Abstract
Increased expression of pituitary tumor-transforming gene 1 (PTTG1) is expressed in many tumors and regulates tumor growth and progression. However, the precise function of PTTG1 in the tumorigenesis of lung adenocarcinoma (LAC) is not defined yet. Here, we examined the expression of PTTG1 in human LAC tissues by immunohistochemical assay using a tissue microarray procedure. A loss-of-function experiment was carried out to investigate the effects of lentiviral vector-mediated PTTG1 shRNA (shPTTG1) on cell growth and invasive potential in LAC cell lines (A549 and LETPα-2), assessed by MTT and Transwell assays. As a consequence, we found that the expression of PTTG1 protein was markedly upregulated in LAC tissues compared with the adjacent non-cancerous tissues (ANCT) (54.0% vs. 28.0%,
Lung cancer is the leading cause of cancer-related mortality with high incidence rates, metastatic propensity, and acquired resistance to therapy. 1 The 5-year overall survival rate of lung adenocarcinoma (LAC) is less than 15%, and has not been improved during recent decades. Metastasis is a main cause of morbidity and mortality in LAC, and surgical resection of LAC is companied by tumor recurrence at distant sites. 2 Thus, the molecular mechanisms of lung cancer metastasis remain unclear.
Many studies have shown that pituitary tumor-transforming gene (PTTG) is an oncogene that is overexpressed in variety of tumors, such as squamous cell carcinomas of the oral cavity, 3 gastric cancer, 4 craniopharyngiomas, 5 aggressive brain tumors, 6 pituitary adenomas, 7 and testicular cancer, 8 exhibits characteristics of a transforming gene 9 and contributes to tumorigenesis of thyroid cancer. 10 These studies suggest PTTG1 may be a potential prognosticator for malignant progression of multiple malignancies. Equally important, PTTG1 is involved in the invasive and metastatic diseases including aggressive clear cell renal cell carcinoma, 11 and is proved to be a functional transforming oncogene promoting tumorigenesis through the loss of p53 9 or facilitating colorectal cancer growth and metastasis. 12 Knockdown of PTTG1 increases drug (doxorubicin or paclitaxel)-induced senescence in cancer cells.13,14 These studies suggest PTTG1 is involved in the development of some cancers and predicts anti-tumor treatment outcomes.
However, few studies show that PTTG1 protein levels, downregulated in breast cancer, are negatively correlated with the tumor grade, and promotes tumor proliferation. 15 To clarify the function and molecular mechanisms of PTTG1 in LAC cells, we detected the expression of PTTG1 by immunohistochemical assay using a tissue microarray procedure and constructed Lv-shPTTG1 vector to observe the effects of PTTG1 knockdown on biological behaviors of LAC cells. We hypothesized that knockdown of PTTG1 might suppress growth and invasion of LAC cells through regulation of the TGFβ1/SMAD3 pathway.
Materials and methods
Materials
The LAC cell lines (A549 and LETPα-2) used for our experiments were obtained from the Institute of Biochemistry and Cell Biology (Shanghai, PR China). Lentivirus-mediated PTTG1 shRNA (Lv-shPTTG1), negative control vector, and virion-packaging elements were purchased from Genechem (Shanghai, PR China). LAC tissues and the corresponding ANCT were collected from Sichuan University. The tissue microarray of LAC was made by Shanghai Outdo Biotech Co. Ltd. (Shanghai, PR China). All the antibodies were purchased from Cell Signaling Technologies (Boston, MA, USA). PTTG1 primer was synthesized by ABI (Framingham, MA, USA).
Drugs and reagents
Dulbecco’s Modified Eagle medium (DMEM) and fetal bovine serum (FBS) were purchased from Thermo Fisher Scientific Inc. (Waltham, MA, USA); TRIzol Reagent and Lipofectamine 2000 were obtained from Invitrogen (Carlsbad, CA, USA); M-MLV Reverse Transcriptase was purchased from Promega (Madison, WI, USA); SYBR Green Master Mix was obtained from Takara (Otsu, Japan); and the ECL Plus Kit was obtained from GE Healthcare (Piscataway, NJ, USA).
