Abstract
The haemodynamic response to the beach-chair position may be affected by the presence of left ventricular hypertrophy where remodelling of cardiac chambers can potentially lead to left ventricular outflow tract obstruction. We present a case report of severe hypotension in the beach-chair position, where focused cardiac ultrasound identified left ventricular hypertrophy and geometric features that contributed to the hypotensive response. This case illustrates that focused cardiac ultrasound has the potential to alert the clinician preoperatively to left ventricular outflow tract obstruction susceptibility during surgery in the beach-chair position, and intraoperatively to direct management should sudden decreases in blood pressure occur.
Keywords
Introduction
For surgery in the beach-chair position (BCP), the placement of a patient into an upright position has an immediate effect on preload, cardiac output (CO) and autonomic reflex activation.1,2 As a result, patients may experience significant and protracted hypotension, potentially leading to cerebral ischaemia. 3 The haemodynamic response to the BCP is also influenced by the presence of left ventricular hypertrophy (LVH), commonly associated with remodelling of cardiac chamber size and shape. 4 In asymptomatic patients, the presence and degree of LVH may not be identified during routine pre-anaesthetic assessment. 5 In this report, with the patient’s consent, we describe the use of focused cardiac ultrasound (FCU) in a patient with LVH, before and during elective BCP surgery.
Case report
A 67-year-old man presented for elective arthroscopic cuff repair of his left shoulder. His past medical history was unremarkable apart from hypertension, which had been treated with telmisartan and metoprolol for five years. Preoperative assessment indicated normal functional capacity with no cardiorespiratory symptoms or restriction of daily activity. His body mass index was 24 kg/m2, and physical examination was unremarkable. Preoperative electrocardiography showed sinus rhythm with minor features suggestive of LVH. Baseline full blood examination was normal. In view of these findings, his physical status was classified as American Society of Anesthesiologists risk classification 2, and he proceeded to surgery.
Preoperative transthoracic echocardiography (TTE)
In our practice, the clinical decision to perform FCU in beach-chair patients is individually based, dependent on age, comorbidity and pre-existing cardiac disease, including hypertension. In this patient, a focused cardiac examination involving parasternal long-axis (PLAX), apical four-chamber (4C) and apical long-axis views was performed (5–1 MHz phased array transducer (P21x) M-turbo; Sonosite, Sydney, Australia). In the PLAX view (Figure 1(a)), left ventricular (LV) posterior wall thickness was measured as 14 mm (normal 6–10 mm), interventricular septum diameter 12 mm (normal 6–10 mm), LV end-diastolic and