Abstract
Background:
The extent to which current prosthetic health economic evaluations inform healthcare policy and investment decisions is unclear. To further the knowledge in this area, existing evidence gaps and method design issues must be identified, thereby informing the design of future research.
Objectives:
The aim of this systematic review was to identify evidence gaps, critical method design and reporting issues and determine the extent to which the literature informs a wide range of policy and investment decisions.
Study Design:
Systematic review.
Methods:
A range of databases were searched using intervention- and health economic evaluation-related terms. Issues with methodological design and reporting were evaluated using the Consolidated Health Economic Checklist – Extended and the Checklist for Health Economic Evaluation Reporting Standards.
Results:
The existing health economic evaluation literature was narrowly focused on informing within-participant component decisions. There were common method design (e.g. time horizon too short) and reporting issues (e.g. competing intervention descriptions) that limit the extent to which this literature can inform policy and investment decisions.
Conclusion:
There are opportunities to conduct a wider variety of health economic evaluations to support within- and across-sector policy and investment decisions. Changes to aspects of the method design and reporting are encouraged for future research in order to improve the rigour of the health economic evaluation evidence.
Clinical relevance
This systematic review will inform the clinical focus and method design of future prosthetic health economic evaluations. It will also guide readers and policy-makers in their interpretation of the current literature and their understanding of the extent to which the current literature can be used to inform policy and investment decisions.
Keywords
Background
People with mobility-related impairments, such as stroke or amputation, use orthoses/prostheses to assist with function, activity and participation. 1
Historically, orthoses/prostheses were provided by highly skilled tradespeople whose primary role was the manufacture and supply of the device.2 –4 Over time, the role of the orthotist/prosthetist has become increasingly clinical3,5 as education and training has shifted to emphasise autonomous professional practice.2,5
The clinical role of the orthotist/prosthetist will continue to evolve in response to new models of care. 6 For example, in Australia, the National Disability Insurance Scheme – a national funding model for disability services and interventions 7 – requires practitioners to use high-level communication skills to support clients in identifying their own treatment goals6,8 and subsequently use that information to inform their decision about orthotic/prosthetic interventions.7,9 Practitioners are also expected to evaluate treatment outcomes using valid and reliable outcome measures10,11 and thereby demonstrate the contribution that the intervention makes towards a client’s goals.
While the clinical role has evolved with contemporary models of orthotic/prosthetic care, it is not clear that health economic evaluations (HEEs) have kept pace with the changes in orthotic/prosthetic practice given that HEEs seem to have focused on the cost-effectiveness of newer technologies (e.g. micro-processor prosthetic knees) compared to the standard of care.12 –16 While these studies are designed to inform technology-related policy decisions, it is not clear whether HEEs have been conducted to inform a wider variety of policy decisions, such as cost-effectiveness of orthotic/prosthetic interventions, compared to other healthcare interventions, such as post-stroke rehabilitation.
If we are to further work in this area, it is prudent to first undertake a systematic review of the orthotic/prosthetic HEE literature to identify evidence gaps, critical method design and reporting issues, and then to determine the extent to which the literature informs a wide range of policy and investment decisions.
Method
A protocol for this systematic review was registered with PROSPERO (CRD42018116910) and published. 17 As such, a summary of the method has been reported here in accord with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 18
Search strategy
National Library of Medicine Medical Subject Headings (MeSH) and search terms for orthotics, prosthetics and health economics were used to search AMED, EMBASE, MEDLINE, PsycINFO, CINAHL, ProQuest Nursing and Allied Health, Web of Science, Cochrane Database of Systematic Reviews, Centre for Reviews and Dissemination Health Technology Assessment and the National Health Service Economic Evaluation Databases.19 –22
Search terms related to the intervention (e.g. prostheses) and HEEs (e.g. cost-utility) were based on previously developed search strings.21 –24 Synonyms and acronyms were used in combination with Boolean operators and wild cards as part of a title, abstract and keyword search. Each search term and string was developed, tested and modified based on a comparison of the yield against a bank of known articles. 17
Searches were restricted to studies published in English between 1 January 2000 and 1 January 2019, given preliminary testing did not yield any HEEs prior to the year 2000. Reference lists of the included studies were hand searched and used for a forward citation search using Google Scholar.
Details of the exact search for one database have been presented (Figure 1).
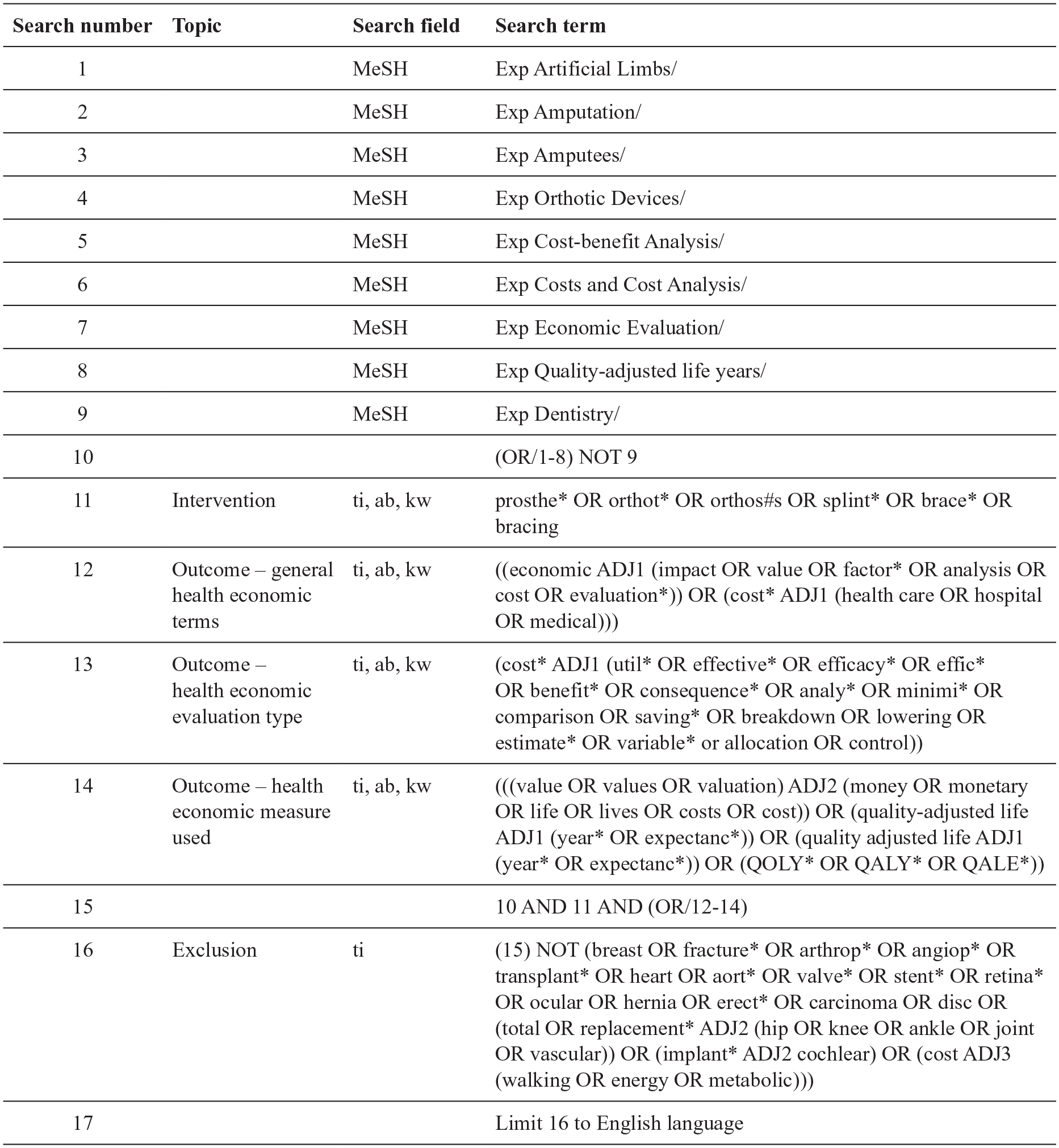
Example search in the MEDLINE database to identify the health economic evaluation literature in orthotics and/or prosthetics.
