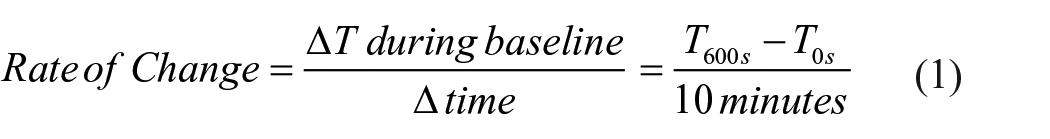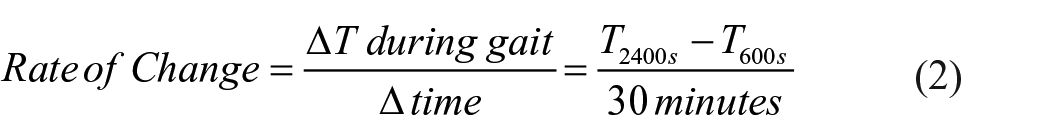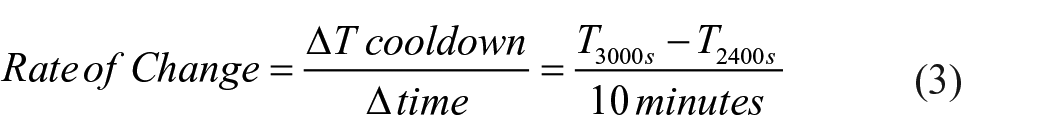Abstract
Background:
Increased foot temperature among individuals with type 2 diabetes can be predictive of diabetic foot ulcer development. A combination of physiological and mechanical deficiencies may contribute to elevations in intact foot temperature during gait for individuals with type 2 diabetes and transtibial amputation.
Objective:
This study evaluated plantar foot temperature differences between individuals with type 2 diabetes with and without transtibial amputation. We hypothesized that individuals with transtibial amputation maintain increased foot temperature compared to those without amputation.
Study Design:
Cross-sectional, case control.
Methods:
A sample of 16 participants with type 2 diabetes and transtibial amputation, and 16 age- and sex-matched participants with type 2 diabetes without amputation were recruited. Foot temperatures were measured during resting, walking, and cooldown periods. Peak temperature, mean temperature, and rate of temperature change were analyzed for each period, and compared between cohorts.
Results:
Participants with amputation exhibited increased mean foot temperature while at rest and during walking. Participants without amputation exhibited increased rate of change of foot temperature during walking. No differences in peak temperature or rate of temperature change were observed during the baseline or cooldown periods.
Conclusion:
The current findings of altered foot temperature for individuals with transtibial amputation and type 2 diabetes suggest a possible reason for the high rates of contralateral limb ulceration and amputation among this population.
Background
Over 422 million people are living with type 2 diabetes (T2D) worldwide, resulting in US$1.3 trillion in healthcare costs each year.1,2 Annually in the United States, US$10.9 billion are spent on treatment and long-term management of diabetic foot ulcers (DFUs). 3 Approximately 19%–34% of individuals with T2D will eventually develop a DFU. 4 Decreased protective sensation combined with altered gait biomechanics, improper footwear, and/or foot deformity increase the likelihood of DFU development. 5 Complications of DFUs may result in infection, gangrene, and amputation.
Approximately 80% of amputations among individuals with T2D are preceded by DFUs. 6 After amputation, one in four individuals with T2D will require more proximal amputation of the ipsilateral limb or contralateral limb amputation. 7 After bilateral amputation, quality of life and independence typically decrease, leading to a more sedentary lifestyle and development of additional comorbidities. 8 As such, preserving and protecting the intact limb is paramount.