end-systolic diameters 3.93 and 3.0 cm (normal 4.2–5.8, 2.5–4.0 cm), respectively, and LV outflow tract diameter index (LVOTI) 8.21 mm/m2 (normal 12–14 mm/m2). The basal septum was also hypertrophied (15 mm). Significant LVH was evident from both wall thickness and calculation of the LV mass index (137.71 g/m2; normal 45–115 g/m2). Contraction of the LV chamber was also reflected in the relative wall thickness (RWT) measurement of 0.68 (normal <0.44). All measurements were in accordance with the American Society of Echocardiography guidelines. 6
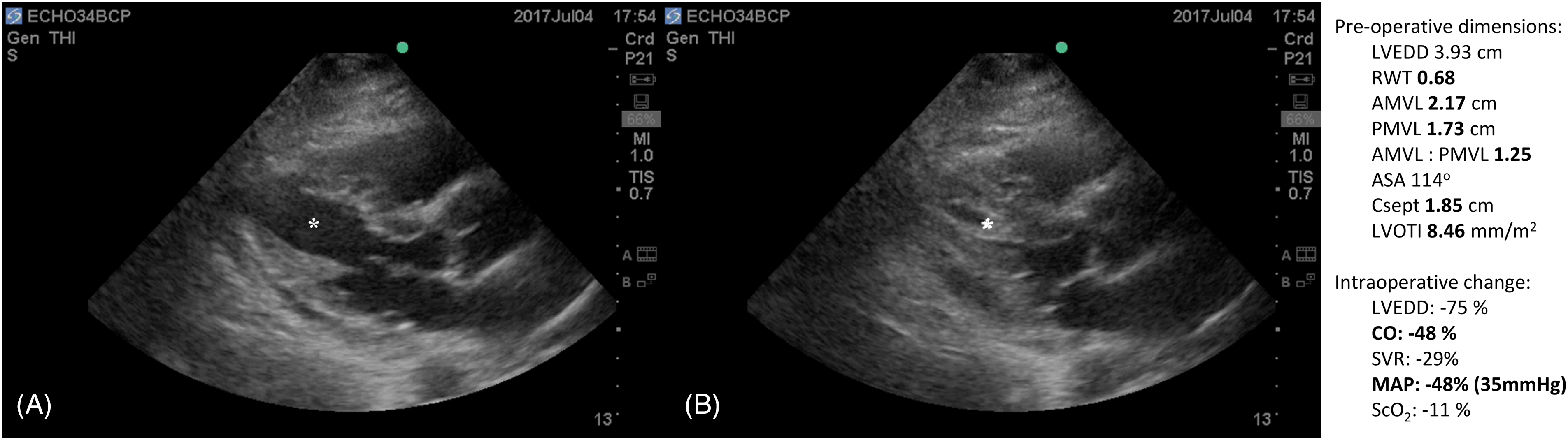
Parasternal long-axis view of a 67-year-old patient undergoing shoulder surgery. Severe hypotension on beach-chair positioning was attributed to acute left ventricular cavity obliteration and distortion of aorto-mitral geometry. (a) Preoperative dimensions of left ventricular outflow tract geometry indicate the presence of risk factors for left ventricular outflow tract obstruction. (b) Acute left ventricular cavity obliteration showing severely contracted chamber (*), with measurements associated with the haemodynamic change.
The aorto-mitral complex was examined and showed an aorto-mitral angle of 119° and aorto-septal angle of 114°. Mitral valve anterior and posterior leaflet heights were 2.2 and 1.6 cm, respectively (ratio <1.3), and the distance between leaflet coaptation point and interventricular septum (Csept) was 1.85 cm. During the cardiac cycle, the left ventricular outflow tract (LVOT) and aortic and mitral valves were examined using colour flow Doppler (CFD). Minor LVOT flow convergence was noted, and valves were functionally normal.
In the 4C view, the contractility of the left and right ventricles was within normal limits. In the apical three-chamber view, pulsed-wave Doppler did not indicate the presence of a significant LVOT flow gradient at rest or during Valsalva manoeuvre. Cardiac index was estimated as 2.2 L/min/m2. Systemic vascular resistance (SVR) was derived using the relation SVR = 80 (mean arterial pressure (MAP)/CO).
Beach-chair placement
On upright positioning (head elevation 75°), there was an immediate decrease in arterial blood pressure to MAP 35 mmHg and a decrease in cerebral oxygen saturation. An immediate FCU examination showed a significant decrease in preload, with LV cavity obliteration and distortion of the aorto-mitral complex (Figure 1(b)). Mild LV global hypokinesis was also evident. The patient was treated aggressively with repeated administration of an alpha1 agonist (metaraminol) and intravenous fluid. Subsequent re-expansion of the LV chamber was observed, and as ventricular shape was normalised, CO and MAP improved. As LV size increased, CFD examination of the LVOT identified flow convergence suggestive of obstruction. Following haemodynamic stabilisation, low-dose metaraminol was infused to maintain arterial pressure, ensuring adequate cerebral and organ perfusion during the surgical procedure.
