Abstract
Background:
Residual limb volume may change after doffing, affecting the limb shape measured and used as a starting point for socket design.
Objectives:
The purpose of this study was to compare residual limb fluid volume changes after doffing for different test configurations.
Study Design:
The study was a repeated measures experimental design with three conditions (
Methods:
Residual limb fluid volume on 30 people with trans-tibial amputation was measured using bioimpedance analysis. Three tests were conducted –
Results:
The percentage fluid volume increase after doffing was significantly higher for
Conclusions:
Activity before doffing intensified the post-doffing limb fluid volume increase. Maintaining a liner after doffing caused limb fluid volume to stabilize faster than removing the liner.
Clinical relevance
To minimize residual limb volume increase before casting or imaging, practitioners should have patients sit with their prosthesis donned for 10 minutes. Leaving a liner on the residuum will not reduce the post-doffing volume increase, but it will help to more quickly achieve a consistent limb fluid volume.
Background
A patient’s residual limb will typically swell after the prosthesis is doffed. Without the socket to constrain the soft tissues, interstitial fluid pressure within the residual limb may decrease, causing an increase in extracellular fluid transport from the arterial vasculature into the interstitium, and a decrease in transport from the interstitium into the venous vasculature. Both mechanisms may contribute to an increase in residual limb volume.
Post-doffing limb volume changes may have important clinical manifestations. They can clinically impact patient care by impacting the residual limb shape captured for socket design. A practitioner preparing to make a prosthetic socket may measure a different shape if the residual limb is imaged or casted right after the prosthesis is doffed compared with later, for example 10 minutes after doffing. On a group of six trans-tibial amputee subjects who had their limb amputation as a result of traumatic injury, Zachariah et al. 1 recorded a median limb volume change of 4.9% at 10 minutes after doffing, compared with 1.0% at 15–30 seconds after doffing.
In the present study we investigated if sitting quietly right before doffing compared with walking beforehand decreased post-doffing fluid volume change, and if so how much. Further, we investigated if post-doff fluid volume change could be reduced by leaving the liner on the residual limb. Substantial differences for different conditions might suggest guidelines for clinical practice.
Methods
Volunteers were considered for inclusion if they had a trans-tibial amputation more than 12 months prior and were at a Medicare Functional Classification Level (MFCL) of K-2 of higher (at least a limited community-level ambulatory). 2 Subjects were required to use a prosthetic limb for an average of at least 4 hours per day, determined by self-report, and capable of treadmill walking at a self-selected walking speed for at least 5 minutes. Subjects were not included if they were currently experiencing skin breakdown or if their residual limb length did not allow at least a 5.5 cm distance between the voltage-sensing electrodes (described below). Human subject approval from a University of Washington Internal Review Board was granted and informed consent was obtained before any study procedures were initiated.
Subjects were asked to refrain from consuming alcohol or caffeine on the day of testing. After arriving at the lab, the subject wore the prosthesis while mass and height were recorded. The subject sat for 10 minutes while the research practitioner queried the subject about smoking habits, presence of diabetes and prosthesis history.
We used a multi-frequency bioimpedance analyzer (Hydra 4200, Xitron, San Diego, CA) to assess residual limb extracellular fluid volume. Four electrodes (Xitron, 77x20 mm contact surface, 0.81 mm thickness) were used. A thin layer of ultrasonic coupling gel (Couplant D, GE Panametrics, West Chester, OH) was placed underside each electrode. We used TegadermTM (Transparent Film Dressing, 3M, St. Paul, MN) to strain relieve wires to the electrodes. We designed a custom, four-pin, Delrin, flat connector (9.0 mm x 11.5 mm, 2.5 mm thickness) that accommodated gold-plated pins (WPI, Viking Electronics, Inc. (division of Cooper Interconnect), Moorpark, CA) to attach the four insulated lead wires from the Xitron instrument cable to the electrodes. This enhancement ensured a stable and consistent signal was recorded while the subject walked on the treadmill.