Data management
Search results were exported to EndNote X8.2 (Clarivate Analytics, Philadelphia, PA) and duplicates removed. EndNote records were exported to a customised Excel spreadsheet (Microsoft Inc., Sydney, NSW). Decisions regarding inclusion/exclusion, data extraction and critical appraisal checklists were recorded (Supplemental Material: Additional file 1).
Selection process
The following criteria were used to determine inclusion:
(a) Research type: original, peer-reviewed;
(b) Language: English;
(c) Publication range: 1 January 2000 to 1 January 2019;
(d) Intervention: externally applied orthoses or prostheses for any part of the human body as defined by the International Standards, ISO 9999:2016 (ISO 8549 1-3 prosthetics and orthotics – vocabulary) 25 and provided by a regulated and/or recognised healthcare professional;26,27
(e) Outcome: a HEE, defined as ‘the comparative analysis of alternative courses of action in terms of both their costs and consequences’. 28
The search yield was screened by one investigator (L.C.) based on a review of title, abstract and the full text as necessary. As required, a second (M.D.) or third opinion (A.S.) was sought and disagreement resolved by discussion until consensus.
Quality appraisal and risk of bias
The Consensus on Health Economic Criteria – Extended (CHEC-Extended) list29,30 was used to assess risk of bias and appraise individual studies. 31 The CHEC-Extended is supported by detailed guidelines. 32 A decision aid specific to the orthotic/prosthetic literature was developed to enhance consistent and transparent decision-making (Supplemental Material: Additional file 2).
The Consolidated Health Economic Evaluation Reporting Standards (CHEERS) checklist 19 was used to assess the quality of reporting of HEEs.
Data extraction and risk of bias assessment process
A customised data extraction spreadsheet (Supplemental Material: Additional file 1) was used to record details of each study and the outcomes of the quality appraisal and risk of bias assessment. 30
A primary reviewer (L.C.) independently extracted data and completed both the CHEC-Extended and CHEERS. A second reviewer (M.D.) independently reviewed these data to confirm accuracy, and disagreements were resolved through discussion with input from a third reviewer (A.S.) as needed. Where aspects of the method design, results or reporting were unclear, the original authors were contacted.
Data summary and reporting
The findings were reported as a narrative without meta-analysis. The appraisal of study methods and reporting were described using examples from the literature. The extent to which issues introduced bias and reduced confidence in the findings was described.
Results
Search results
The search yielded 1137 studies (Figure 2). Following the removal of 295 duplicates, 842 studies were vetted against the inclusion criteria. A total of 64 articles underwent full-text review: 42 studies did not meet the inclusion criteria, and 1 was excluded after efforts to contact the authors failed to confirm eligibility. Hand searching the reference lists yielded one further article that met the inclusion criteria.
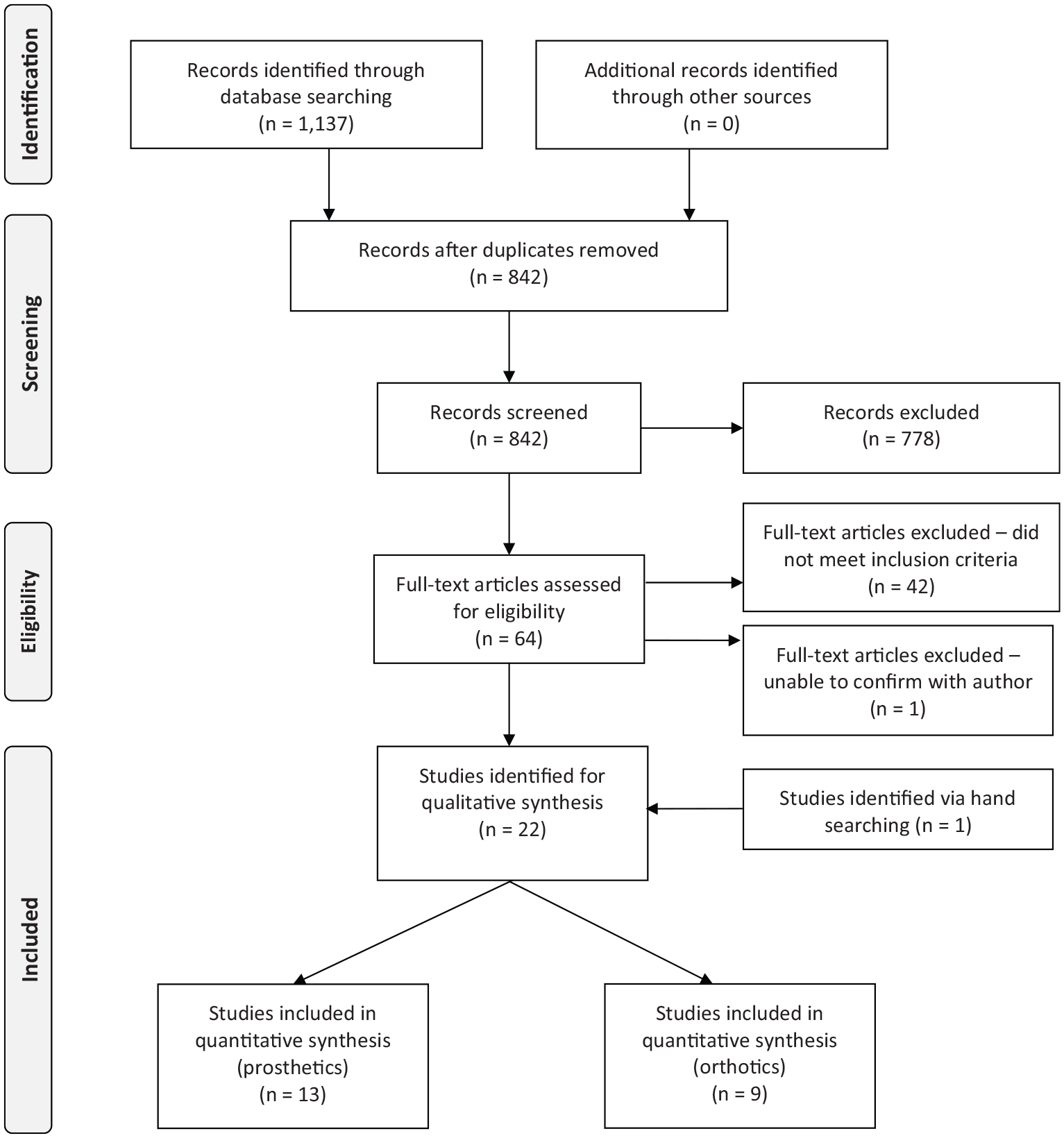
PRISMA flowchart of the systematic search yield.
