Abstract
Summary
Dabigatran is an oral anticoagulant used for atrial fibrillation and venous thromboembolism. While an effective antibody reversal agent is available, its cost precludes routine use and the mainstay of preoperative management is timely dabigatran interruption. Unlike warfarin, there are no universally accepted protocols for interruption of dabigatran in the preoperative period and there is uncertainty around the interpretation of standard coagulation tests in the presence of dabigatran. We performed a prospective, observational pilot study in patients presenting for elective surgery to examine: 1) the preoperative plasma dabigatran concentrations on day of surgery associated with the local dabigatran interruption protocol, 2) the potential utility of dabigatran concentrations on day of surgery, and 3) the utility of standard coagulation tests in determining whether dabigatran concentrations were below a ‘safe’ threshold for surgery. We recruited patients presenting to pre-admission clinics for elective surgery. Dabigatran concentrations below 30 µg/L were considered adequate for proceeding with surgery. Data were obtained and analysed from 21 patients with a median (range) age of 70 (20–86) years. Median (range) dabigatran concentrations on the day of surgery were 5 (0–59) µg/L. Two patients had day of surgery concentrations exceeding 30 µg/L. Of the standard coagulation tests examined, only the thrombin clotting time (TCT) was abnormal for these two patients. Our interruption protocol was associated with safe dabigatran concentrations in most patients on the day of surgery. A minority of patients had dabigatran concentrations above the safe threshold, which were detectable by abnormal TCT results.
Dabigatran is a non–vitamin K oral anticoagulant (NOAC) approved for use in non-valvular atrial fibrillation and venous thromboembolism, exerting its effect as a direct thrombin inhibitor. It has been fully subsidized with no requirements for specialist consultation in New Zealand since 2011 and has since rapidly achieved significant market penetration. 1 This expanding patient group caused major concern in the perioperative setting as interruption guidelines were scarce and there was no effective reversal agent. There was also widespread uncertainty around interpretation of coagulation tests in the setting of dabigatran therapy. While issues regarding emergency reversal have been largely addressed with the introduction of idarucizumab, its high cost means that the mainstay of surgical preparation remains timely interruption of dabigatran therapy.
Both dabigatran dosing and perioperative interruption guidelines rely on population data rather than providing individualized therapy based on some measure of pharmacodynamic effect. While this approach has been shown to be non-inferior to warfarin in the setting of stroke prevention in atrial fibrillation, 2 there may be better approaches for the perioperative patient. 3
Standard coagulation tests are affected to varying degrees by dabigatran. 4 The thrombin clotting time (TCT) is the most sensitive measure and is thus expected to be useful for gauging low concentrations of the drug. Therefore, instead of measuring dabigatran concentrations directly, the TCT may be used as a surrogate to determine whether dabigatran concentrations post-interruption are low enough to proceed safely with surgery.3–5 The TCT is also a test that is both more widely available and familiar to clinicians than a dabigatran concentration assay.
There is a dearth of corresponding Australasian data regarding preoperative dabigatran interruption and biomarkers such as dabigatran concentrations and coagulation tests. In this prospective observational study, we aimed to describe the plasma dabigatran concentrations on the day of surgery in the context of the local dabigatran interruption protocol. Further, we investigated the potential utility of these concentrations in informing decision-making on the day of surgery. The identification of patients on the day of surgery with dabigatran concentrations above a safety threshold of 30 µg/L may be useful to guide anaesthetists and surgeons about whether to proceed with surgery. 6 Finally, we examined the utility of standard coagulation tests in determining whether plasma dabigatran concentrations were above or below the threshold of 30 µg/L.
Materials and methods
We conducted a prospective, observational study in elective surgical patients scheduled for procedures at two local tertiary referral hospitals (Christchurch Hospital and Burwood Hospital) from December 2016 to December 2017. The study was approved by the national ethics committee (17/STH/5).
