Abstract
Class III obese (body mass index ≥ 40 kg/m2) patients, now regularly encountered clinically, have increased perioperative risks, including potentially from suboptimal drug dosing. However, current dosing guidelines are based on low-level evidence and may not be widely accepted. This study aimed to investigate anaesthetists’ dosing practices for class III obese surgical patients, explore if they had experienced an increased incidence of adverse events potentially related to drug dosing with these patients and assess which resources they consulted for dosing advice in this population. An electronic survey was emailed to 1000 randomly selected members of the Australian and New Zealand College of Anaesthetists. Data were summarised and the Pearson’s χ2 test was used to compare respondents’ genders, geographic locations and seniority designations with the greater Australian and New Zealand College of Anaesthetists’ membership. There were 230 completed responses (response rate 23%). A large proportion (46%–76%) of respondents indicated they dose class III obese patients in keeping with current recommendations; however, substantial heterogeneity in dosing practices was found. Lean body weight was the most frequently used regimen for dosing propofol, non-depolarising muscle relaxants, sugammadex and opioids, whereas total body weight was most frequently used for suxamethonium. Nearly 70% of respondents reported using at least one resource to assist their dosing practices in obesity. Importantly, increased incidences of adverse events in class III obese patients related to drug dosing were commonly experienced by respondents. Until higher-level evidence is available for dosing class III obese patients, anaesthetists should consider current recommendations and exercise increased attention to dosing. Further clinician education may assist in optimising dosing in this patient group.
Introduction
The prevalence of obesity in developed countries has rapidly increased in the last two decades.1,2 Obese patients are more likely than non-obese to undergo surgery, including for bariatric procedures and obesity-related comorbidities. 3 Obesity-related physiological changes, combined with altered pharmacokinetics and pharmacodynamics, may increase the risk of drug dosing problems perioperatively in obese patients. 4
The combined guidelines of the Association of Anaesthetists of Great Britain and Ireland (AAGBI) and the Society for Obesity and Bariatric Anaesthesia (SOBA), mainly based on small-scale pharmacokinetic studies rather than large-scale clinical studies, state that dosing based on total body weight (TBW) increases the risk of relative overdose and suggests some dosing strategies.5,6 Various expert reviews and commentaries suggest similar perioperative drug dosing strategies in obesity to the AAGBI/SOBA guidelines.4,7–11 Pharmacokinetic studies can provide important baseline dosing knowledge; however, this knowledge may not always be clinically practical. 12 There are no drug dosing recommendations based on Level 1 evidence available for class III obese (body mass index ≥ 40 kg/m2) patients undergoing anaesthesia. 13 There are also few studies exploring anaesthetists’ actual dosing practices for these patients.
This survey study aimed to investigate current perioperative drug dosing practices for class III obese patients, explore if anaesthetists have experienced more adverse events potentially related to dosing with these patients and identify the dosing resources they used.
Methods
Ethical approval to conduct this survey was obtained from the Tasmanian Human Research Ethics Committee (H0017165). Participation in this survey was considered implied consent, as was outlined in the participant information sheet. The survey’s initial draft was developed by the research team (comprising a consultant anaesthetist and five pharmacists) based on an extensive literature review. This first draft was emailed to a small group of other consultant anaesthetists (n=10) for face and content validation. The feedback was extensively reviewed by the research team and suggested changes were incorporated. This process was similarly repeated with the second draft (n=10) to assist in producing the final survey. The final survey (Appendix 1 (available as online supplementary materials)) consisted of four sections designed to capture anaesthetists’: (a) demographic information; (b) dosing scalars used for common intravenous peri-operative medications in class III obesity; (c) relative incidence, compared with non-obese individuals, of adverse events related to drug dosing experienced with class III obese patients; and (d) resources regularly referenced for dosing advice in class III obese patients. The dosing regimens assessed were TBW = weight in kilograms; lean body weight (LBW) = TBW minus body fat weight; ideal body weight (IBW) = weight corrected for sex, height and frame size; and adjusted body weight (ABW)/corrected body weight = correction factor of excess weight above IBW added to IBW.
