Abstract
Background:
Diabetes mellitus is among the most common causes of peripheral neuropathy worldwide. Sensory impairment in diabetics is a major risk factor of plantar ulcers and neurogenic arthropathy (Charcot joints) causing severe morbidity and high health-care costs.
Objective:
To discuss the different patterns of sensory alterations in diabetic neuropathies and their anatomical basis.
Study design:
Literature review.
Methods:
Review of the literature discussing different patterns of sensory impairment in diabetic neuropathies.
Results:
The different varieties of diabetic neuropathies include typical sensorimotor polyneuropathy (lower extremity predominant, length-dependent, symmetric, sensorimotor polyneuropathy presumably related to chronic hyperglycemic exposure, and related metabolic events), entrapment mononeuropathies, radiculoplexus neuropathies related to immune inflammatory ischemic events, cranial neuropathies, and treatment-related neuropathies (e.g. insulin neuritis). None of these patterns are unique for diabetes, and they can occur in nondiabetics. Sensory alterations are different among these prototypic varieties and are vital in diagnosis, following course, treatment options, and follow-up of treatment effects.
Conclusions:
Diabetic neuropathies can involve any segment of peripheral nerves from nerve roots to the nerve endings giving different patterns of abnormal sensation. It is the involvement of small fibers that causes positive sensory symptoms like pain early during the course of disease, bringing subjects to physician’s care.
Clinical Relevance
This article emphasizes on the fact that diabetic neuropathies are not a single entity. They are rather different varieties of conditions with more or less separate pathophysiological mechanisms and anatomical localization. Clinicians should keep this in mind when assessing patients with diabetes on the first visit or follow-up.
Keywords
Background
The first notion that diabetes mellitus (DM) could involve the nervous system was made by Marchal de Calvi 1 in 1864, and from then, the salient clinical features of diabetic neuropathy were delineated. The loss of tendon reflexes in the legs was described by Bouchard, 2 and the occurrence of spontaneous pains and hyperesthesia with diabetes was first emphasized by Pavy. 3 With recognition of the highly variable nature of diabetic neuropathies, the first classifications were suggested by Leyden 4 and Pryce 5 in 1893.
DM is associated with a wide spectrum of neuropathy syndromes, ranging from a mild asymptomatic distal sensory neuropathy to a severe disabling radiculoplexus neuropathy. 6 A population survey reported that 30% of type 1 and 36%–40% of type 2 diabetic patients experienced neuropathic symptoms. 7 It should be noted that diabetes may not necessarily be the cause of the neuropathy in all diabetic patients. Indeed, other causes such as hereditary, inflammatory, and other metabolic neuropathies may coexist and should be actively excluded. 8 In this article, we shed more light on the different patterns of sensory alterations in diabetic neuropathies and their anatomical correlation.
Altered sensations (loss or increased sensory phenomena) are usually early and prominent manifestations of varieties of polyneuropathy associated with diabetes. 9 In questioning patients about abnormal sensation, it is necessary to make the distinction between sensation loss (negative neuropathic sensory symptoms) of “numbness” and increased sensory phenomena (positive neuropathic sensory symptoms (PNSS)) of “prickling” or varieties of pain. 10 Unlike inherited neuropathies, diabetic neuropathies are more often associated with PNSS. Nonetheless, clinicians should be aware that pain symptoms can improve and completely resolve while progression of neuropathy continues; diminution of pain can mean worsening of sensory function, as more pain receptors, free nerve endings, die off. 11
