Abstract
Stress ulcer prophylaxis is commonly prescribed in the intensive care unit but can be inappropriately commenced or continued on discharge, exposing patients to potential harm. We aimed to evaluate whether a prescribing guideline, education program and pharmacist oversight would reduce inappropriate continuation of stress ulcer prophylaxis. This was a multicentre pre- (2014) and post- (2016) quality improvement study across five Australian intensive care units. Cost data were estimated using local information about prescribing patterns, and the relationship between long-term use and adverse events. A total of 531 patients were included in the pre- and 393 in the post-implementation periods. The proportion of hospital survivors inappropriately continued on stress ulcer prophylaxis reduced from 78/184 (42.4%) to 11/143 (7.7%) in the post-implementation period (odds ratio = 8.83; 95% confidence interval 4.47–17.45; P < 0.0001). Clostridium difficile–associated disease reduced from 10 patients to one in the pre- to post-implementation groups. The extrapolated direct savings to all Australian intensive care units from reduced proton pump inhibitor prescribing are relatively small (AUD$2.08 million/year), but the reduction in complications has both benefits for patients and indirect savings of AUD$16.59 million/year nationally. In patients admitted to the intensive care unit, the introduction of a simple, bundled intervention resulted in a significant decrease in inappropriate continuation of stress ulcer prophylaxis at hospital discharge and a reduction in recognised complications, and substantial cost savings.
Keywords
Introduction
Stress ulcer prophylaxis (SUP) is commonly prescribed in the intensive care unit (ICU) to reduce the risk of gastrointestinal bleeding.1–3 The prescription and continuation of SUP often exceeds clinical guideline recommendations.4,5 Furthermore, a substantial proportion of patients commenced on SUP in the ICU continue on these medication at hospital discharge without clear ongoing indication. 6 The resultant over-utilisation of SUP is costly and exposes patients to the potential side-effects of these medications.4,7,8 These include an association with increased risk of Clostridium difficile infection, ventilator-associated pneumonia, interstitial nephritis, dementia, osteoporosis and mortality.9,10 Therefore, there is an unmet need to assess the effectiveness of interventions aimed at decreasing inappropriate prescription and continuation of SUP in patients admitted to the ICU. The aim of this study was to assess whether the introduction of a bundled approach, including a pharmacist-guided de-escalation protocol, could improve SUP prescription compliance and reduce costs in patients admitted to the ICU.
Methods
A multicentre pre- and post-implementation quality improvement study was conducted in five adult ICUs in Western Australia (two tertiary, two metropolitan and one regional ICU). The SUP prescribing practices were compared before and after an intervention aimed at improving compliance with optimal initiation and duration of SUP therapy. All sites had pharmacists present in the ICU in both study periods and used paper-based records throughout. There was no SUP protocol or guideline at any of the study sites in the pre-implementation period. The post-implementation period involved the introduction of a bundle that included: (a) site-based dissemination of locally produced SUP prescription guidelines for medical staff that included recommendations for initiating and ceasing SUP and documentation of the indication and suggested duration of SUP based on the best available evidence (Appendix 1); and (b) ICU pharmacist-led discontinuation of SUP prior to ICU discharge if an indication for ongoing SUP was no longer present. 11
Data were collected prospectively using a standardised data extraction form for consecutive patients admitted at each participating site with a pre-specified sample size of 50–150 participants (depending on unit size). Due to the differing time for approvals at each participating site, the data for the pre-implementation phase were collected between September 2013 and January 2015. The post-implementation data were collected from all sites between May and October 2016, and due to resource constraints at some sites, they collected 50–100 patients. Patients <18 years of age were excluded. Risk factors for stress ulcer bleeding were classified as major and minor, in accordance with the current literature. 12 A complete list of risk factors and the prescribing guideline is provided in Appendix 1. Proton pump inhibitor (PPI) therapy for the treatment of upper gastrointestinal (UGI) bleeding as the indication for ICU admission or another documented indication for long-term PPI was differentiated from SUP that was initiated in the ICU. For the purposes of this study, pre-hospital PPI or H2 receptor blocker (H2RB) without clear indication for long-term therapy was still considered an acceptable indication for ongoing use post discharge from the ICU. Participants who died prior to hospital discharge were considered to have ceased SUP. Hospital- or ventilator-acquired pneumonia was defined by the initiation or escalation of antibiotic therapy for a suspected or confirmed clinical diagnosis of pneumonia >48 hours after ICU admission. A significant gastrointestinal bleed was defined as sufficient to require the clinicians to organise either a gastroscopy or transfusion of blood products. New C. difficile infection was defined as the occurrence of relevant symptoms in ICU patients with a positive stool toxin and/or polymerase chain reaction (PCR) after commencement of SUP in the ICU. The default SUP therapy at all study sites was once daily administration of intravenous pantoprazole 40 mg. Institutional approval was obtained for all sites prior to commencement of the study (Sir Charles Gairdner Hospital Ethics 2015-009, Joondalup Hospital Ethics #1512).