An increased rate of contralateral limb amputation among individuals with previous amputation is multifactorial.9,10 For example, dialysis, presence of T2D, and impaired cellular wound healing can all contribute to contralateral limb amputation. 9 Generally, individuals with T2D demonstrate increased global foot temperature compared to healthy persons, and elevated foot temperature tends to precede a DFU.11–14 This may be a physiological marker indicating diminished thermoregulatory capabilities. Obesity, hypertension, and cardiovascular disease are associated with T2D and may compromise thermoregulatory mechanisms, especially in the distal extremities. 15 In addition, poor glycemic control and peripheral neuropathy negatively impact sweat glands, resulting in suboptimal thermoregulation. 16
Microcirculatory dysfunction in the diabetic foot is directly related to poor glycemic control.17,18 Microcirculatory dysfunction limits the foot’s vasodilatory response after stress or trauma, making the foot functionally ischemic. As such, ischemia is commonly found in conjunction with microcirculatory dysfunction. Ischemia, which is a contributing factor in 90% of lower extremity (LE) amputations, may contribute to prolonged inflammatory response in the foot. 19 This ischemic response, combined with microcirculation, can lead to blood vessel thickening, compromising transmission of healing nutrients and activated leukocytes to a wound. 19 Furthermore, increased rigidity within capillary walls may reduce vasodilatory response to local injury, resulting in functional ischemia. 19 Ultimately, these factors contribute to overall elevations in foot temperature and the development of DFUs.17,18
Previous literature has demonstrated value in comparing foot temperature asymmetries to identify individuals at elevated risk for DFUs. 20 This “asymmetry analysis” in conjunction with offloading plantar foot pressures may decrease the likelihood of DFU development among individuals with T2D without amputation. 20 While asymmetry analysis is unfeasible after loss of a LE, monitoring intact foot temperature among individuals with LE amputation may be important to protect the intact foot through offloading, activity modification, and prosthetic component selection.
As cardiovascular disease and T2D advance, likelihood of LE amputation increases. After amputation, individuals experience biomechanical deficiencies associated with loss of the plantarflexor muscle group. Commonly prescribed prosthetic components are currently unable to fully replicate lost muscle function. 21 Subsequently, persons with LE amputation experience reduced ankle power during push off, resulting in decreased ability to deflect the body’s center of mass from a forward and downward trajectory to a forward and upward path during contralateral initial contact during gait. 22 This asymmetry results in greater collision forces and energy absorption within the intact limb. 23 Although not directly measured in the current study, it is plausible increased collision force may contribute to elevated intact foot temperature as kinetic energy is transferred to thermal energy.
A combination of physiological and mechanical deficiencies may contribute to elevations in foot temperature on the intact foot during gait for individuals with T2D and LE amputation. In light of these physiological and mechanical deficiencies, the purpose of this study was to determine whether foot temperature and thermodynamic behavior differences exist between persons with T2D and LE amputation and persons with T2D without amputation, at rest and during walking. We hypothesized that given the potential physiological and mechanical alterations, individuals with T2D and LE amputation would have increased foot temperature and increased rate of change in temperature compared to those with T2D without amputation.
Methods
All participants were required to have capacity to provide consent, be aged between 35 and 65 years, have a confirmed diagnosis of T2D, and have the mobility profile of a typical independent community ambulator. Participants were excluded from the study if they were utilizing an LE compression garment or assistive device. Participants were also excluded if they had Charcot Arthropathy, active ulcerations, type I diabetes, or any additional movement disorders (stroke, traumatic brain injury, atraumatic brain injury, Parkinson’s Disease, etc.). Participants with amputation had a unilateral TTA and utilized their personal, custom prosthesis during the protocol. All participants provided written informed consent in accordance with the University of Houston Institutional Review Board approved protocol.
Cadence measurement
Gait cadence was measured with a Biometrics FS4 Contact Switch (DataLOG Biometrics Ltd., Gwent, UK), as there is evidence cadence influences foot temperature. 24 For participants with TTA, the contact switch was placed inside the shoe, under the heel of his or her prosthetic foot. For participants without amputation, the contact switch was placed between the diabetic insert and sole of the shoe under the calcaneus of his or her left foot. Cadence data were collected during a 30-min walking trial.
Health state metrics
Glycated hemoglobin (%HbA1c) was obtained via commercially available point-of-care monitoring kits (PTS Diagnostics, Indianapolis, IN, USA). Overall body temperature was taken at rest and during 10 min intervals throughout the walking trial with a commercially available tympanic thermometer (Braun ThermoScan, Kronberg, Germany).
Foot temperature measurement
Foot temperature was measured via a single iButton® temperature monitor (model DS1921H-F5, Maxim Integrated, San Jose, CA, USA) 25 embedded into the medial longitudinal arch (MLA) of an off-the-shelf plastizote diabetic shoe insert customized to the participant’s foot (Figure 1). The iButton® temperature monitor has been validated for human skin temperature monitoring. 25 Temperature data were recorded at 1 Hz. The iButton’s® position along the MLA was selected to ensure a total contact fit between the device and participant’s skin, and to reduce risk of skin breakdown under bony prominences. Each participant wore identical nylon stockings and Medicare-approved diabetic shoes (SureFit Apex Walker; Surefit, Alpharetta, GA, USA).