Advanced diabetic sensorimotor polyneuropathy (DSPN) is associated with elevated vibration and thermal perception thresholds that progress to sensory loss, occurring in conjunction with degeneration of all fiber types in the peripheral nerve. 12 A proportion of patients with DSPN also describe abnormal sensations such as paresthesias, allodynia (pain due to a stimulus that does not normally cause pain, for example, stroking), hyperesthesia (increased sensitivity to touch), hyperalgesia (increased sensitivity to painful stimuli), and spontaneous pain that sometimes seriously affects their quality of life. 13 Allodynia and hyperalgesia start to happen when the disease starts to extend from the smaller unmyelinated pain transmitting nerve fibers (type C fibers) to the larger myelinated fibers (Αβ and Αγ fibers) that transmit touch pressure and vibration senses. 14
Early recognition and appropriate management of neuropathy in the patient with diabetes is important for a number of reasons, including the fact that up to 50% of DSPN may be asymptomatic, and patients are at risk of insensate injury to their feet. As 80% of amputations follow a foot ulcer or injury, early recognition of at-risk individuals, provision of education, and appropriate foot care may result in a reduced incidence of ulceration and consequently amputation. 15 Therefore, periodic assessment of the sensation of feet of patients with diabetes with a 10-g monofilament is strongly recommended to be performed by health-care professionals. 9
Classification of diabetic neuropathies
Because of the wide range of clinical presentations, many different classifications of diabetic neuropathies have been proposed over the years. 6 The authors accept the Boulton et al. separation of diabetic neuropathies into generalized polyneuropathies, focal neuropathies (e.g. CrIII neuropathy and median neuropathy at the wrist (MNW) from carpal tunnel syndrome), and multifocal varieties (e.g. multiple mononeuropathies, lumbosacral, thoracic, and cervical radiculoplexus neuropathies).16 –18 The underlying mechanisms of these major neuropathy syndromes are not well understood. Some of them appear to relate to metabolic derangement within the peripheral nerve, others to altered immunity and nerve ischemia (the radiculoplexus neuropathies), and others with entrapment and compression, and in these, it is unclear whether it is due to excessive compression or altered response to compression. It is also still unclear how excessive and rapid institution of insulin therapy can cause insulin neuritis.
Although none of these varieties are uniquely associated with DM, all varieties are more prevalent in diabetes. Underlying mechanisms are thought to be different among these varieties. 9 In typical DSPN, pathological degeneration of receptors and nerve fibers begins symmetrically and distally and spreads proximally. 19 In compression and entrapment, nerve-fiber degeneration begins and is maximal at the site of compression or entrapment. In the radiculoplexus neuropathies, nerve-fiber degeneration is multifocal with involvement of nerve roots, spinal ganglia, plexuses, and peripheral nerves. 20
The three main patterns of sensation loss mirror the fiber class vulnerability in peripheral nerve disease: selective involvement of large sensory fiber function of touch pressure and vibration (e.g. as found in pseudotabes diabetica), selective involvement of small sensory and often also of autonomic nerve fibers (atypical small-fiber diabetic peripheral neuropathy (DPN)), and involvement of both large and small fibers (e.g. typical DSPN). 21
DSPN
Diabetic distal symmetric sensorimotor polyneuropathy, typically referred to as DSPN, affects at least 50% of diabetic patients in the United States, 22 probably being the most common form of the diabetic neuropathies. 6 It is seen in both type 1 and type 2 DM with similar frequency, and may be already present at the time of diagnosis of type 2 DM. 23 Staged diagnostic criteria for DSPN are illustrated in Table 1.
Staged diagnostic criteria for DSPN. 18

DSPN: diabetic sensorimotor polyneuropathy; NC: nerve conduction.