A total of 22 HEEs met the inclusion criteria including 9 orthotic-focused33 –41 and 13 prosthetic-focused studies.12 –16,42 –49
The orthotic- and prosthetic-focused studies were different in terms of their HEE method design, and thus the findings have been presented in two separate reviews with the remainder of this review based on the 13 prosthetic HEEs that met the inclusion criteria.12 –16,42 –49
Study characteristics
In order to differentiate the different types of HEEs included in this review, we provide introductory definitions of cost-utility analyses (CUA), cost-effectiveness analyses (CEA) and cost-consequence analyses (CCA).
Both CUA and CEA bring together the benefits and costs of interventions to establish an Incremental Cost-Utility Ratio (ICUR) or an Incremental Cost-Effectiveness Ratio (ICER). The equation used to calculate the ICER/ICUR is
In both CUA and CEA, the costs describe the expenses incurred in providing the intervention and may include the cost of the prosthesis or a practitioner’s labour, as illustrative examples. The primary difference between the CUA and CEA is the approach taken to measure benefits: a CUA uses a generic measure of benefit, such as health-related quality of life (HR-QoL) that allows the results to be compared across different interventions and sectors. By comparison, a CEA uses a benefit specific to the interventions under investigation (e.g. temporospatial parameters). Finally, CCA does not bring the costs and benefits together as a ratio, but reports them in a disaggregated form. 28
Of the 13 included studies, 6 were CUA,12 –15,42,46 6 were CCA,16,43 –45,48,49 and 1 was a CEA 47 (Table 1) (Supplemental Material: Additional file 1, Tab 1).
Summary of study design of the included prosthetic HEE studies.
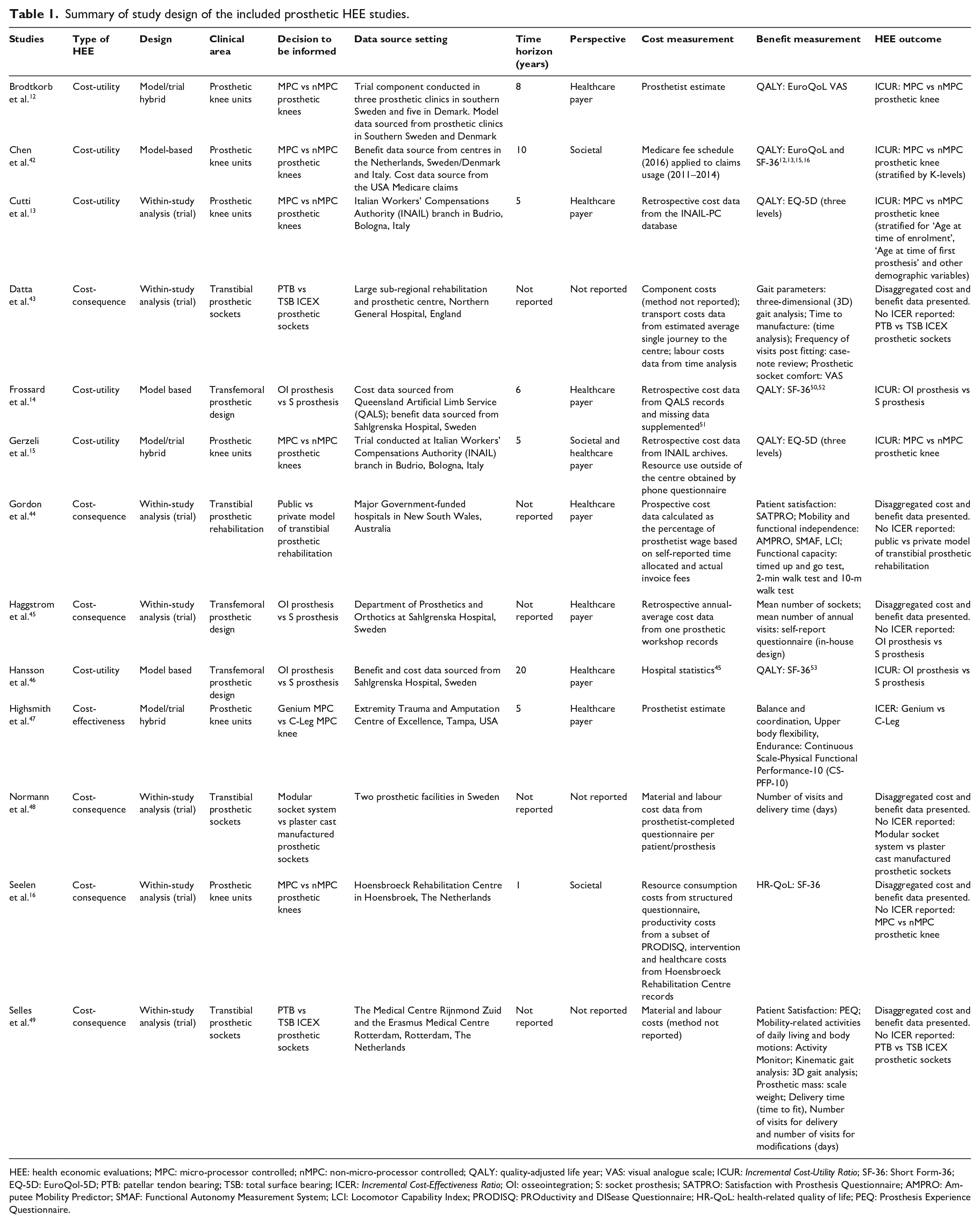
HEE: health economic evaluations; MPC: micro-processor controlled; nMPC: non-micro-processor controlled; QALY: quality-adjusted life year; VAS: visual analogue scale; ICUR: Incremental Cost-Utility Ratio; SF-36: Short Form-36; EQ-5D: EuroQol-5D; PTB: patellar tendon bearing; TSB: total surface bearing; ICER: Incremental Cost-Effectiveness Ratio; OI: osseointegration; S: socket prosthesis; SATPRO: Satisfaction with Prosthesis Questionnaire; AMPRO: Amputee Mobility Predictor; SMAF: Functional Autonomy Measurement System; LCI: Locomotor Capability Index; PRODISQ: PROductivity and DISease Questionnaire; HR-QoL: health-related quality of life; PEQ: Prosthesis Experience Questionnaire.