Clinical data
Tissue microarray was prepared for IHC test. Human LAC tissues and the corresponding ANCT were obtained from biopsy in a total of 50 consecutive LAC cases admitted in our hospital from January 2011 to December 2013. The baseline characteristics of the patients before neo-adjuvant chemotherapy were summarized. The study was approved by Medical Ethics Committee of Sichuan University and written informed consent was obtained from the patients or their parents before sample collection. Two pathologists respectively reviewed all of the cases.
Tissue microarray
The advanced tissue arrayer (ATA-100, Chemicon International, Tamecula, CA, USA) was used to create holes in a recipient paraffin block and to acquire cylindrical core tissue biopsies with a diameter of 1 mm from the specific areas of the ‘donor’ block. The tissue core biopsies were transferred to the recipient paraffin block at defined array positions. The tissue microarrays contained tissue samples from 50 formalin-fixed paraffin-embedded cancer specimens with known diagnosis, and corresponding ANCT from these patients. The block was incubated in an oven at 45°C for 20 min to allow complete embedding of the grafted tissue cylinders in the paraffin of the recipient block, and then stored at 4°C until microtome sectioning.
Immunohistochemical staining
Tissue microarray sections were processed for IHC analysis of PTTG1 protein as follows. Immunohistochemical examinations were carried out on 3 mm thick sections. For anti-PTTG1 immunohistochemistry, unmasking was performed with 10 mM sodium citrate buffer, pH 6.0, at 90°C for 30 min. For anti-PTTG1 immunohistochemistry, antigen unmasking was not necessary. Sections were incubated in 0.03% hydrogen peroxide for 10 min at room temperature, to remove endogenous peroxidase activity, and then in blocking serum (0.04% bovine serum albumin, A2153, Sigma-Aldrich, Shanghai, PR China and 0.5% normal goat serum X0907, Dako Corporation, Carpinteria, CA, USA, in PBS) for 30 min at room temperature. Anti-PTTG1 antibody was used at a dilution of 1:200. The antibody was incubated overnight at 4°C. Sections were then washed three times for 5 min in PBS. Non-specific staining was blocked with 0.5% casein and 5% normal serum for 30 min at room temperature. Finally, staining was developed using diaminobenzidine substrate, and sections were counterstained with hematoxylin. Normal serum or PBS was used to replace anti-PTTG1 antibody in negative controls.
Quantification of protein expression
The expression of PTTG1 was semi-quantitatively estimated as the total immunostaining scores, which were calculated as the product of a proportion score and an intensity score. The proportion and intensity of the staining was evaluated independently by two observers. The proportion score reflected the fraction of positive staining cells (0, none; 1, ⩽10%; 2, 10% to ⩾25%; 3,
Cell culture and transfection
LAC cell lines were cultured in DMEM medium supplemented with 10% heat-inactivated FBS, 100 U/mL of penicillin, and 100 μg/mL of streptomycin. Cells in this medium were placed in a humidified atmosphere containing 5% CO2 at 37°C. Cells were subcultured at a 1:5 dilution in medium containing 300 µg/mL G418 (an aminoglycoside antibody, commonly used stable transfection reagent in molecular genetic testing). On the day of transduction, LAC cells were replated at 5×104 cells/well in 24-well plates containing serum-free growth medium with polybrene (5 mg/mL). When reached 50% confluence, cells were transfected with recombinant experimental virus or control virus at the optimal MOI (multiplicity of infection) of 50, and cultured at 37°C and 5% CO2 for 4 h. Then supernatant was discarded and serum containing growth medium was added. At 4 days post transduction, transduction efficiency was measured by the frequency of green fluorescent protein (GFP)-positive cells. Positive and stable transfectants were selected and expanded for further study. The Lv-shPTTG1 vector-infected clone and the negative control vector-infected cells were named as Lv-shPTTG1 group and NC group.