Sensory symptoms are more prominent than motor symptoms and usually involve the lower extremities; these include pain, paresthesias, hyperesthesias, deep aching, burning, or sharp stabbing sensation. 24 In addition, patients may experience negative symptoms such as numbness in the feet and legs, leading in time to painless foot ulcers and subsequent amputations if the neuropathy is not promptly recognized and treated. Unsteadiness is also frequently seen, owing to abnormal proprioception and muscle sensory function. 25 Alternatively, some patients may be completely asymptomatic, and signs may be only discovered by a detailed neurologic examination. 24 On physical examination, a symmetric stocking-like distribution of sensory abnormalities in both lower extremities is usually seen. Hands may be involved in more severe cases. All sensory modalities can be affected, particularly vibration, touch, and position perceptions (large Αα, Aβ fiber damage), and pain, with abnormal heat and cold temperature perception (unmyelinated C fibers and small thinly myelinated Αδ fibers damage, respectively). 14
Pain associated with a peripheral nerve injury has several distinct clinical characteristics. Some describe bees stinging through the socks, whereas others talk of walking on hot coals. The pain, worse at night, keeps the patient awake and is associated with sleep deprivation. 26 Patients volunteer allodynia or pain from normal stimuli, such as the touch of bedclothes, and may have hyperesthesias or hyperalgesia. These symptoms may be paradoxic, with differences in sensation to one or other modality of stimulation. Unlike animal models of DSPN, the pain is spontaneous and does not need provocation. It has a glove-and-stocking distribution. Pain usually occurs at rest and improves with ambulation, in contrast to osteoarthritic pain, which is worsened with ambulation and decreased with rest. Pain may persist over several years, causing considerable disability and impaired quality of life in some patients, whereas it remits partially or completely in others with further deterioration in small-fiber function. 24
Toronto classification of distal symmetric diabetic polyneuropathies 17
Possible DSPN. The presence of symptoms or signs of DSPN may include the following: symptoms—decreased sensation, PNSS (e.g. “asleep numbness” prickling or stabbing, burning or aching pain) predominantly in the toes, feet, or legs—or signs—symmetric decrease of distal sensation or unequivocally decreased or absent ankle reflexes.
Probable DSPN. The presence of a combination of symptoms and signs of neuropathy including any two or more of the following: neuropathic symptoms, decreased distal sensation, or unequivocally decreased or absent ankle reflexes.
Confirmed DSPN. The presence of an abnormality of nerve conduction (NC) and a symptom or symptoms, or a sign or signs, of neuropathy confirm DSPN. If NC is normal, a validated measure of small-fiber neuropathy (SFN) (class 1 evidence) may be used.
Subclinical DSPN. The presence of no signs or symptoms of neuropathy are confirmed with abnormal NC or a validated measure of SFN. Definitions 1, 2, or 3 can be used for clinical practice, and definitions 3 or 4 can be used for research studies.
SFN. SFN should be graded as follows: (1) possible: the presence of length-dependent symptoms and/or clinical signs of small-fiber damage; (2) probable: the presence of length-dependent symptoms, clinical signs of small-fiber damage, and normal sural NC; and (3) definite: the presence of length-dependent symptoms, clinical signs of small-fiber damage, normal sural NC, and altered intraepidermal nerve-fiber (IENF) density at the ankle and/or abnormal thermal thresholds at the foot on Quantitative Sensory Testing (QST).
