Abstract
This study focuses on the recycling of mill scale of AISI 4330 V grade using induction furnace melting. The prime objective of the work is to establish a viable pathway for efficient recovery of iron, nickel and molybdenum from briquettes made of mill scale using carbothermic reduction utilising Calcined Petroleum Coke (CPC) as a reducing agent. Briquettes were prepared by grinding mill scale into powder and mixing it with CPC and molasses as a binder. Chemical composition analysis, X-ray diffraction (XRD), and thermogravimetric analysis (TGA) were conducted to assess the effectiveness of the recovery of metals from mill scale. Both stoichiometric calculations and TGA analysis signified that, briquettes with 10% to 12% carbon can give best reduction results. Subsequently, induction furnace experiments were conducted, varying the proportion of charge constituents for making melt pool. Results indicated that using mild steel (MS) plates for making melt pool improved recovery rates compared to using pig iron. A respective recovery of 99.36%, 75.52% and 97.01% was achieved for iron, nickel and molybdenum. A rise of 24.44%, 14.45% and 47.47% was observed in recovery of iron, nickel and molybdenum, respectively, in route using MS plate for making melt pool in comparison with route using PI for making melt pool. XRD study of slag produced in various routes was conducted to assess the presence of phases which indicates loss of elements into the slag. Substantial recovery of iron, nickel and molybdenum was observed in the final ingot contributing towards circular economy. Furthermore, energy requirements for the recycling process were calculated and it was observed that significant portion of energy (78.55%) was consumed in the reduction process.
Introduction
The steel making industry is one of the major contributors of solid waste in the world, which contains several valuable elements that can be reused if recovered economically. Approximately 600 kg of solid wastes are produced by steel making industry per ton of steel produced. About 70% to 80% of the waste is in the form of slag, which is used in cement making for construction purposes. The remaining solid wastes can be categorised as mill scale, furnace dust and sludge.1,2 Mill scale, formed by oxidation of steel surface during solidification and reheating for hot rolling or forging, reduces overall yield and increases costs. Although not hazardous, it poses environmental risks if left unprocessed. 3 Mill scale primarily contains oxides of iron and various other valuable elements.4,5 Studies have been carried out by why researchers and industries to explore reduction methods using certain reducing agents like C, Al, Si, H2 and CO to name a few. Generally, these mill scales are fed in the form of cold-pressed briquettes or pellets in blast furnace, which has a reducing atmosphere with CO as the major reducing agent. 6 However, blast furnaces are a major source of CO2 emission. As per the reports, it has been estimated that 1816 kg CO2 is emitted per ton of final product for the blast furnace (BF)/converter route. This huge value of CO2 emission makes BF a major contributor of carbon footprints. Owing to this, steel making companies are rapidly shifting towards electric arc furnaces (EAF) and induction furnaces (IF) which decrease the CO2 emission significantly with steel production using scrap melting route. In EAF and IF, the CO2 emission is estimated to be 517 and 680 kg, respectively, per ton of steel produced. 7 As far as mill scale is concerned, they are consumed in sinter-based BF to reduce it to iron. However, valuable elements like nickel, molybdenum and chromium are lost during BF melting, thus value of mill scale is substantially decreased. Additionally, extra transportation cost is incurred for the transfer of mill scale from EAF/IF steel making plant to remotely located BF steel making plants. As far as EAF is concerned, there is an oxidising environment due to oxygen lancing and hence oxides present in mill scale cannot be reduced efficiently. Hence, oxides of high value elements like chromium and vanadium remain unreduced and are lost in the slag. 8 IF having neutral atmosphere can provide a required pathway to reduce these oxides as reported by various researchers provided some suitable reducing agent is added.9,10,11 Some reducing agent like charcoal and Calcined Petroleum Coke (CPC), has been reported to be agglomerated using a binder12,13 such as bentonite clay, sodium silicate, molasses and starch-based binders have been used to produce briquettes using press. Magdziarz et Al. 14 performed series of experiments to determine the optimal composition of the briquettes and the best binder to be used for getting zinc free ingots. It was found that the best binder was a mixture of hydrated lime and sugar molasses in terms of reducibility and compressive strength. Mombelli et al. 15 added starch as a binder to jarosite sludge and blast furnace sludge for making briquettes by using pressure of 20 and 40 MPa. The results showed that the briquettes made using 10% starch under 40 MPa pressure had more strength and best mechanical properties.
Several researchers have worked in characterising briquettes to get the desired combination of reducibility, compressive strength and impact strength. Wu et al. 16 Similar results were obtained by Yang et al. 17 found that briquettes of 60 mm×45 mm×30 mm were best suited for EAF smelting, with a charging rate of 10 briquettes per minute, with minimum silicon content to enhance carbon reduction. This is because, silicon can react with carbon to form silicon carbide, which is a non-metallic compound that does not melt at high temperature. We et al. 18 studied the reduction of carbon composite briquettes made from EAF dust and mill scale at temperatures ranging from 1000°C to 1550°C. They found that EAF dust reduced more effectively than mill scale below 1250°C, but both behaved similarly above 1250°C. Similar studies have been reported by Khaerudini et al. 19 who achieved over 80% metallization of mill scale at 1000°C in 2 hours. Flores et al. 20 and Bagatini et al. 21 found that both mass loss and metallization increase with temperature, reaching 76% at 1400°C. Hussiny et al. 22 investigated the use of briquetting to recycle waste materials from iron and steel plants and found that briquettes made from a mixture of blast furnace flue dust and mill scale could be reduced to metallic iron at temperatures as low as 1100°C using carbothermic reaction. Davydenko et al. 23 conducted a large-scale trail and found that the briquettes could be reduced at 1500°C. The recovery of iron, nickel and chromium from the briquettes was up to 93% to 100%. It was reported that up to 330 kg of Fe, 28 kg of Ni and 66 kg of Cr per ton of added briquettes can be recovered in EAF.
From the above literature review, it can be concluded that the wastes of steel making plants have rich proportion of metal present in it which can ensure better waste management to contribute towards reduction of environmental pollution and circular economy. Several attempts have been made to find out a viable way to recover metallic components from by-products of steel making plant. However, most of these works are confined to lab scale production of self-reducing briquettes and intended to be used in EAF in which recovery of high value metal is not that feasible. Very limited work has been carried out using IF for recovery of metals from mill scale using carbothermic reaction. As IF has neutral atmosphere, it might offer viable pathway for mill scale reduction, provided an appropriate reducing agent is utilised. In the current work, an attempt was made to recycle mill scale of AISI 4330 V grade produced at Saarloha Advanced Materials Private Limited and Bharat Forge Limited using small scale IF of 100 kg capacity. The prime objective of the work is to establish a viable pathway for efficient recovery of iron, nickel and molybdenum from briquettes made of mill scale using carbothermic reduction utilising CPC as a reducing agent. The proportion of CPC was fixed using stoichiometric calculations and TGA. The chemical composition and X-ray diffraction (XRD) pattern of ingot and slag was studied for each route along with their respective metal recovery. Finally, the energy requirement calculation has been done to predict the amount of electricity in different processes during melting.
Materials and methodology
Mill scale of AISI 4330 V steel grade was collected from different sites such as steel melting shop, rolling and forging mill shop at Saarloha Advanced Materials Private Limited. It is approximated that 100 to 200 kg mill scale is generated during rolling and hot forging per ton of steel processed, depending upon the specific method employed and the grade of steel being manufactured. To find out the composition of mill scale in terms of their respective oxides, X -ray fluorescence (XRF) analysis was done and are presented in Table 1. Based on molar calculations, the percentage of various elements was evaluated, which is also given in Table 1.
Chemical composition of by-products collected at steel-making shop, rolling and forging shop.

It can be seen from Table 1, that mill scale obtained from of steel making plant is a rich source of iron (Fe), nickel (Ni) and molybdenum (Mo), which if recovered economically can contribute remarkably towards sustainable steel making. To further confirm the nature of iron present in mill scale, wet analysis was conducted. Initially, a 250 ml conical flask with a stopper is prepared by removing the air within. And 0.1 g of the powdered sample is added to the flask, followed by the addition of 15 ml of water and 35 ml of 25% FeCl3 aqueous solution. The flask is immediately closed, placed on a magnetic stirrer, and stirred gently for 45 minutes while ensuring it is covered to prevent air ingress. Subsequently, 50 ml of water and 25 ml of a 0.1 N H2SO4 solution are added, along with a few drops of indicator. The solution is titrated quickly against 0.1 N K2Cr2O7 until a purple endpoint is reached. The presence of a purple colour indicates the presence of metallic iron in the sample which can be calculated by formula: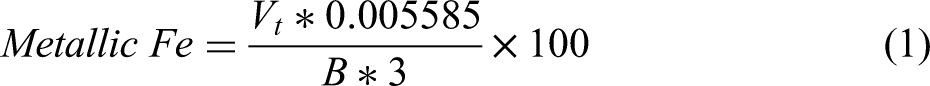
Using the above method, the metallic Fe content in mill scale was calculated to be 61.21% which closely matches with the value obtained by XRF analysis presented above.