The subject doffed the prosthesis, and we prepared the skin for the bioimpedance electrodes. The skin was rubbed gently with sandpaper (Red DotTM Trace Prep 2236, 3M, St. Paul, MN) to achieve good electrical coupling. 3 Two outer electrodes injected current while two inner electrodes sensed voltage (Figure 1). The proximal voltage-sensing electrode was positioned at the level of the patellar tendon proximal to the fibular head. The distal current injecting electrode was placed as far distally as possible but still on the curved cylindrical portion of the residual limb. The distal voltage-sensing electrode was positioned at least 3.5 cm proximal to the distal current-injecting electrode and always proximal to the distal end of the tibia. The proximal current-injecting electrode was placed at least 7.0 cm proximal of the proximal voltage-sensing electrode such that it was outside of the socket brim but under the liner or suspension sleeve. Care was taken to ensure no loss of suction from air escaping along the lead wires extending out at the thigh from under the liner or sleeve. The Xitron instrument applied current at between 50µA and 700µA across 50 frequencies (5k Hz to 1 MHz) each second, and measured amplitude and phase differences between the injected and sensed signals at a 1 Hz sampling rate. We plotted the bioimpedance data in approximately real time (3 s delay) using custom Matlab (Mathworks, Natick, MA) code that implemented a Cole model, 4 similar to that used in the Xitron post-processing program, 5 so that we could identify set up problems if they existed.
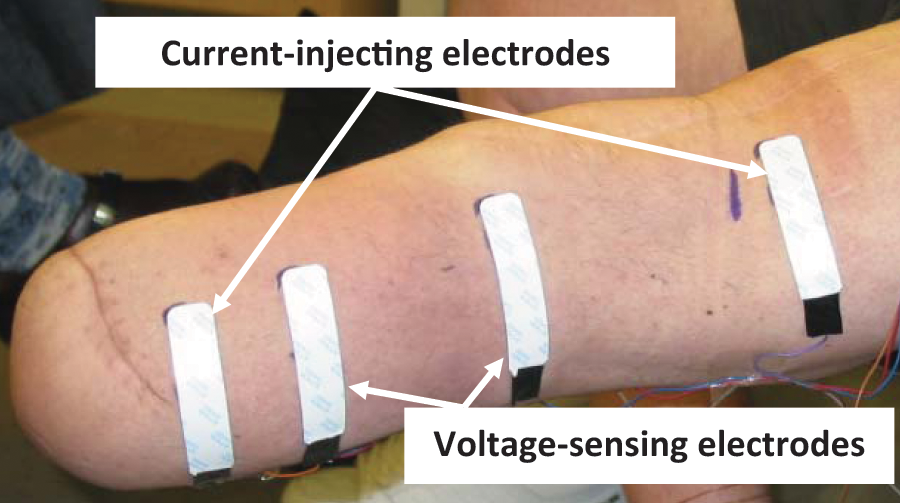
Bioimpedance measurement. Four electrodes were placed on the residual limb.
After the electrodes were applied and the instrument was started, the subject donned the prosthesis, standing and weight-bearing for several seconds until comfortable. Three different protocols were executed with no order preference except that the
Using standard medical testing procedures, orthostatic blood pressure (OBP) was measured on the day of the test, and on a different day ankle brachial index (ABI) and segmental limb pressure (SLP) were assessed.6–8 We used an electronic blood pressure measurement unit (HEM-775, Omron, Kyoto, Japan) to determine OBP. A commercial cuff inflator (TD312 Cuff Inflator, MV10 Manifold Selector, and SC12 and SC10 cuffs, Hokanson, Bellevue, WA) and a Doppler flow meter (MD6 Doppler, Hokanson) were implemented to evaluate ABI and SLP. Collected data were interpreted for presence of high blood pressure and arterial disease by a practising endocrinologist using standard clinical procedures.6–8 Subject health records were consulted to identify presence of a major medical condition (e.g. congestive heart failure, kidney failure, diabetes, cancer).
Body mass index (BMI) was calculated as the quotient of mass (kg) and the square of height (m2). 9 Because the subjects wore their prosthesis while we measured mass, no correction was made to BMI for the lack of an intact limb.