Discussion
This report illustrates how FCU can play an important role in the perioperative management of patients in the BCP. First, preoperative FCU examination identified the presence of LVH and features of cardiac geometry known to predispose to dynamic LVOT obstruction (LVOTO). The pattern of concentric hypertrophy of the LV resulted in both chamber size and outflow dimension being relatively small, reflected in the measurement of RWT (0.68) and LVOTI (8.25 mm/m2). Structural narrowing of the LVOT was evident with the presence of basal septal hypertrophy. Further, the geometry of the aorto-mitral angle indicated features specific for LVOTO risk—a decreased Csept distance <2.5 cm, anterior:posterior mitral valve leaflet ratio <1.3 and aorto-septal angulation <120°. These echocardiographic features have been established from studies investigating patients with hypertrophic cardiomyopathy (HCM), during stress echocardiography, or following post-surgical mitral valve repair.7–15
Patients with LVOTO geometry are susceptible to developing sudden decreases in CO, usually with acute changes in preload, 16 as occurs in a number of clinical situations, including beach-chair positioning. The mechanism of obstruction relates to the interaction between predisposing cardiac geometry and dynamic cardiac contraction. A hypercontractile empty ventricle can itself result in distortion of the LVOT, LV chamber, papillary muscles and mitral valve. Systolic contraction increases both LVOT velocity and movement of the mitral valve, particularly the leaflet co-aptation point, towards the septum. Systolic anterior motion of the anterior leaflet (SAM) into the LVOT may then occur, exacerbating flow obstruction and causing mitral incompetence. In patients with HCM, the mechanism of SAM is a predominant component of LVOT obstruction. With LVOTO, hypotension and low CO syndrome are best treated with aggressive fluid therapy, vasoconstrictors and beta-adrenoceptor antagonism. Importantly, administration of inotropes and vasodilators can aggravate the condition.
In this patient, the dramatic effect of acute preload reduction, with collapse of the LV cavity, distortion of outflow geometry and associated dramatic decrease in CO, was clearly demonstrated by intraoperative FCU examination. Although an LVOT gradient was not measured, outlet deformation and obstruction was believed to be the primary cause for hypotension and cerebral desaturation. During treatment, monitoring provided real-time evaluation of repeated vasopressor and fluid therapy, and with LV cavity re-expansion, contractility and ventricular ejection was seen to improve. The alpha1 agonist metaraminol had a direct effect on both systemic vascular impedance and venous return, facilitating LV expansion, and was used in preference to ephedrine, a sympathomimetic with mixed chronotropic and inotropic action. Importantly, in hypotensive BCP patients unresponsive to therapy, rapid placement into a supine position is necessary to restore systemic perfusion. 17
According to current guidelines, preoperative TTE is only indicated for evaluation of unexplained dyspnoea, limitation of function or previous cardiac disease in preoperative patients. 5 The American Society of Echocardiography, however, has recognised the rapidly growing role of focused transthoracic cardiac ultrasound, 18 and defines this approach as an examination directed towards a specific clinical question or evaluation ‘appropriate for the clinical setting’. The role of FCU in the perioperative evaluation of patients is gaining momentum,19–22 but there remains the question of what actually defines a minimal focused examination in patients undergoing surgery. Importantly, accurate measurement and interpretation in TTE is reliant on operator experience and accreditation. Expertise in focused imaging requires multiple skill sets, including correct transducer orientation and avoidance of foreshortening, as well as appropriate settings for calliper and Doppler measurement. Expertise also requires recognition and interpretation of variant morphology, as well as an understanding of when formal assessment is indicated. Operator inexperience can be a major limitation for FCU, with misdiagnosis having the potential to affect patient management adversely.
Finally, hypertension is a prevalent condition, and many patients may present for surgery with unrecognised LVH 23 and propensity to develop LVOTO. In experienced hands, an FCU examination has the potential to identify and evaluate the significance of LVH before surgery. LVOTO risk may be suspected when one or more key sonographic features are present. These include the presence of LVH with a small LV chamber, a prominent basal septum with narrowing of the LVOT, flow convergence on CFD (with Valsalva) or abnormal anterior mitral valve leaflet movement suggestive of SAM. In BCP surgery, this approach may alert the clinician to anticipate LVOTO, and consider it as a possible cause of intraoperative hypotension.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