Characterisation of mill scale
The size distribution of mill scale particles was initially assessed through simple sieve analysis and found to be predominantly in the range of 3 and 5 mm. However, for briquette preparation, mill scale was used in powdered form, as powders provide a greater surface area-to-volume ratio, which enhances the kinetics of reduction. Bagatini et al. 24 showed that finely grinding mill scale leads to a faster metallization rate as it increases the availability of reactive sites at the interface. Additionally, powders when agglomerated to form briquettes give better strength. Studies by Wu et al. 16 and Yang et al. 17 have shown that briquettes made of uniformly sized particles have better strength both at low and high temperatures as uniformly sized particles can be agglomerated more efficiently creating a briquette with higher strength. For this reason, mill scale was grinded to make powders of average size of 0.5 mm and rammed to make briquettes.
Briquette making and formulation
After mill scale had been powdered, briquettes were made which could be finally charged in IF. For this purpose, the powder of mill scale was dried in muffle furnace for 2 hours at 200°C. This was done to take out moisture content and volatile substances present in the mill scale powder. Post drying, the powder was mixed with different proportions of CPC was agglomerated using a molasses (4%–6%) as a binder. The agglomerated mixture was rammed and pressed for 50 to 55 seconds using a hydraulic press (pressing stress of 20 MPa). CPC was added in a specific proportion to reduce the FexOy and other metallic oxides to their corresponding metallic elements (Figure 1).
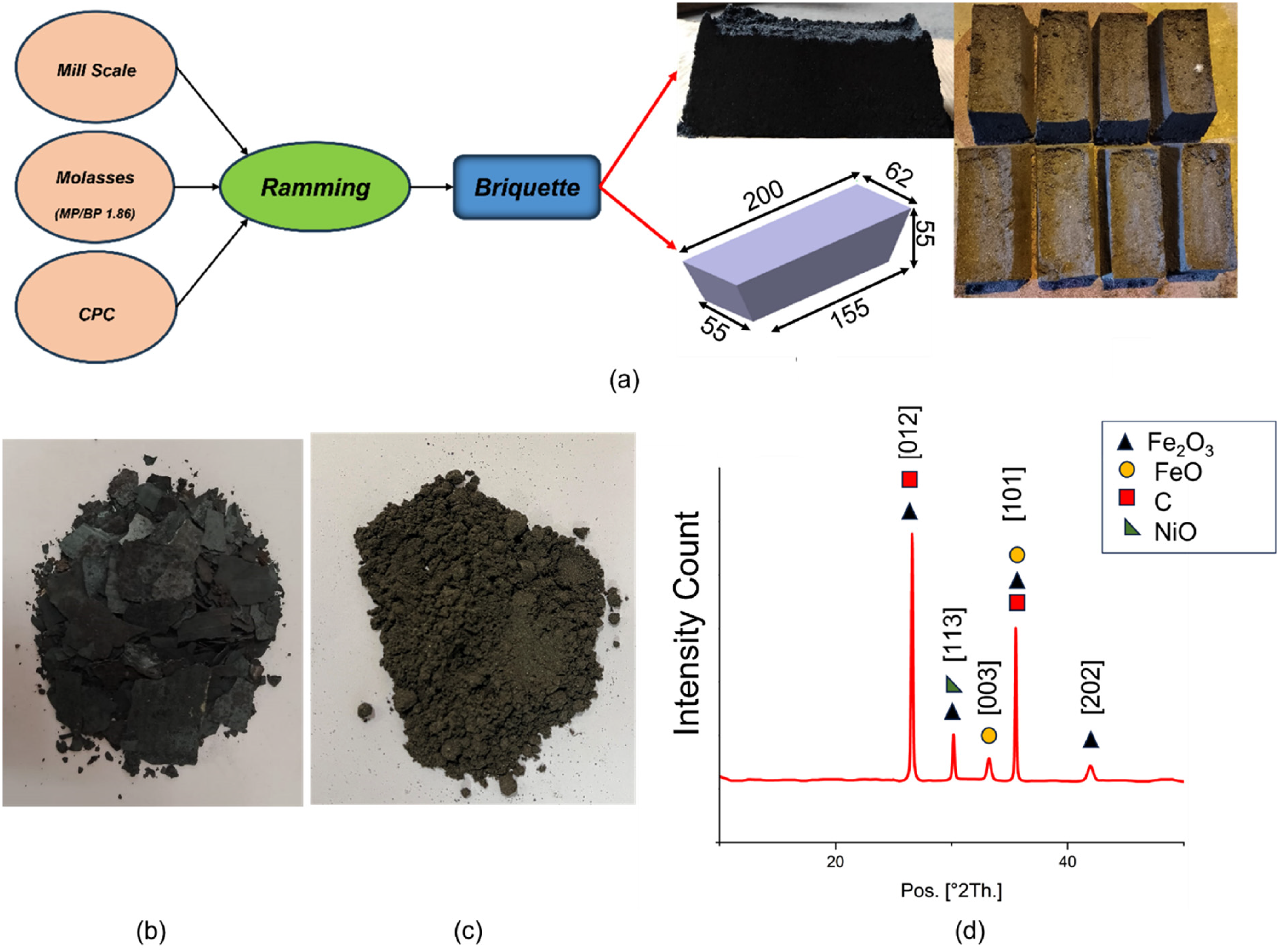
(a) Process for briquette making and briquettes with dimensions; (b) collected mill scale; (c) powdered mill scale; and (d) XRD pattern of mill scale briquette. XRD: X-ray diffraction.
Once the mill scale powder, CPC and binders were mixed, the XRD of the mixture was performed to find out the presence of dominant phases in the briquette. It was observed that, the mill scale powder primarily consists of Fe2O3 (00-001-1053) and FeO (00-002-1180). Peaks corresponding to Carbon (00-001-0640) and NiO(00-002-1216) were also obtained.25,26
As can be inferred from the Ellingham Diagram 27 as shown in Figure 2, that carbon can act as a very good reducing agent for reducing ferrous, nickel and molybdenum oxides. The proportion of CPC to be added to form self-reducing briquettes was theoretically calculated based on chemical reactions taking place at different temperatures. The amount of carbon required for carbothermic reduction of ferrous oxides only was taken, as the amount of C required for other oxides will be very less due to their presence in very small amount in mill scale.
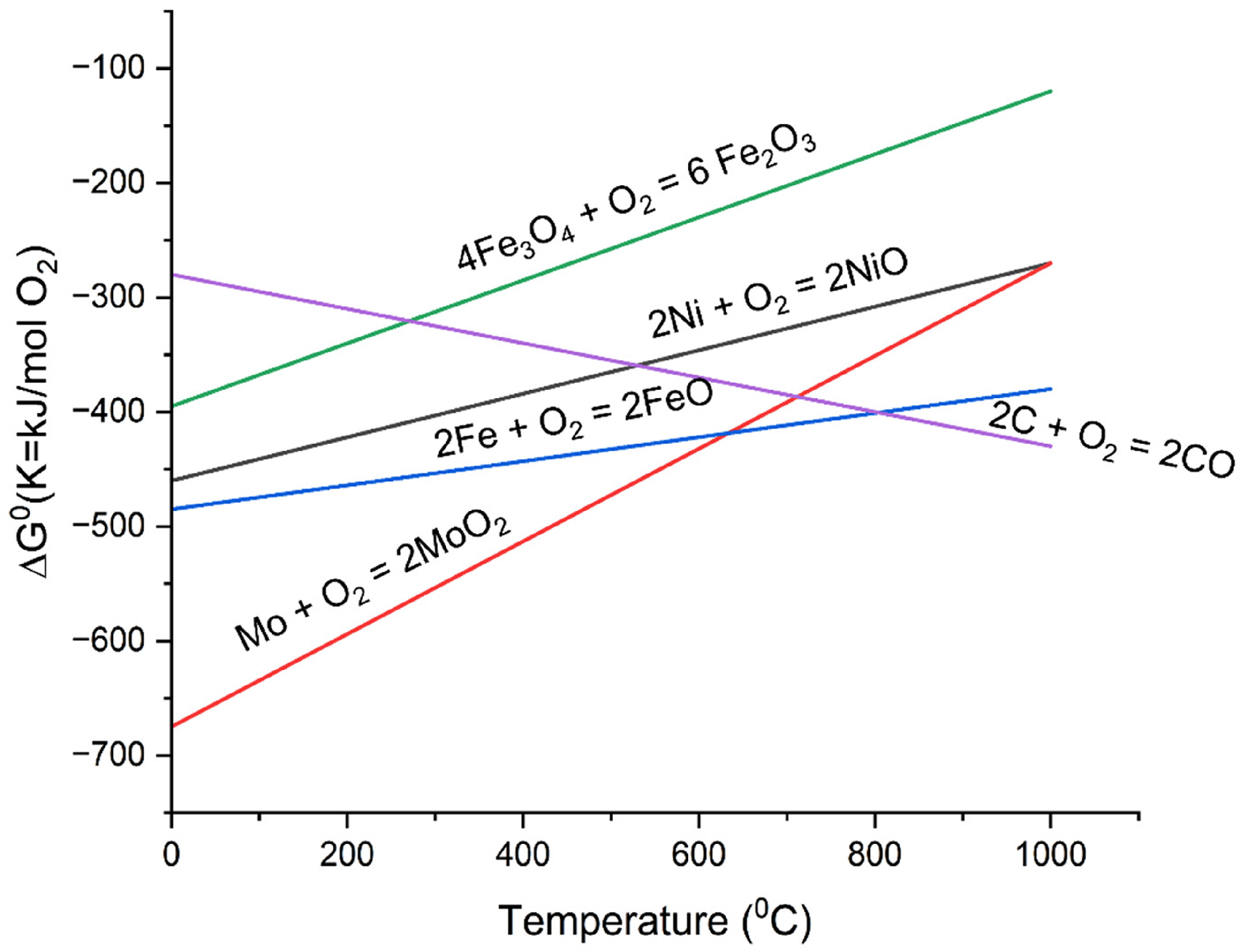
Ellingham diagram for various oxides and carbon. 27
Stoichiometric calculation and thermogravimetric analysis
As it can be seen from Ellingham diagram in Figure 2, Carbon can act as a reducing agent for reducing iron oxides at higher temperature (typically > 983 K), at higher temperature. Hence, at temperature above 1000 K, it was assumed that the carbothermic reduction takes place by reaction given in equations (2), (3), and (4)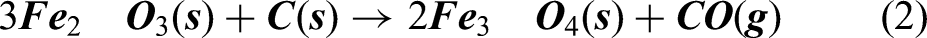
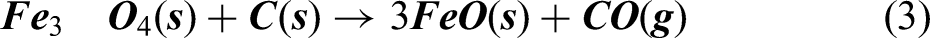
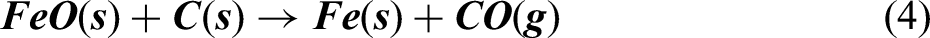
According to reactions represented in equations (2), (3) and (4), it can be said that carbon monoxide gas escapes out of the furnace. Combining equations (2), (3) and (4) for reduction of Fe2O3 converges to equation (5).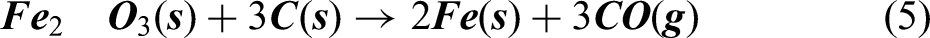
As from the thermodynamic data,
28
the standard entropy for Fe2O3, C, Fe and CO are 87.40, 5.75, 34.76, and 197 J/K, respectively, making the standard entropy of reaction to be 545 J/mol-K. Similarly, 
