Abstract
Background:
Children with hemiparesis are commonly prescribed ankle foot orthoses to help improve gait; however, these orthoses often result in only small and variable changes in gait. Research with adult stroke survivors has suggested that orthoses that extend beyond the ankle using long, passive tendon-like structures (i.e. exotendons) can improve walking.
Objectives:
The aim of this study was to quantify the impact of an exotendon-based exoskeleton on pediatric gait.
Study design:
Repeated-measures study.
Methods:
Two typically-developing children and two children with hemiparesis completed a gait analysis, walking without and with the exoskeleton. The exotendon was tested at three stiffness levels.
Results:
All children were able to walk comfortably with the exoskeleton, with minimal changes in step width. Walking speed increased and lower limb joint symmetry improved for the children with hemiparesis with the exoskeleton. Each participant had changes in muscle activity while walking with the exoskeleton, although the impact on specific muscles and response to exotendon stiffness varied.
Conclusion:
Exotendon-based exoskeletons may provide an alternative solution for optimizing gait in therapy and in the community for children with hemiparesis. Determining the optimal stiffness and configuration for each child is an important area of future research.
Background
Hemiparesis is a neurologic condition in which half the body is affected by motor impairments. There are many causes for hemiparesis, and the largest cause among children is cerebral palsy (CP), which is diagnosed in 2–3.8 out of every 1000 children born in the United States.1,2 These children participate in 13% to 53% less habitual physical activity than their typically-developing (TD) peers. 3 There is a need for new devices or interventions that can improve mobility for these children and allow them to better participate in activities of daily living.
Current solutions to assist walking for children with hemiparesis include clinic-based tools (treadmill training and robotic exoskeletons) and community-based assistive mobility devices (walkers, crutches, and orthoses). Bodyweight-supported treadmill training4,5 and robotic-assisted gait training 6 can have positive effects for providing repetitive, task-specific walking practice, but these devices can only be used in the clinic and require large commitments of both time and resources. In the community, walkers and crutches often provide critical support to improve balance and mobility, but present challenges for navigating tight spaces and uneven terrain, and do not encourage improved gait. Ankle foot orthoses (AFOs) are often prescribed to assist motion and can improve spatiotemporal outcomes.7,8 Passive orthoses that assist other joints are rarely used, and typically only assist with proper positioning or support, such as hip abduction orthoses or the standing, walking, and sitting hip (SWASH) orthosis.9,10 Recently, several powered orthoses have been developed for children with paresis that have demonstrated promising changes in gait.11,12 Lerner and colleagues have demonstrated that a powered knee exoskeleton can improve knee extension and a powered AFO can improve metabolic costs among children with CP.12,13 However, powered devices still require regular charging and include the extra weight of batteries and actuators, which can make daily use outside of the laboratory challenging.
Recent research has suggested that drawing inspiration from biological systems may enhance passive exoskeleton designs.14,15 Systems with long tendons spanning multiple joints, such as a horse’s leg, exhibit extremely efficient locomotion.15,16 Simulations by van den Bogert 15 suggested that using “exotendons,” long spring-like devices that mimic these tendons by passively storing and releasing energy, could assist human locomotion and reduce energy costs. This concept was further developed in a commercially-available exoskeleton (Kickstart, Cadence Biomedical, Seattle, WA, USA) that has been shown to increase walking speed and endurance for adults recovering from neurological injuries. 14 Whether these principles can also be applied to improve gait of children with hemiparesis remains unclear.
The primary aim of this research was to quantify changes in gait with an exotendon-based pediatric exoskeleton among TD children and children with hemiparesis. For the TD children, we sought to examine the impact of an exotendon on pediatric walking symmetry and stability (i.e. step width) and provide a peer comparison using our selected outcome measures. For the children with hemiparesis, we hypothesized that use of the exotendon-based exoskeleton would improve walking, similar to results observed in adult stroke survivors. 14
Methods
Device design
The pediatric exoskeleton (PlayGait) was designed to span the hip, knee, and ankle and use an exotendon to store and return energy over the gait cycle (Figure 1). Energy is stored during stance and returned during the swing phase of walking. To make the design compatible with a pediatric population, the leg supports are adjustable to accommodate leg length between 45.2 and 71.1 cm, corresponding to the leg length of 5th percentile 3-year-olds to 95th percentile 6-year-olds. 17 Each leg of the exoskeleton can be removed independently to accommodate unilateral or bilateral users. For unilateral configurations, PlayGait allows 5 degrees of freedom (DoF): knee and ankle flexion/extension in the sagittal plane and full hip range of motion. The exotendon comprises a spring in series with a cable that connects between pulleys at the hip, knee, and ankle. The pulley radii were 34.0, 4.8, and 26.9 mm for the hip, knee, and ankle joints, respectively. These sizes were based on the commercially available adult exotendon-based exoskeleton (Kickstart) pulley sizes but scaled within manufacturability to child size. A footplate and flat metal stirrup which fit over the shoe were used for this study. A unilateral configuration (i.e. pelvic support and one leg) weighs 1.45 kg (0.32 kg belt, 0.45 kg hip joint, 0.17 kg upper leg support, 0.05 kg knee joint, 0.15 kg lower leg support, 0.10 kg ankle joint, 0.07 kg cable, and 0.14 kg footplate).