Off-the-shelf, custom-fit plastizote diabetic shoe insert with iButton™ temperature monitor integrated into the medial longitudinal arch of the insert.
Experimental procedure
All data collection occurred in one location over 18 months. Each participant walked from the parking lot to a temperature-controlled room set at 21.1ο C (a distance of approximately 45 m). Each participant was seated and removed their shoes and socks. Prior to the baseline period, health state metrics were obtained to control for potential confounding effects. The participant was then fit by a certified orthotist with properly sized diabetic shoes and a custom-fit insert with the integrated iButton®. All participants wore identical shoes and inserts. Baseline foot temperature data were recorded for 10 min while the participant remained in a seated position wearing the provided shoes and inserts. The baseline period was implemented to mitigate any impact of prior activity on foot temperature, allow comparison of baseline foot temperature between groups, and assess thermodynamic behavior. The participant then transferred to a treadmill and walked at a self-selected, comfortable, walking speed for 30 uninterrupted minutes (Figure 2(a)). During the 30-min treadmill walk, raw temperature data were gathered via the iButton® temperature monitor for subsequent export and analysis within Matlab (2017 Version 9.3; The MathWorks, Inc., Natick, MA, USA). Footswitch and overall body temperature data were also collected at this time. After the walking trial, participants completed a 10-min cooldown period. During cooldown, each participant spent 10 min in a seated position while wearing the provided diabetic shoes and inserts.
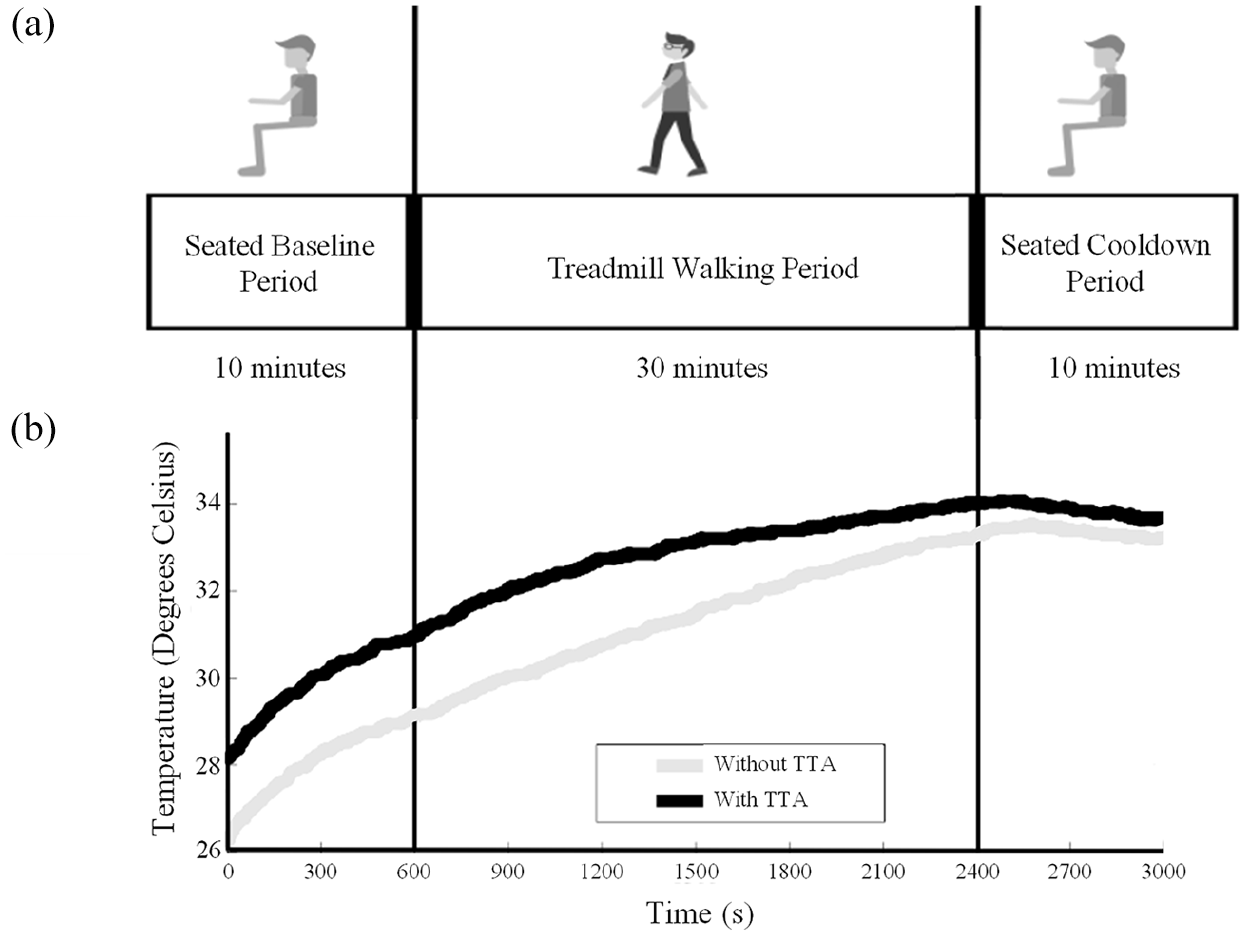
(a) Experimental protocol consisting of seated baseline period (10 min), treadmill walking period (30 min), and seated cooldown period (10 min). (b) Mean foot temperature change over time for both cohorts.
Data analysis