The required temperature comes out to be 1013 K. Hence above this temperature the reaction can go forward producing CO. The CO produced will rise upwards in the furnace and will again encounter metal oxide which will be at relatively lower temperature (700–900 K) and will again combine with it to give Fe and finally liberate CO2 in atmosphere as represented by equation (7).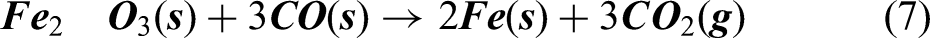
Based on chemical reactions and stoichiometric analysis, it was calculated that 10% to 12% of CPC is required to completely reduce the oxides present in a briquette (predominantly). To verify the proportion of CPC to be added and to examine the kinetics of reducibility, TGA was carried out. It is worth noting that, the purpose of this analysis is to check the feasibility of the considered reaction as mentioned in equations only equations (2)–(6) and (7). To conduct TGA, 4 different briquette compositions were chosen for analysis. The composition of all such briquettes is given in Table 2.
Composition of different types of briquettes used for TGA.

Calcined Petroleum Coke; TGA: thermogravimetric analysis.
The samples B1, B2, B3 and B4 were loaded into a crucible and placed in a muffle furnace initially at room temperature and were heated at a rate of 25 K/min till 873 K and 10 K/min till 1273 K. Finally, the samples were held for 30 minutes at 1373 K.29,30 The weight of the briquette was measured after every 100°C. These measurements are graphically represented in a thermogram as shown in Figure 3.
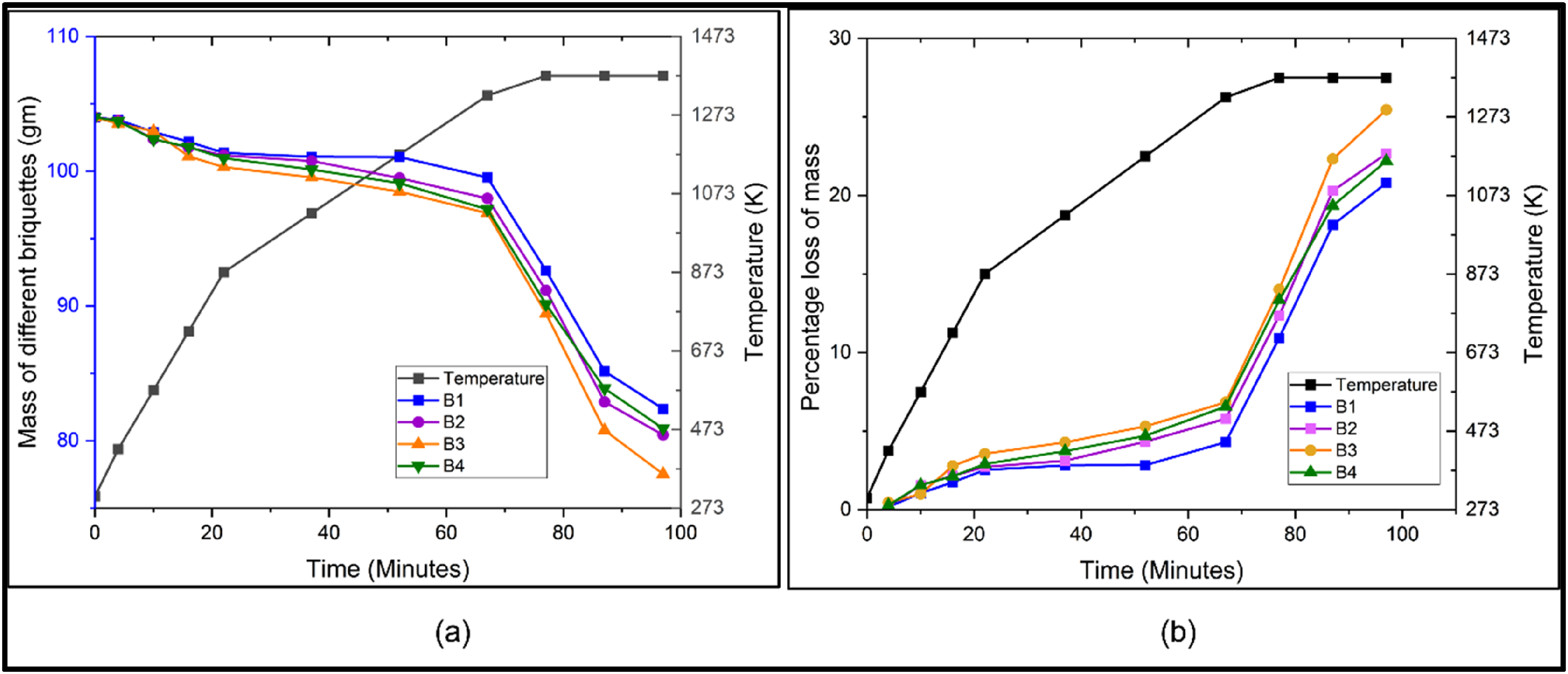
TGA analysis: (a) variation of mass of different briquettes with time and (b) variation of percentage mass loss with time. TGA: thermogravimetric analysis.
It can be seen from Figure 3(a), that similar reduction trend is followed in all four cases. It can be seen from Figure 3(a) that initially the rate of reaction is slow till 573 K, due to evaporation of moist content or sublimation of volatile materials. Post this temperature, reduction reaction starts above 773 K and continues with a slow rate till 973 K. In this temperature regime, carbon monoxide acts as a reducing agent. As can be seen from Ellingham diagram in Figure 2 at approximately 983 K, the line corresponding to reaction 2CO + O2 = 2CO2 and 2C + O2 = 2CO intersect. Post this temperature, the line corresponding to 2C + O2 = 2CO gets below the line corresponding to 2CO + O2 = 2CO2. Hence C itself becomes a better reducing agent than CO at higher temperature and can reduce metal oxides. Hence post 973 K, accelerated reaction was observed leading to maximum mass loss. It can be said that carbothermic reduction of mill scale in IF primarily happens due to carbon acting as a reducing agent. Maximum reduction was observed in case of B3 in which CPC percentage lied between 10% and 12%. These results are in agreement with stoichiometric analysis carried out considering equations (2) to (6). The loss percentage versus time graph is shown by Ellingham diagram in Figure 2, in which maximum mass loss corresponding to B3 is nearly 25%. Percentage loss of metal is simply calculated as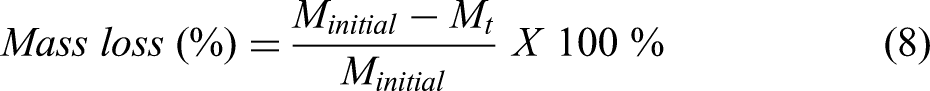
As maximum mass loss was observed in case of B3 briquettes and was also backed by stoichiometric calculations, this composition was chosen for further analysis. At temperature above 1273 K the reduction of FexOy is most accelerated with increased rate of reduction. After 1373 K, the reaction almost stabilises, suggesting a complete reduction of FexOy, the region where point of inflexion is observed in the mass versus temperature plot. Similar trends were reported earlier by various researchers.16,17,18,21 In order to analyse the rate of reaction of B3 briquette a plot of dm/dt versus temperature was made for B3 which is shown in Figure 4(a).