We processed the bioimpedance data using custom code that implemented a Cole model algorithm similar to the manufacturer’s (v.2.2, Xitron). We converted the data to extracellular fluid volume using limb circumference and segment length measurements in a well accepted geometric limb model. 10 Using the fluid volume measured immediately after doffing as a reference, we calculated the percentage fluid volume change over time during the 10-minute sitting period. We present percentage fluid volume change instead of fluid volume in mL because of the dependence of the results on limb length and size. Use of percentage fluid volume change normalized the data so that comparisons among subjects could be made.
Descriptive analyses (summary statistics and visual displays) were performed for all variables. The linear association between variables was assessed by Pearson correlation. Due to the exploratory nature of the study and the small sample size, the data analysis focused on exploratory and descriptive methods.
Results
A total of 22 males and eight females participated in this study. Their mean age was 50 years (sd 13). Twenty subjects had their limb amputation as a result of trauma, seven from vascular disease, one from Larsson’s syndrome, one from cancer, and one from spina bifida. All but three subjects had a unilateral amputation. Subject mass averaged 90 kg (sd 20), height averaged 177 cm (sd 10), and BMI averaged 28.7 kg/m2 (sd 5.9). Eleven subjects were obese (defined as BMI ≥ 30 kg/m2) and another six subjects were overweight (defined as BMI ≥ 25 kg/m2). Ten subjects were at a MFCL of K-2, 14 at K-3, and six at K-4. Nine subjects were diabetic, 20 were not, and one subject’s diabetic status was not known. Eight subjects were smokers, and 22 were not. Eighteen subjects used an elastomeric liner with pin suspension, while 12 used a different type of suspension.
Fourteen subjects had high blood pressure, 15 did not, and one subject’s status was not known. Twelve subjects had an ankle brachial index (ABI) and clinical history indicative of peripheral arterial complications,6,7 17 did not, and one subject’s status was not known.
All test sessions were started between 9:30 am and 2:00 pm. Tests were started during morning hours (before noon) for 16 subjects, and after noon for 14 subjects.
How much does residual limb fluid volume change after doffing, and how long does it take to stabilize?
Residual limb fluid volume change after doffing ranged from −1.1% to 8.3% with a mean of 1.9% (sd 1.6). The time to achieve peak fluid volume ranged from 36 seconds to 10.0 minutes with a mean of 6.0 minutes (sd 4.0).
The shape of the response curve depended on the test performed. In general, for all three tests (
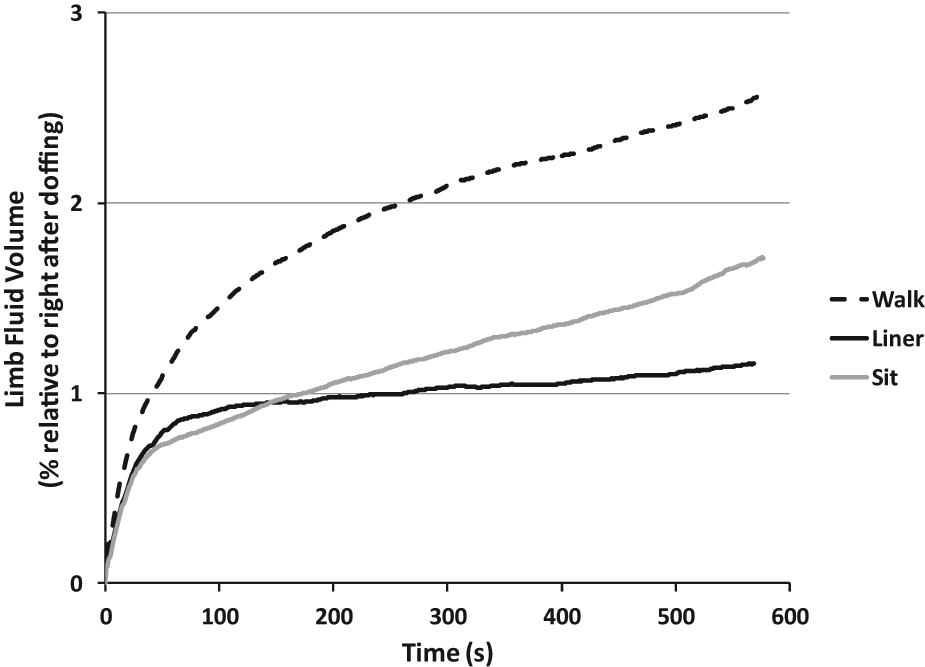
Residual limb fluid volume change over time after doffing. Average response curves for all subjects are shown for the three test protocols:
Is fluid volume change different after walking than after sitting?