The following findings should alert the physician to consider causes for distal sensory neuropathic symptoms other than diabetes and referral for a detailed neurologic workup: (1) pronounced asymmetry of the neurologic deficits; (2) predominant motor deficits, mononeuropathy, or cranial nerve involvement; (3) rapid development or progression of the neuropathic impairments; (4) progression of the neuropathy despite optimal glycemic control; (5) symptoms starting at the upper extremities; (6) family history of a nondiabetic neuropathy; and (7) a diagnosis of DSPN that cannot be ascertained by clinical examination. 27
The last position statement of the American Diabetes Association recommends that all patients with diabetes be screened for diabetic neuropathies at diagnosis in type 2 DM and 5 years after diagnosis in type 1 DM. Diabetic neuropathy screening should be repeated annually and must include sensory examination of the feet and ankle reflexes. 15 One or more of the following can be used to assess sensory function: pinprick (using safety pins, Wartenberg wheel, or similar instrument), temperature, vibration perception (using 128–256 Hz tuning fork), or 1-g and 10-g monofilament pressure perception at the distal halluces. Combinations of more than one test have more than 87% sensitivity in detecting DSPN.15,28 Longitudinal studies have shown that these simple tests are good predictors of risk for foot ulcer. 29 Numerous composite scores to evaluate clinical signs of diabetic neuropathies, such as the neurologic impairment score (NIS), are currently available. In combination with symptom scores, these are useful in documenting and monitoring neuropathic patients in the clinic. 30 The feet should always be examined in detail to detect ulcers, calluses, and deformities, and footwear must be inspected at every visit. 24
Unfortunately, clinical assessment of decreased sensation by physicians is generally inadequate because it is not performed or is performed badly. 9 Even expert clinical physicians, specialists in neuromuscular diseases, without pertaining or consensus development, and although showing good test–retest reproducibility, markedly over-reported abnormality of neurological signs (including assessment of sensation) in a masked cohort study of patients without and with DSPN. 31 When the study was repeated, asking the same physicians to judge as abnormal only unequivocal abnormality, taking age, sex, and physical fitness into account, proficiency improved remarkably. 32 Therefore, physicians should use these more specific approaches in their clinical assessment, and they should be used in the conduct of therapeutic trials. 9 In a series of trials of a large cohort of diabetic patients, investigators in Toronto, Ontario, Canada assessed the utility of using the 10-g monofilament test for detection of loss of foot sensation and of DSPN. They concluded that although there were limitations of its specificity, a simple threshold of 5 of 8 was predictive of DSPN. 33
However, screening of sensation with the 10-g monofilament is not an adequate assessment of the kind, severity, or distribution of sensation loss. It is thought that computer-assisted (smart) QST may meet this need. 9 In a recent proficiency trial of technologists from three medical centers, it was shown that these smart QSTs provided “accurate assessment of sensation loss without intra- or inter-test differences; therefore useful for multicenter therapeutic trials.” 34 A recent consensus panel, sponsored by the International Association for the Study of Pain (NeuPSIG), confirmed the utility of QST for assessment and monitoring of somatosensory deficits and for other indications. 35
In testing for and estimating the body surface distribution of sensation loss in a condition like DSPN, the knowledge that the disease process is symmetrical and length dependent is used to limit the amount of testing needed to a minimum. Thus, of the 20 selected anatomical sites that are used to represent the body surface area, it may be possible to estimate sensation loss from the evaluation of as few as four sites and only a slightly larger number of sites in more severe sensory loss. A similar approach to that for the entire body can be used to estimate sensation loss of an extremity (e.g. in diabetic lumbosacral radiculoplexus neuropathy (DLRPN) (Bruns–Garland syndrome)). 9