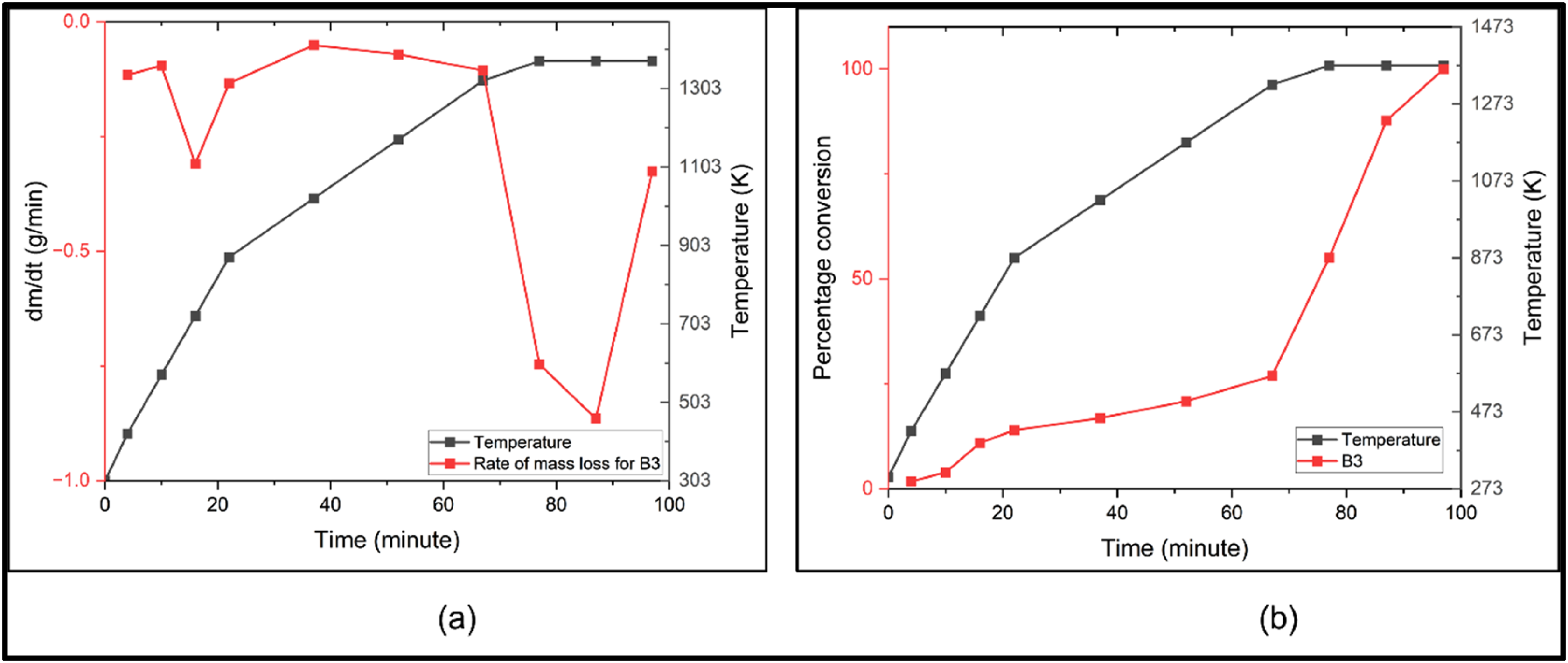
(a) Rate of mass loss for B3 versus time and (b) percentage conversion versus time for B3 versus time.
It can be seen from Figure 4(a), that after sublimation of volatile material in low temperature ranges (< 573 K), the rate of mass loss is almost constant till 1273 K after which it increases till it becomes maximum. Subsequently, the reaction rate starts decreasing till 1373 K suggesting the complete reduction till this temperature. Figure 4(b) gives an idea of percentage conversion with time. Conversion % can be calculated as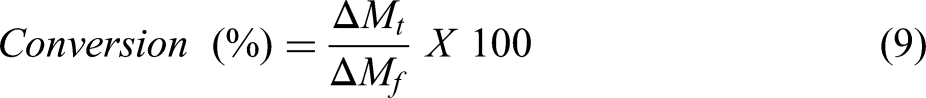
where,
It can be seen from Figure 4(b) that, rate of percentage conversion increases most steeply after 1273 K and then falls with subsequent time increase. It can be concluded from thermometric analysis, that carbon-based carbothermic reduction of FexOy to Fe is carried out at the maximum rate above 1273 K. After 1373 K, the rate of reaction almost stabilises, and this is the temperature at which maximum oxides are converted in pure metal. It can also be said that 10% to 12% CPC can be a reasonably good addition in briquette for getting optimal reduction which is backed by both stoichiometric calculations as well as Muffle furnace-based experiments. It can be observed that at temperatures above 1273 K, the reduction of iron oxides (FexOy) to metallic iron becomes thermodynamically feasible. Under the same conditions, the carbothermic reductions of NiO and MoO₃ using carbon monoxide also proceed with negative standard Gibbs free energy changes (ΔG°), indicating favourable reaction thermodynamics.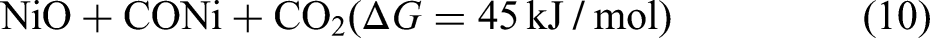
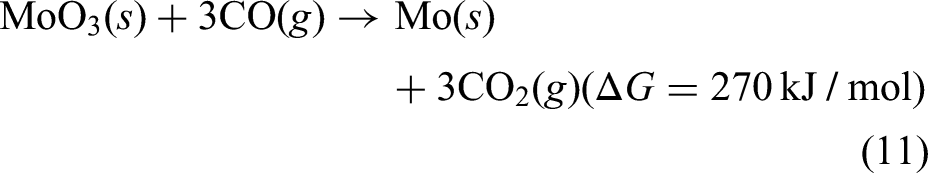
These values confirm the thermodynamic feasibility of reducing NiO and MoO3 alongside FexOy under the experimental conditions employed in this study. Based on the results obtained from TGA analysis, experiments were planned for metallisation of corresponding metallic oxides, so that a viable pathway could be established for the reutilisation of mill scale using IF which is the prime objective of current work.
Experiments with induction furnace
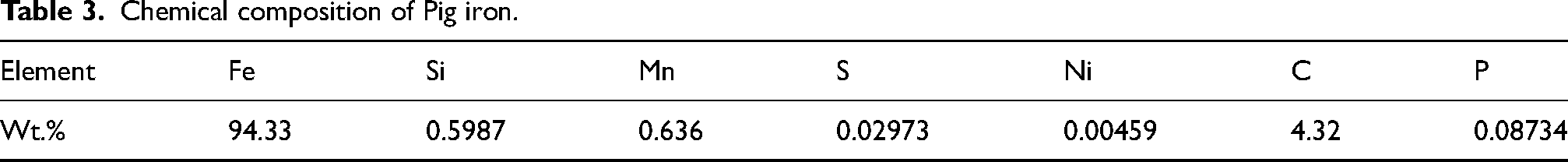
Laboratory-scale IF facility at Saarloha Advanced Materials Private Limited was used for reduction of mill scale briquettes and subsequent melting. Due to poor electrical conductivity and para magnetic nature of FexOy, the briquettes made of mill scale and CPC, cannot be directly melted. Hence, liquid metal pool of Pig iron or mild steel (MS) scrap was made initially to provide an electrically conductive medium which further serves as a heat reservoir to carry out subsequent reactions. 31 Briquettes absorb heat from liquid pool and attain required temperatures for carbothermic reduction. The composition of Pig iron and MS scrap are given in Tables 3 and 4, respectively.
Chemical composition of Pig iron.
Chemical composition of MS scrap.
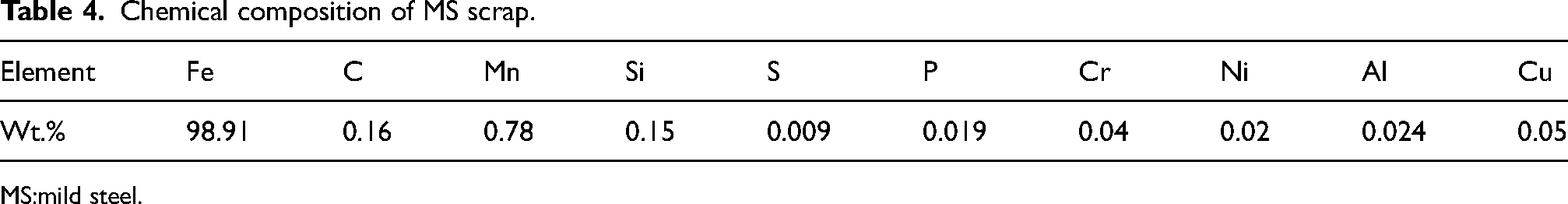
mild steel.