Descriptive statistics were based on frequency distributions for categorical data and medians and interquartile ranges for continuous data. Chi-square tests were used for categorical group comparisons based on pre and post groups. Data were analysed using IBM SPSS Statistics for Windows v24.0 (IBM Corp., Armonk, NY) and STATA/IC v14.2 (StataCorp, College Station, TX). P-values <0.05 were considered statistically significant.
To estimate the cost impact of reducing inappropriate SUP at discharge, we augmented the clinical data presented here with estimates around longer-term use and events from the literature. To estimate the number of patients discharged on an inappropriate SUP, we applied the proportion of such patients in the two arms to estimates of the national ICU population. 13 To estimate attrition, data from New Zealand were used, which suggested that 71% of patients receiving SUP at ICU discharge continued to receive it in the long term. 6 We do not have an estimate for how long such patients remain on SUPs; we assumed the mean was ten years, but acknowledge there will be significant outliers in both directions. To estimate the cost of this ongoing prescribing, we assumed the SUP was pantoprazole 40 mg, and applied the Dispensed Price for Maximum Quantity (DPMQ), giving an annual cost of AUD$166.07. Regarding cost savings from reduced adverse events, a number of assumptions are required to produce realistic cost implications. Due to data limitations, we focused exclusively on C. difficile–associated disease (CDAD). Using the C. difficile proportions from our data, and again applying these to the estimated Australian ICU population, we estimated the number of cases that are potentially prevented. The cost of managing a case is uncertain and variable, but a recent systematic review suggested the cost of primary infection ranged from US$5243 to US$8570. 14 Conservatively assuming the lower end of this range, and applying October 2017 exchange rates, this gives a cost per case of AUD$6867. This was then multiplied by the number of cases potentially averted.
Results
The study included 924 participants: 531 in the pre-implementation period and 393 in the post-implementation period. The derivation of the cohort is provided in Figure 1.
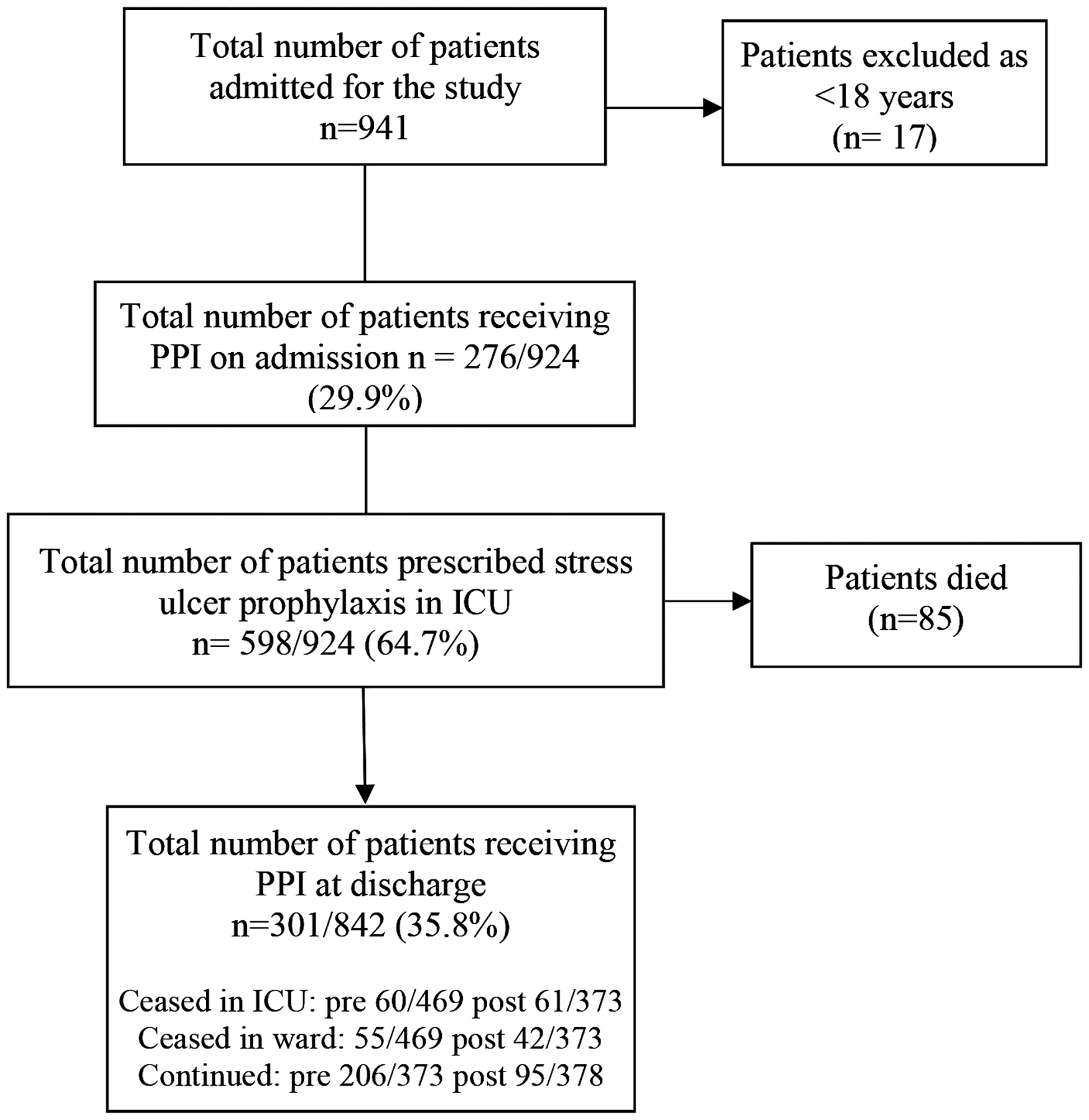
Derivation of the cohort (pre and post).
SUP initiation and continuation at hospital discharge
A comparison of the participant characteristics and prescribing practices in the two study periods is described in Table 1.
Patient demographics and SUP prescribing.
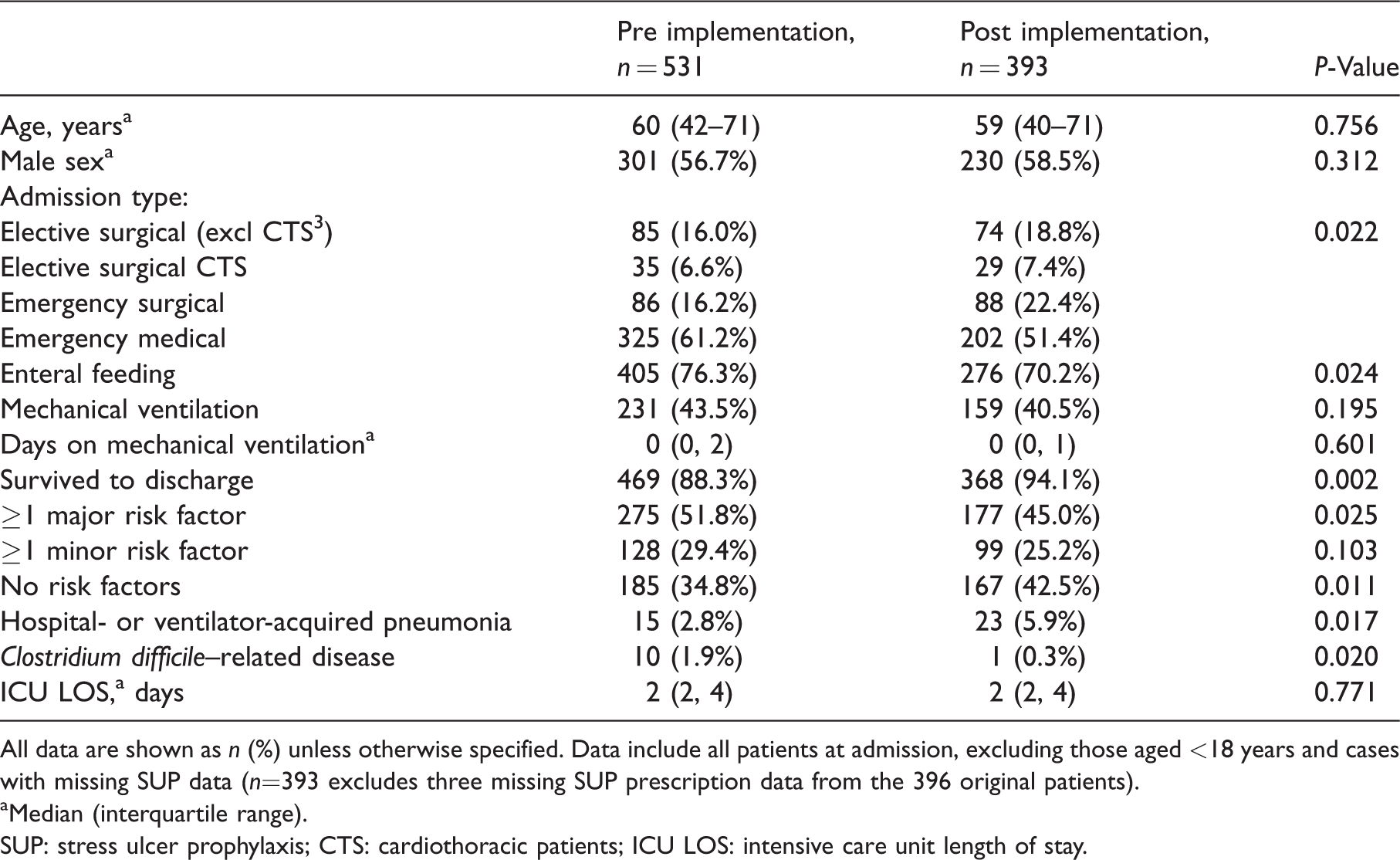
All data are shown as n (%) unless otherwise specified. Data include all patients at admission, excluding those aged <18 years and cases with missing SUP data (n=393 excludes three missing SUP prescription data from the 396 original patients).
Median (interquartile range).
SUP: stress ulcer prophylaxis; CTS: cardiothoracic patients; ICU LOS: intensive care unit length of stay.
A large proportion of patients (pre: 170/531 (32%); post: 105/393 (26.7%)) were already receiving a PPI or H2RB on admission to the ICU, and a significant proportion of patients in both study periods had at least one major risk factor for stress ulceration (pre: 275/531 (52%); post: 177/393 (45%)).
A comparison of the frequency of inappropriate SUP initiation or continuation in the two study periods is provided in Tables 2 and 3. The proportion of patients without any risk factors who were prescribed SUP in the pre-implementation phase was 42/212 (19.8%) compared to 39/153 (25.5%) in the post-implementation phase (P = 0.123). The proportion of hospital survivors inappropriately continued on SUP at hospital discharge in the pre-implementation period was 78/184 (42.4%) compared to 11/143 (7.7%) in the post-implementation period (odds ratio = 8.83; 95% confidence interval 4.47–17.45, P < 0.0001). Four (2.7%) patients in the pre-implementation group had a major risk factor but did not receive SUP. There were no such patients in the post-implementation group.
Inappropriate SUP initiation in ICU/continuation on hospital discharge (all patients).