Quantitative real-time PCR
To quantitatively determine the mRNA expression level of PTTG1 in LAC cells, real-time PCR was performed. Total RNA was extracted for each clone using TRIzol according to the manufacturer’s protocol. Reverse transcription was carried out using M-MLV and cDNA amplification was performed using the SYBR Green Master Mix kit according to the manufacturer’s guidelines. The PTTG1 gene was amplified using a specific oligonucleotide primer and the GAPDH gene was used as an endogenous control. Data were analyzed using the comparative Ct method (2−ΔΔCt). Three separate experiments were performed for each clone.
Western blot assay
LAC cell lines were harvested and extracted using lysis buffer (Tris-HCl, SDS, mercaptoethanol, and glycerol). Cell extracts were boiled for 5 min in loading buffer, and then an equal amount of cell extracts was separated on 15% SDS-PAGE gels. Separated protein bands were transferred onto polyvinylidene fluoride (PVDF) membranes, which were subsequently blocked in 5% skim milk powder. Primary antibodies against PTTG1, p-TGFβ1, p-SMAD3, CyclinD1, and MMP-2 were diluted according to the manufacturer’s instructions and incubated overnight at 4°C. Subsequently, horseradish peroxidase-linked secondary antibodies were added at a dilution of 1:1,000 and incubated at room temperature for 2 h. The membranes were washed three times with PBS, and the immunoreactive bands were visualized using the ECL Plus Kit according to the manufacturer’s instructions. The relative protein levels in different cell lines were normalized to the concentration of GAPDH. Three separate experiments were performed for each clone.
Cell proliferation assay
Cell proliferation was analyzed using the MTT assay. Briefly, cells infected with Lv-shPTTG1 virus were incubated in 96-well-plates at a density of 1×105 cells per well with DMEM medium supplemented with 10% FBS. Cells were treated with 20 μL of MTT dye at 0, 24 h, 48 h, 72 h, and subsequently incubated with 150 μL of DMSO for 5 min. The color reaction was measured at 570 nm using an Enzyme Immunoassay Analyzer (Bio-Rad, Hercules, CA, USA). The proliferation activity was calculated for each clone.
Transwell invasion assay
Transwell filters were coated with Matrigel (3.9 µg/µL; 60–80 µL) on the upper surface of a polycarbonate membrane (diameter, 6.5 mm; pore size, 8 µm). After incubating at 37°C for 30 min, the Matrigel solidified and served as the extracellular matrix for analysis of tumor cell invasion. Harvested cells (1×105) in 100 µL of serum-free DMEM were added into the upper compartment of the chamber. A total of 200 µL of conditioned medium derived from NIH3T3 cells was used as a source of chemoattractant, which was placed in the bottom compartment of the chamber. After 24 h of incubation at 37°C with 5% CO2, the medium was removed from the upper chamber. The non-invaded cells on the upper side of the chamber were scraped off with a cotton swab. Cells that had migrated from the Matrigel into the pores of the inserted filter were fixed with 100% methanol, stained with hematoxylin, then mounted and dried at 80°C for 30 min. The number of cells invading through the Matrigel was counted in three randomly selected visual fields from the central and peripheral portion of the filter by using an inverted microscope (200× magnification). Each assay was repeated three times.