Results and discussions
Metal recovery in various routes
For recovery of metals from mill scale, briquettes were charged in IF in different ways. The flow diagram for melting in IF is shown in Figure 5.

Melting route adopted for melting of mill scale in induction furnace.
For melting mill scale 4 different trials were done. In route 1, pig iron was used for making melt pool. Pig iron was used since it contains 4.32 wt.% of carbon, and separate CPC addition can be avoided. However, the obtained ingots were found to have higher amount of phosphorus (wt.% ∼ 0.07) which cannot be controlled if the ingots are used in ladle furnace for alloying. To overcome this issue in route 2, MS scrap plates were used along with Pig iron (1:1) to dilute the phosphorus content present in the final ingot. However, the dilution obtained was not significant as phosphorus still was present in significant amount (wt.% ∼ 0.062%). So, in route 3, the melt pool was completely made from MS plates and CPC was used to reduce the oxides present in the mill scale. The phosphorus content in this route was found to be within acceptable limits (wt.% ∼ 0.014%). Finally in route 4, the melt pool was made from ingots obtained in route 3, so that final ingots could be further enriched in nickel and molybdenum. The quantity details of input charge, output ingot and slag are given in Table 5.
Details of various melting routes adopted.
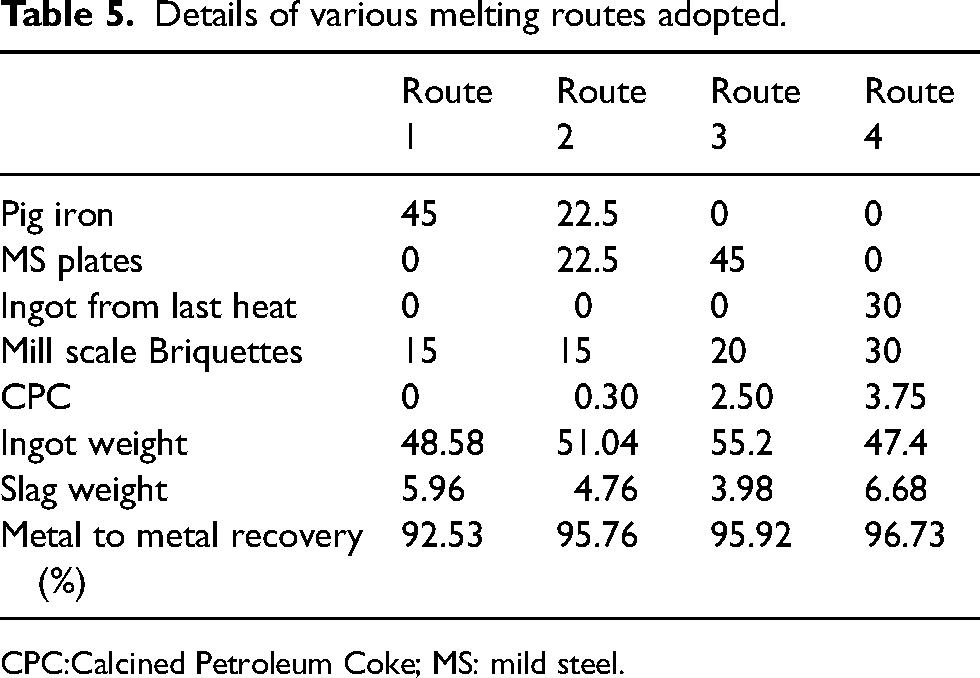
Calcined Petroleum Coke; MS: mild steel.
It can be seen from Table 6 that metal to metal recovery is descent in almost all cases. The metal to metal recovery in route 4 was found to be maximum at 96.73%. It can be recommended from above study that ratio of mill scale to initial scrap in IF-based melting, gives optimal results near unity. Further, loss of metal in recovery is attributed to unreduced form of oxides which were present in the slag produced during melting. A detailed analysis of slag is presented in the forthcoming sections.
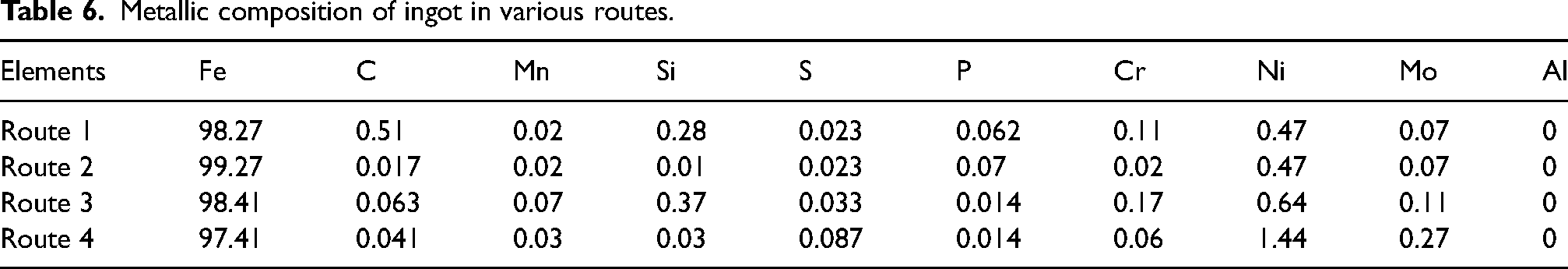
Metallic composition of ingot in various routes.
Ingot composition obtained in different routes
After the Mill scale briquettes were completely melted in IF, a lollypop sample was taken for determining chemical composition. These samples were analysed using mass spectroscopy technique on Thermo scientific ARL 3460 metals analyser provided by M/S Thermo Fisher Scientific, Switzerland for their respective chemical compositions, which is presented in Table 6.
It can be seen from Table 7, that the ingots produced in different routes are rich in terms of iron content. Additionally, the low carbon content shows the almost complete utilisation of carbon present in the initial charge. Final ingot contains descent quantity of Ni (1.44%) and molybdenum (0.27%). Hence these ingots can be used to produce low alloy steel grades containing Ni and Mo, for example, AISI P series, 32 46XX, 48 XX grades directly. 33
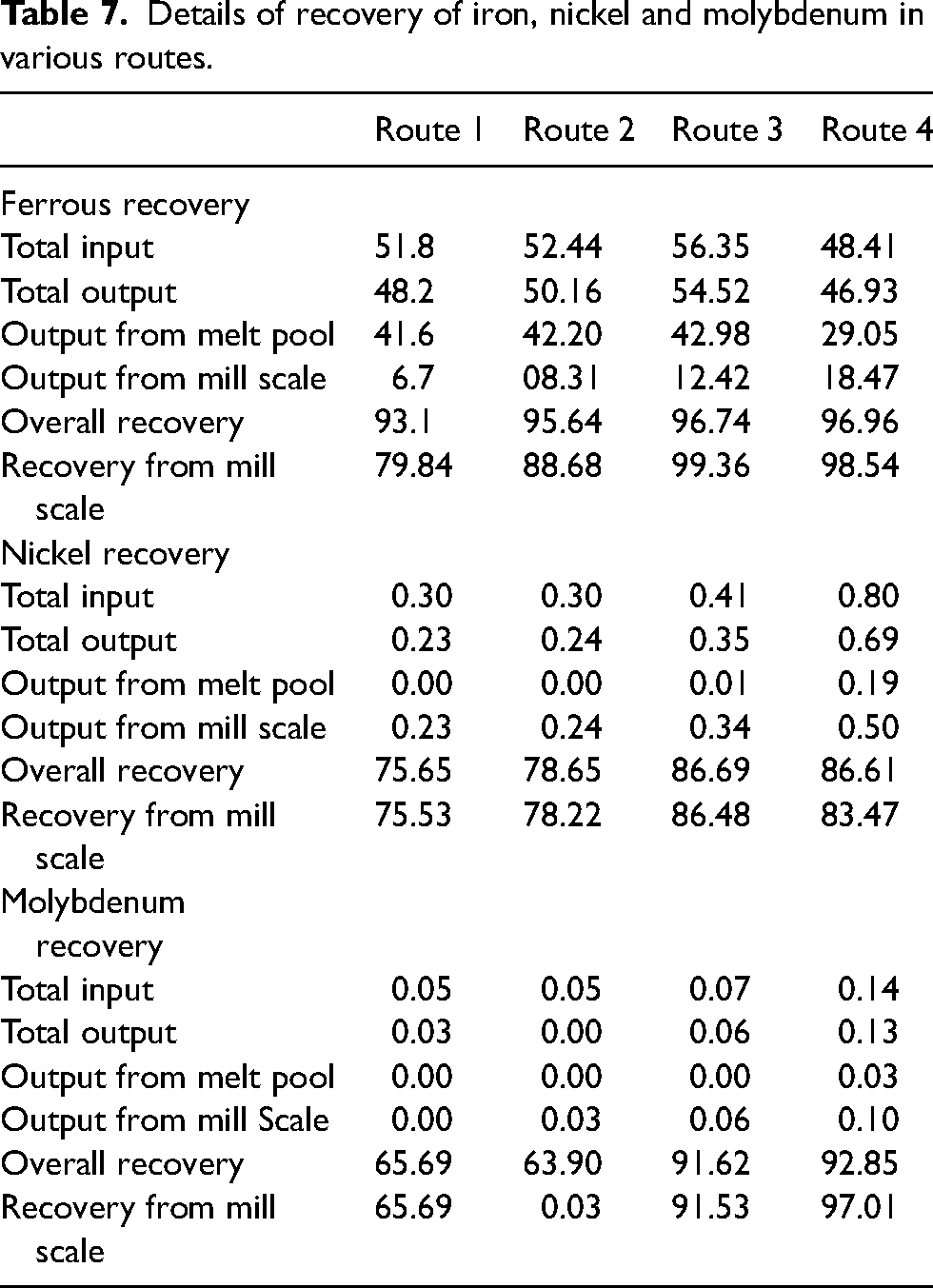
Details of recovery of iron, nickel and molybdenum in various routes.