Impaired glucose tolerance
This condition is applied to persons having either fasting blood glucose of 100–125 mg/dL or, preferably, a 2-h oral glucose tolerance test of 140–199 mg/dL. 36 Impaired glucose tolerance (IGT) was found at a rate three times higher in patients with otherwise idiopathic sensory neuropathy than in age-matched general population. The painful sensory neuropathy common to these patients is indistinguishable from sensory symptoms typically encountered in early DSPN. 37 Most patients with IGT and neuropathy have a length-dependent distal small-fiber sensory neuropathy, often with prominent neuropathic pain. 38 Skin biopsy with IENF density measurement as a morphometric quantification of small sensory fibers has recently been advocated as a reliable and efficient technique to confirm the clinical diagnosis of SFN with a level A recommendation. 39
Sumner et al. found that patients with IGT have less severe neuropathy than diabetic subjects as demonstrated by a significant difference in the distal leg IENF density, sural NC velocity and sensory action potential amplitude, and superficial peroneal sensory amplitude between the two groups. Furthermore, patients with IGT more often had neuropathy restricted to small nerve fibers compared to patients with diabetes who had more involvement of both large and small nerve fibers. Based on these results, they suggested that a dose–response relationship exists between the severity of glucose dysregulation and the severity of neuropathy. 40 These results also supported the earlier notion that small nerve fibers are damaged early in the course of glycemic dysmetabolism. 41 Skin punch biopsy studies for IENF density determination suggested that diet therapy, counseling, and weight loss can induce some reinnervation of epidermal nerve branches, correlating with improved acral discomfort. 42
DLRPN
Most authors attribute the original description of DLRPN to an article written by Bruns 43 in 1890. The syndrome was largely forgotten until Garland described it again in 1953, first calling it “diabetic myelopathy” 44 and then “diabetic amyotrophy,” 45 because of uncertainty about the pathoanatomical localization. Hence, it is called the Bruns–Garland syndrome. 46 It is now clear that the disease process involves root, lumbosacral plexus, and peripheral nerve (i.e. a lumbosacral radiculoplexus neuropathy). Frequently, there are associated thoracic radiculopathies. 47
Typically DLRPN develops in patients with type II DM in middle or old age. In general, patients had not been diabetic for a long period of time, and the glycemic dysregulation was not severe. Accordingly, patients with DLRPN usually do not have many of the long-term complications of DM, such as coexisting retinopathy or nephropathy, which are frequently seen in DSPN. 48 Frequently, there is a large, concomitant weight loss. 49 The condition begins abruptly and focally, and patients often recall the exact day their symptoms began, whereas DSPN begins insidiously and symmetrically. A nondiabetic analogue of DLRPN exists (LRPN) and is almost indistinguishable clinically and pathologically from DLRPN. 50
The condition usually starts with pain that may be described as sharp or lancinating, deep aching, burning, or contact allodynia. The disorder begins asymmetrically (usually unilaterally) and focally, involving the hip and thigh somewhat more frequently than the foot and leg. However, it quickly spreads to involve the initially unaffected segments and the contralateral side so that by the time of evaluation, the disorder usually appears more widespread and symmetric than when it first began. 47 Most of the patients had sensory loss in both proximal and distal segments, and all classes of sensory fibers were affected.20,49,51
