Abstract
We report a case of pseudohypertrophy of the left ventricle secondary to hypovolaemia from anaphylaxis. The patient was a healthy young female who developed anaphylaxis during a general anaesthetic. A transthoracic echocardiogram performed during the anaphylaxis crisis demonstrated significant left ventricular hypertrophy. However, when the anaphylaxis had resolved, a follow-up transthoracic echocardiogram demonstrated a normal left ventricular wall thickness. This left ventricular hypertrophy was a pseudohypertrophy as the left ventricular mass was normal and the wall thickness normalised when the hypovolaemia and anaphylaxis had resolved. Pseudohypertrophy of the left ventricle is an echocardiographic finding that suggests hypovolaemia. Furthermore, this echocardiographic finding supports the importance of volume resuscitation in the management of anaphylaxis. This phenomenon may be more frequently reported in the future due to the wider availability of point-of-care ultrasonography along with an increase in the number of physicians trained in echocardiography.
Introduction
Pseudohypertrophy of the left ventricle (LV) is an echocardiographic finding of LV hypertrophy that is not associated with an increase in the LV muscle mass. Pseudohypertrophy of the LV has been reported in patients with cardiac tamponade. 1 It has also been reported in pigs, 2 cats 3 and horses 4 secondary to hypovolaemia. In these reports, the increased LV wall thickness returned to normal when the cardiac tamponade and hypovolaemia had resolved. We present a case of LV pseudohypertrophy secondary to hypovolaemia from anaphylaxis. Pseudohypertrophy of the LV is an easy sign to recognise on echocardiography and suggests hypovolaemia. Furthermore, this sign supports the critical role of aggressive fluid resuscitation in the management of anaphylaxis.
Case report
A healthy 15-year-old female presented for a tendon repair of her little finger. Following an uneventful induction of general anaesthesia with fentanyl and propofol, a laryngeal mask airway was inserted. Thereafter, cefazolin was administered for surgical antimicrobial prophylaxis and the operative site was cleaned and prepared with chlorhexidine. Then, ten minutes after induction, a digital ring block with bupivacaine was performed by the surgeon prior to incision.
Next, 15 minutes after induction, the patient developed tachycardia (heart rate 140/minute) and hypotension (systolic blood pressure 50 mmHg). Boluses of metaraminol and phenylephrine were administered without effect.
A focused transthoracic echocardiogram (TTE) revealed an underfilled and vigorously contracting LV. It also revealed significant LV hypertrophy (LVH) (Figure 1). The LV end-diastolic wall thickness was significantly increased with the anteroseptal wall thickness measuring 1.47 cm (normal 0.6–0.9 cm) and the inferolateral wall thickness measuring 1.66 cm (normal 0.6–0.9 cm). The small LV end-diastolic dimension of 2.82 cm (normal 3.8–5.2 cm) indicated an underfilled LV with a decreased preload. It was difficult to exclude the possibility of hypertrophic cardiomyopathy (HOCM) with dynamic LV outflow tract (LVOT) obstruction due to the significant tachycardia. However, no mitral regurgitation was observed on colour flow Doppler.