Custom Matlab algorithms were implemented to assess cadence and foot temperature data. Baseline foot temperature was calculated by obtaining the mean temperature of each participant’s foot for the first 10 min of the seated baseline period (0–600 s). The average temperature during the walking period was calculated as mean foot temperature during the total time walked (600–2400 s). Mean walking temperature was then calculated during first (600–1200 s), second (1200–1800 s), and third (1800–2400 s) 10-min intervals during the walking period. Peak temperature was determined by identifying the highest foot temperature value during the entire walking period (600–2400 s). Rate of foot temperature change during the seated baseline, walking, and cooldown periods was calculated and reported as rate of temperature (ΔT) change in οC·min (Equations 1, 2, and 3)
Cooldown foot temperature was calculated by obtaining mean foot temperature during the final 10 min of the seated trial (2400–3000 s).
Statistics
Data were reported as (mean (SD)) and compared between groups using one-way multivariate analysis of covariance (one-way MANCOVA) via SPSS 26 (IBM Corporation, Armonk, NY, USA). The primary between-subject factor was Group (two levels; participants with TTA vs participants without amputation). The primary outcome variables were mean temperature, peak temperature, and rate of change of foot temperature. Measures were all assessed during baseline, walking, and cooldown periods. A secondary one-way MANCOVA analysis was completed for foot temperature differences between groups during 10 min sub-intervals of the walking trial. Both one-way MANCOVA analyses included covariates of cadence and %HbA1c.24,26
Results
Sixteen adults with T2D and transtibial amputation (TTA) and 16 age- and sex-matched individuals with T2D without amputation volunteered to participate; see Table 1 for summary demographics.
Demographic and clinical characteristics of participants (mean (SD)).
