar dosing regimen for up to 24 h, modified to a LBW approach. To evaluate the scope for further precision in lidocaine delivery, pre-specified sub-studies will fully explore autoinhibition and pharmacogenomic effects.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X231194833 - Supplemental material for Serum lidocaine (lignocaine) concentrations during prolonged perioperative infusion in patients undergoing breast cancer surgery: A secondary analysis of a randomised controlled trial
Supplemental material, sj-pdf-1-aic-10.1177_0310057X231194833 for Serum lidocaine (lignocaine) concentrations during prolonged perioperative infusion in patients undergoing breast cancer surgery: A secondary analysis of a randomised controlled trial by Andrew J Toner in Anaesthesia and Intensive Care
Supplemental Material
sj-pdf-2-aic-10.1177_0310057X231194833 - Supplemental material for Serum lidocaine (lignocaine) concentrations during prolonged perioperative infusion in patients undergoing breast cancer surgery: A secondary analysis of a randomised controlled trial
Supplemental material, sj-pdf-2-aic-10.1177_0310057X231194833 for Serum lidocaine (lignocaine) concentrations during prolonged perioperative infusion in patients undergoing breast cancer surgery: A secondary analysis of a randomised controlled trial by Andrew J Toner in Anaesthesia and Intensive Care
Footnotes
Author Contributions
Acknowledgements
At Royal Perth Hospital, Perth, the following research fellows assisted with data collection: F Droste, K Fisher, A Ganska, E Sethi-Chaudhary, M De Avila, D Ishak, C Fellingham, Y Yew. In-kind equipment support was received from REM Systems (North Ryde, Australia), including loan of Niki T34 syringe drivers and associated disposables, with no involvement in study design, data collection, analysis or interpretation.
Data sharing statement
Data are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Australian and New Zealand College of Anaesthetists (19/040); the Royal Perth Hospital Research Foundation (Springboard Grant 2019); and a Raine Medical Research Foundation/Department of Health Clinician Research Fellowship (CRF011-R7) to AT.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