Statistical analysis
SPSS 20.0 was used for the statistical analysis. Kruskal-Wallis H test and Chi-square test were used to analyze the expression rate in all groups. One-way analysis of variance (ANOVA) was used to analyze the differences between groups. The LSD method of multiple comparisons was used when the probability for ANOVA was statistically significant. Statistical significance was set at
Results
The expression of PTTG1 in LAC tissues
The expression of PTTG1 protein was examined by IHC staining in LAC tissues. The expression level of PTTG1 protein was evaluated in LAC and ANCT tissues (Figure 1). Positive PTTG1 immunostaining was mainly localized in the nucleus and cytoplasm of LAC tissue cells. According to the PTTG1 immunoreactive intensity, the positive expression of PTTG1 was significantly increased in LAC tissues compared with that in ANCT (
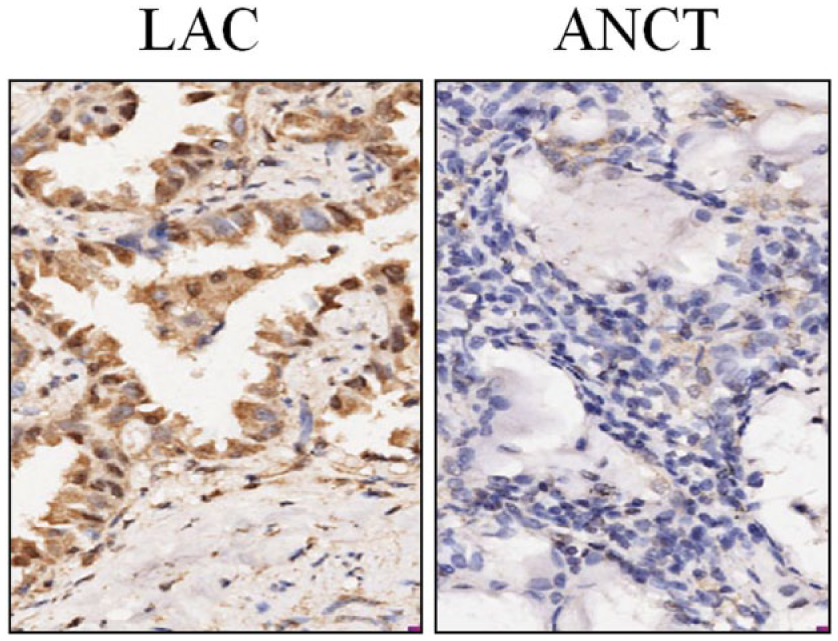
The expression of PTTG1 in LAC tissues (× 200). Expression of PTTG1 protein was examined by IHC staining. PTTG1 expression was found increased in LAC tissues compared to that in ANCT.
Expression of PTTG1 protein in LAC tissues.

ANCT: adjacent non-cancerous tissues; LAC: lung adenocarcinoma; PTTG1: pituitary tumor-transforming gene 1.
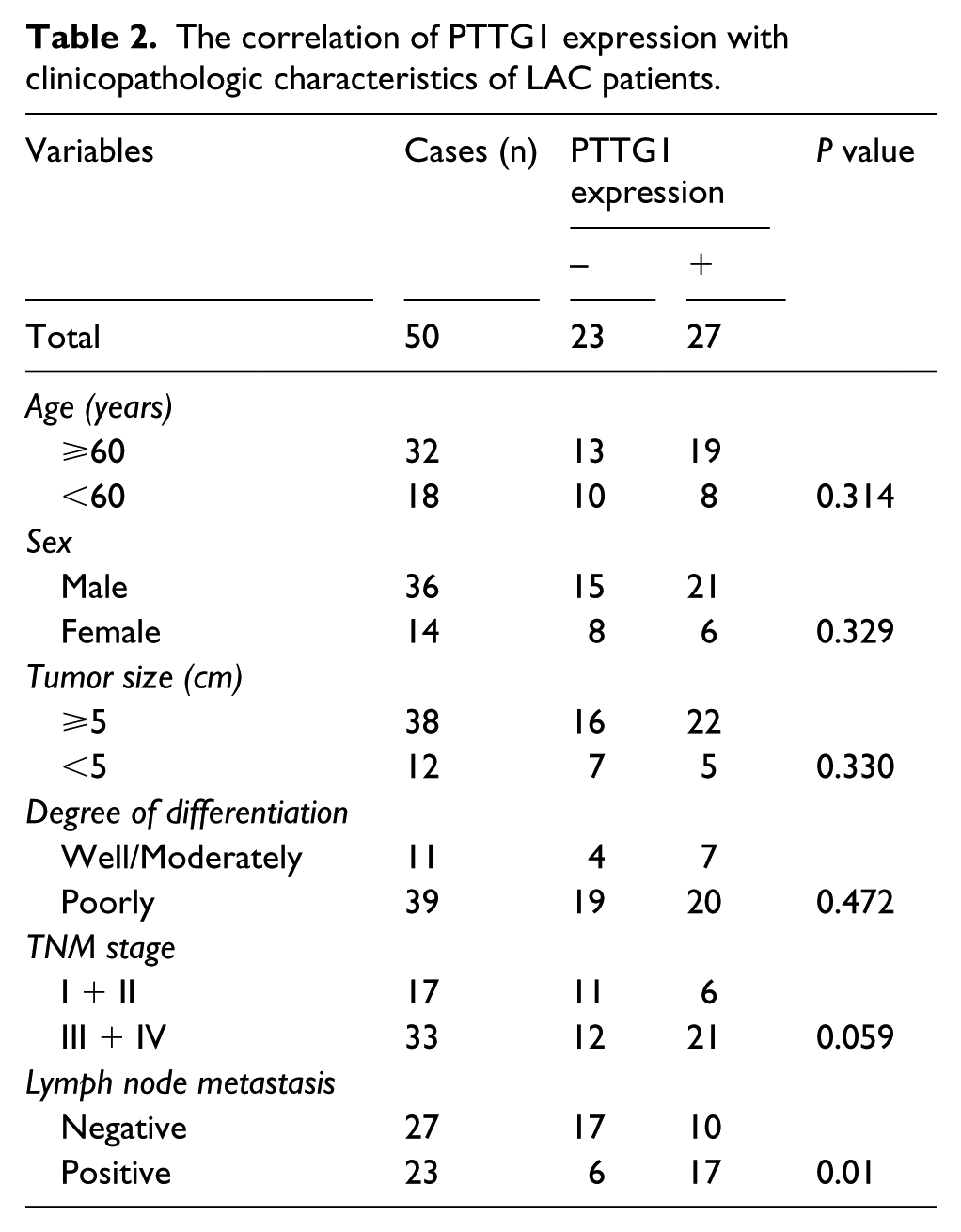
The correlation of PTTG1 expression with clinicopathologic characteristics
The correlation of PTTG1 expression with several clinical and pathologic factors was analyzed. As shown in Table 2, increased expression of PTTG1 was closely correlated with lymph node metastases of LAC (
The correlation of PTTG1 expression with clinicopathologic characteristics of LAC patients.
The effect of PTTG1 knockdown on the expression of TGFβ1/SMAD3 signaling
After Lv-shPTTG1 was transfected into LAC cell lines (A549 and LETPα-2) for 24 h, the mRNA expression levels of PTTG1, TGFβ1, and SMAD3 (Figure 2A and B) and those protein levels (Figure 2C and D) were detected by real-time PCR and western blot assays, indicating a decreased expression of PTTG1 and increased expression of p- TGFβ1 and p- SMAD3 in Lv-shPTTG1 group compared with the NC group in LAC cells (**

Effect of PTTG1 knockdown on TGFβ1 and SMAD3 expression. (A, B) The mRNA expression of PTTG1 was decreased but that of TGFβ1 and SMAD3 was increased, indicated by real-time PCR assay in Lv-shPTTG1 group compared with the NC group (each **
The effect of PTTG1 knockdown on cell proliferation