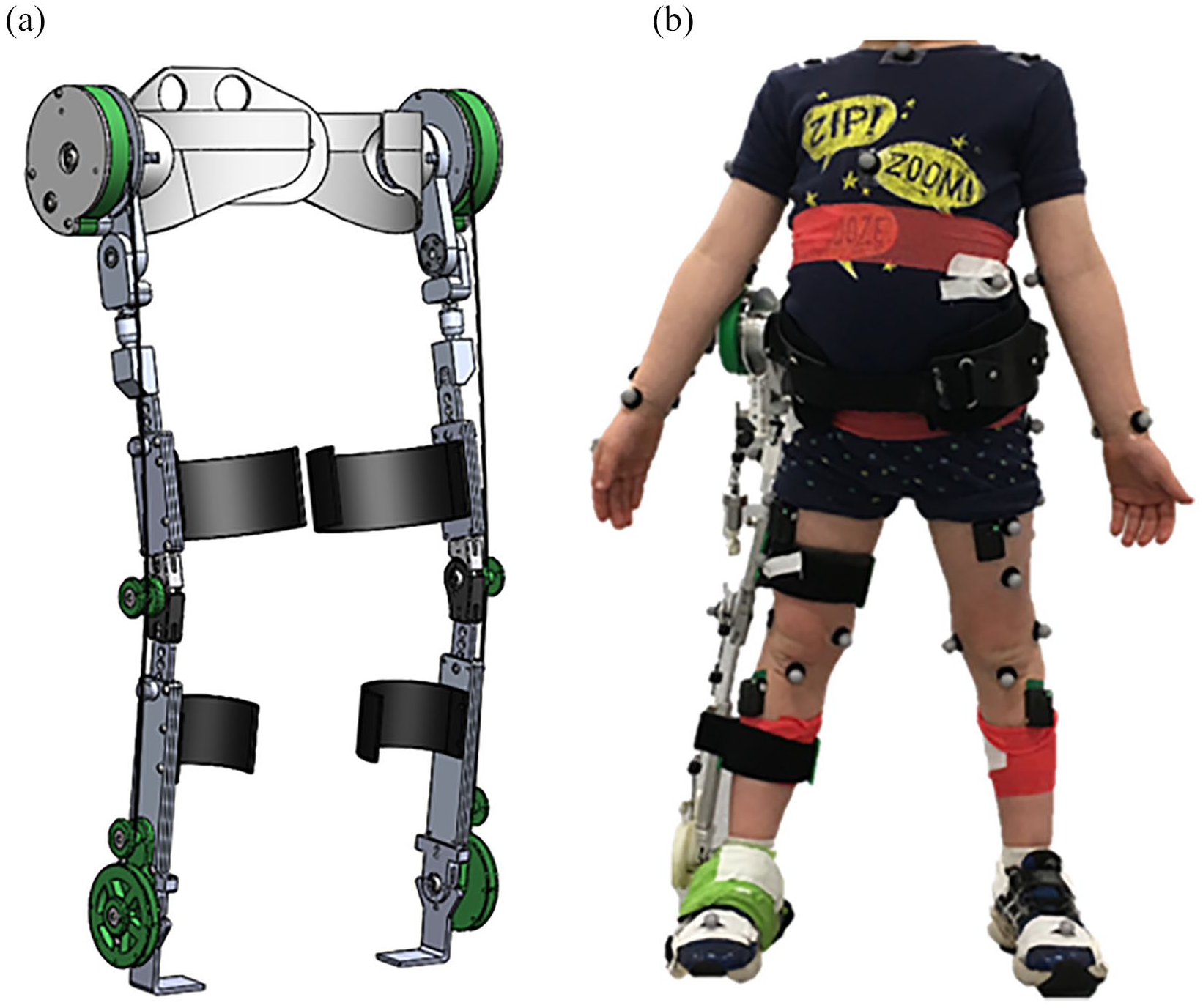
(a) CAD rendering of PlayGait demonstrating the bilateral configuration, with an exotendon connecting the hip, knee, and ankle joints. Adjustable struts were used to customize the fit to each child and an extension spring was connected to the exotendon parallel to the shank segment for the various stiffness levels. The participants in this research all wore PlayGait in a unilateral configuration. (b) One of the participants wearing the exoskeleton in the unilateral configuration on the right leg. Reflective markers and electromyography (EMG) sensors were used to track motion and muscle activity for all trials.
We varied exotendon stiffness using different extension springs normalized to body weight and leg length
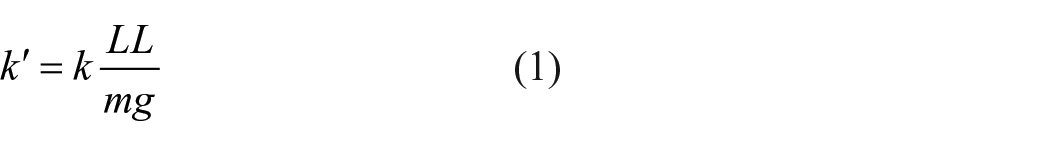
where
Participants
For this repeated-measures study, we recruited TD children and children with hemiparesis from the local community, including clinics and advocacy groups. Inclusion criteria were 3 years of age or older; ankle range of motion to neutral position and hip extension range of motion greater than 10 degrees; no surgery, hospitalization, or musculoskeletal injury in the past year; and cognitive abilities to follow simple instructions. Children with hemiparesis also were Gross Motor Function Classification System (GMFCS) Levels I–III and had no dystonia or other restrictive muscle tone. Parents provided informed consent and patients provided verbal assent for use of participant images.