Although pain is the most prominent early symptom, weakness soon becomes the major issue. Patients often complain of both proximal weakness (difficulty in going upstairs or getting off the toilet, or one of their legs giving way beneath them) and distal weakness (foot drop and tripping). The weakness is severe, and about half of patients might become wheelchair-bound at some point during their illness. Almost everybody requires aids for ambulation. 47 In addition to pain and weakness, about half of DLRPN patients develop new autonomic symptoms with their illness, which could include orthostatic intolerance, change in sexual function, and change in bowel and bladder function. 49
QSTs show that there are unequivocal patchy and often ill-defined sensory abnormalities in the foot, leg, and thigh and that all fiber classes are involved. 50 The electrophysiologic abnormalities tend to be much more widespread than the clinical deficits. There are marked reductions in amplitude of the compound muscle action potentials and sensory nerve action potentials, with only mild slowing of NC velocities. Needle electromyography (EMG) shows fibrillation potentials, decreased recruitment, and long-duration, high-amplitude motor unit potentials in muscles innervated by multiple nerve roots and different peripheral nerves. Paraspinal muscles EMG often shows fibrillation potentials as evidence of proximal denervation.47,50 From the pathologic standpoint, we believe that DLRPN and LRPN most likely are forms of nonsystemic vasculitis of nerve—microvasculitis. 47 That entails epineurial perivascular mononuclear inflammation; vessel wall inflammation, ischemia and previous bleeding (Figure 1) that were not found in DSPN or in healthy control nerves. These pathologic alterations involve small arterioles, venules, or capillaries (i.e. a microscopic vasculitis). 49
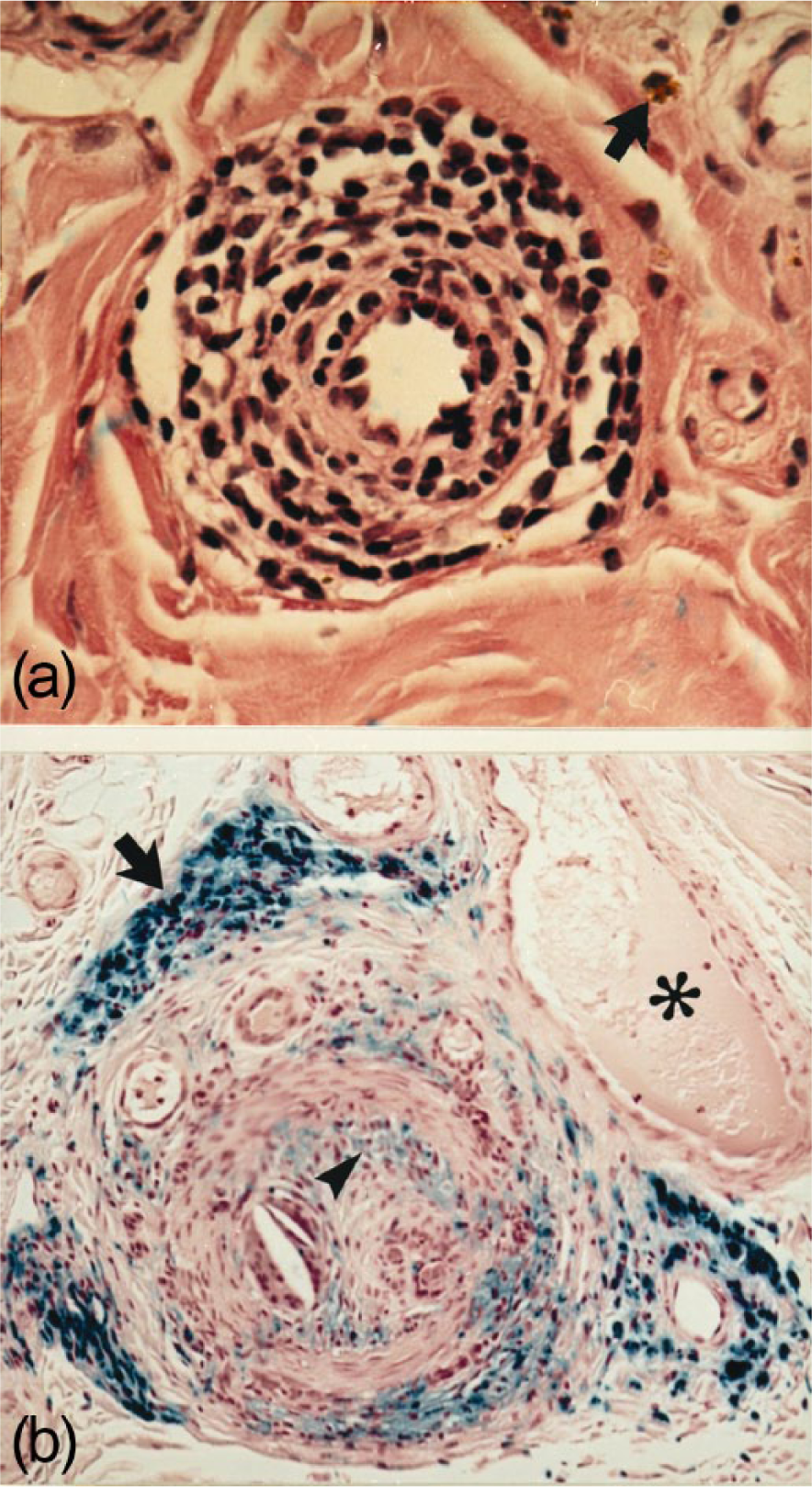
Transverse paraffin sections of sural nerves from patients with diabetic lumbosacral radiculoplexus neuropathy (DLRPN) showing epineurial microscopic vasculitis. (a) (hematoxylin–eosin) mononuclear cell infiltration and separation of smooth muscle of a small epineurial arteriole. The arrow shows a deposit of hemosiderin. (b) (turnbull blue) an epineurial arteriole with intimal thickening, adventitial scarring, recanalization, and perivascular hemosiderin deposition. The asterisk shows fresh blood (pink), whereas the arrow shows previous bleeding (hemosiderin) that stained blue. The arrowhead shows intimal proliferation. 49
Diabetic thoracic radiculopathy
Diabetic thoracic polyradiculopathy (DTP) can be an unusual cause of severe, chronic abdominal pain. Since afflicted patients seek care for abdominal pain, gastroenterologists and surgeons encounter these subjects first. 52 In the population-based, prospective Rochester Diabetic Neuropathy Study (RDNS), out of the 105 patients with diabetic polyradiculopathy, 14 patients had flank or abdominal involvement. 48 In these patients, other diabetic complications, including retinopathy and other forms of neuropathy are common, but they may not be present. 47
Pain may occur in the upper or lower abdomen and is sometimes bilateral. It can begin suddenly or gradually and is usually severe. Paresthesia alone is unusual. 53 Many patients complain of abdominal protrusion, usually unilateral, due to motor denervation of the abdominal wall muscles. 54 As may occur with lumbosacral polyradiculopathy, weight loss is common. 55
Physical examination may reveal unilateral abdominal wall paresis. This finding may only be evident with the patient standing or contracting the abdominal muscles by raising the head or lower extremities off the examining table. After palpation reveals no abdominal mass, cutaneous sensory examination should be the next step. This procedure usually reveals hyposthesia or hyperesthesia in multiple adjacent dermatomes. Identification of the affected dermatomes localizes the diseased spinal nerve roots.