Deregulated cell proliferation is a hallmark of cancer. To confirm the effect of PTTG1 knockdown on cell growth, we assessed cell proliferative activities by MTT assay. We found that knockdown of PTTG1 decreased the proliferative activities of LAC cell lines in a time-dependent manner compared to the NC group (**

Effect of PTTG1 knockdown on cell proliferation. (A, B) We investigated the growth of LAC cells by MTT. Knockdown of PTTG1 significantly inhibited proliferative activities of LAC cells in a time dependent manner compared with the NC group (each **
The effect of PTTG1 knockdown on cell invasion
To determine the effect of PTTG1 knockdown on cell invasion, a Transwell assay was performed. The invasive potential of tumor cells in Transwell assay was determined by the ability of cells to invade a matrix barrier containing laminin and type IV collagen, the major components of the basement membrane. Representative micrographs of Transwell filters can be seen in Figure 4A. We found that the invasive potential of LAC cells was reduced in Lv-shPTTG1 group compared to the NC group (**

Effect of PTTG1 knockdown on cell invasion. (A) The invasive potential assessed by Transwell assay was determined on the basis of the ability of cells to invade a matrix barrier containing laminin and type IV collagen, the major components of the basement membrane. (B) The invasive potential of LAC cells was reduced in Lv-shPTTG1 group compared with the NC group (**
Discussion
PTTG1 as an identified proto-oncogene is implicated in a wide variety of human cancers. Overexpression of PTTG1 promotes the pathogenesis and development of endometrial carcinoma, 16 and is expressed in primary ductal breast carcinoma, lymph node infiltration, and distant metastases, indicating a powerful biomarker of breast cancer cells. 17 PTTG1 overexpression is associated with the gender of patients with pancreatic cancer 18 and poor survival in adrenocortical carcinoma, 19 and serves as a negative prognostic factor for progression in advanced prostate cancer. 20 It is also a proliferation marker associated with mutated p53 protein but does not correlate with the prognosis in cutaneous squamous cell carcinomas. 21 In the present study, we examined the expression of PTTG1 in LAC tissues and found that PTTG1 was upregulated in the nucleus and cytoplasm of LAC tissue cells compared with the ANCT, and it was positively correlated with lymphatic invasion of the tumor, suggesting that intracellular accumulation of PTTG1 might participate in tumorigenesis of LAC.