After the survey questionnaire was finalised, an application was made to the Australian and New Zealand College of Anaesthetists’ (ANZCA) Clinical Trials Network (CTN) to seek assistance in distributing the survey to its members. After review and approval by the CTN, a cover letter and online survey link (using Lime Survey version 2.06, Hamburg, https://www.limesurvey.org/) was emailed by ANZCA to 1000 randomly selected members (786 Fellows and 214 trainees) in August 2018. This strategy had been used in several previous ANZCA CTN surveys.14,15 The survey was blinded, as investigators were not involved in the randomised recipient-selection process by ANZCA. Therefore, also, information about non-respondents could not be collected. The survey was anonymous and no internet protocol addresses were collected. The survey remained active for a period of two months, with email reminders sent by ANZCA to all recipients at one week and then again at six weeks following the initial invitation.
Categorical variables were presented as counts and percentages. Pearson’s χ2 test was used to compare the respondents’ genders, geographic locations and seniority designations to the greater ANZCA membership. A P value <0.05 was considered statistically significant. Analyses were performed using SPSS version 24 (IBM Inc., Chicago, IL, USA).
Results
Of 1000 invitations sent to ANZCA members, 230 complete responses were received (response rate 23%). The demographics of respondents were representative of the majority of the ANZCA membership, with no significant differences between the distribution of respondents and ANZCA members for gender, the majority of geographical locations, and seniority designation (Table 1). Respondents were mostly male (69%), Fellows (85%), working full-time (76%) and had more than ten years of anaesthesia experience (49%).
Details of survey respondents and comparison with the Australian and New Zealand College of Anaesthetists’ membership.
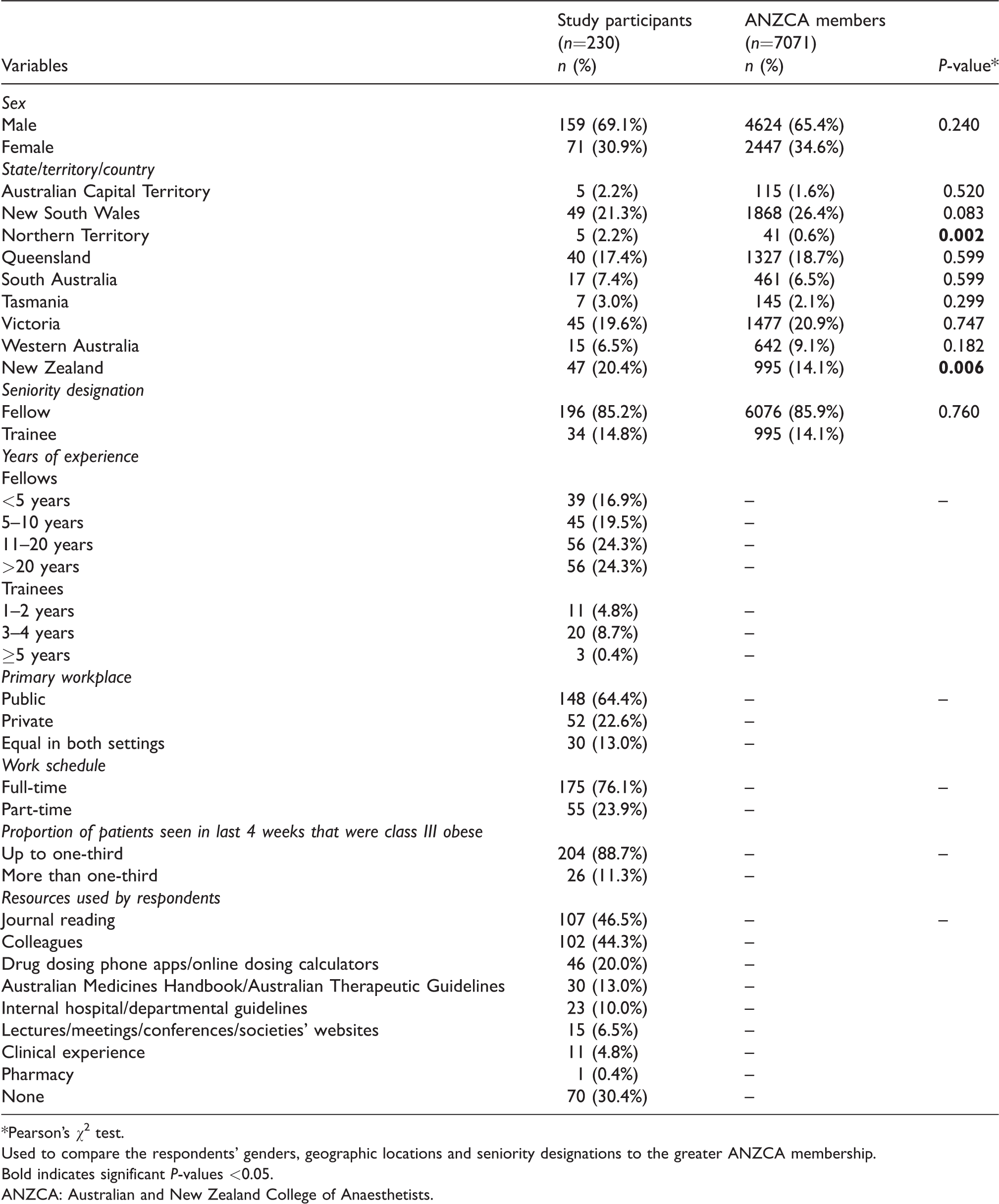
*Pearson’s χ2 test.Used to compare the respondents' genders, geographic locations and seniority designations to the greater ANZCA membership.
Bold indicates significant P-values <0.05.ANZCA: Australian and New Zealand College of Anaesthetists.
With the exception of TBW for dosing suxamethonium, LBW was the most frequently selected regimen for dosing all other drugs asked about in the survey (Figure 1). Still, for all drugs, a heterogeneous pattern of dosing practices was observed. Most respondents (70%) reported using at least one resource to assist their dosing practices in obesity (Table 1). Journal reading (46%) (where guidelines are published) and consultation with colleagues (44%) were the most common.
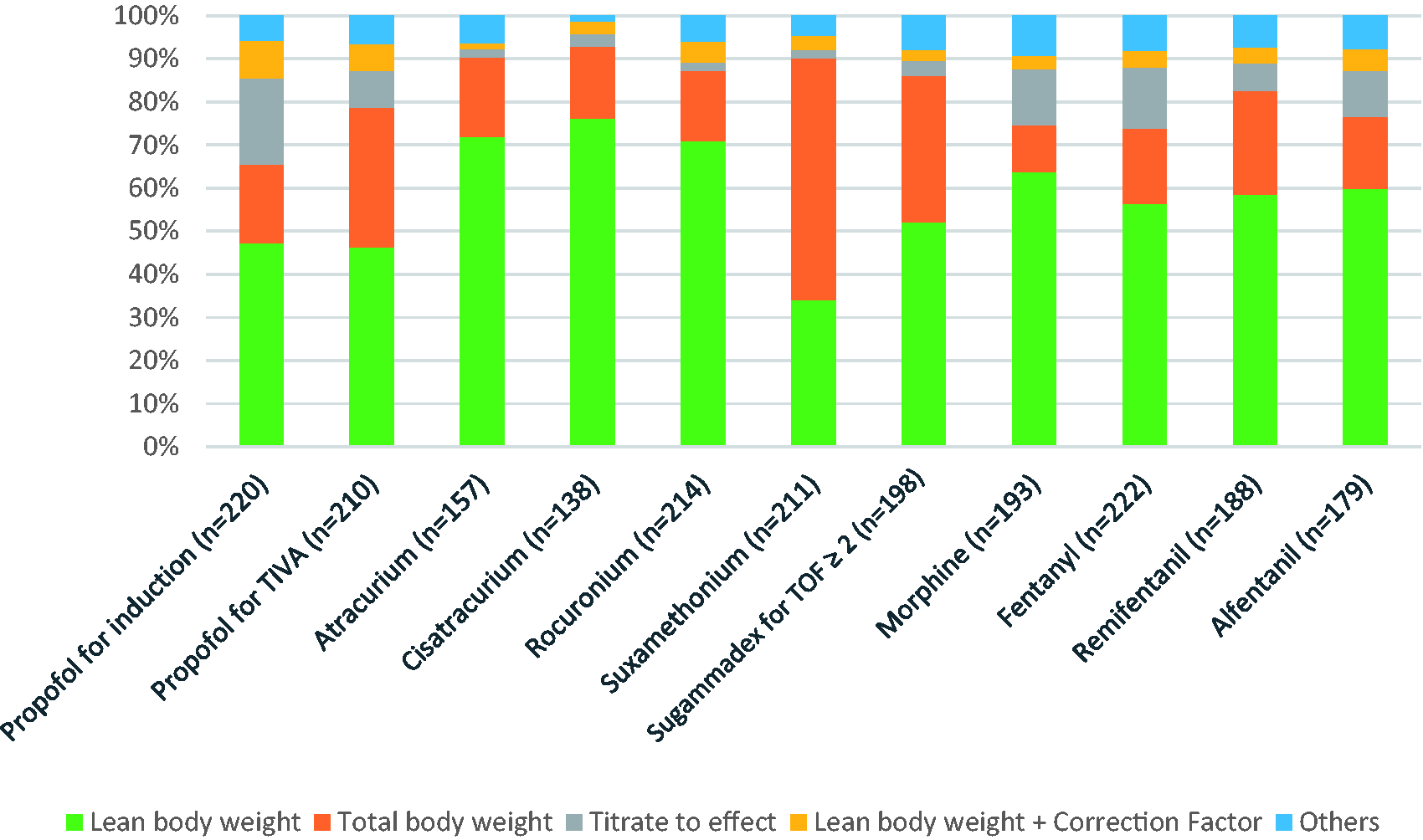