The included studies used model,14,42,46 trial13,16,43 –45,48,49 or hybrid designs12,15,47 whereby trial results were extrapolated to extend the time horizon (Table 1). Modelling studies sourced input data from a small pool of studies,12,13,15,16,45,50–54 of which several were included in this review12,13,15,16,45 (Supplemental Material: Additional file 1, Tab 2). Most studies took a payer perspective12 –14,16,43 –49 rather than a societal perspective.15,16,42 Time horizons were typically less than 6 years13 –16,47 or not reported43 –45,48,49 (Table 1).
Seven studies in this review used population-level utility measures (i.e. HR-QoL) to measure benefits, including EQ-5D (EuroQoL),12,13,15,42 and Short Form Health Survey (SF-36).14,16,42,46 The remaining studies used population-specific effectiveness measures, including physical function,44,47,49 gait,43,44 patient satisfaction, 44 time to prosthetic delivery,48,49 number of prosthetic sockets 45 and number of visits43,45,48,49 (Table 1).
Participants included in these studies were typically young men with lower limb amputation due to trauma (Table 2). Only one study reported comorbid conditions 42 (Table 2).
Summary of participant demographic characteristics of the included prosthetic HEE studies.
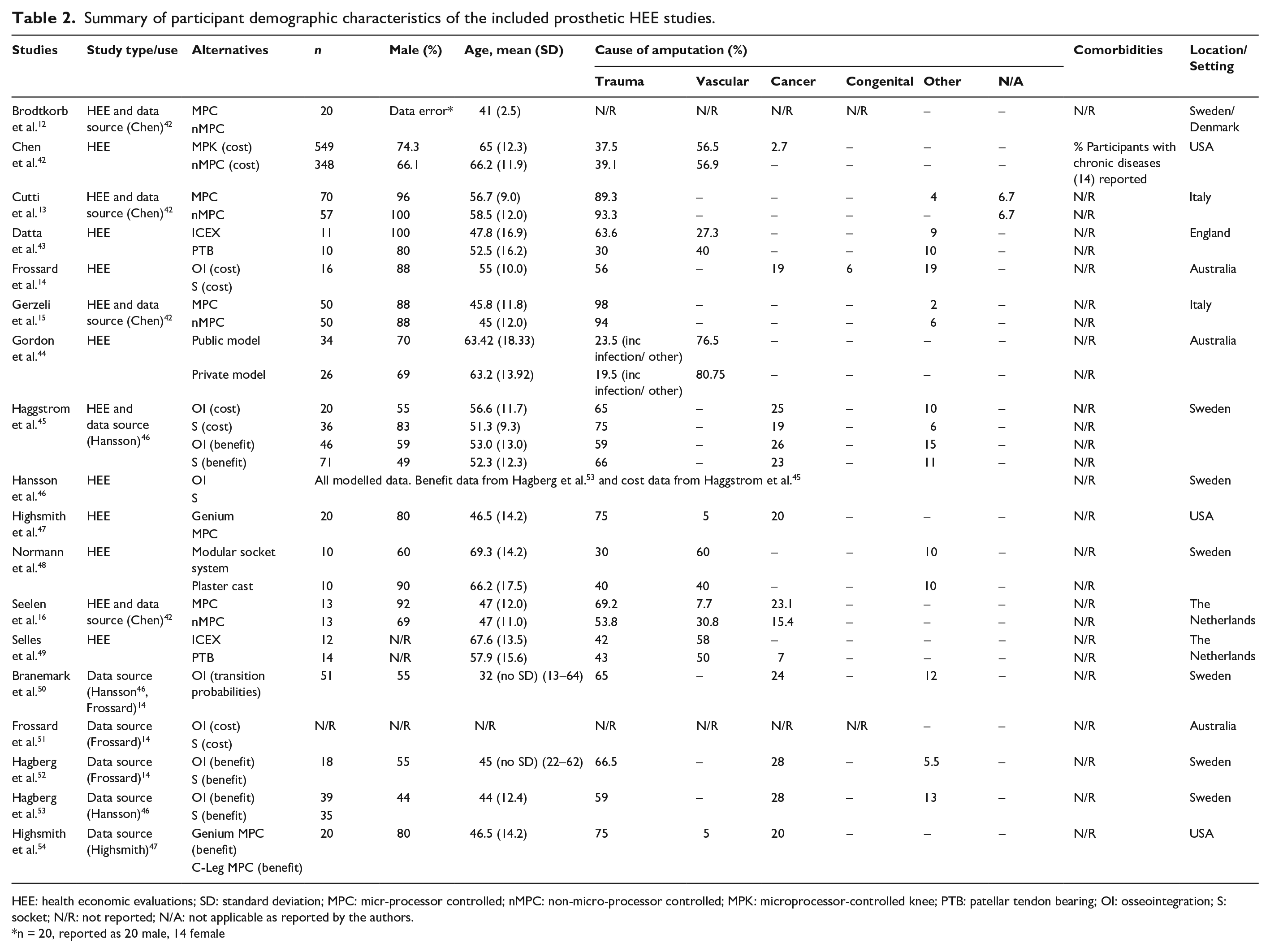
HEE: health economic evaluations; SD: standard deviation; MPC: micr-processor controlled; nMPC: non-micro-processor controlled; MPK: microprocessor-controlled knee; PTB: patellar tendon bearing; OI: osseointegration; S: socket; N/R: not reported; N/A: not applicable as reported by the authors.
n = 20, reported as 20 male, 14 female
Studies were conducted in countries with well-developed healthcare systems including: Sweden,12,45,46,48 USA,42,47 Australia,14,44 Italy,13,15 The Netherlands16,49 and England 43 (Table 2).
Evidence gaps
The HEEs included in this review focused on evaluating a few discrete areas, comparing new prosthetic technologies to the standard of care including:
Osseointegration- (OI) and socket- (S) prosthesis for people living with transfemoral amputation;14,45,46
Micro-processor-controlled (MPC) and non-micro-processor-controlled (nMPC) prosthetic knees included in transfemoral prostheses;12,13,15,16,42,47
Direct- versus traditional- methods for the manufacture of transtibial prosthetic sockets;43,48,49
Public- versus private- models of prosthetic rehabilitation. 44
There were no HEEs designed to evaluate the cost-effectiveness of a wider range of prosthetic interventions common in contemporary clinical practice.
Method design and reporting appraisal
Several method design issues were common across studies included in this review: selection bias, allocation bias, choice of competing interventions, methods used to calculate costs and benefits, length of the time horizon, quality of the model input data and the management of uncertainty (Table 3) (Supplemental Material: Additional file 1, Tab 1). Common issues with reporting were also identified; in particular, the lack of details about the interventions or setting, and insufficient clarity in the reporting of the input data (Table 4) (Supplemental Material: Additional file 1, Tab 1). Each of these issues have been explored in the following subsections.
CHEC-Extended appraisal of included prosthetic HEE studies.