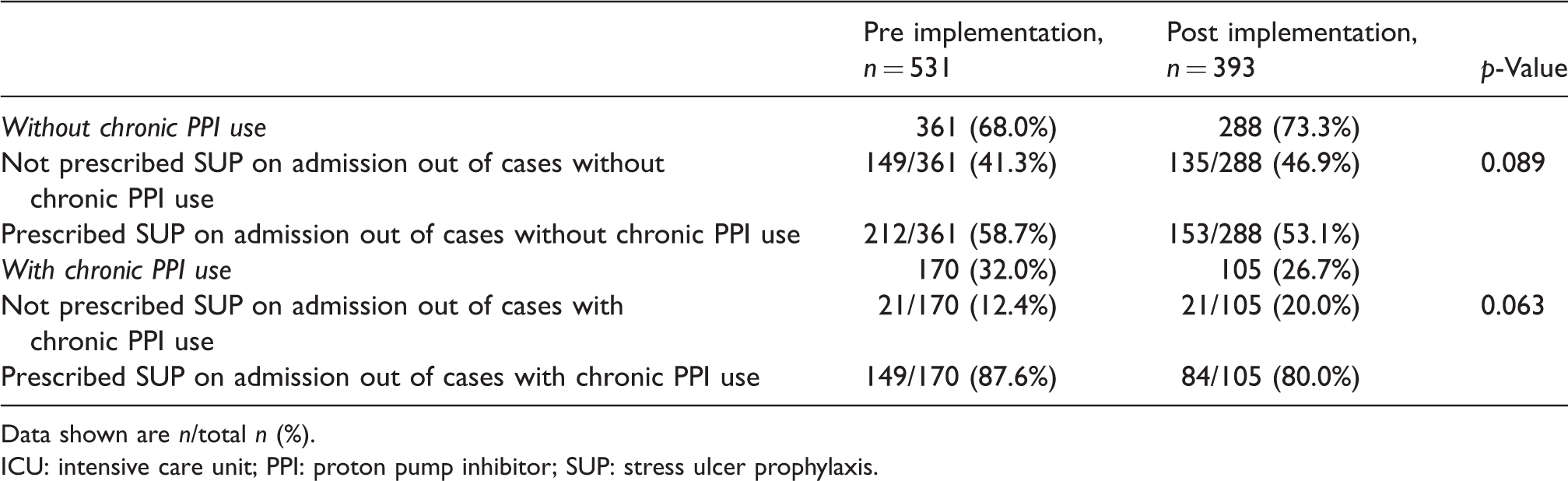
Data shown are n/total n (%).
ICU: intensive care unit; PPI: proton pump inhibitor; SUP: stress ulcer prophylaxis.
Inappropriate SUP initiation in ICU/continuation on hospital discharge (patients with no chronic PPI use).
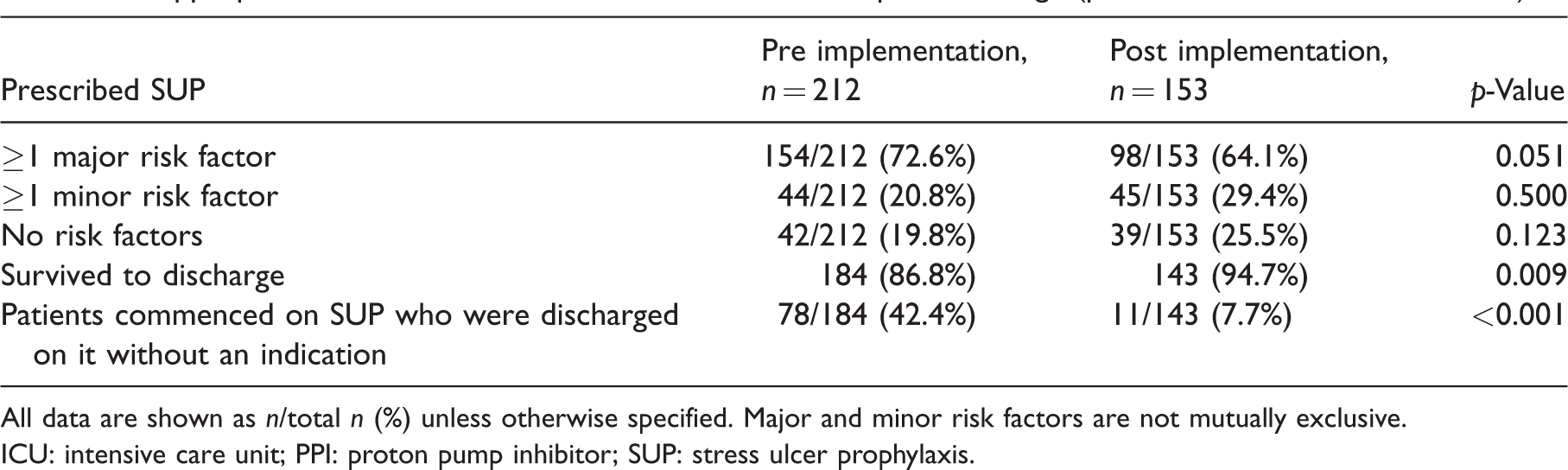
All data are shown as n/total n (%) unless otherwise specified. Major and minor risk factors are not mutually exclusive.