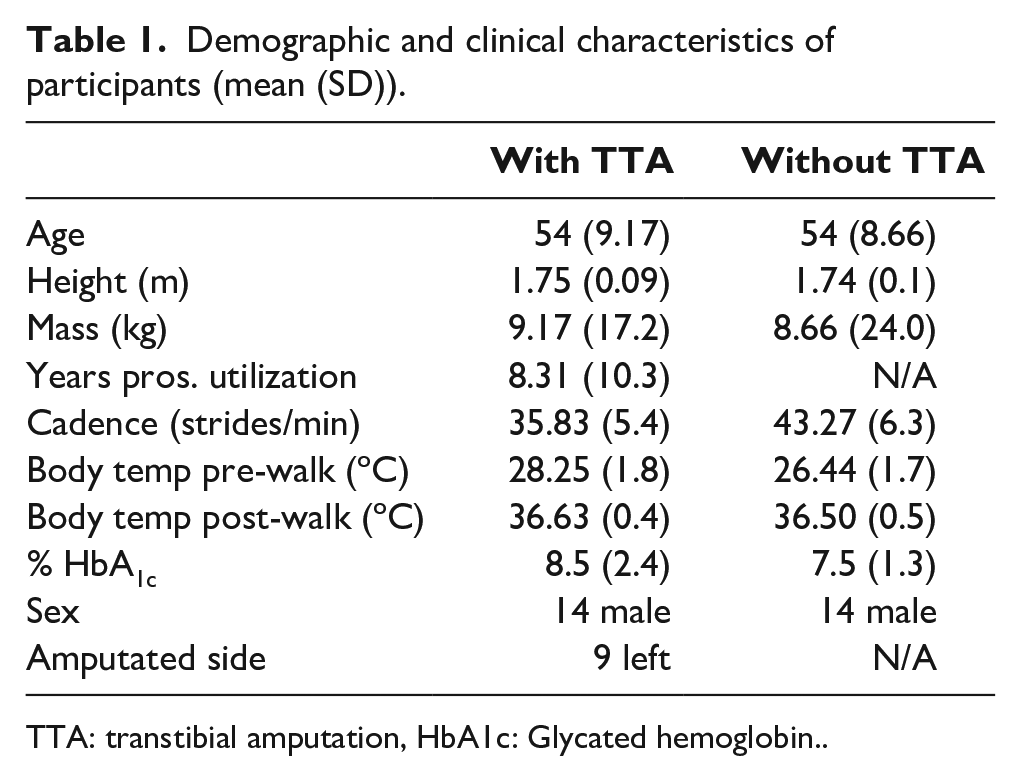
TTA: transtibial amputation, HbA1c: Glycated hemoglobin.
Walking foot temperature
Figure 2(b) depicts mean ensemble curves of temperature change for both groups during each phase of the protocol. Participants with TTA exhibited overall elevated mean foot temperature during walking compared to participants without amputation (Table 2). Main effect of Group was present (F1,15 = 5.86, p < 0.05). No significant covariates were found.
Foot temperature differences between groups (mean (SD)).
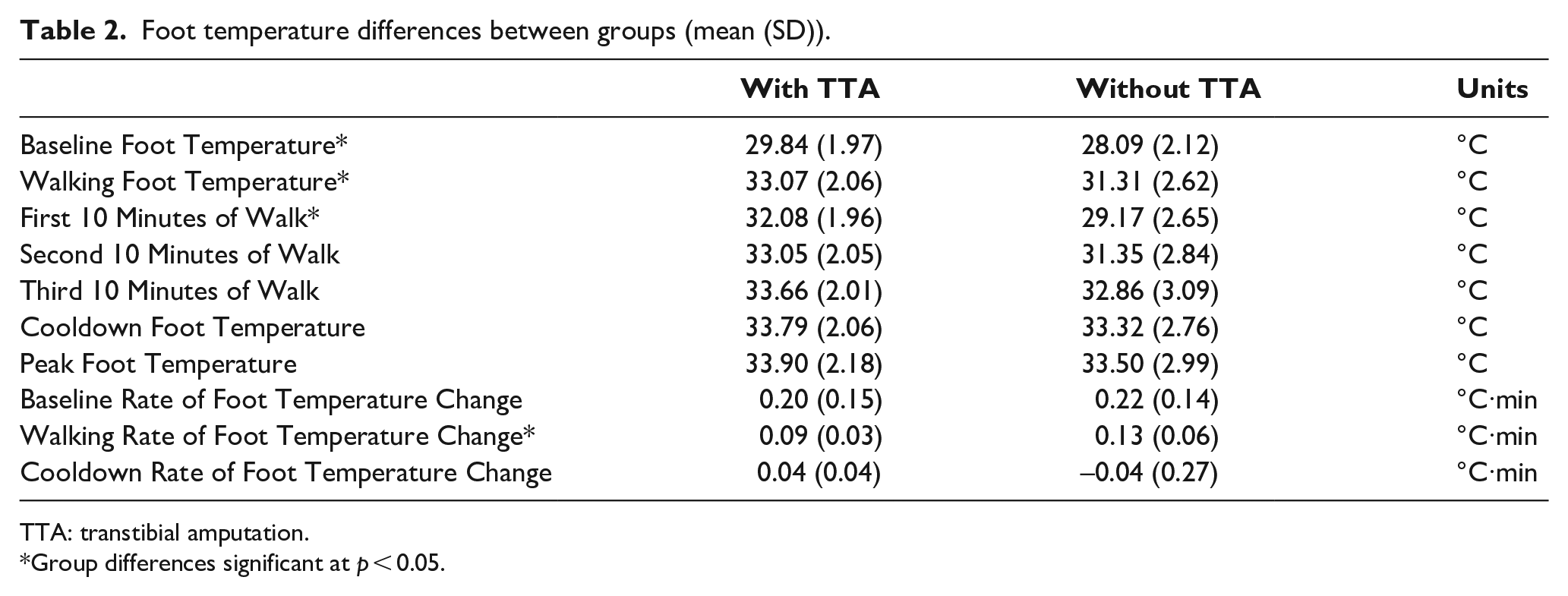
TTA: transtibial amputation.