Composition of obtained ingots in different routes
It was concluded from the composition report that the obtained ingots had rich content of iron indicating its optimum reduction due to dense structure of the briquettes felicitating their efficient integration with the melt pool. The richness of ingots in terms of iron content is also attributed to negligibly small carbon present in the ingots. The Fe and C content in the obtained ingots is shown in Figure 6. Carbon content in the ingot obtained in route 1 is 0.51%. Out of 45 kg of Pig iron, 1.9 kg (4.34%) was available for utilisation of reduction of various oxides. However, out of total available carbon, 247.78 g of carbon remains unutilised. Hence utilised carbon is 1.65 kg, which accounts for 11% of mill scale (15 kg). Hence, the amount of carbon required for oxidation is in close agreement with the calculated values presented in the briquette making and formulation section. In the subsequent trial, same proportion of carbon was used in initial charge. In these routes, the proportion of C in obtained ingots was decreased. In such ingots, a negligible quantity of carbon (0.017%, 0.063%, 0.041%) was obtained showing the effective utility of carbon for reduction of metal oxides as shown in Figure 6.
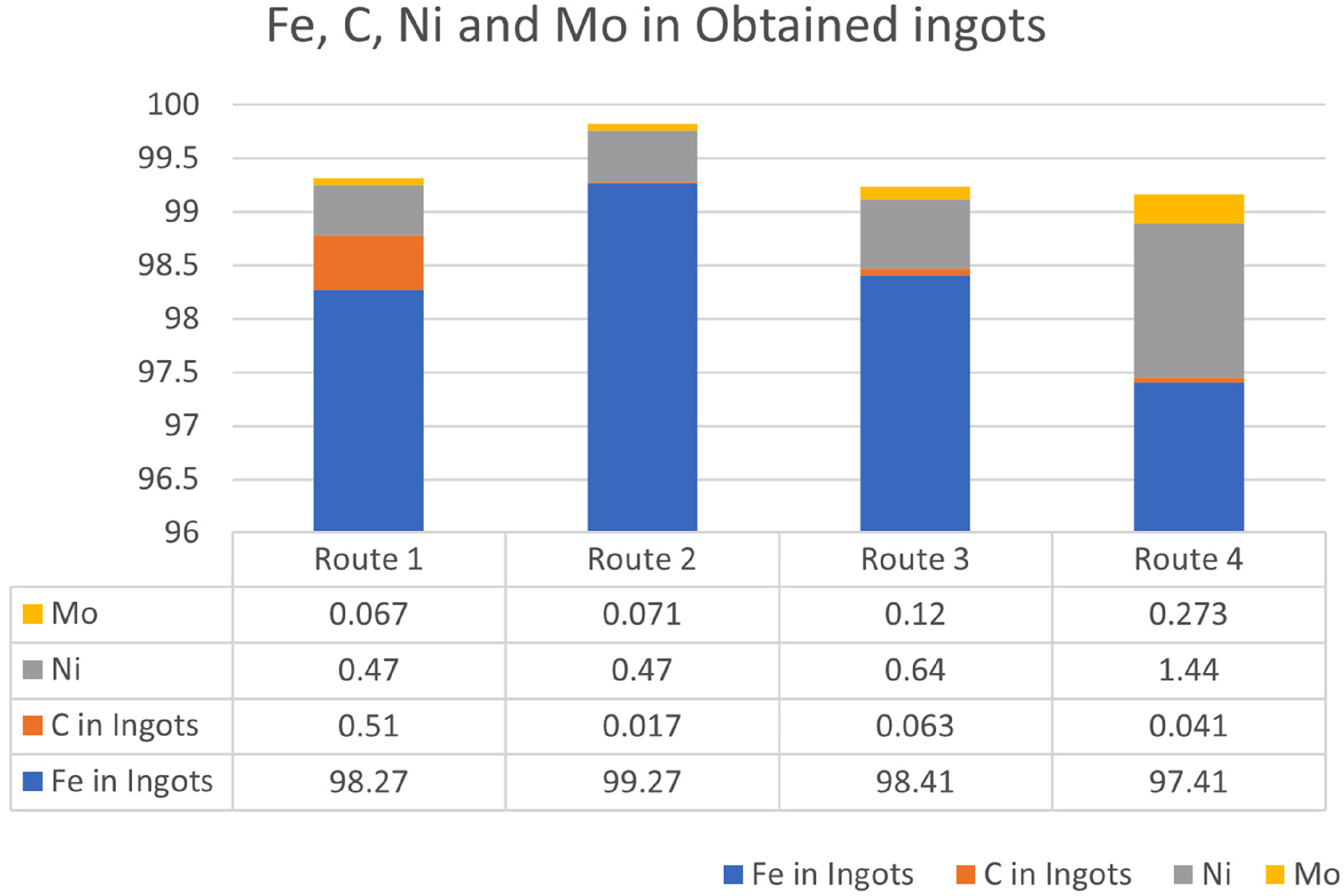
Details of ingots in various routes with Fe, C, Ni and Mo percentage in all routes.
It can be observed that adding CPC helps increase carbon utilisation for reduction reaction than carbon present in Pig iron. Further, it can also be observed from Figure 6 that, Ni and Mo was efficiently recovered from the mill scale briquettes. The percentage of Ni (0.67%) in ingots was found to be better when MS plate-based plates were used for making melt pool instead of Pig iron. Additionally, the Ni can be further enriched by using ingots of route 3 which was done in route 4 leading to 1.34 wt.% Ni in the ingot obtained. An almost similar trend was observed for molybdenum with 0.12% and 0.273% Mo-rich ingots obtained in routes 3 and 4, respectively. As both nickel and molybdenum can be easily reduced, due to low standard-free energy of formation of oxides (450 kJ/mol O2 for Ni and 661 kJ/mol O2 for Ni), they can be efficiently recovered using IF route. For such processes, a cleaner melt pool made of MS plate gives significantly better result than that of PI.
Recovery of iron, nickel and molybdenum
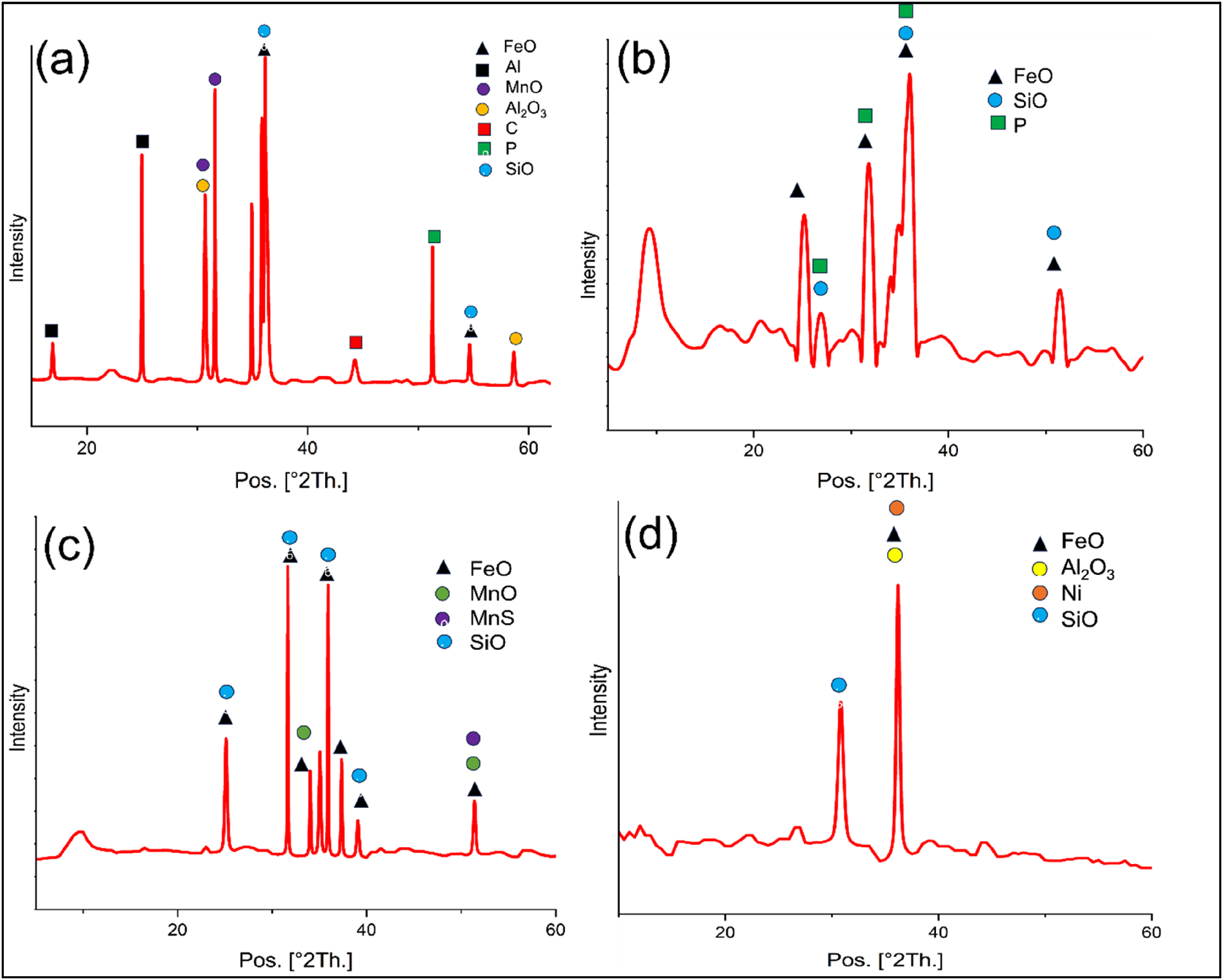
As it is evident from the analysis of obtained ingots, they are rich in Fe, Ni and Mo. The recovery of these elements in all routes is pivotal in establishing the feasibility of the process. The elements present in the ingot come from two sources, that is, mill scale and material used for making melt pool. In the current work, for the purpose of mass balance it has been assumed that the recovery of metals coming directly from melt pool material is 96%, and rest of the material has come from mill scale.