We included patients presenting for elective surgery, who were currently taking dabigatran and would require interruption of anticoagulation prior to their procedure. All patients were recruited after obtaining written informed consent. As this was an observational study, the perioperative advice given to the study participants was left entirely up to the treating clinicians. While we expect appropriateness of dabigatran treatment was part of the routine anaesthetic assessment, this aspect was not formally examined in this study, as it was beyond the scope of the aims. The local dabigatran interruption protocol recommends that dabigatran is ceased for 72 h before surgery in patients with a creatinine clearance above 50 mL/min, and ceased for 96 h in those with a creatinine clearance of 30–50 mL/min. 7 Further, bridging with enoxaparin is recommended in the following: atrial fibrillation with previous ischaemic stroke, previous venous thromboembolism.
In addition to routine preoperative bloods (e.g. complete blood count, renal function), plasma dabigatran concentrations were measured and standard laboratory coagulation tests were performed at the pre-admission clinic. The timing of this in relation to dabigatran dosing was expected to vary (e.g. if morning clinic, then the sample may be a few hours after a dose). Dabigatran was measured at the local hospital laboratory using a validated liquid chromatography–mass spectrometry (LC-MS/MS) assay. 8 Standard coagulation tests included the International Normalized Ratio (INR), activated prothrombin time (aPTT) and TCT. On the day of surgery, following interruption of dabigatran, drug concentrations and standard coagulation tests were repeated preoperatively. Treating clinicians were made aware of these additional day of surgery blood tests that may not have been part of their routine, and were able to access the results as they became available in the electronic results system. Turnaround for dabigatran concentrations (1–7 days) and coagulation tests (1–4 hours) were the standard timeframes as per any other clinical request. Samples for the dabigatran assay were batched for weekly analysis, and hence results for samples taken on the day of surgery were not available until after the procedure.
Additional patient data were collected from the clinical record including demographics, renal function, indication for anticoagulation, type of surgery, use of parenteral anticoagulation bridging during interruption of dabigatran, intended duration of dabigatran interruption (measured in hours), and the immediate postoperative outcome (to the point of discharge from hospital). Intended duration of interruption was calculated by assuming that the patient stopped taking dabigatran for the number of days stated in the pre-surgical anaesthetic plan, and that the last dose was at 2000 h on the day prior to interruption.
Descriptive data are presented as median (range). Scatter plots were constructed for the various coagulation tests against dabigatran concentrations. For day of surgery samples, we calculated the fractions of results within and above the normal reference range for each of the three coagulation tests (INR 0.8–1.2, aPTT 24–38 s, TCT 15–21 s) for plasma dabigatran concentrations below and above 30 µg/L, respectively. Further, as the TCT was expected to be the most useful coagulation test for identifying patients with a dabigatran concentration ≤30 µg/L, to assess this, a receiver operating characteristic (ROC) plot was constructed, and associated statistics calculated, for day of surgery samples. Values ≤30 µg/L were classified as ‘1’, and the rest as ‘0’.
Statistical analysis was performed using GraphPad Prism (version 7.04 for Windows, GraphPad Software, La Jolla, CA, USA, www.graphpad.com), except for ROC analysis, which was performed with MedCalc Statistical Software version 18.10.2 (MedCalc Software bvba, Ostend, Belgium; http://www.medcalc.org; 2018).
Results
Twenty-one patients were included, all of whom were on standard doses of dabigatran (Table 1). Orthopaedic surgery made up 10/21 cases, with total knee replacement being the most common operation. Five patients had preoperative enoxaparin bridging therapy.
Patient characteristics and dabigatran biomarkers.