We found that the fluid volume change after
Comparison of

Is the amount of limb fluid volume increase reduced when a liner is maintained after doffing compared with no liner?
The fluid volume change from the
Comparison of

We conducted an exploratory analysis to investigate if the magnitudes of limb fluid volume change and the time they were achieved were related to qualities of the subjects and prostheses. The following qualities showed no significant correlation (using a test statistic of 0.05): subject gender, BMI, presence of peripheral arterial complications, high blood pressure, diabetes or smoking. However, we noticed that there were common shapes in the response curves for different subjects. Thus we investigated if there were relationships between curve shapes and features of the subjects and prostheses.
We grouped the results according to the four curve shapes illustrated in Figure 3. Curve shapes #1 and #2 demonstrated a concave inflection point within the first two minutes after doffing while #3 and #4 did not.

Shapes of post-doffing response curves. Curve shapes #1 and #2 had convex inflection points soon after doffing. Curve shapes #3 and #4 lacked this characteristic.
Subjects tended to have the same type of curve for

Curve type results. The numbers of subjects with each combination of curve types are shown. Boxes with high values are highlighted. (A)
We observed that subjects with curve types #2 and #3 tended to have higher magnitude volume changes than subjects with curve types #1 and #4, whether for
Eighty-six per cent of subjects with type #3 and #4 curves for
Discussion
The residual limb fluid volume increase that occurs after doffing is relevant to clinical practice and advancing limb prosthetics. Post-doffing volume change can affect the shape of the cast or the image scan that is used as a starting shape for socket design. By understanding how much and over what time course a residual limb will increase in fluid volume upon doffing, we may be able to better factor the expected per cent increase into the socket modification to ensure a proper socket fit. It may be possible to develop best practices to facilitate socket design.
In this study, we measured residual limb extracellular fluid volume using bioimpedance analysis. Bioimpedance measures primarily through muscle and skin tissues, the primary tissues expected to undergo volume change in the residual limb over the short term. 1 We did not report intracellular fluid volume changes in the present study because of noise in the bioimpedance measurement at high frequencies. We did not have confidence in the quality of intracellular fluid volume data. We report limb fluid volume change as a percentage of conductive tissue volume instead of absolute fluid volume because of the need to normalize the data to account for different distances between voltage-sensing electrodes among subjects.
We placed voltage-sensing electrodes on the residual limb to measure the maximum length possible. However, this configuration did not allow us to measure from the very distal region beyond the tibia, thus conclusions drawn from this investigation do not necessarily apply to the most distal area of the residual limb. Results apply to the cylindrical portion where most of the weight bearing is tolerated.
In this investigation we did not control the time of day that the studies were conducted because of logistical challenges with subject schedules. However, time of day was not expected to be an important variable in the present study since all data were collected within a 1.25 hour timeframe, and all data were normalized to limb fluid volume right after the prosthesis was doffed.
The residual limb typically enlarges after doffing because of a change in interstitial fluid pressure. While the residual limb is within the socket, interstitial pressure is elevated because of the pressure applied by the socket wall on limb soft tissues. When the socket is doffed, however, this constraint is released and interstitial fluid pressure decreases. As a result, the arterial to interstitial pressure gradient increases, and more fluid enters the interstitial space. The interstitial to venous pressure gradient decreases, reducing fluid transport from the interstitium into the venous vasculature out of the limb. Further, the reduction in interstitial tissue pressure may cause vessels to enlarge and more blood to enter the residual limb. Thus both interstitial fluid and blood may contribute to the fluid volume increase after doffing.
Residual limb fluid volume increased more after walking than after sitting presumably because of the increased arterial fluid drive after exercise. The increased arterial drive caused a greater blood volume flow rate through the vasculature. Pressure-induced vasodilation has been described in the literature and may be at work here. 11 There was greater driving force to move fluid across from the arteries into the interstitial space and as a result limb fluid volume increased more after walking than after sitting.