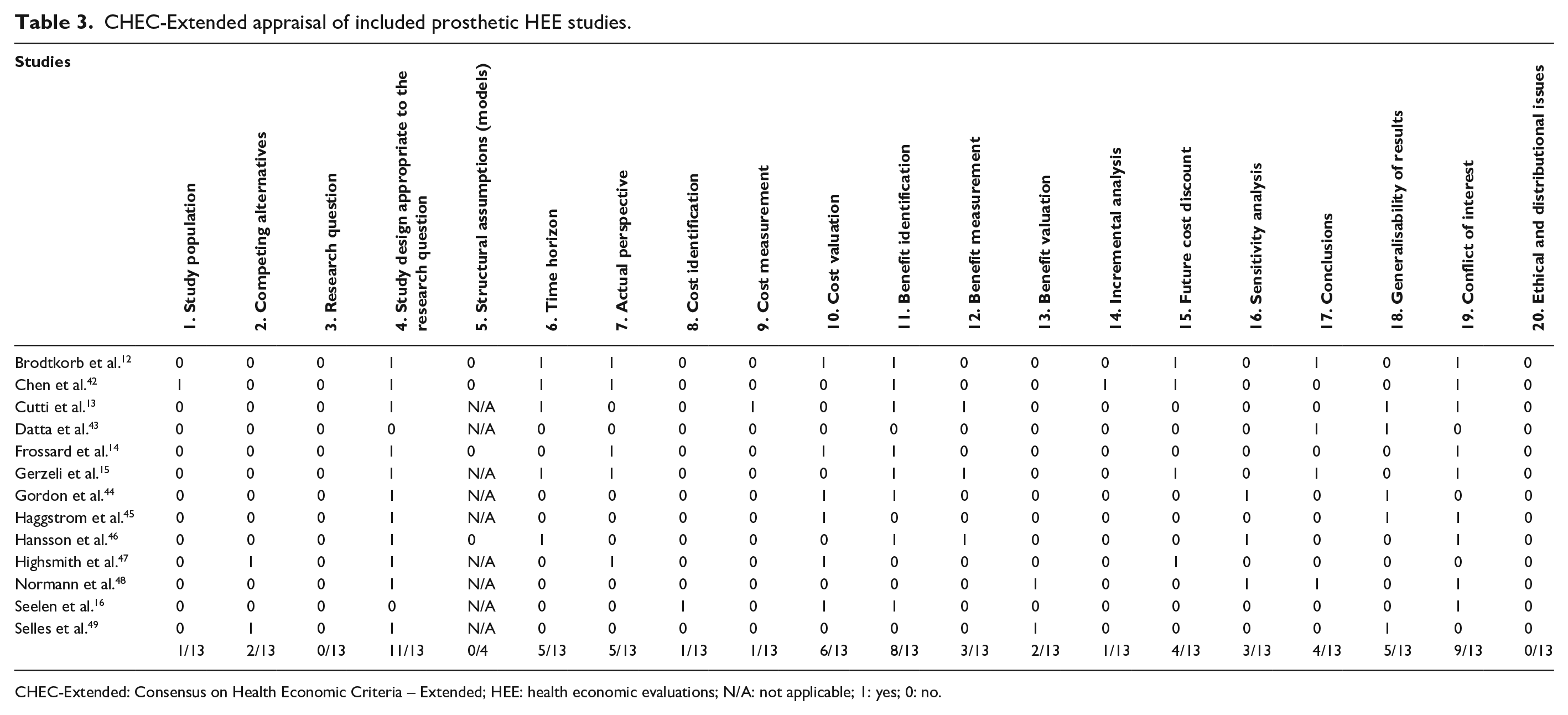
CHEC-Extended: Consensus on Health Economic Criteria – Extended; HEE: health economic evaluations; N/A: not applicable; 1: yes; 0: no.
CHEERS (reporting) appraisal of the included prosthetic HEE studies.
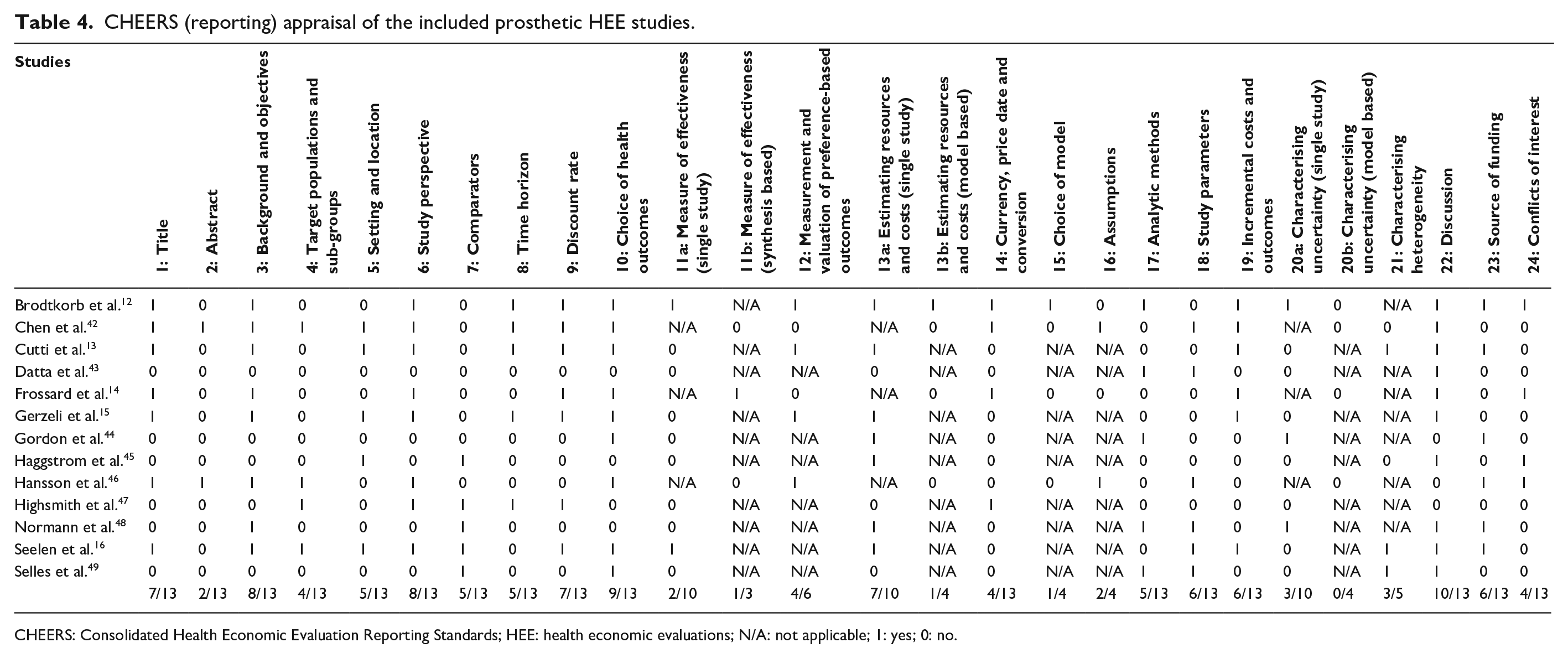
CHEERS: Consolidated Health Economic Evaluation Reporting Standards; HEE: health economic evaluations; N/A: not applicable; 1: yes; 0: no.