34
This data has been taken from a long-time average of recoveries obtained in induction furnace at Saarloha Advanced Materials Private Limited. Using the assumption, the recovery from mill scale is given by: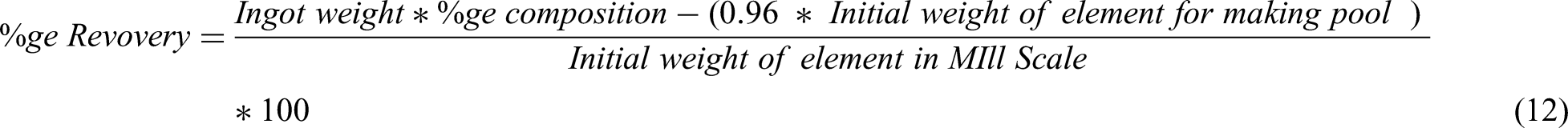
Using the above formula, the recovery for Fe, Ni and Mo has been calculated and given in Table 7.
It can be seen from Table 8 that, the efficiency of reduction in case of route 1 is reduced significantly. This can be because, the melting point of PI is significantly low (approximately 1473 K). Since the mill scale obtains heat from melt pool for its reduction and subsequent melting the melting point plays a crucial role. As the melting point of PI is low, the efficiency of reduction is lowered. The recovery of iron from mill scale improved to 99.36% in route 3 as compared to 79.84% in route 1. As far as nickel and molybdenum is concerned, the recovery has been quite descent routes 3 and 4. The recovery of nickel in route 3 stood at 86.44% in comparison with route 1, where the nickel recovery was 75.52%. Similarly, recovery of molybdenum increased from 65.69% in route 1 to 97.01% in route 3.
Slag composition in various routes.
A rise of 24.44%, 14.45% and 47.47% was observed in recovery of iron, nickel and molybdenum, respectively, in route 3 in comparison with route 1. Hence it can be inferred that, for better recovery of metals from mill scale, iron-rich and carbon-deficient material-based melt pool gives better recovery.
Study of slag in various routes
Slag generated in various routes was segregated as they float at the top of the melt pool because of their lower density. The slags in all routes were analysed for their composition using XRF, which are given in Table 8.
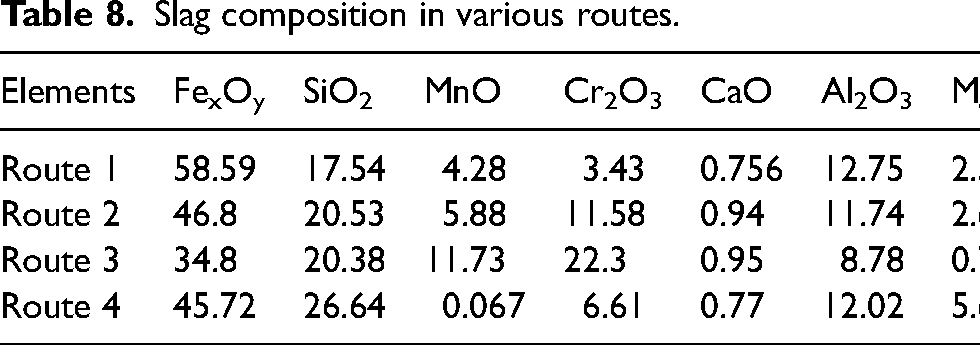
It can be seen from Table 9 that route 1 contains the highest amount of ferrous oxide accounting unreduced iron oxides during melting. The observations also align with recovery results as reported in Table 8. XRD was conducted to evaluate the presence of dominant phases present in slags of various routes which is presented in Figure 7.
XRD pattern of slags obtained in various routes: (a) route 1, (b) route 2, (c) route 3 and (4) route 4. XRD: X-ray diffraction.
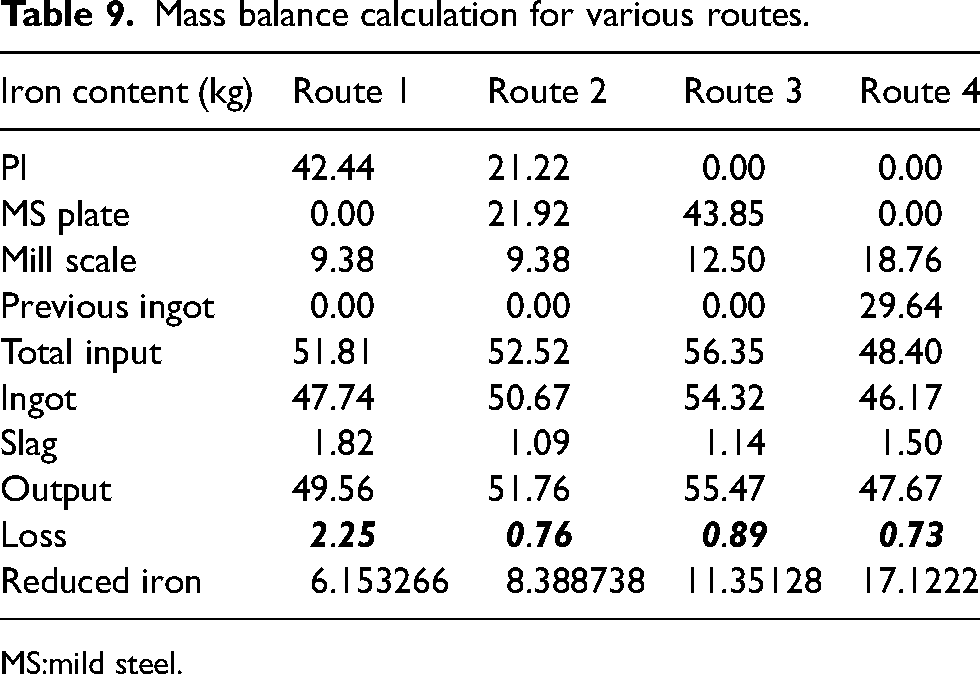
Mass balance calculation for various routes.
mild steel.
It can be observed from the characteristics XRD curves that FeO (00-002-1180) and SiO (00-030-1127) are the dominant phases present in all types of slag. Similar results were reported by earlier researchers like Vaverka et al. 35 and Balamurugan et al. 36 where they found the presence of Wüstite as the major contributing phase in slag. Additionally, it can be seen from Figure 7, that in route 1 there is presence of C (00-001-0640) and P (00-002-0266) phase. This result was expected as a significant portion of carbon coming from PI was unutilised and the same can be said for the presence of P in routes 1 and 2. In routes 3 and 4, apart from FeO (00-002-1180) and SiO presence of Mn (00-001-1234), Ni (00-001-1258) and Mo (00-001-1205) were also found which accounts for the loss of respective metals during melting. Using wet analysis presented in the second section, the quantity of Fe was analyzed in all four routes for calculating final loss of iron. The iron content in routes 1, 2, 3 and 4 were found to be 28.51%, 20.96%, 22.96% and 21.50%, respectively. The mass balance calculation has been given in Table 9.
Table 9 clearly depicts the effectiveness of routes 2, 3 and 4 for recovery of iron from mill scale with minimal loss. It is highly recommended to use IF to process mill scale as a source of iron and other valuable elements (Ni and Mo in the current work) for sustainable production of different steel grade. The proposed methodology of sustainable steel production is shown in Figure 8. There are two possibilities of using mill scale using IF route. Some steel grades can be directly produced by using IF melting. In addition to this, ingots produced from IF can be used in ladle furnace as alloying addition for production of various grades of steel.