Group differences significant at p < 0.05.
Differences in mean foot temperatures during the first, second, and third 10-min intervals of the walking period were noted (Table 2). One-way MANCOVA analysis indicated a significant difference in temperature during the first 10 min of the walking trial (F1,15 = 5.70, p < 0.05). No statistical differences were found between groups during the second and third 10-min walking intervals.
Baseline foot temperature
Participants with TTA exhibited increased baseline foot temperature compared to participants without amputation (Table 2). One-way MANCOVA analysis indicated a significant main effect of Group was present (F1,15 = 8.47, p < 0.05) without significant covariates.
Peak foot temperature
No differences in peak foot temperature were found between groups (p > 0.39; Table 2).
Rate of foot temperature change
No differences in seated baseline period rate of change in foot temperature were found between groups (p > 0.69; Table 2). Participants with TTA exhibited decreased rate of foot temperature change compared to participants without amputation during the walking period (F1,15 = 4.99, p < 0.05) without significant covariates (Table 2). No differences in the seated cooldown period rate of change in foot temperature were found between groups (p > 0.18; Table 2).
Cooldown period foot temperature
No differences in foot temperature during the cooldown period were found between groups (p > 0.61; Table 2).
Discussion
The purpose of this study was to evaluate if differences in foot temperature and thermodynamic behavior existed between individuals with T2D and TTA and individuals with T2D without amputation. Our findings partially supported our hypotheses. Participants with TTA began with higher baseline foot temperature but experienced a slower rate of temperature increase during the walking period. Near the end of the 30 min walking bout and continuing into the cooldown phase, both groups had similar foot temperatures. Furthermore, no difference in peak foot temperature was noted between cohorts.
Baseline temperature differences
It is well established that increased foot temperature may serve as a marker for DFU development.12–14,27–29 However, previous studies investigating the relationship between foot temperature and DFUs have focused on individuals with T2D without amputation.12–14,24,27–30 A main finding from the current study was that participants with T2D and TTA exhibited increased foot temperature during the initial baseline period. Preliminary findings indicated that 10 of 16 participants with T2D and TTA presented with higher average baseline foot temperature when compared with the highest average baseline temperature found among participants without TTA (average baseline temperature range: 25.6οC–33.2οC T2D and TTA vs 21.6οC–30.8οC T2D). This initial difference in temperature may be attributed to physiological differences including advanced disease state of T2D among participants with TTA, which would be at least partially supported by the increased HbA1C levels found in this group (Table 1).
Individuals with advanced T2D experience microvascular dysfunction that can be observed clinically by increased foot temperature, palpable pulses, and distended veins. 31 More advanced diabetes tends to result in decreased microvascular reactivity in the foot, which may retard the foot’s ability to vasodilate. Furthermore, diabetic neuropathy can disrupt subpapillary arteriovenous shunts innervated by the sympathetic nervous system, resulting in a functionally ischemic foot. 31 While it is anticipated these factors may be common within both groups, the decrements may have been more pronounced in participants with T2D and TTA, as this cohort was found to have further diminished health state metrics (increased %HbA1c) compared to participants without TTA.
Unfortunately, the baseline period in the present study had been also intended to normalize foot temperature to constant temperature. The noted rate of change in foot temperature makes it clear that this was not accomplished and instead individuals experienced rising foot temperature during a seated non-moving activity. Participant activity prior to entering the facility was not recorded. It is possible that activity performed prior to the onset of the study protocol may have impacted baseline foot temperature more than originally assumed. As such, a more controlled and extended baseline period may provide more insight into these findings. While conclusions regarding causation for the elevated baseline foot temperatures found in this group are not possible, the higher foot temperatures are consistent with the groups’ status as higher risk for ulceration.