Selection bias
Selection bias affected seven studies included in this review,12,14,15,42,45 –47 including three studies that evaluated the cost-effectiveness of OI prostheses versus S prostheses.14,45,46 People seeking to transition from an S prosthesis to an OI prosthesis often have a long history of fitting problems with their S prosthesis 55 and use their prosthesis a small proportion of the time, as evidenced by data showing that 15% of participants report using their S prosthesis less than 1 day per week. It is therefore not unexpected that the SF-36 physical function score of S prosthesis users who transitioned to an OI prosthesis was half that (x̅w, 35.3 ± 21.7)50,52,53 reported in another study of S prosthesis users (68.2 ± 23.3) who had not sought an OI prosthesis. 16 The selection of people with a long history of fitting problems with the S prosthesis has likely overvalued the benefit of the OI prosthesis and as such, generalising the results to people that have not had a history of problems with the S prosthesis requires care in order to be confident that the costs and benefits associated with transitioning from an S prosthesis to an OI prosthesis will be as reported in the literature.
Allocation bias
Allocation bias may have been present in four studies that evaluated the cost-effectiveness of MPC versus nMPC prosthetic knees, due to participants being allocated to an intervention based on ‘medical necessity’.13,15,16,42 We contend that those allocated to the nMPC prosthetic knee were likely less healthy than those who received the MPC prosthetic knee. For example, a modelling study 42 provides supplementary material that includes a comprehensive description of the MPC and nMPC cohorts from which the cost data were collected. The supplementary material highlights that the cohort that received the nMPC prosthetic knee had a higher proportion of people with diabetes (nMPC 54.6% vs MPC 45.2%), double the number of participants with myocardial infarction (nMPC 6.0% vs MPC 3.1%), more participants with chronic obstructive pulmonary disease (nMPC 37.9% vs MPC 27.7%) and heart failure (nMPC 36.2% vs MPC 25.7%) and double the mortality rate in the 1 (nMPC 13.5% vs MPC 6.0%) and 2 years (nMPC 26.1% vs MPC 12.2%) that followed. 42 The likely allocation bias partly explains the lower HR-QoL,13,15,16,42 higher healthcare usage and costs12,16 and greater productivity costs 16 in the group allocated the nMPC prosthetic knee compared to those allocated the MPC prosthetic knee. As such, the greater cost-effectiveness associated with the MPC prosthetic knee is likely exaggerated by allocation bias.
Competing interventions
In five studies included in this review, the comparator or competing intervention was poorly considered.13,15,16,42,48 For example, consider one of the studies 16 that compared a specific MPC prosthetic knee (i.e. C-Leg) with a group of disparate nMPC prosthetic knees (i.e. Total Knee, Proteval, Otto Bock 3R80 or 3R92), each with a different control mechanism. 16 Consequently, there is considerable variability in the SF-36 Physical Function subscale score among the group of patients with nMPC prosthetic knees (65.0 ± 27.3) which masks the comparative benefit of the MPC knee (84.0 ± 11.4), rendering the difference statistically nonsignificant. 16
Calculating costs
There were two key issues with cost calculation: identification of all relevant costs12 –15,42 –49 and measurement of actual costs.12 –15,42 –49
In terms of identifying all relevant costs, many studies accounted only for the cost of the discrete prosthetic component that was the focus of the HEE (e.g. MPC and nMPC knee), excluding the cost of other component parts of the prosthesis or the clinical service.12 –15,42,43,45 –48 The rationale for this approach was proffered with varying degrees of rigour, with some studies indicating that the prosthetic foot15,42 and fitting costs 13 were the same between the MPC and nMPC interventions. While this rationale may be reasonable in today’s clinical practice, given that in many settings the prosthetic foot prescribed is determined by K-level classification, this was not the case at the time the cost data were collected in these investigations. At the time of cost measurement (1999–2003),13,15 there is evidence that foot prescription was influenced by knee unit configuration and warranty requirements 56 or the policy requirements of funding agencies. 13 Where there are potential differences in cost items between interventions, it is recommended that these costs are captured in the HEE. Failure to capture all costs associated with the provision of the nMPC and MPC prosthetic knees, such as the prosthetic foot and fitting costs, reduces confidence in the results and their generalisability to other settings.
In terms of measuring actual costs, a number of studies used a retrospective survey of practitioners12,16,47 or patients,14,15 that reduced confidence in the accuracy of the reported costs. For example, a modelling study 14 sought to fill gaps in the cost data for the S prosthesis intervention using data from a prior HEE 45 that asked participants using an OI prosthesis to recall how many prosthetic sockets they were provided with when they used a transfemoral S prosthesis. Given that the average time since using an S prosthesis was nearly 7 years, it is unlikely that participants were able to accurately recall the number of sockets provided over time. During the years participants wore the S prosthesis, they reported receiving about one socket each year. 45 This is in stark contrast to a study that actually recorded the number of sockets provided over a 10-year period, where participants were provided with one socket about every 3 years. 57 In this case, the recall bias may have artificially inflated the number of prosthetic sockets that were costed, also biasing the cost-effectiveness in favour of the OI prosthesis intervention.
Calculating benefits
There were three main issues with the way the benefits were determined in studies included in this review: choice of outcome measure appropriate to the purported benefit of the intervention,43,45,47 –49 bias in data collection12,14,16,42 –45,47 –49 and issues with the way the ICURs/ICERs were calculated.12,14,47
First, five studies utilised outcome measures that may not be considered appropriate to capture the benefit of the intervention.43,45,47 –49 For example, studies comparing two methods of transtibial prosthetic socket manufacture (i.e. conventional plaster cast method vs direct manufacture onto the residual limb) used a variety of benefit measures with limited rationale for the choice.43,48,49 Many of the benefit measures used (e.g. temporospatial parameters) are unlikely to be sensitive to the impact of differences in the way the socket was manufactured43,49 or represent the benefits of most importance to healthcare funders and patients. The direct manufacture technique is likely to be less burdensome on the patient (e.g. requiring fewer visits) and allow more timely provision of the prosthesis; consequences that are likely to be of benefit to patients independent of the clinical outcome. Ideally, the economic evaluation would capture such benefits in its assessment of cost-effectiveness.
Second, a number of studies administered benefit measures in a way that introduced bias.12,14,16,42 –45,47 –49 For example, one study12(p.27) asked participants currently using an MPC prosthetic knee to complete the EuroQoL visual analogue scale (VAS) while they ‘pictured themselves in their current situation of life but using a prosthesis fitted with their prior knee’. It is difficult to have confidence in these data given that participants were asked to imagine their current life situation assuming that they were using an nMPC knee that, on average, they had ceased using 45 months prior. 12
Finally, there were issues with the way utility was determined in a number of investigations included in this review.12,14,47 For example, the aforementioned study 12 administered only the VAS component of the EuroQoL. Without first administering the complete EuroQoL, participants lack a standardised definition of the health states that are important to inform their self-assessed utility on the VAS. 28 The bias introduced by this approach may be evidenced by the threefold increase in the incremental quality-adjusted life years (QALYs) at 1 year between this study (0.3 incremental QALYs) 12 and other HEEs that compared the same interventions but used the EuroQoL and VAS as intended (0.09 incremental QALYs; 13 0.08 incremental QALYs). 15 This issue has artificially inflated the incremental QALY in favour of the MPC prosthetic knee, which is unfortunate given that these data have been widely cited,58,59 used in a subsequent modelling study 42 and likely influenced policy related to the provision of MPC prosthetic knees.