Data acquisition
All participants were instrumented with reflective markers following a modified Helen Hayes marker set, including five additional markers on the exoskeleton: one centered on each of the hip, knee, and ankle pulleys and on the thigh and shank segments. Since the anterior and posterior superior iliac spine (ASIS and PSIS) pelvic markers on the posterior and anterior superior iliac spine (i.e. ASIS and PSIS) had to be removed for trials with the exoskeleton, the left and right gluteus maximus, iliac crest, and greater trochanter markers were used to track pelvic position. Electromyography (EMG) sensors (2000 Hz, Trigno, Delsys, Boston, MA, USA) were placed on the left and right rectus femoris (RF), biceps femoris (BF), medial gastrocnemius (MG), and tibialis anterior (TA). Linear measurements were taken of body segments. Motion capture data were collected with an eight-camera optical motion capture system (120 Hz, Qualisys, Goteborg, Sweden).
The exoskeleton was fit to the impaired leg of the participants with hemiparesis, which was the right leg for both participants, and to the right leg of TD participants, for consistency. During donning, the exoskeleton joints were aligned with the participants’ joints, and the exotendon was given a preload similar to the Kickstart by rotating a hip ratcheting mechanism that stretched the exotendon by 1.3 to 2.7 cm beyond the slack length when the child was standing upright.
Participants walked over level ground at a self-selected speed for three trials in each of four walking conditions. Participants wore shoes for all trials (including with the exoskeleton) but did not wear prescribed AFOs, if applicable. Trials with and without the exoskeleton were completed in a nonrandomized format to minimize experimental time and support participant comfort. Exotendon stiffness continually increased with walking condition from (1) no exoskeleton (No Exo) to (2) exoskeleton with low stiffness (Exo Low), (3) medium stiffness (Exo Med), and (4) high stiffness (Exo High). Before the exoskeleton trials, participants voiced their readiness to begin within a 2- to 5-min acclimation period with the device.
Data analysis
Motion capture data were initially processed in Qualisys and MATLAB (MathWorks Inc. Natick, MA USA), and kinematic processing was completed in OpenSim using the inverse kinematics algorithm. 19 A participant-specific model was created in OpenSim by scaling segment lengths of a 37 degrees-of-freedom (DoF) model 20 that included all markers, including the five exoskeleton markers, using the participant’s No Exo static trial. This No Exo model was then scaled using a static trial from an Exo condition to generate an Exo model with correct placement of the five markers on PlayGait. All markers were fixed except those that were removed or moved to accommodate donning PlayGait (ASIS, PSIS, and thigh markers). The EMG recordings for each muscle were high-pass filtered at 40 Hz, rectified, low-pass filtered at 10 Hz, and divided into gait cycles.
Our primary outcomes were walking speed, step width, and symmetry. Walking speed was evaluated by dividing the fore-aft distance traveled of the sternum marker by the time for each trial. Walking speed was evaluated as the average nondimensionalized walking speed21,22
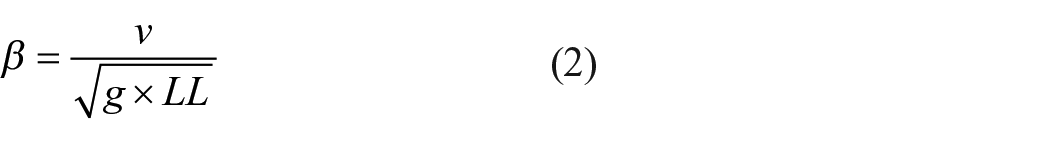
where
Results
Participants
Data were collected for four participants (Table 1; age: mean = 5.5 (standard deviation (SD) = 1.7) years; height: mean = 1.2 (SD = 0.11) m; mass: mean = 21.3 (SD = 5.81) kg; and one female and three males): two TD children (TD1, TD2) and two children with hemiparesis (H1, H2). Both H1 and H2 were diagnosed with CP due to hemispherectomy.
Spring stiffness.
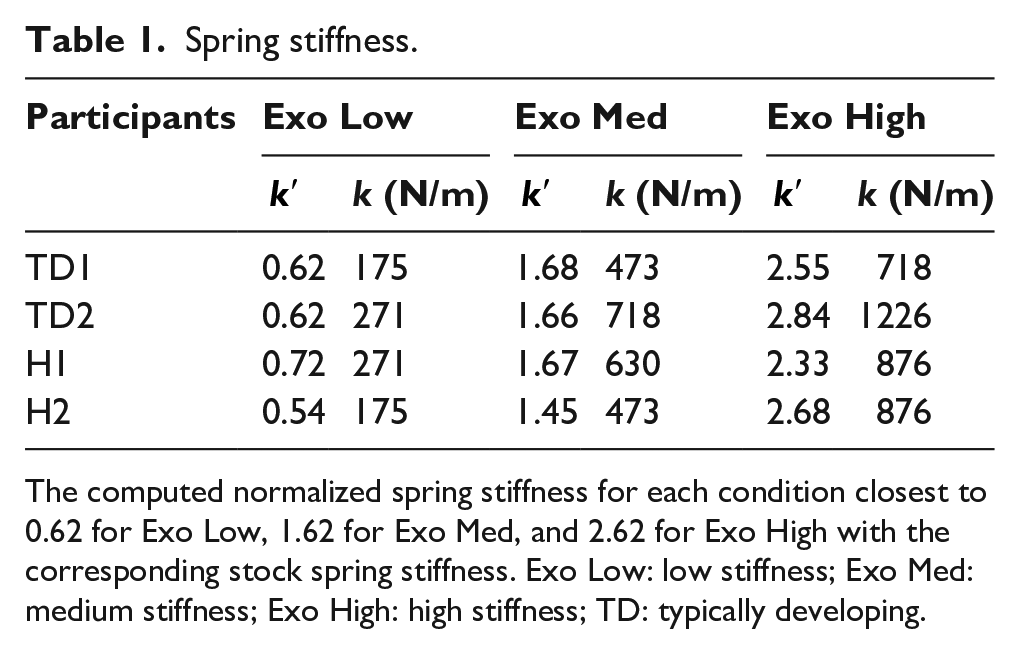
The computed normalized spring stiffness for each condition closest to 0.62 for Exo Low, 1.62 for Exo Med, and 2.62 for Exo High with the corresponding stock spring stiffness. Exo Low: low stiffness; Exo Med: medium stiffness; Exo High: high stiffness; TD: typically developing.