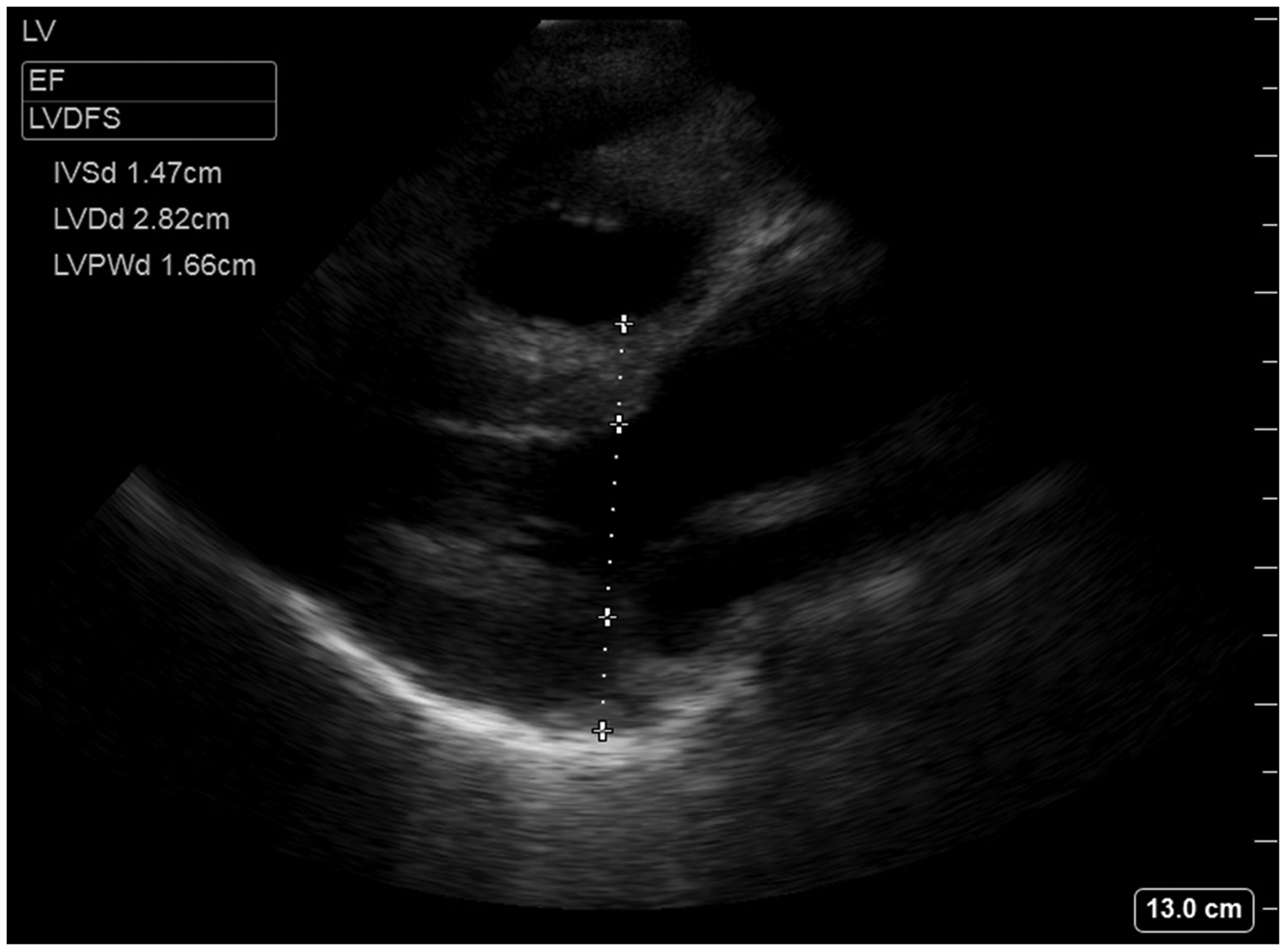
Transthoracic echocardiogram parasternal long axis view showing pseudohypertrophy of the left ventricle (LV) secondary to hypovolaemia from anaphylaxis. The LV end-diastolic wall thickness is significantly increased, with the anteroseptal wall thickness measuring 1.47 cm and the inferolateral wall thickness measuring 1.66 cm. The small LV end-diastolic dimension of 2.82 cm indicates an underfilled LV with a decreased preload.
The differential diagnosis for the shock included anaphylaxis and dynamic LVOT obstruction.
Treatment was initiated with a rapid intravenous infusion of crystalloid fluid as this is a constituent in the management for both anaphylaxis and dynamic LVOT obstruction. Boluses of esmolol did not improve the patient’s haemodynamic status.
A 12-lead electrocardiogram was performed and did not show signs of LVH. The patient was intubated and epinephrine (adrenaline) was administered, which resulted in haemodynamic stability. The haemodynamic improvement, subsequent to the epinephrine, suggested a diagnosis of anaphylaxis rather than dynamic LVOT obstruction. Twenty minutes following the initiation of epinephrine, the patient’s skin became erythematous and she developed angioedema. She remained intubated and ventilated and was transferred to the intensive care unit having received seven litres of intravenous crystalloid fluid and still requiring an epinephrine infusion. The following day, the epinephrine infusion was weaned and the patient was extubated.