PTTG is also a well-studied oncogene for its function in tumorigenesis and serves as a marker of malignancy in several cancers. PTTG1 gene is upregulated in hepatocellular carcinoma (HCC) and inhibiting PTTG1 expression decreases cell proliferation and induces apoptosis. 22 Importantly, PTTG1 oncogene promotes lymph node metastasis 23 cell invasion and metastasis 24 and epithelial to mesenchymal transition (EMT)25,26 in multiple cancer cells. Knockdown of PTTG1 suppresses the proliferation and invasive potential of human cancer cells.27,28 But, the function of PTTG1 in LAC cells is not comprehensively clarified. In the present study, we investigated the biological behaviors in Lv-shPTTG1-transfected LAC cells and found that knockdown of PTTG1 could inhibit the proliferation and invasion of LAC cells. In addition, Cyclin D1 and MMP-2 have been implicated in the proliferation and invasion of lung cancer cells. 29 We found that PTTG1 knockdown decreased the expression of Cyclin D1 and MMP-2 in LAC cells. These findings indicated that that PTTG1 may play a tumor-promoting role in LAC.
Transforming growth factor-beta (TGFβ) family regulates a series of biological processes including cell proliferation, migration, apoptosis, and extracellular matrix deposition. 29 Accumulating evidence show that TGFβ signaling can inhibit tumor growth in early-stage tumors, but promotes tumor invasiveness and metastasis in late-stage tumors. 30 Moreover, PTTG1 promotes the growth of cancer cells via the inhibition of TGFβ1/SMAD3 signaling.31,32 In the present study, we found that knockdown of PTTG1 could significantly upregulate the expression of TGFβ1 and SMAD3, suggesting that PTTG1 might be involved in the progression of LAC through regulation of the TGFβ1/SMAD3 signaling.
In conclusion, our findings demonstrate that high expression of PTTG1 is correlated with the tumor metastasis of LAC patients, and knockdown of PTTG1 suppresses the growth and invasion of LAC cells through regulation of the TGFβ1/SMAD3 signaling, suggesting that PTTG1 may be a potential target for developing an effective immunotherapeutic strategy for LAC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the grants from National Natural Science Foundation of China (Nos. 30960439 and 81101693), Yunnan National Natural Science Foundation (No. 2011FB206), and National Key Clinical Specialty (Oncology) Fund.