Spatiotemporal
Nondimensionalized walking speed was higher for TD participants than for participants with hemiparesis, but the response to the exoskeleton was variable across participants (Figure 2). Without the exoskeleton, the average nondimensionalized speed of the TD children was 0.52 versus 0.31 for the children with hemiparesis. TD1 exhibited a 17% decrease in speed with the exoskeleton (0.48 No Exo to average 0.40 with Exo), while TD2 slightly increased speed with each consecutive walking condition (0.56 No Exo to 0.62 Exo High). H1 walked fastest during the Exo High condition (0.46, compared to minimum 0.29 with Exo Med), and H2 had an increased walking speed during the Exo Med and Exo High conditions (average 0.33 for Exo Med and Exo High, compared to average 0.25 for No Exo and Exo Low).
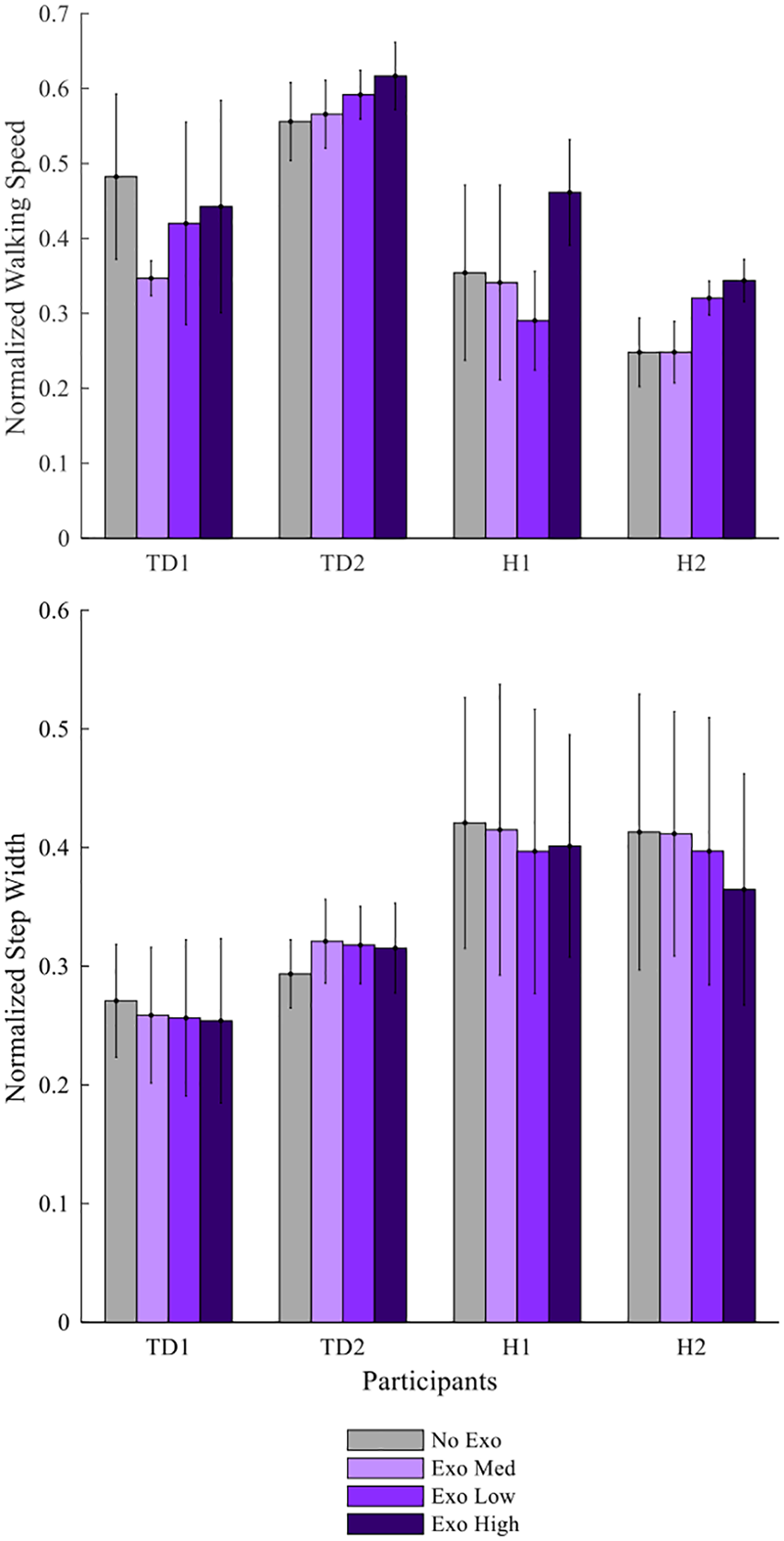
Spatiotemporal effects of exoskeleton assistance. Nondimensionalized walking speed and normalized step width averaged across all trials for each subject and stiffness level. Walking speed increased with exoskeleton use for H1 and H2 at the highest exotendon stiffness. Step width was similar across conditions for all participants. Error bars represent +/- 1 standard deviation.