Abdominal imaging procedures, such as computerized tomography, are usually performed first to exclude malignancy. Paraspinal EMG confirms the diagnosis by displaying abnormalities corresponding to the involved thoracic nerve roots. 52 Some authors have diagnosed the disorder with EMG of anterior abdominal wall muscles, 56 thermoregulatory sweat testing, 57 or skin biopsy. 58 Symptomatic therapy remains the mainstay of treatment. Eventually, the pain subsides, but its duration can exceed 2 years and can be disabling. 52
Focal extremity neuropathies (entrapment neuropathies)
Compression and entrapment neuropathies were recognized relatively late as a form of diabetic neuropathies. 59 These include MNW, carpal tunnel syndrome, ulnar neuropathy at the elbow (UNE), and peroneal neuropathy at the fibular head. Radial, obturator, and lateral cutaneous nerves of the thigh have rarely been described in diabetic patients. 6 Electrophysiologic diagnosis of median nerve entrapment in diabetics may be challenging because of effects attributable to coexistent polyneuropathy. Treatment and operative decisions concerning median nerve compression in diabetes are the same as those for the general population. Diabetic mononeuropathy at non-entrapment appendicular sites is unusual. 36
Based on 19 reports composed of 40–1215 patients, the number of patients with MNW who had coexisting DM ranging from 2.6% to 20%. If the data from all these studies are combined, then of 6647 patients with MNW, 467 (7%) had DM.6,60 –78 Among eight series of patients with DM composed of 64–995 patients, 1.2%–20.8% had MNW.19,50,79 –83 Then if the data are pooled, of 2652 patients with DM, 153 (5.8%) had MNW. 84 The definition of MNW varies and most of these studies are not population-based epidemiologic studies although. 6 In the RDNS cohort, electrophysiologic evidence of MNW without symptoms was found in 22% of type 1 DM patients and in 29% of type 2 DM patients. Clinical evidence for MNW was found in 9% of type 1 and 4% of type 2 DM patients with stage 2a (mild symptomatic) neuropathy and in 2% of type 1 and 2% of type 2 DM patients with stage 2b (problematic) neuropathy. 48
The causes for mononeuropathies in DM are probably multifactorial. Obesity is a predisposing factor for MNW.61,64,65,85,86 Some have suggested that fat accumulation in the carpal tunnel could result in compression of the median nerve. 65 Axonal transport was reported to be reduced in DM. 87 Another possible explanation focuses on the local environment surrounding nerves. Findings such as Dupuytren disease, palmar flexor tenosynovitis, and limited joint mobility are often detected in hands of DM patients and may contribute to MNW. 88
UNE was first attributed to DM in 1945. 89 In four DM series that included 64–490 patients, the percentage of DM patients with UNE ranged from 1.2% to 4.9%. When the data are pooled, 16 DM patients from 756 total (2.1%) had UNE.6,90 –93 Again (as for MNW), epidemiologic problems exist with these data. Based on the RDNS study, symptomatic UNE was found in 2% of type 1 and 2% of type 2 DM patients. 32 Diabetic UNE and MNW should always be differentiated from the more extensive cervical radiculoplexus neuropathy. 6
Isolated cranial neuropathies
It is commonly believed that the incidence of cranial neuropathies is higher in patients with DM when compared with the nondiabetic population. However, the incidence of cranial neuropathies in patients with DM is not known due to the lack of population-based incidence studies. 6 In the RDNS cohort, no patient presented with cranial neuropathies. 32 In a retrospective study of 4278 cases of paralysis of the oculomotor, trochlear, or abducens nerve, only 2%–3% of cases were patients with DM. 94
It is unclear whether other cranial nerves, with the possible exception of the seventh, are ever affected in isolation. Onset is usually abrupt; oculomotor neuropathy is often painful, but abducens palsy may be painless. Both nerves can be affected concomitantly. Orbital discomfort, sometimes severe, often precedes oculomotor palsy by several days. 36