ICU: intensive care unit; PPI: proton pump inhibitor; SUP: stress ulcer prophylaxis.
SUP complications
CDAD occurred in 10 (1.8%) patients in the pre-implementation study period, of whom three were on a PPI prior to admission and seven received SUP. CDAD occurred in one (0.4%) patient in the post-implementation study period, also associated with SUP. Incidence of UGI bleeding complicating hospitalisation was similar in the two study periods. A comparison of the complications associated with SUP is provided in Table 4. When combining the two cohorts, and comparing exposure to PPI or not, those exposed to PPI were more likely to suffer from pneumonia, but there was no significant difference for CDAD and UGI bleeding.
Complications associated with SUP.
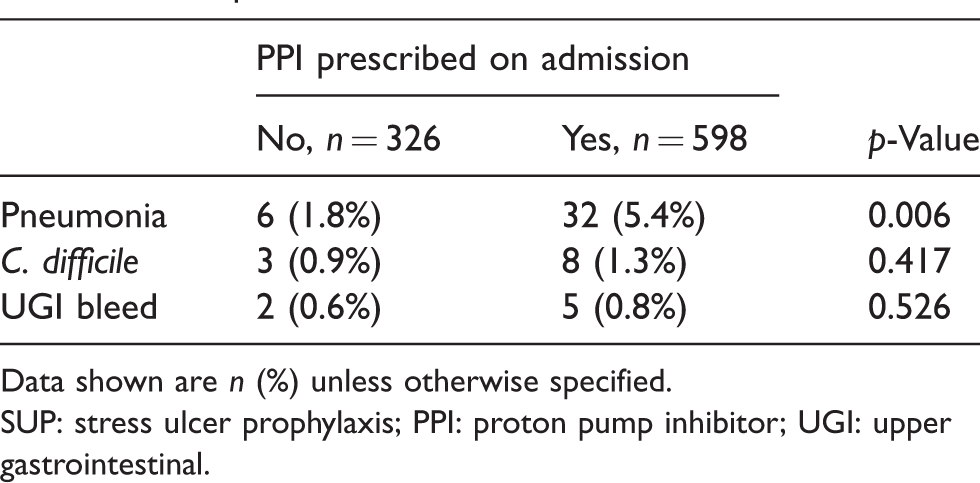
Data shown are n (%) unless otherwise specified.
SUP: stress ulcer prophylaxis; PPI: proton pump inhibitor; UGI: upper gastrointestinal.
Costs
In the pre-implementation group, of the 531 initial patients, 78 (14.69%) were ultimately discharged receiving a SUP, despite not having a documented indication for ongoing therapy. This contrasts with 11/396 (2.78%) in the post-implementation group. To extrapolate this to Australian ICUs, data from 2016 suggested there to be 148,254 patients admitted to an ICU in Australia in that year, which yields an additional 17,656 patients receiving SUP. 13 This would cost 17,656 × 71%×AUD$166.07 each year, which is AUD$2.082 million per year. The additional lifetime cost (assuming 10-year endurance) of inappropriate SUP continuation post ICU in a year is AUD$20.82 million. Under the shorter scenarios of two- or five-year continuation, this figure reduces proportionally to AUD$4.164 million and AUD$10.41 million, respectively.