Possibly this finding can be used as a clinical advantage. For subjects who experience limb volume loss over the day, a means to help increase volume back up to levels experienced earlier in the day may be to remove the prosthesis temporarily right after walking. Some practitioners recommend periodic doffing to their patients. If these doffing intervals were of appropriate duration and timed properly then they may help the subject keep limb volume within an acceptable range and negate the need for sock addition or other accommodation. Further, with new controllable socket adjustment technologies emerging in prosthetics,12–17 if sockets were designed to increase in volume during rest after activity then limb volume over the day may stabilize.
As the liner restricted the limb and kept interstitial pressure elevated (though not as elevated as with the socket donned), the subject leaving the liner on tended to stabilize limb fluid volume faster than the subject removing the liner. Interestingly, while keeping the liner on influenced the time to stabilization, it did not influence the volume at stabilization compared to without wearing the liner (
Subjects had different shaped post-doff curves because they have different fluid transport capabilities. It is unknown, however, to what degree socket design and health variables affected post-doff curve shapes. Unlike in a previous investigation where we observed a relationship between limb fluid volume change from sock addition/removal and presence of peripheral arterial complications, 18 in the present study no strong relationship between post-doffing limb fluid volume change and presence of peripheral arterial complications was found. We expect that this result reflects, in part, the influence of socket fit. Socket fit may have had as much or more influence as vascular health effects. A research area of possible relevance is to investigate relationships between post-doff limb volume change and variables that influence how well the socket supports the skeletal structures and enhances movement within the surrounding soft tissues.
We expect that the reason some subjects showed a limb fluid volume decrease upon doffing (group #4 curve shapes) was because of their co-morbidities. The initial decrease may have reflected the time needed to increase local arterial pressure high enough to open occluded arteries. This delayed vascular flow response has been discussed by Meijer et al. 19 who noted that thermal response curves showed a similar characteristic on bedridden subjects at high risk of decubitus ulcer formation.
We expect that the early part of the fluid volume response curve, a fast volume increase on most subjects, reflected primarily blood volume returning to the limb, while the later part, a slower volume change, reflected primarily interstitial fluid increase. The interaction and magnitudes of these two flow mechanisms may dictate the shape of the post-doff volume curve. We noticed that overweight and obese subjects tended to have type #3 and #4 curves; they lacked a concave inflection point after doffing but instead showed gradual changes in fluid volume change. This result may reflect complications associated with being overweight, for example a delayed vascular response. The lack of a locking pin might have accentuated this effect since a locking pin tends to pull distal limb tissues distally during swing phase and may accentuate interstitial fluid draw into the residual limb.
Part of why pursuing the shape of the post-doffing response curve is meaningful is that commercial imaging systems may be capable of collecting these data. If relevant relationships between socket fit and post-doffing volume results were found then those findings could be applied directly to clinical practice to help improve patient care.
Clinically, for the practitioner to capture a residual limb shape of volume closest to ambulatory limb volume, we recommend casting or scanning right after doffing. Notably, the residual limb will be increasing in volume. Fast scanning procedures, for example optical scanning,
20
will reduce measurement error. An alternative strategy is to wait for stabilization (see
Conclusion
The results from the present study suggest that practitioners preparing to cast or image a residual limb for socket design should have their patient sit quietly for 10 minutes before doffing. Patient history and fit of the current prosthesis can be discussed during this 10-minute period. In the present study, this quiet interval reduced fluid volume increase about 36% compared with walking before doffing. It also reduced continued limb expansion over the subsequent 10 minutes.
Residual limb fluid volume will stabilize faster if the liner is maintained rather than removed. A stable limb shape may be important when using imaging systems that take more than a few seconds to image the limb, for example laser scanners. Imaging systems that acquire residual limb shape quickly and right after doffing should be encouraged so as to reduce the detrimental impact of limb fluid volume change on limb shape measurements.
Research investigations are needed to establish best practices for factoring expected per cent limb volume increases after doffing into socket modification.
Footnotes
Funding
This work was supported by the National Institute of Child Health and Human Development at the National Institutes of Health [grant number R01HD060585].