Following the cranial nerves controlling external eye movements, comes the trigeminal (ophthalmic and maxillary), in the frequency of cranial nerves involvement in DM. 95 Arap et al. found abnormal pain thresholds on performing facial QST, in the maxillary branch territory. It could be an evidence of the initial sensory compromise of trigeminal small fibers. They also found a high prevalence of orofacial pain (55.2%), and burning mouth was the most common complaint (17.2%) of their 29 diabetic subjects with orofacial pain, which could indicate orofacial diabetic neuropathy. 96 However, on orofacial evaluation, other etiologies that were observed include masticatory myofascial pain and periodontal disease, which themselves could cause the burning sensation.96 –98 The absence of correlation of orofacial pain with hyperglycemia or glycated hemoglobin supported other etiologies rather than painful diabetic cranial neuropathy. 96
Acute painful diabetic neuropathy
Acute painful neuropathy with weight loss is considered by some authors to be a clinical entity separate from DSPN. 16 This syndrome, also known as diabetic cachexia, is not associated with duration or severity of DM and usually has a monophasic course over months. Severity of pain is not related to the severity of objective neurologic impairment. The illness begins with precipitous and profound loss of weight, followed by severe, unremitting, burning pain and allodynia distally in the lower limbs, most troublesome at night. The association of pain, weight loss, and a monophasic course is more reminiscent of DLRPN. This syndrome could be viewed as one end of the spectrum of DLRPN, with involvement of pain fibers in the lumbosacral dermatomes, without involvement of motor fibers. No specific treatment has been suggested or advocated for this condition. 6 Restoration of glycemic control and weight gain are associated with diminished pain and gradual recovery over many months. 36
Insulin neuritis
The development of an acute painful sensory neuropathy after initiating insulin therapy was first described by Carvati 99 in 1933. Patients develop distal sensory symptoms, mainly in the lower limbs on commencing insulin for the first time. 81 The term insulin neuritis describes the condition of neuropathic symptoms developing soon after institution of insulin therapy or also oral hypoglycemic agents. It was reported to be a reversible disorder characterized by severe pain, autonomic dysfunction, and unmyelinated nerve-fiber damage after rapid and sustained glucose regulation in individuals with historically poor glycemic control. 100 There may be mild stocking–glove sensory loss and allodynia, or no findings on either physical examination or electrodiagnostic tests. Pain gradually dissipates within a year of onset. 36 The distribution of sensory alteration is poorly characterized due to lack of studies implementing QST in this condition. The cause is not known. It may be due to too tight glycemic control causing prolonged hypoglycemia, too rapid shift in the glucose level, or a response induced by the insulin itself. 82
Conclusion
Of all the clinical manifestations of diabetic neuropathies, sensory alteration is the most important. This applies also to primarily motor syndromes like DLRPN which starts with severe neuropathic pain. Moreover, diabetic neuropathies can involve any segment of the peripheral nerves from nerve roots to the smallest nerve fibers giving different patterns of abnormal sensation. It is the involvement of the latter that causes positive sensory symptoms like pain early during the course of disease, bringing subjects to physician’s care. Thus, diabetic neuropathies differ from other categories of neuropathies that present with slowly progressive negative sensory symptoms, like hereditary polyneuropathies. Indeed, we want to emphasize on the diverse nature of the multiple diabetic neuropathy syndromes and how they differ in clinical context, quality and distribution of symptoms and signs, pathophysiology, and hence treatment options.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
Mohamed Kazamel has no disclosures or conflict of interest. Peter J Dyck has received financial support from National Institutes of Health (NIH) and Food and Drug Administration (FDA) grants in the recent past, receives an honorarium as co-editor of journal Diabetes, and his laboratory receives financial support for conduct of research from Alnylam, Inc.; ISIS, Inc.; Pfizer Japan, Inc.; and within the last 5 years from other pharmaceutical houses.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