Step width was similar with and without the exotendon for all participants (Figure 2). The average nondimensionalized step width for TD1 and TD2 was 0.28 and 0.29 with and without the exoskeleton, respectively. H1 and H2 had an average step width of 0.42 and 0.40 with and without the exoskeleton, respectively.
Kinematics
TD1 and TD2 demonstrated high levels of symmetry between the right and left legs with and without the exoskeleton (Figure 3 and Figure 4). Without the exoskeleton, the average symmetry (R2) was 0.98 (SD = 0.01) at the hip, 0.97 (SD = 0.01) at the knee, and 0.94 (SD = 0.01) at the ankle. Symmetry was similar with the exoskeleton: 0.98 (SD = 0.02) at the hip, 0.93 (SD = 0.05) at the knee, and 0.88 (SD = 0.08) at the ankle.
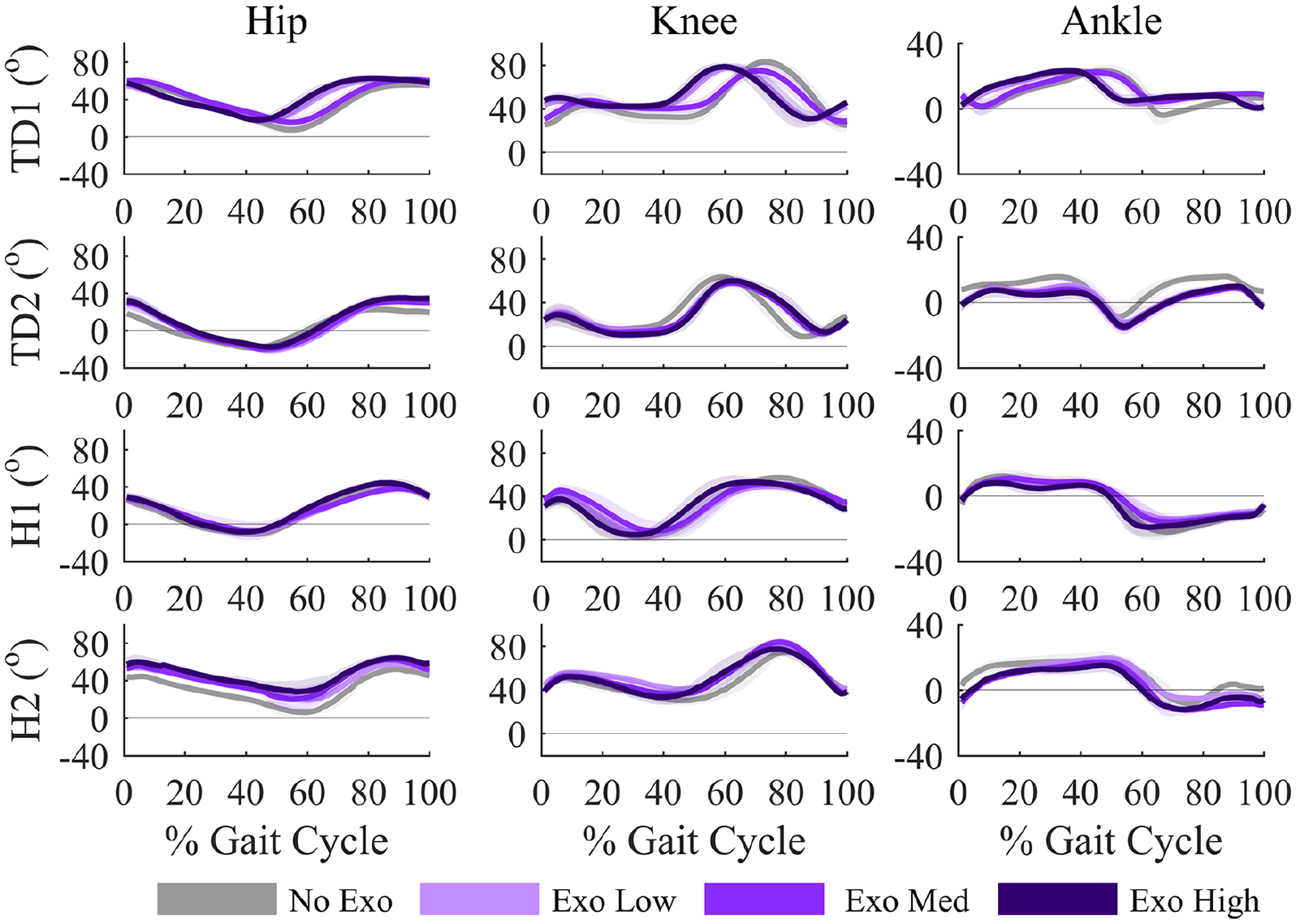
Kinematic effects of exoskeleton assistance. Right leg kinematics (leg with exoskeleton) for each participant at the hip, knee, and ankle across the gait cycle. Kinematic symmetry improved for H1 and H2 with exoskeleton use, particularly at the ankle. Shaded areas represent ±1 standard deviation.