Assuming a causal link with PPI prescription, the decrease in CDAD associated with the implementation suggests the potential to reduce the number of CDAD cases by 2417 cases nationally on an annual basis. Preventing 2417 cases would yield a cost saving of AUD$16.59 million per year. This figure is uncertain for a number of reasons, but indicates the possibility for substantial cost savings by reducing C. difficile cases by better use of SUP.
Discussion
Our study found that the implementation of a SUP prescription bundle, including a pharmacist-led de-escalation protocol, resulted in a significant reduction in inappropriate SUP continuation at hospital discharge. With the recent focus on ‘unnecessary’ or ‘low-value’ treatments provided in medicine, prescribing medications that are of no benefit and have the potential to cause harm, such as SUP without an approved indication, would qualify as unnecessary.15,16 This study, while focused on one medication class and one department, shows what is possible for ‘active de-prescribing’ within the hospital. The study also suggests that the results are mostly the effect of the pharmacist component and not the educational component.
Although the specific components of the bundle that were most effective is unknown, the use of a clinical pharmacist in the ICU has previously been successful in critical care quality improvement initiatives.17,18 Just as antibiotic stewardship is now an established component in the delivery of high-quality care in hospital, there may be a role for dedicated critical care stewardship rounds that consider SUP, as well as other elements of standard supportive care in the ICU such as the ongoing requirements for central venous access. 19 The rate of inappropriate initiation of PPIs was unchanged in our study, suggesting that the educational program, and perhaps having it directed to the multiple different prescribers (rather than to the small number of pharmacists), is less effective.
We found that the decrease in inappropriate SUP therapy was associated with a decrease in complications, namely hospital-acquired pneumonia and CDAD. Although SUP has an established role in selected patients admitted to the ICU, use is based largely on historical data and may not be needed in modern ICU practice.20,21 Indeed, the recent SUP-ICU randomised controlled trial showed a reduction in clinically significant bleeding between pantoprazole and placebo but no changes between groups when a composite outcome (bleeding, pneumonia, C. difficile infection or myocardial infarction) was used. 22
Our study suggests that improved prescribing practice has both direct and indirect savings. The extrapolated national direct savings from reduced PPI prescribing are relatively small (AUD$2.08 million/year), and potentially less if the period of continuation is <10 years. However, the reduction in complications has both benefits for patients and economic benefits from indirect savings of AUD$16.59 million/year. There are other potential complications from continuing on a PPI long term, including associations with gastric cancer, dementia, osteoporosis and interstitial nephritis—longer-term complications that we do not account for in our analysis.23,24 Furthermore, we do not have information on the actual duration of SUP continuation post hospital for our patients, which would potentially increase the exposure and risk for patients.
Our study has several limitations. First, the results reflect the practice of selected Western Australian ICUs. Other units may already have established guidelines and enforcement of SUP policy. Nonetheless, the sites involved represent the majority of ICU beds in the state and include different-sized units. A previous Australian study has shown that unit guidelines for SUP are uncommon. 25 Second, causal inference is limited by the pre and post study design. It may be that other factors including temporal trends, the differences in enteral feeding or types of admissions (more patients survived in the post-implementation group) could have influenced our results. However, there were no changes to unit policies or prescribing methods over our data-collection period, and the results were consistent between study sites. Third, by sampling consecutive patients, we may have inadvertently captured a ‘non-standard’ patient population. Fourth, our cost analysis is based on a number of assumptions, including using US-derived estimates of CDAD. Finally, there may also be opportunities to de-prescribe PPIs further than those found in this study. For example, some research has suggested that patients receiving enteral feeds may not need SUP due to the mucosal protection from feeding. 26
Conclusions
In patients admitted to the ICU, the introduction of a simple, bundled intervention resulted in a significant decrease in inappropriate continuation of SUP at hospital discharge. The decrease in inappropriate SUP therapy was associated with a reduction in recognised complications, including CDAD infection, and potentially resulted in substantial cost savings.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