Time horizon
The time horizon is the period over which costs and benefits are measured and should be long enough to capture the costs and benefits of the competing interventions. 28 In a number of studies, the time horizon was too short14,16,43,47 –49 or differed between the collected cost and benefit data.14,43,45
Six studies had time horizons that were too short.14,16,43,47 –49 Consider, for example, a cost-consequence study that compared the costs and benefits of the nMPC and MPC prosthetic knees over a 1-year time horizon. 16 While the 1-year time horizon was sufficient to capture the initial cost of the MPC prosthetic knee, it was too short to capture the ongoing costs associated with replacement of the nMPC prosthetic knee given that the usable life of an nMPC knee is in the order of 3 years.12,60 Neither was the 1-year time horizon long enough to capture the full benefits of the upfront investment that would be realised over the usable life of the MPC knee intervention which, for the MPC prosthetic knee unit in these studies, is between 5 and 8 years.12,15
There were four studies where the time horizons differed between the cost and benefit data.14,16,43,45 Using a modelling study 14 that compared OI prostheses and S prostheses as an illustrative example, the synthesised benefit data were captured over 2 years 53 and cost data captured over 6 years post-surgery. 14 This assumes that the benefits observed over 2 years were sustained over 6 years, 14 which is unlikely since some participants would have experienced complications – implant loosening (6%), 50 implant failure (3%), 61 periprosthetic fracture (5%), 61 and infection (18%) 62 – more than 2 years after surgery. As a result, the costs are likely understated, and the benefits overstated, in favour of the OI prosthesis.
Model design based on fixed state
Due to the absence of long-term trial data, seven studies extrapolated data to extend the time horizon of the evaluation.12,14,15,42,46,47 By way of example, consider a HEE modelling study that compared OI and S prosthesis, 46 where the benefits 53 and transition probabilities 50 were based on input data collected over 2 years and extrapolated over a 20-year time horizon assuming a fixed state. The fixed-state assumption is problematic since a proportion of prosthesis users would normally transition to another state of health in future years, as is evidenced by the 10-year cumulative risk of osteomyelitis (20%) and associated implant removal (9%). 63 Without a more sophisticated model that acknowledges people continue to transition between health states, there is no opportunity to accurately reflect the risk of complications, or their associated costs and impact on the benefit over time. The fixed-state assumption used in OI modelling studies14,46 overstates the benefits and understates the costs of the OI prosthesis condition.
Managing uncertainty
Three studies conducted appropriate sensitivity analyses to explore uncertainty, such as the impact of varying time, cost or benefit inputs, on the cost-effectiveness results.44,46,48 One of these studies conducted exemplary sensitivity analysis that showed the impact of time on the cost per QALY gained for the OI prosthesis compared to the S prosthesis with a 20-year time horizon (€83,374) compared to shorter time horizons of 15 years (€98,519), 5 years (€243,322) and 1 year (€2,578,563). 46 The large differences in cost per QALY over time are meaningful for policy-makers seeking to inform decisions for groups of people, whereby age influences the number of years over which the benefit may be obtained.
Quality of reporting
A number of reporting issues were identified in this review including limited details about the competing interventions, study settings and the quality of the model input data (Table 4).
Eleven studies did not provide sufficient details about the competing interventions,12 –16,42–46,48 often only reporting the prosthetic component that was the focus of the HEE (e.g. MPC prosthetic knee). Without a complete description of all component parts of the prosthesis, it is difficult for readers and policy-makers to determine the exact intervention that led to the costs and benefits observed. 28 In turn, this makes it challenging for readers to judge the applicability of the results to their policy or practice settings. 28
Eight studies did not sufficiently describe the study setting12,14,43,44,46 –49 and the influence this may have had on cost and benefit data. For example, in a study comparing the costs and benefits of MPC and nMPC prosthetic knees, the out-of-pocket cost covered by the patient (i.e. a 20% patient contribution for prosthetic costs) were not included. 42 While these results might inform policy decisions from the payer perspective in countries with similar payer–patient co-payment, the results are not generalisable to settings where payers cover the entire cost of the prosthesis, or where the patient contribution is substantially higher. Hence, it is important that authors describe specific features of the setting so that readers can make informed decisions about the applicability of the results to their healthcare setting.
In the three modelling studies,14,42,46 there were limited details about the quality of the model input data and, as such, issues that introduced bias were not transparent to readers (Supplemental Material: Additional file 1, Tab 2). For example, a modelling study 14 required readers to review three additional studies45,51,55 to establish that cost data for the OI prosthesis were based on patient estimates of the number of visits and suffered from recall bias. 45 While this issue affecting the cost calculation was well described in the original research, 45 this limitation was not transparent in subsequent modelling studies that utilised these data.14,46
Discussion
HEEs of prosthetic interventions and services are in their infancy. Early prosthetic HEEs used a CCA approach as part of small trials.43,49 As the literature became more established, prosthetic HEEs used CEA and CUA approaches, such as those evaluating MPC prosthetic knee units.12,15,16 In recent times, the first model-designed prosthetic HEEs emerged;14,42,46 however, these were limited by the availability of high-quality and rigorously conducted experiments that are typically used to provide input data into HEE models. Given the short time frame over which these studies have been produced, there has been limited opportunity to build on the work of others, because much of this work has been done by different groups, in parallel.
At this juncture, there are opportunities to build on the work of others, by first identifying evidence gaps, critical method design and reporting issues, and determine the extent to which the literature informs a wide range of policy and investment decisions.
Evidence gaps
There are significant evidence gaps given that HEEs have focused on just a few discrete areas of new and high-cost prosthetic interventions (e.g. OI prostheses) compared to the standard of care (e.g. S prostheses).
It is interesting to consider that there are similarly new and high-cost interventions that have not been subject to HEE, including upper limb prosthetic technologies such as myoelectric prostheses. Perhaps, these technologies have not been a focus of HEEs given that the prevalence of upper limb prosthesis users is small compared to transfemoral or transtibial prosthesis users. There are likely many comparatively low-cost interventions that may be highly cost-effective (e.g. silicone liners) that have also not been the subject of HEE.
The narrow focus of current prosthetic HEEs impacts the extent to which policy and investment decisions can be informed by evidence, something discussed in a later subsection.
Method design and reporting issues
Given the method design issues identified through this appraisal, the available HEE literature provides limited evidence to inform decisions about the cost-effectiveness of competing prosthetic interventions.