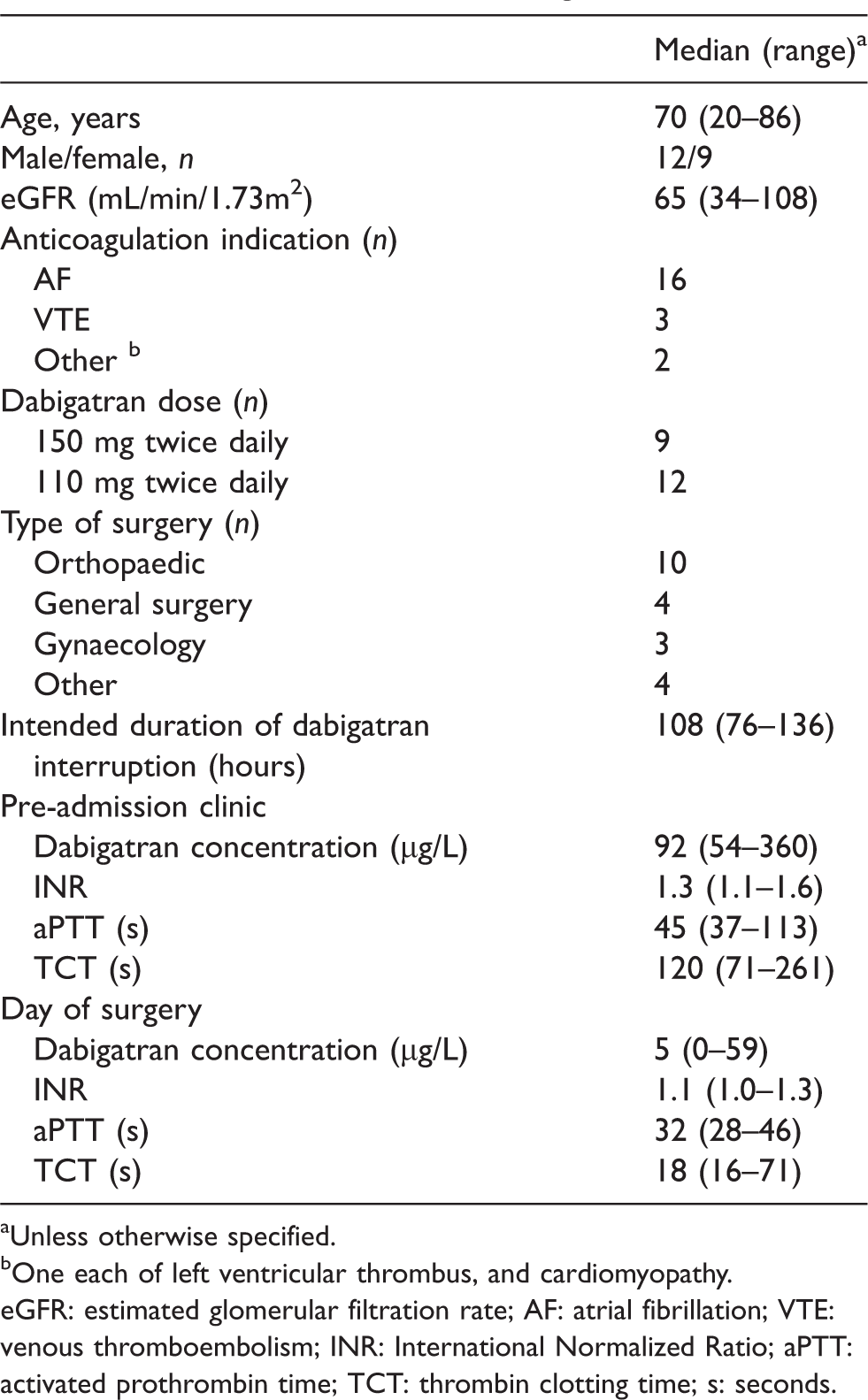
aUnless otherwise specified.
bOne each of left ventricular thrombus, and cardiomyopathy.
eGFR: estimated glomerular filtration rate; AF: atrial fibrillation; VTE: venous thromboembolism; INR: International Normalized Ratio; aPTT: activated prothrombin time; TCT: thrombin clotting time; s: seconds.
Following interruption, two patients had plasma dabigatran concentrations >30 µg/L on the day of surgery. One patient underwent an elbow replacement (33 µg/L) with no bleeding complications, while the other had total laparoscopic hysterectomy and bilateral salpingo-oophorectomy (59 µg/L) without perioperative bleeding complications.