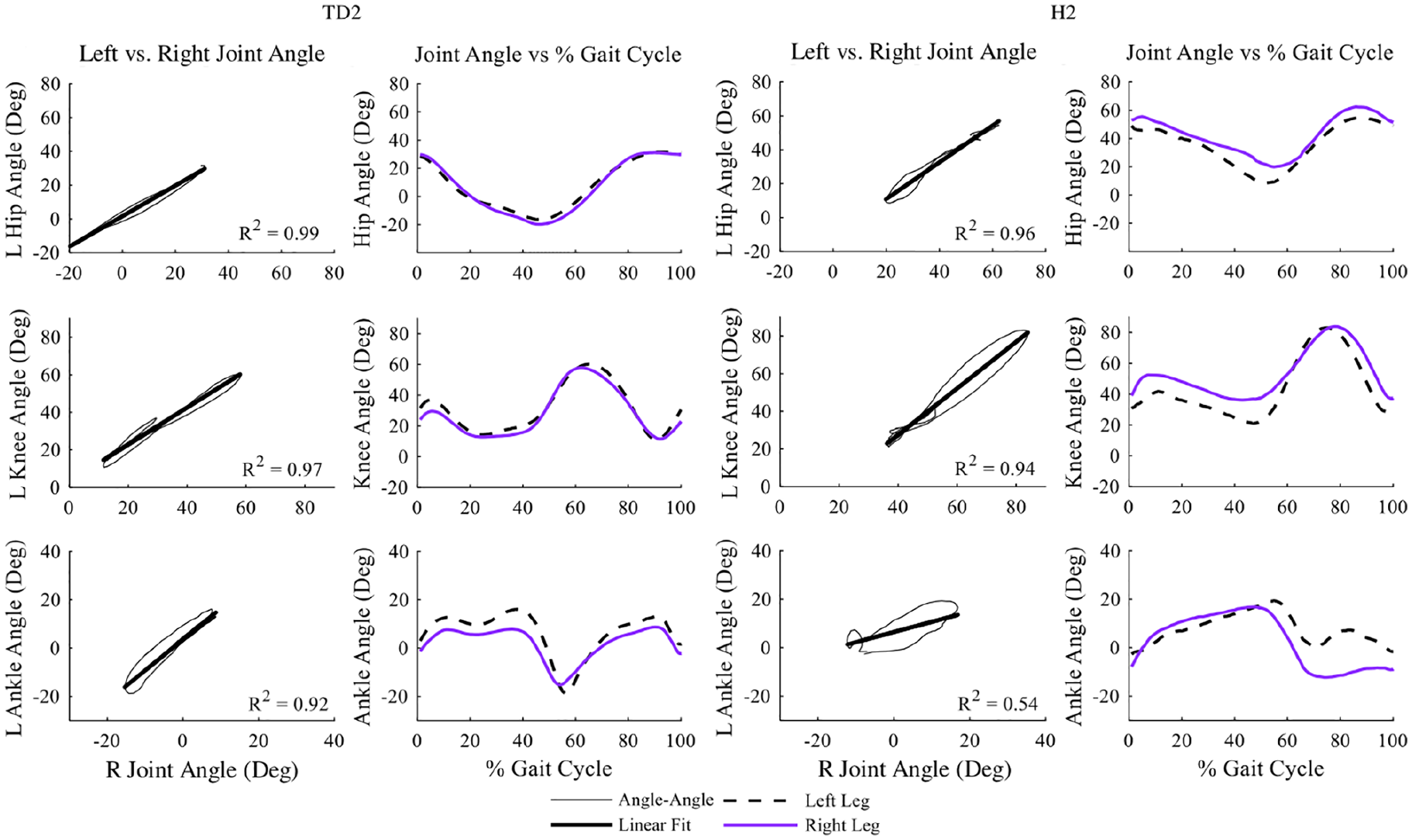
Symmetry (left) and corresponding kinematics (right) between the right (exoskeleton) and left limbs at the hip, knee, and ankle for TD2 and H2 during the Exo Med condition. Symmetry is lower for participant with hemiparesis than for TD participant.
As expected with hemiparesis, symmetry was lower for H1 and H2 compared to the TD children. Without the exoskeleton, H1 had a symmetry (R2) of 0.14, 0.63, and 0.003 at the hip, knee, and ankle, respectively. Hip and knee symmetry were similar during the Exo conditions (range: 0.11–0.13 for the hip, 0.34–0.41 for the knee), but there was an increase in ankle symmetry for the Exo High condition, from 0.003 for No Exo to 0.20 for Exo High. H2 demonstrated highest symmetry at the hip for all conditions, 0.90 without the exoskeleton and an average 0.92 (SD = 0.04) with the exoskeleton. Knee and ankle symmetry increased with the exoskeleton for H2. Knee symmetry increased from 0.85 without the exoskeleton to 0.97 for the Exo Low condition (range: 0.94–0.97 with exoskeleton). Ankle symmetry increased from 0.24 without the exoskeleton to 0.54 for the Exo Med condition (range: 0.36–0.54 with exoskeleton).