As detailed in the Results section, selection and allocation bias were common. More complex issues were also identified including: the choice of time horizon did not reflect the useful life of the intervention, measurement of costs and benefits that incorrectly valued or costed the interventions, models that did not include transitions to account for changes in health state over time and omission of sensitivity analyses to manage uncertainty.
Reporting issues were also identified that made it difficult to know which interventions were being compared or how the results might be generalised to other healthcare settings with different policies. Similarly, the quality of the model input data was often inadequately reported and, as such, issues that introduced bias were not made transparent.
Extent to which the literature informs policy and investment decisions
Most of the existing literature is focused on component parts of a prosthesis and therefore predominantly informs within- and across-intervention policy decisions. Within-intervention studies seek to establish the cost-effectiveness of different component parts of a prosthesis (e.g. MPC and nMPC prosthetic knees) for the same type of intervention (e.g. a transfemoral prosthesis), population (e.g. people with transfemoral amputation) and setting. By contrast, across-intervention studies seek to establish the cost-effectiveness of different types of interventions (e.g. OI vs S prosthesis management) for the same population (e.g. people with transfemoral amputation) and the same setting. Within- and across-intervention HEEs are of interest to third-party payers and those responsible for managing a finite budget for prosthetic services, given their responsibility for funding decisions affecting individual prosthesis users.
Within- and across-sector decisions are important to inform higher-level decisions, say by state or federal governments, about where healthcare resources should be directed. While there are a small number of studies that inform within- and across-sector decisions in the literature, there are opportunities to take a societal perspective and include a comprehensive range of costs in future research, which can better inform within- and across-sector decisions into the future. These types of HEEs are required to make an informed decision about the cost-effectiveness of prosthetic services compared to other competing healthcare demands, such as a community vaccination programme.
Recommendations
The findings of this review underpin recommendations for stakeholders and researchers in order to progress knowledge beyond the existing pool of early prosthetic HEEs.
Stakeholders: policy-makers, funding agencies and end-users
It will be important to engage policy-makers and funders in the design of future HEE research given that these stakeholders are best positioned to describe the types of evidence they need. With this engagement, we may see more HEEs that support within- and across-sector comparisons and can therefore inform federal healthcare policy and investment decisions.
There are also opportunities to engage end-users to understand their perspective regarding the benefit of a prosthetic intervention or service to ensure that the right benefits are being measured.
Researchers
The findings of this review point to changes that could improve the quality and rigour of future prosthetic HEEs. By way of example, future prosthetic HEE studies could:
Compare one type of intervention to one next best alternative appropriate to the outcome of interest (e.g. one specific MPC prosthetic knee vs one specific nMPC prosthetic knee) and in this way avoid using categories of heterogeneous interventions (e.g. nMPC prosthetic knees) as a comparator;
Where ethical, randomly assign participants to competing interventions to control for the influence of comorbid health conditions. Where randomisation is not ethical (e.g. randomisation to OI intervention), studies should detail participant demographics and outcomes likely to influence costs and benefits, and explore their effect through sensitivity analysis;
Choose time horizons that reflect the usable life of the competing interventions (e.g. the full warranty period of an MPC prosthetic knee) or capture costs and benefits over the period of time in which they are expected to differ;
Incorporate transition states into modelling studies to reflect the likelihood of complications (e.g. implant removal in OI prosthesis intervention) that may increase costs and reduce benefits.
We encourage researchers to use existing resources to support the design of rigorous HEEs including CHEC-Extended and CHEERS checklists.29,30
The intended audience for HEE research includes funding agencies and policy-makers across different healthcare settings. Researchers are encouraged to discuss issues affecting generalisability, to assist stakeholders to appropriately translate their research to different healthcare settings.
We acknowledge that many of the method design features that introduce bias are likely driven by the financial and time constraints of conducting HEEs. If we are to produce a rigorous evidence base in the years to come, there will need to be significant investment; particularly given the high-cost of many prosthetic interventions, and the lengthy time horizons over which research should be conducted.
Limitations
A number of scoping and systematic reviews have been designed to determine which prosthetic interventions are the most cost-effective.59,64-66 We felt it important to identify evidence gaps and critical method design and reporting issues to guide future research in this area.
We highlight that this review only reports on prosthetic HEEs despite the initial protocol 17 and search strategy designed to yield both prosthetic and orthotic studies. Our intent is to publish a companion systematic review focused on orthotic HEEs and, in doing so, reach a different target audience and provide a detailed appraisal of the same rigour for a body of literature with different HEE method design features.
As described in the Method section, we developed a decision aid to support consistent use of the CHEC-Extended. 19 We acknowledge that the decision aid has not undergone rigorous testing or review. We provide this decision aid as supplementary material (Supplemental Material: Additional file 2) so our work is transparent and available to others that might wish to appraise HEEs in prosthetics and orthotics into the future.
Conclusion
In line with the aim of this systematic review, significant evidence gaps in the prosthetic HEEs were identified, noting the focus on a few discrete new and high-cost prosthetic technologies. There are opportunities to undertake studies that support within- and across-sector decisions, and therefore help inform a broader range of investment and policy decisions.
A wide range of method design issues were identified, and the available HEE literature currently provides limited evidence to inform decisions about the cost-effectiveness of competing prosthetic interventions. We acknowledge the early research in this area as well as the opportunity to extend the current evidence base through the design of rigorous HEEs that meet contemporary standards as described in guidelines such as the CHEC-Extended. Investment will be required to support researchers to extend current knowledge through rigorous HEEs that provide sufficient evidence of the most cost-effective prosthetic interventions and services, therefore informing prosthetic funding and policy into the future.
Supplemental Material
10.1177_0309364620935310_Supplementary_Material_File1 – Supplemental material for A systematic review of health economic evaluations in orthotics and prosthetics: Part 1 – prosthetics
Supplemental material, 10.1177_0309364620935310_Supplementary_Material_File1 for A systematic review of health economic evaluations in orthotics and prosthetics: Part 1 – prosthetics by Leigh Clarke, Michael P Dillon and Alan Shiell in Prosthetics and Orthotics International
Supplemental Material
10.1177_0309364620935310_Supplementary_Material_File2 – Supplemental material for A systematic review of health economic evaluations in orthotics and prosthetics: Part 1 – prosthetics
Supplemental material, 10.1177_0309364620935310_Supplementary_Material_File2 for A systematic review of health economic evaluations in orthotics and prosthetics: Part 1 – prosthetics by Leigh Clarke, Michael P Dillon and Alan Shiell in Prosthetics and Orthotics International
Footnotes
Acknowledgements
The authors wish to thank the Australian Orthotic Prosthetic Association for supporting this work.
Author contributions
L.C. conceptualised the review. L.C. and M.D. designed and tested the search strategy. L.C. executed the search strategy and prepared the spreadsheets for vetting of articles against the inclusion criteria. L.C. and M.D. vetted the articles for inclusion. L.C. and M.D. appraised the articles, extracted the data and checked entries for accuracy. L.C., M.D. and A.S. each participated in the preparation of the results narrative. L.C. led the drafting of the manuscript with input from M.D. and A.S. All authors reviewed the manuscript for important intellectual content and provided critical review. The guarantor of the review is L.C. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