Figure 1 shows the scatter plots of the standard coagulation tests against dabigatran concentrations for both pre-admission and day of surgery samples. For day of surgery samples, the INR, aPTT and TCT were within the normal reference range in 18/19, 16/19 and 16/19 patients, respectively, for dabigatran concentrations ≤30 µg/L. The INR, aPTT and TCT were above the normal reference range in 0/2, 0/2 and 2/2 patients, respectively, for dabigatran concentrations above 30 µg/L. Figure 2 shows the ROC plot of a normal TCT for predicting concentrations ≤30 µg/L. The sensitivity and specificity (95% confidence intervals, CI) were 84% (60% to 97%) and 100% (16% to 100%), respectively. The positive and negative predictive values (95% CI) were 100% (not calculated) and 40% (19% to 65%), respectively.
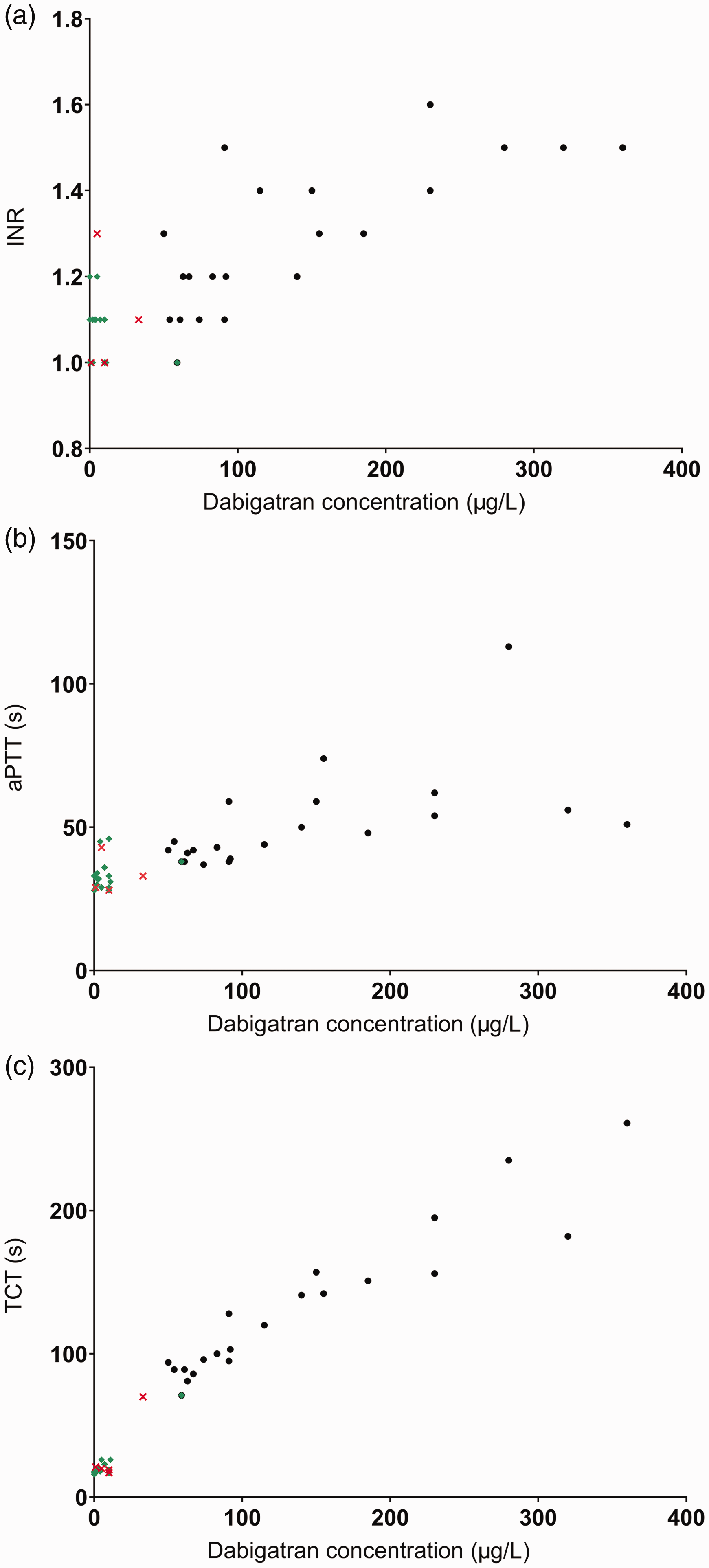
Scatter plots of INR (1a), aPTT (1b) and TCT (1c) against plasma dabigatran concentrations. Black filled circles are pre-admission clinic random samples, green filled diamonds are day of surgery preoperative samples in patients without preoperative enoxaparin bridging, red crosses are day of surgery preoperative samples in patients with preoperative enoxaparin bridging. INR: International Normalized Ratio; aPTT: activated partial thromboplastin time; TCT: thrombin clotting time.