EMG
The exoskeleton induced changes in muscle activity, although the changes were variable across participants (Figure 5). TD1 had the greatest reduction in muscle activity with the exoskeleton, with reductions in RF, BF, and MG activity with the exoskeleton, although these changes could be due to the reduced walking speed. 24 TD2 had minimal changes in EMG activity with the exoskeleton, except an increase in MG activity during terminal swing for all Exo conditions. H1 had increased RF and BF activity in late swing and early stance with the exoskeleton. H2 also demonstrated increased BF activity with the exoskeleton. There were minimal changes in muscle activity for the left legs.
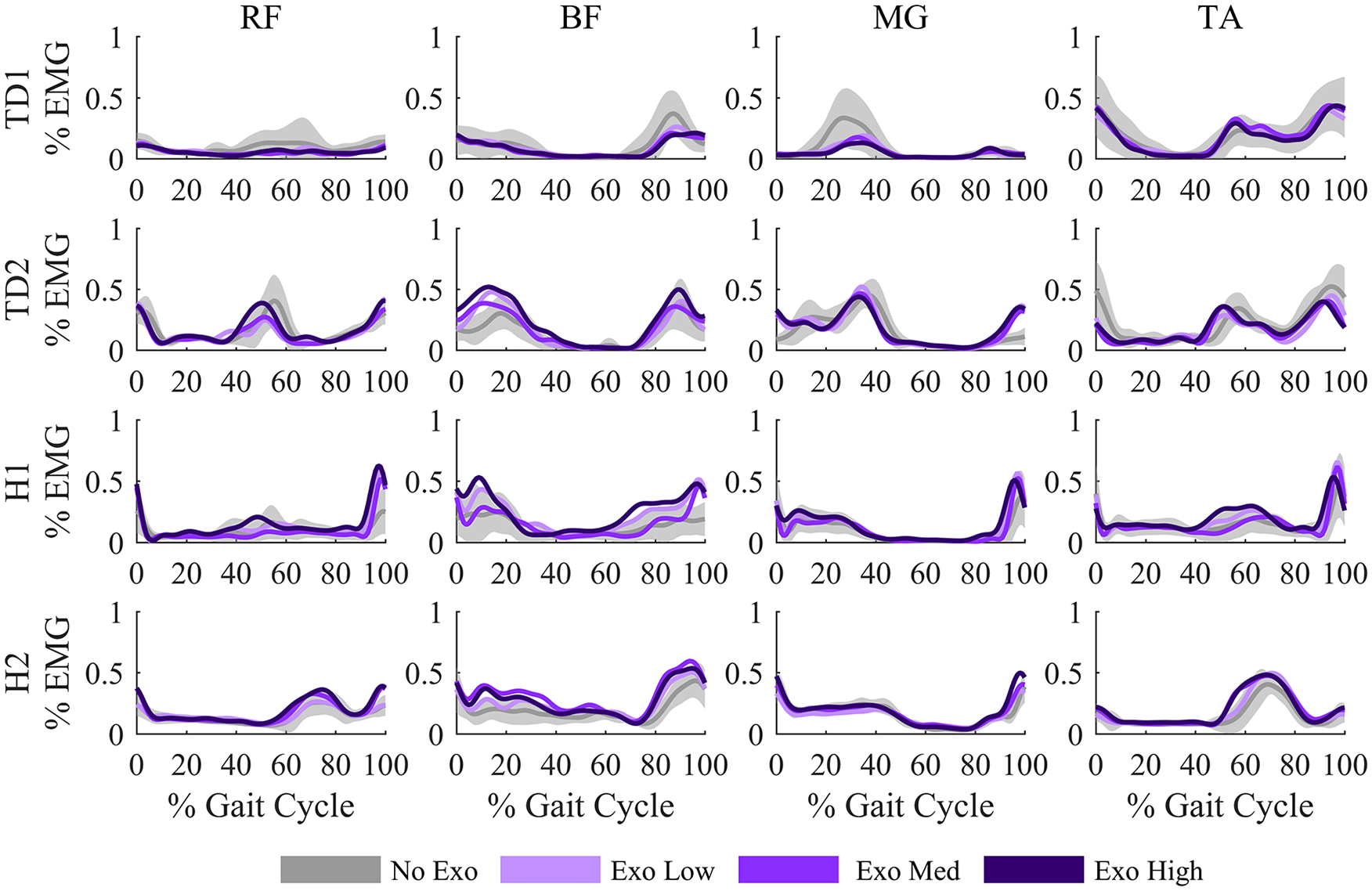
Muscle activity with and without the exoskeleton on the right limb. Electromyography (EMG) data were normalized to the maximum value across all trials for each participant. Muscle activity was altered with exoskeleton use, with the changes variable across participants and muscles. Gray shaded areas represent ±1 standard deviation of the No Exo condition. RF: rectus femoris; BF: biceps femoris; MG: medial gastrocnemius; TA: tibialis anterior.
Discussion
This study aimed to evaluate changes in gait for TD children and children with hemiparesis with a passive exoskeleton—specifically the impact on walking speed, step width, kinematic symmetry, and muscle activity. The exoskeleton had minimal impact on gait for TD children, indicating the device did not hinder unimpaired walking. For children with hemiparesis, we observed improvements in spatiotemporal and kinematic outcomes, as well as changes in muscle activity that varied with exotendon stiffness.