The 4-hour mast cell tryptase was elevated at 33.3 μg/L (baseline 2.7 μg/L). Allergy testing has revealed that cefazolin was the trigger for the anaphylaxis.
On the day following the anaphylaxis crisis, when our patient had completely recovered, a follow-up TTE was performed (Figure 2). The follow-up TTE showed no signs of LVH, which indicated the LVH seen during the anaphylaxis crisis was a pseudohypertrophy. This follow-up TTE showed a normal LV wall thickness, with the anteroseptal wall thickness measuring 0.84 cm and the inferolateral wall thickness measuring 0.75 cm. The LV end-diastolic dimension had also normalised at 4.81 cm, which indicated the patient was now euvolaemic.
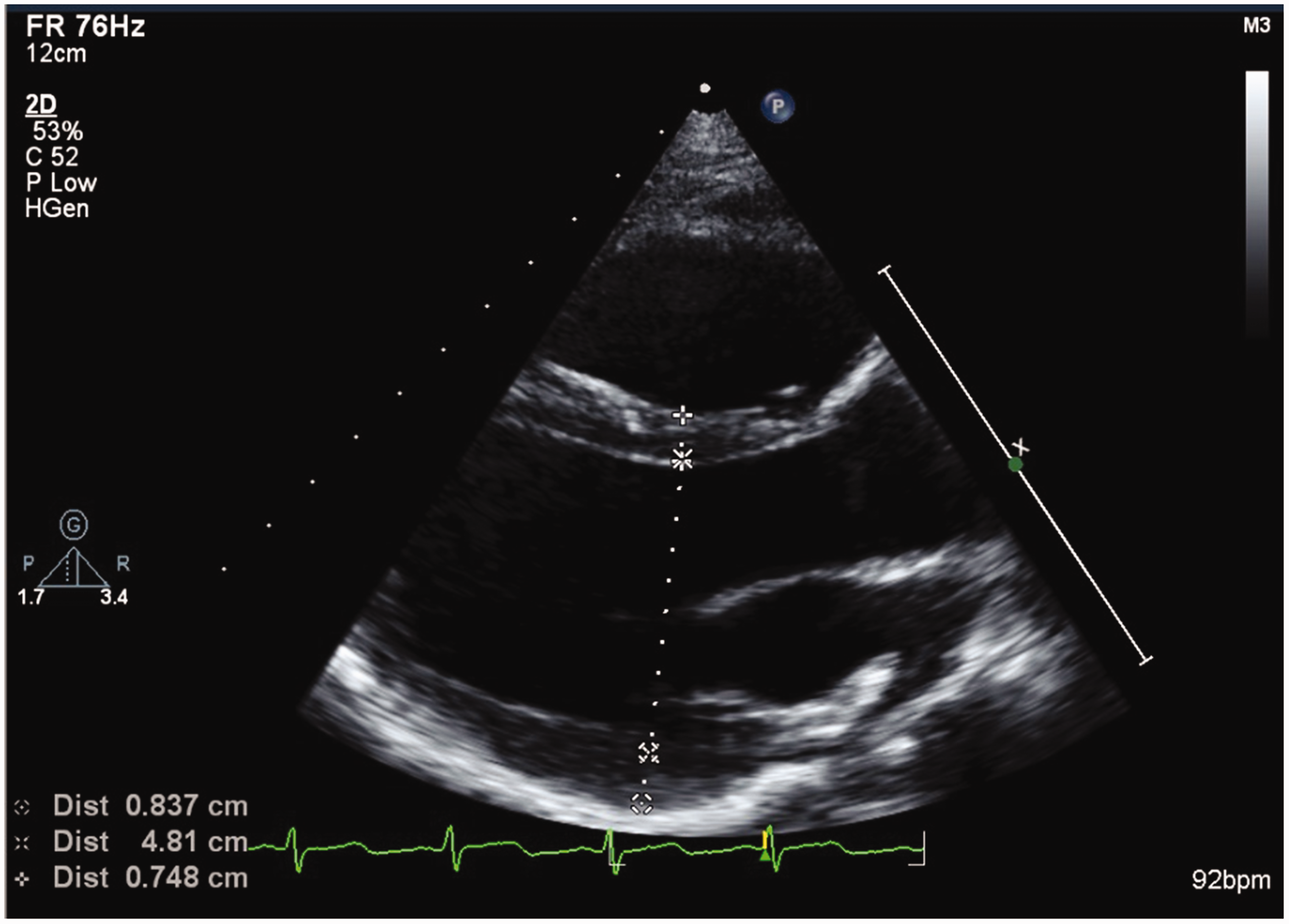
Transthoracic echocardiogram parasternal long axis view following complete clinical recovery of the anaphylaxis and hypovolaemia. The left ventricular (LV) wall thickness is normal (anteroseptal wall 0.84 cm, inferolateral wall 0.75 cm). The LV hypertrophy has resolved, indicating this was a pseudohypertrophy. In addition, the LV end-diastolic dimension has normalised (4.81 cm), indicating the hypovolaemia has resolved.
Consent has been obtained from the patient’s mother for publication of the case report.
Discussion