Dosing practices of respondents in class III obese patients. Do not use/not specified survey responses for propofol (induction) = 10 (4.3%), propofol (TIVA) = 20 (8.7%), atracurium = 73 (31.8%), cisatracurium = 92 (40.0%), rocuronium = 16 (6.9%), suxamethonium = 19 (8.3%), sugammadex = 32 (13.9%), morphine = 37 (16.0%), fentanyl = 8 (3.5%), remifentanil = 42 (18.2%) and alfentanil = 51 (22.2%). TIVA: total intravenous anaesthesia; TOF: train-of-four.
Respondents reported an increased incidence of adverse events potentially related to drug dosing for class III obese patients seen in anaesthetic practice (Table 2).
Reported increased incidence of adverse events in class III obese patients.

Discussion
This study provides insights into dosing practices for commonly used intravenous perioperative drugs in class III obese patients in Australia and New Zealand. Most respondents (46%–76%) employed dosing practices (for propofol, common muscle relaxants, sugammadex and common opioids) in keeping with current advice. However, for all these drugs, there was substantial heterogeneity in dosing practices. Most (70%) respondents described using some published reference for dosing advice in this patient group.
The AAGBI/SOBA guidelines recommended LBW and ABW as suitable dosing scalars, respectively, for propofol induction and total intravenous anaesthesia (TIVA).5,6 The survey results showed that LBW was the most commonly used regimen for both induction (47%) and TIVA (46%), yet over half of the respondents used a variety of other dosing methods.