This study demonstrated that PlayGait did not adversely affect locomotion and has the potential to alter kinematics and muscle activity. We observed a slight decrease in walking speed when first walking with PlayGait, which may be attributed to insufficient training time before evaluation. Using step width as a measure of stability,25,26 the relatively constant step width across conditions suggests that PlayGait did not adversely affect stability. The kinematic results indicated that PlayGait was not a benefit or detriment to the joint symmetry of TD participants, but improved symmetry at specific joints for the children with hemiparesis. For example, H1 walked in equinus on his paretic side and PlayGait improved his ankle symmetry.
Examining the response to different exotendon stiffness values demonstrates the challenges of identifying optimal stiffness and normalizing stiffness values for a given individual. For TD1 and TD2, the Exo Med stiffness was most like their No Exo joint correlation symmetry results. H1 had the best kinematic symmetry across joints for Exo High, while H2 had the best correlation symmetry for Exo Med. This is similar to prior AFO research that reported variable responses to AFO stiffness between participants and the challenges in determining optimal stiffness for each individual.27,28 The interindividual differences stem from the heterogeneity of gait abnormalities and accompanying pathology among individuals with hemiparesis, 29 highlighting the need for assistive walking devices that can be customized to each individual.
The EMG results confirmed that children with hemiparesis walk with increased muscle demand compared to TD peers,30–32 although the exoskeleton did not lead to substantial changes in muscle activity. Prior simulation research predicted reductions in muscle activity for unimpaired adults walking with an exotendon-based exoskeleton. 15 While TD1 and H1 had small reductions in muscle activity, these results demonstrate the challenges in altering muscle coordination patterns during gait, even for TD participants. However, we allowed participants to self-select their walking speed and provided only limited acclimation time with the exoskeleton. Greater training and controlling walking speed may lead to more consistent changes in muscle activity and represents an important area for future research.
This study represents the first evaluation of exotendon-based exoskeletons to assist gait among children. Our conclusions are limited by the small population of participants and the relatively short data collection period. Longer training periods should be provided so that the results are not affected by the nonrandomized order of walking conditions, which most certainly allowed a training effect as this study progressed from the Exo Low to the Exo High conditions. However, the Exo High condition did not consistently outperform other Exo conditions across outcome measures. The gait patterns of H1 and H2 often deviated from straight line walking, which led to the wide variability in step width and symmetry outcomes. In addition, H1 showed his comfort with the device by becoming more playful as his trials progressed, consequently increasing his self-selected walking speed across trials. Future research should examine the long-term training effects of walking with exotendon-based exoskeletons in both laboratory and community settings. Energy storage and release could also be improved by optimizing timing of the exotendon’s release for the unique gait pattern of each child. We also observed unintended changes in gait due to device design. In particular, the placement and size of the hip pulley impacted natural arm swing among the TD children (see videos in Supplementary Material). While this had minimal impact on their lower extremity kinematics and did not impact the hemiparetic children due to their arm position during walking, reducing these effects is important for future device design.
Conclusion
The results from this study demonstrate that young children can walk with an exotendon-based exoskeleton and offer potential for improving gait symmetry and walking speed for children with hemiparesis. Passive exoskeletons may offer a method for gait rehabilitation outside the clinic that, if optimized correctly, could increase practice of symmetric gait patterns. Adjustable exotendons could also support exoskeletons that are easily customized for each individual with growth and development. Future work with additional study participants, longer training, and evaluation in the community will inform the long-term benefits of passive exoskeletons.
Supplemental Material
10.1177_0309364620956868_Supplementary_Material – Supplemental material for Evaluation of a passive pediatric leg exoskeleton during gait
Supplemental material, 10.1177_0309364620956868_Supplementary_Material for Evaluation of a passive pediatric leg exoskeleton during gait by Jessica Zistatsis, Keshia M Peters, Daniel Ballesteros, Heather A Feldner, Kristie Bjornson and Katherine M Steele in Prosthetics and Orthotics International
Footnotes
Acknowledgements
We thank Brianna Goodwin, Brian Glaister, Chris Richburg, Andrea Willson, Alex Gong, Daniel Parrish, Jeffrey Bergeson, and Kira Newman for their assistance with this research and prototype development.
Author contributions
JZ: Conceptualization, Funding Acquisition, Methodology, Investigation, Formal Analysis, Visualization, Writing - original draft.
KMP: Investigation, Formal Analysis, Visualization, Writing - review & editing.
DB: Investigation, Writing - review & editing.
HAF: Investigation, Writing - review & editing.
KB: Investigation, Resources, Writing - review & editing.
KMS: Conceptualization, Funding Acquisition, Investigation, Methodology, Project Administration, Resources, Supervision, Writing - review & editing.
Ethics Review and Approval
This study was approved by the University of Washington Institutional Review Board (STUDY00001789).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the New England Pediatric Device Consortium (NEPDC) and Food & Drug Administration (FDA) (P50FD004907); the Center for Translation of Rehabilitation Engineering Advances and Technology (TREAT) and National Institutes of Health (NIH) (R24HD065703, P2CHD086841); the Orthotic and Prosthetic Education and Research Foundation, Inc. (OPERF-2017-FA-1); the CoMotion Innovation Fund; and a 2017 Developmental Grant from the University of Washington Global Center for Integrated Health of Women, Adolescents, and Children (Global WACh) and the University of Washington Coulter Translational Research Partnership Program.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