We report a case of pseudohypertrophy of the LV secondary to hypovolaemia from anaphylaxis. The patient was a healthy young female who received a general anaesthetic for a simple hand procedure and subsequently developed anaphylaxis to an antibiotic. A TTE performed during the anaphylaxis crisis demonstrated significant LVH. However, when the anaphylaxis had resolved, a follow-up TTE demonstrated a normal LV wall thickness. This indicated the LVH seen during the anaphylaxis crisis was a pseudohypertrophy. Pseudohypertrophy of the LV is an echocardiographic finding that suggests hypovolaemia.
LV pseudohypertrophy secondary to hypovolaemia has been previously reported in pigs, 2 cats 3 and horses. 4 In these animal studies, hypovolaemia had been induced experimentally by bleeding, 2 water deprivation3,4 and the administration of frusemide.3,4
Recently, Sadleir et al. 5 published a case report describing LVOT obstruction and systolic anterior motion (SAM) of the mitral valve with mitral regurgitation in the setting of anaphylaxis. No comment was made about pseudohypertrophy of the LV. However, on the transoesophageal echocardiogram, significant hypertrophy of the anteroseptal LV wall was present during the anaphylaxis crisis (Figure 3). On the following day, when the patient had fully recovered, the TTE demonstrated a normal anteroseptal LV wall thickness (Figure 4). Therefore, the LVH seen during this anaphylaxis crisis was also a pseudohypertrophy. In addition to our description of LV pseudohypertrophy observed during an anaphylaxis crisis, we have confirmed this finding in a recently published case.