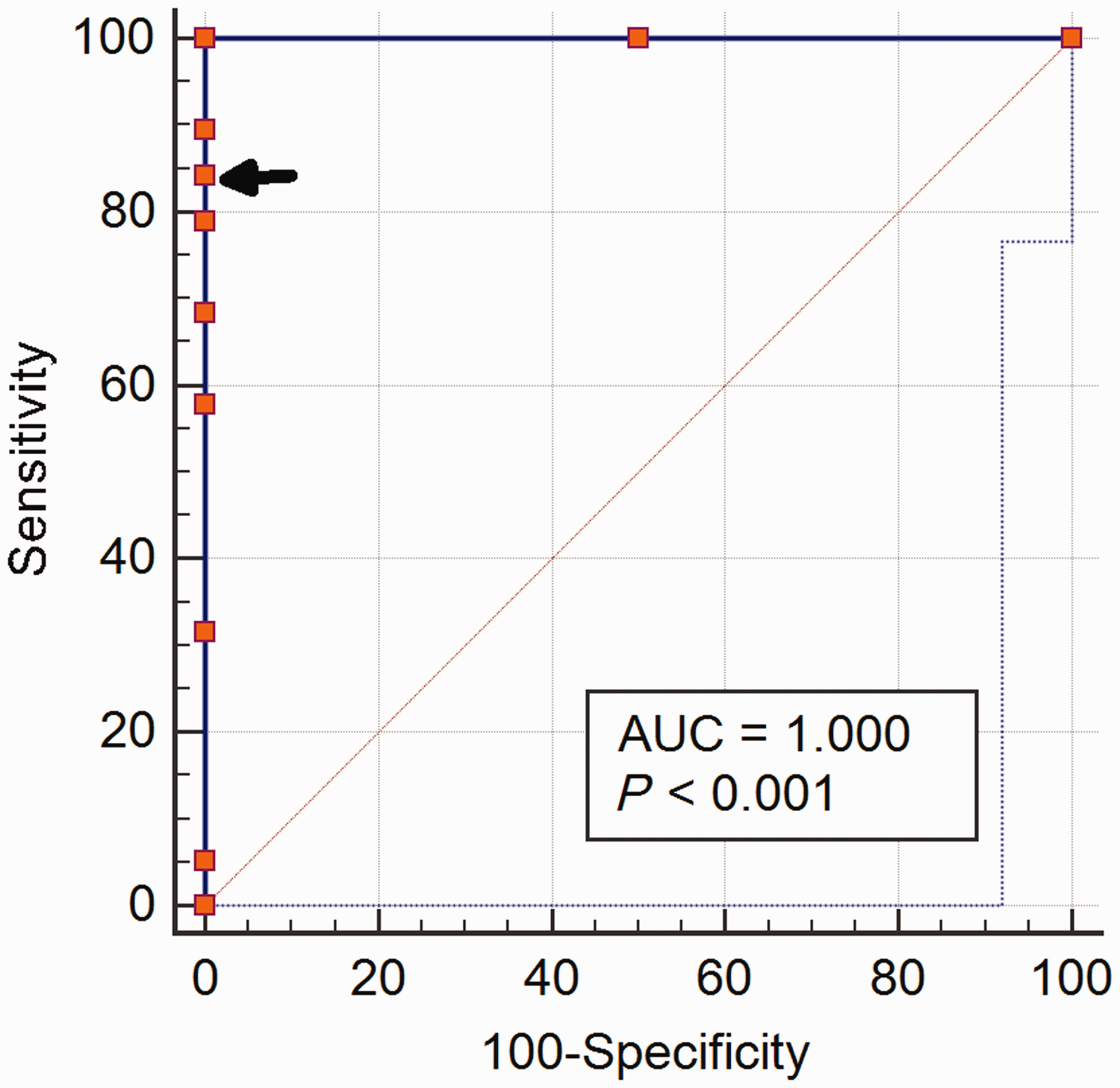
Receiver operating characteristic plot relating thrombin clotting time with dabigatran concentration ≤30 µg/L. AUC: area under curve.