Walking temperature differences
While no differences were noted between the cohort’s peak foot temperature, participants with T2D and TTA demonstrated significantly increased mean foot temperature during the 30 min treadmill walking trial. Group temperature differences were significantly higher during the first 10 min of the walking period. Although participants with TTA demonstrated increased mean foot temperature during the second and third 10 min periods, differences were not found to be statistically significant. This suggests that the overall mean temperature difference observed occurred early in the walking trial. Participants without TTA exhibited increased rate of change in foot temperature during the walking period.
Klute et al. 32 reported prosthesis users typically perform numerous short bouts of walking through the day, mainly consisting of less than 17 steps per minute. In the same study, it was noted that this population rarely performs bouts of walking greater than 15 min, or far less than the artificial 30-min walking bout that participants were required to perform. For participants with TTA, the rapid temperature rise and overall elevated average temperature during the first 10 min of walking is critical as this, combined with the baseline differences, may most align with the natural conditions occurring throughout the day for these individuals. This finding supports the importance of monitoring foot temperature during acute bouts of walking and the potential benefit of activity modification for this population.
Cooldown temperature differences
No differences in mean or rate of change in foot temperature differences were found during the cooldown period. Moreover, neither cohort’s foot temperature returned to baseline temperature at the conclusion of the 10 min cooldown period. It is anticipated that foot temperature would continue to decrease had the cooldown period been extended. This was a known limitation in the current protocol that was accepted to minimize individuals’ participation time. The primary focus of this study was to investigate foot thermodynamic characteristics and temperature differences between persons with T2D and TTA and persons with T2D without TTA while walking. However, the cooldown period of this protocol may provide direction for future research aimed at examining how the diabetic foot responds and recovers after an extended walking bout.
In healthy individuals, physiological mechanisms are in place to reduce foot temperature and repair tissue microtrauma after situations of increased activity, including extended bouts of walking. This “down-regulation” allows the foot to return to equilibrium by processing signals to and from the central nervous system and executing repair responses. This defense mechanism provides inherent protection to the foot, returns the foot to normal temperature, and combats tissue breakdown. In the diabetic foot, vascular insufficiency and local infection contribute to thermal changes, which may become more pronounced after an extended bout of walking. 33 Furthermore, increased local skin temperature due to inflammation and enzymatic autolysis of tissue tend to precede ulcer development in high stress areas on the plantar foot.26,33 This increase in physical trauma and amplified inflammatory response may yield increased foot temperature, ultimately resulting in tissue breakdown and DFU development. After a bout of walking, these characteristics may be exacerbated. Providing an extended cooldown period may potentially provide insight into these mechanisms and provide further insight into the benefit of activity modification for high-risk individuals.
Limitations
There are limitations to the current study worth noting. First, each participant ambulated at a self-selected walking speed rather than a predetermined cadence. A self-selected walking speed was implemented given the discrepancies in endurance levels among participants and the requirement to ambulate for 30 continuous minutes. Second, participants should have been given a longer period without walking to establish baseline temperature. The lack of available literature in this area, however, made it challenging to design a protocol that could account for a long enough baseline to ensure a constant temperature. Future investigations should attempt to quantify pre-testing biomechanical loading and/or perform a longer pre-walking temperature measurement to assess baseline plantar temperatures.
Conclusion
Participants with TTA demonstrated evidence of increased foot temperature, as well as altered thermodynamic behavior, when compared to those without amputation. Given that increased foot temperature may be a warning sign of impending DFU development, results from this study could prove clinically relevant. The results from this study further highlight the importance of skin temperature monitoring for individuals at high risk for lower limb amputation.11,13 In addition, in consideration of differences in foot temperature and thermodynamic behavior for individuals at what may be an advanced disease state, or a potential mechanical disadvantage, clinicians may start to consider activity modification protocols in attempt to maintain the diabetic foot at an appropriate temperature. This is valuable, as healthcare professionals consider methods to prevent contralateral limb amputation for those with existing unilateral amputation.
Footnotes
Acknowledgements
We also appreciate the guidance of and scientific discussion with Dr. Stacey Gorniak throughout the course of this project.
Author Contributions
All authors contributed equally in the preparation of this article.
Declaration of Conflicting Interests
The author(s) disclosed no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Orthotic and Prosthetic Education Research Foundation (OPERF) Fellowship Award.