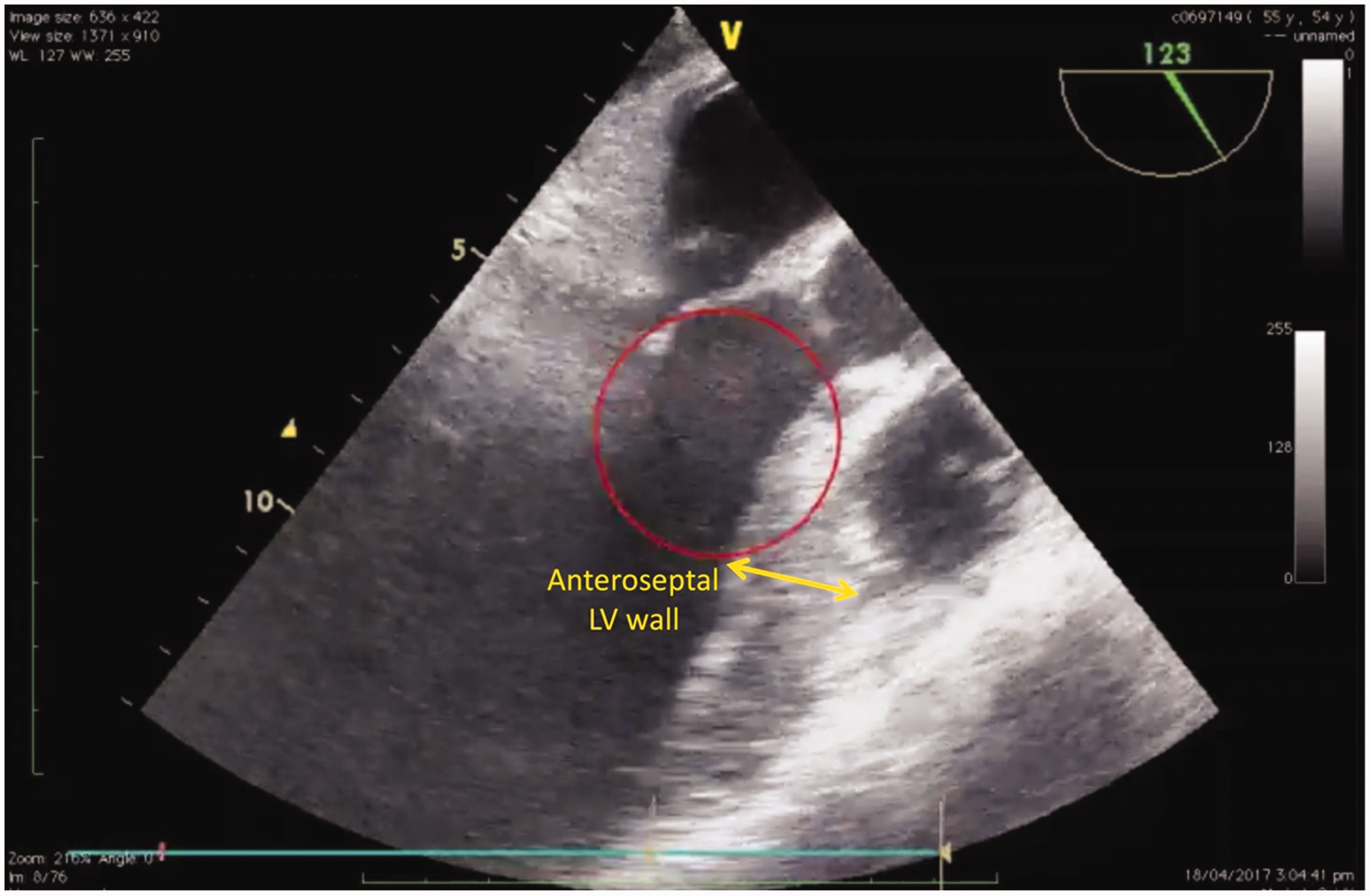
Transoesophageal echocardiogram mid-oesophageal long axis view of Sadleir et al.’s 5 patient showing a significantly increased anteroseptal left ventricular (LV) wall thickness. This LV pseudohypertrophy observed during the anaphylaxis crisis suggests hypovolaemia. Reproduced with permission from Sadleir et al.5
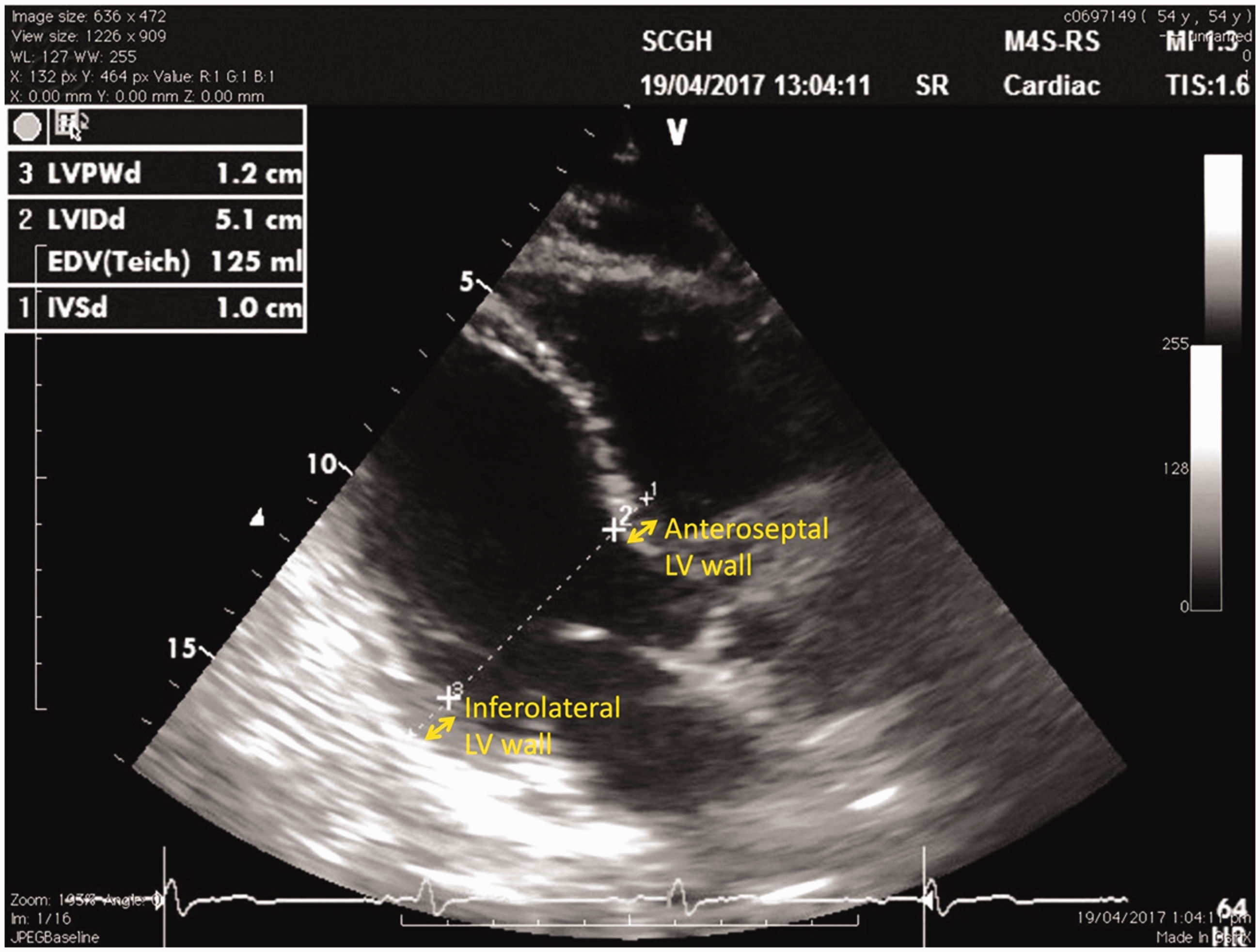
Transthoracic echocardiogram parasternal long axis view of Sadleir et al.’s5 patient following complete clinical recovery of the anaphylaxis and hypovolaemia. The anteroseptal left ventricular (LV) wall thickness is normal (1.0 cm). The inferolateral LV wall thickness is also normal (note the papillary muscle/chordae tendineae has been included in the measurement, which will overestimate the wall thickness). The LV hypertrophy has resolved, indicating that this was a pseudohypertrophy. In addition, the LV end-diastolic dimension is normal (5.1 cm), indicating the hypovolaemia has resolved. Reproduced with permission from Sadleir et al.5
Sadleir et al. 5 have noted that anaphylaxis causes a reduction in both the preload and afterload, which predisposes a patient in anaphylactic shock to LVOT obstruction. LV pseudohypertrophy is another risk factor that predisposes a patient in anaphylactic shock to LVOT obstruction. LV pseudohypertrophy narrows the LVOT area, which will increase the velocity of blood travelling through the LVOT and therefore increase the risk of SAM of the mitral valve. Moreover, the echocardiographic finding of LV pseudohypertrophy suggests the importance of a large volume of intravenous fluid for the immediate treatment for anaphylaxis, as it is for LV pseudohypertrophy in hypovolaemia.