With only one exception, patients were instructed by anaesthetists to interrupt dabigatran for longer than recommended by our protocol. This additional time ranged from 4 h to 64 h, with an average of 26 h.
Two patients suffered an ischaemic stroke following surgery, including one that was fatal. One of these patients was bridged with enoxaparin preoperatively, and then resumed dabigatran postoperatively. The other had a prolonged period of 16 days off dabigatran following orthopaedic surgery, at the surgeon’s request. There were no major bleeding or ischaemic events during the immediate postoperative period in the other 19 patients.
Discussion
In this study of patients undergoing preoperative dabigatran interruption at tertiary hospitals using a local protocol, most had dabigatran concentrations below the nominal safety threshold of 30 µg/L, with a range of 0–59 µg/L. However, two patients were above this threshold. This indicates the potential value in measuring preoperative concentrations, instead of assuming that adequately low concentrations had been achieved.
The value of preoperative laboratory monitoring of dabigatran following interruption has been debated. 3 An argument against laboratory monitoring is the often-repeated assertion that dabigatran pharmacokinetics are more predictable than those of warfarin. 9 However, data from the major randomized controlled trial of dabigatran in atrial fibrillation revealed a six-fold variability in the 10th to 90th percentiles of dabigatran concentrations. 10 Even greater variability is expected when considering the remaining 20% of patients, and furthermore in real-world populations outside the context of a trial. 5 Measurement of dabigatran concentrations would help minimize some of the uncertainty associated with this variability when making clinical decisions, such as whether or not to proceed with surgery.
Dabigatran concentrations may be measured either directly or by using surrogates such as coagulation tests. Given the limited availability of dabigatran assays, the use of common tests such as those comprising the standard coagulation screen should be considered. The aPTT and INR have been found to be insensitive to dabigatran. 4 These two tests are typically unable to distinguish between an absence of dabigatran and concentrations at the lower end of the therapeutic range, with values within the normal reference range for both scenarios. This is reflected by the two patients in our study with day of surgery concentrations above 30 µg/L having normal aPTT and INR. In comparison, the TCT can distinguish between these two scenarios, 6 and was abnormal for these patients. However, we found that the TCT was not the perfect test, as it was also abnormal in three of 19 patients with concentrations below 30 µg/L. This may have led to unnecessary deferral of surgery, if the decision to proceed had hinged upon the preoperative TCT. Our findings are consistent with those of a recent multicentre European study of preoperative interruption of NOAC drugs (the CORIDA study). 6 While 100% of normal TCT values were associated with a dabigatran concentration ≤30 µg/L, 65% of abnormal TCT values were also associated with concentrations below this threshold. Further, in relation to the use of normal TCT to predict concentrations ≤30 µg/L, the CORIDA study authors also reported similar values with tighter 95% CI for sensitivity (49%, 95% CI 39% to 60%), specificity (100%, 91% to 100%), positive predictive value (100%, 95% to 100%) and negative predictive values (35%, 24% to 46%).