For non-depolarising muscle relaxants, the majority of respondents reported using the LBW regimen (72% for atracurium, 76% for cisatracurium and 71% for rocuronium), which is in concordance with the AAGBI/SOBA recommendations.5,6
The AAGBI/SOBA guidelines, however, do not provide recommendations for dosing suxamethonium or sugammadex. For suxamethonium, survey results showed that most respondents (56%) used TBW, which is in keeping with the advice of other current literature.7,11 For sugammadex, more than half of respondents (52%) used LBW. There is controversy in the literature regarding the appropriate dosing regimen for sugammadex in class III obese patients, with some studies suggesting IBW, ABW or TBW.16–20
According to the AAGBI/SOBA guidelines, LBW is the preferred dosing regimen for most opioids, except alfentanil.5,6 Our survey results found that most respondents use LBW for morphine (64%), fentanyl (56%), remifentanil (59%) and alfentanil (60%). Nearly 40% reported using a variety of other dosing strategies. Importantly, more respondents stated increased incidences of adverse effects in class III obese patients (versus non-obese patients) with opioids than with any other drugs included in the survey. This finding may be multifactorial (including due to inappropriate drug dosing, drug selection and post-operative ward monitoring), combined with pre-existing obesity-related pathophysiology. There may be no dosing regimen that is always safest for dosing these drugs in class III obese patients, especially when considering age differences, comorbidities such as obstructive sleep apnoea and renal impairment, as well as opioid tolerance, dependence and addiction. 21 Furthermore, opioids are generally higher risk in class III obese patients because of the risk of obesity hypoventilation syndrome.22,23
This study has some limitations. Firstly, the response rate of 23% was relatively low, possibly due to survey fatigue in recipients (from frequent requests for survey completion), or a degree of disinterest in the survey’s subject (as might be the case with recipients who do not encounter a high number of obese patients in their practice). In addition, ANZCA have advised that a small proportion of survey recipients may not have been practising anaesthetists (i.e. purely pain medicine practitioners), but the exact number of these recipients could not be provided. Therefore, this study’s findings may not represent the majority of survey recipients, and so the potential for responder bias exists. Nevertheless, a broad representation of ANZCA’s anaesthetic membership was achieved.14,15 Secondly, not all drugs used in anaesthetic practice were included in the survey (such as volatile anaesthetics and sympathomimetics), which may have biased some factors surveyed (for example, residual muscle weakness could be exacerbated by volatile anaesthesia). In addition, as an exploratory study, results would need to be confirmed by future studies.
In conclusion, this study has shown that, although 46%–76% of respondents dose drugs for class III obese patients in a manner consistent with AAGBI/SOBA guidelines, a substantial proportion of dosing practices are heterogeneous. This heterogeneity could reveal either a dosing knowledge gap amongst respondents, or inadequate application of dosing knowledge in this population. Further, this heterogeneity may be contributing in part to respondents reporting a higher incidence of adverse events related to drug dosing in class III obese patients. Ongoing clinician education may assist with optimising drug dosing in class III obese patients. The AAGBI/SOBA guidelines consider the dosing regimens it recommends as being the safest; however, there remains a lack of high-level evidence for them. Establishing higher-level evidence over time may also improve drug-dosing safety in class III obese patients. Until higher-level evidence is available for dosing class III obese patients, anaesthetists should carefully consider current recommendations as well as exercise increased attention around dosing for these high-risk patients.
Supplemental Material
AIC886596 Supplemetal Material - Supplemental material for Anaesthetists’ drug dosing practices in class III obese surgical patients: A bi-national survey
Supplemental material, AIC886596 Supplemetal Material for Anaesthetists’ drug dosing practices in class III obese surgical patients: A bi-national survey by Zahid Hussain, Karl Gadd, Colin Curtain, Corinne Mirkazemi, Gregory M Peterson and Syed Tabish R Zaidi in Anaesthesia and Intensive Care
Footnotes
Acknowledgements
We are grateful to the ANZCA CTN for distributing this survey to its members.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