With pseudohypertrophy, the LV walls appear thickened. This was not a true hypertrophy as the LV wall thickness normalised following correction of the hypovolaemia. Furthermore, LV mass can be estimated using the Devereux formula: 0.8 × {1.04 × [(LVEDD + AS + IL)3 – LVEDD3]} + 0.6; where LVEDD is the LV end-diastolic dimension, AS is the anteroseptal LV wall thickness and IL is the inferolateral LV wall thickness. 6 Indexing of LV mass is recommended to adjust for different body sizes. 7 When our patient (weight 79 kg, height 1.73 m) was hypovolaemic during the anaphylaxis crisis, her estimated indexed LV mass was normal at 81 g/m2 (normal 43–95 g/m2). Furthermore, when she had recovered from her hypovolaemia and anaphylaxis, her estimated indexed LV mass remained normal at 65 g/m2. Using the Devereux formula to estimate LV mass has been well validated 7 and some ultrasound machines will automatically calculate the estimated LV mass which would have been useful during our anaphylaxis crisis. A normal estimated LV mass, despite an increase in the LV wall thickness, suggests the possibility of a pseudohypertrophy and therefore hypovolaemia. In addition, the only echocardiographic method of directly measuring LV mass is with 3-dimensional echocardiography (3DE). However, 3DE is not a practical tool during a crisis as 3DE probes are not commonly found on point-of-care ultrasound machines, 3DE images are more difficult to acquire, and normal values for LV mass measured on 3DE are not well established.
LV pseudohypertrophy has previously been described in humans as a sign of cardiac tamponade. In Di Segni and colleagues’ case reports, 1 the mean LV end-diastolic wall thickness was 1.5–1.6 cm while the patients were in a shocked state. Following removal of the excess pericardial fluid, the LV wall thickness normalised to 0.9–1.0 cm. Feinberg and colleagues 8 also described patients with cardiac tamponade and LV pseudohypertrophy that resolved following pericardiocentesis. The LV pseudohypertrophy related to cardiac tamponade was postulated to be secondary to myocardial congestion and interstitial oedema. However, the LV pseudohypertrophy associated with cardiac tamponade was also probably secondary to hypovolaemia. As seen in our case, the LV end-diastolic dimensions in the tamponade state were significantly smaller, which indicates a decreased preload.
Our case presented a diagnostic and management dilemma. The differential diagnosis included anaphylaxis and HOCM with dynamic LVOT obstruction. To complicate the differential diagnosis further, anaphylaxis can induce dynamic LVOT obstruction by decreasing both the preload and afterload. 5 Epinephrine is the first-line treatment for anaphylaxis. However, epinephrine exacerbates a dynamic LVOT obstruction, which further impairs cardiac output. 9
Although this may be the first description of pseudohypertrophy of the LV recognised during anaphylaxis, this phenomenon may be more frequently recognised in the future. Point-of-care ultrasonography is now widely available in operating theatres, intensive care units and emergency departments. Furthermore, in addition to cardiologists, there are an increasing number of anaesthetists, intensivists and emergency physicians proficient in performing TTE.
In conclusion, we report a case of LV pseudohypertrophy visualised on echocardiography during an anaphylaxis crisis. Pseudohypertrophy of the LV is an echocardiographic sign that suggests hypovolaemia. This LV pseudohypertrophy was not a true hypertrophy as the estimated LV mass was normal and the LVH resolved following correction of the hypovolaemia and anaphylaxis. In addition, we have identified this phenomenon in a recently published case report on anaphylaxis. It is possible that pseudohypertrophy of the LV may be more frequently identified in future cases of severe hypovolaemia due to the increased availability of point-of-care ultrasonography and TTE skills. Furthermore, this sign supports the critical role of aggressive fluid resuscitation in successfully managing an anaphylaxis crisis.
Footnotes
Author contributions
Christopher AHG Wong: concept, background research, manuscript revision, manuscript final approval.
Sivasundari Arumugam: manuscript draft, manuscript final approval.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