The CORIDA study reported that only 5% of anticoagulant concentrations were above 30 µg/L at the time of the invasive procedure following interruptions of 49–72 h. 6 In addition to duration of interruption, creatinine clearance <50 mL/min and use of particular antiarrhythmic drugs (amiodarone, verapamil, diltiazem) were also found to predict concentrations above 30 µg/L. Hence, one approach to the clinical use of dabigatran concentration measurement would be to target patients with these risk factors for higher preoperative concentrations. However, this approach will not identify patients without risk factors who have been non-adherent to the intended duration of interruption. Just as the warfarinized patient requires demonstration of an acceptable INR before major surgery, the patient on dabigatran may also be better served by a laboratory measure of drug concentration or effect rather than simply relying on a time-based population average. Testing every patient on dabigatran on the day of surgery impacts on perioperative workflow for a variety of reasons, such as the laboratory turnaround for these tests. This impact should be assessed and compared with the potential benefits by any institution considering the implementation of this approach. Quantification of these benefits for a given institution with a particular surgical case-mix may be informed by data from the CORIDA study, which showed that creatinine clearance <50 mL/min, concomitant antiplatelet therapy, and procedure-associated high bleeding risk were significant covariates of periprocedural bleeding events. 6
We found that anaesthetists at our institution tended to interrupt dabigatran for longer than is recommended in the local dabigatran interruption protocol (median of 26 h longer). This is consistent with a recent audit of elective surgical patients on NOAC drugs. 11 While this was associated with most patients in our cohort having normal coagulation profiles and negligible dabigatran concentrations on day of surgery, the downside to this approach is that there may an increased risk of thromboembolic complications. Further, even though our sample size is too small to make meaningful comments on complication rates, it should be noted that two of our patients suffered ischaemic strokes in the perioperative period, which would far exceed numbers predicted for such a population. 12
Our study was performed in the New Zealand context, which is distinct from Australia where dabigatran is not fully subsidized. These cost implications need to be considered with that of a dabigatran assay, which is variably available. There are also a number of other limitations. Firstly, the dataset is small with 21 patients, with correspondingly limited statistical power for insights into clinical outcomes. However, other than the multicentre CORIDA study, which included 131 patients on dabigatran, we are not aware of any larger published cohorts. Secondly, the threshold of 30 µg/L is conservative, and higher thresholds such as 50 µg/L have been proposed, which may be appropriate for operations with inherently lower risk of significant bleeding. An abnormal TCT is expected to be less specific for dabigatran concentrations exceeding a given threshold, with higher threshold concentrations. Thirdly, unlike the INR and warfarin, there is substantial variability between coagulation tests such as the TCT in the dose-response relationship with dabigatran concentrations, which limits the applicability of our coagulation results to those from other centres. 13 We also did not assess other coagulation tests such as the dilute TCT and ecarin clotting time, as these are not widely available. Fourthly, the positive and negative predictive values of normal TCT for identifying patients on day of surgery with dabigatran concentrations ≤30 µg/L is predicated upon the prevalence of 90% (19/21) of patients with such concentrations, which limits the applicability of this result to other patient populations. For example, lower prevalence would have led to a lower positive predictive value. Finally, the estimation of duration of dabigatran interruption was based on what was stated in the pre-surgical anaesthetic plan, which patients may not have followed.
Conclusion
This study found that a minority of patients had day of elective surgery dabigatran concentrations above the conservative safety threshold of 30 µg/L, despite the durations of intended dabigatran interruption being longer than locally recommended. Hence, there is value in the measurement of day of surgery dabigatran concentrations, to identify patients with higher concentrations. The TCT is not a perfect surrogate for dabigatran concentrations as it is associated with false positive results. Prospective studies are required to determine whether clinical decisions based on preoperative dabigatran concentrations, such as postponement of surgery and the use of idarucizumab, lead to improved outcomes.
Footnotes
Acknowledgement
The authors would like to acknowledge colleagues from the Department of Anaesthesia who assisted with recruitment of patients.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