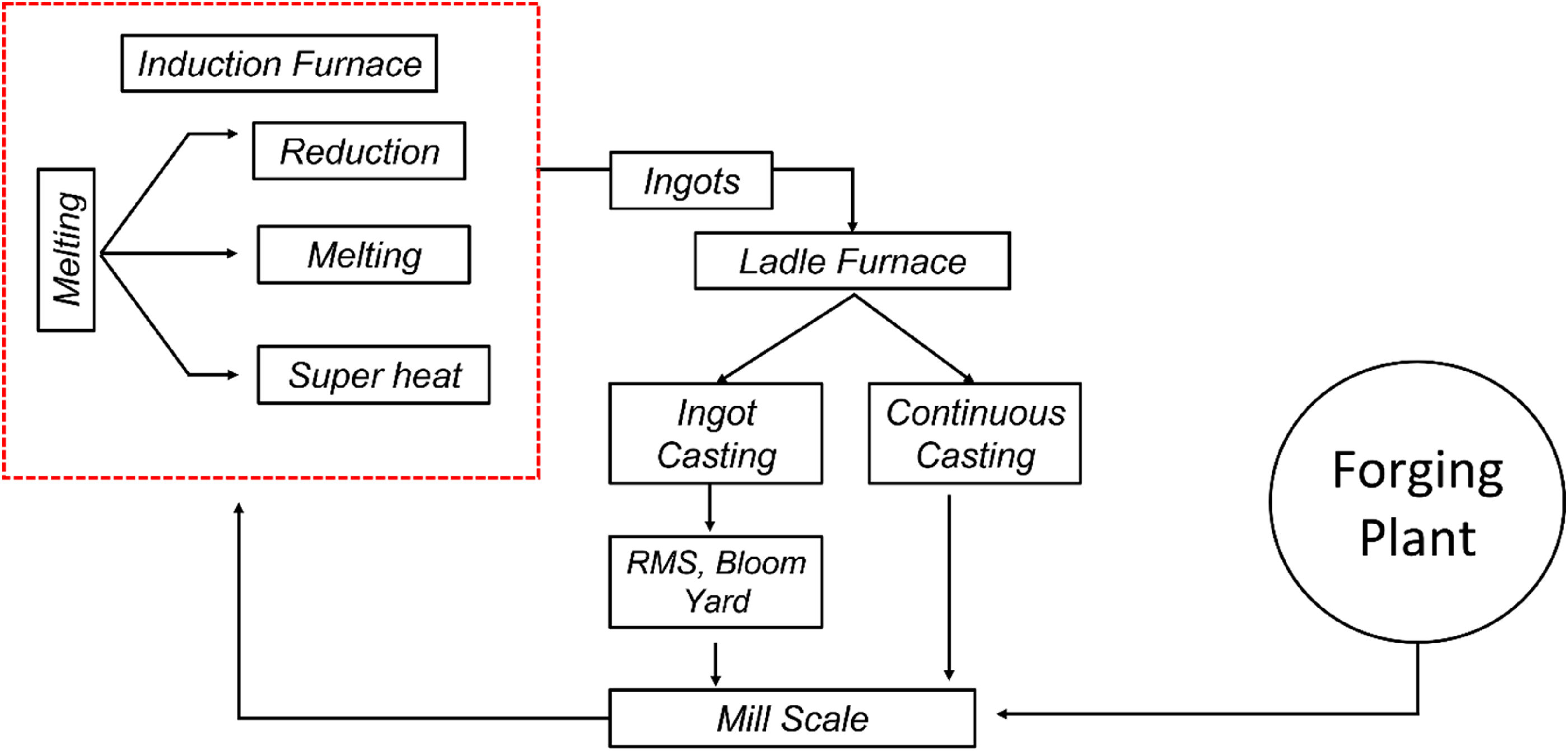
Proposed methodology for mill scale utilisation.
Heat estimation for route 3
It is quite crucial to calculate the energy requirements for melting of charge containing briquette and pool making material. Several attempts have been made by researchers to model the energy consumed in IF.37,38 A simplified heat balance model has been presented to calculate the heat required for melting. The heat is required for making the molten pool and for melting the briquettes. The heat required for making melt pool can be straight forward calculated by the formula: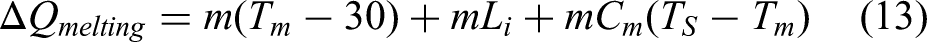
It is worth noting over here that the material used for making melt pool contains some amount of carbon which on combining with oxygen can give some heat due to the exothermic nature of the reaction. However, this value of heat has not been considered due to negligibly small amount of C loss observed during making of initial melt pool. Additional heat is required for melting of briquettes. For calculating the heat involved in the process, it is most crucial to define the underlying mechanism of the processes taking place during melting in a chronological fashion. The proposed mechanism of melting is shown in Figure 9.
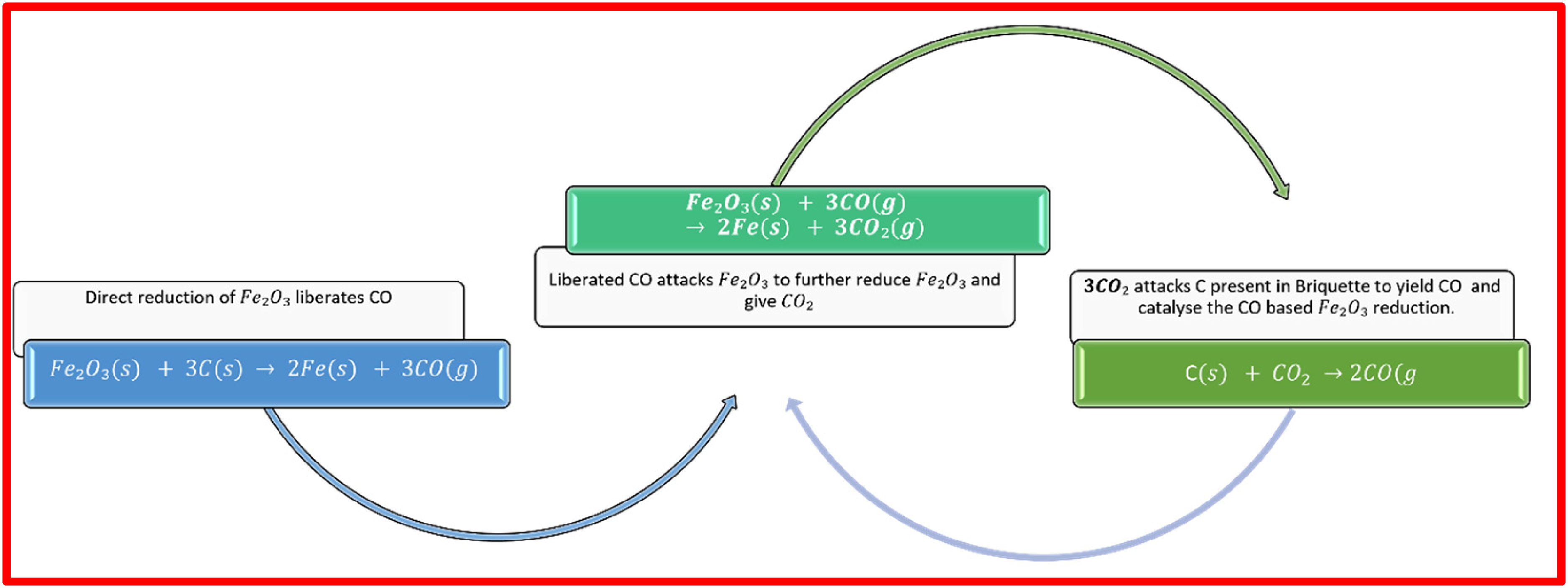
Proposed mechanism of reduction and melting in induction furnaces (IF).
The reactions taking place in the IF for reducing of briquettes are given by: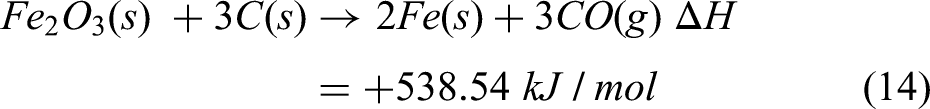
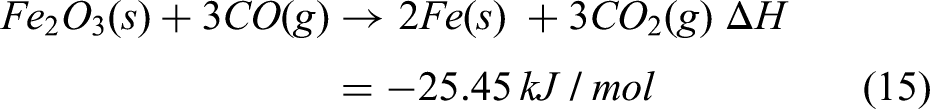
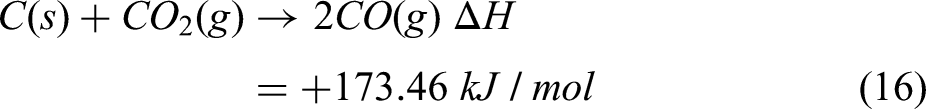
From TGA analysis, it is quite evident that the rate of reduction is most pronounced within the temperature range from 1273 to 1373 K. Above 1273 K, the reduction rate stabilises, signifying complete reduction of oxides and further heat is required to raise the temperature to the melting point. It has been assumed that reactions represented in equations (13) and (15) are contributing to the reduction process which continues till 1373 K. Heat of reduction till 1373 K has been calculated by using equation (16).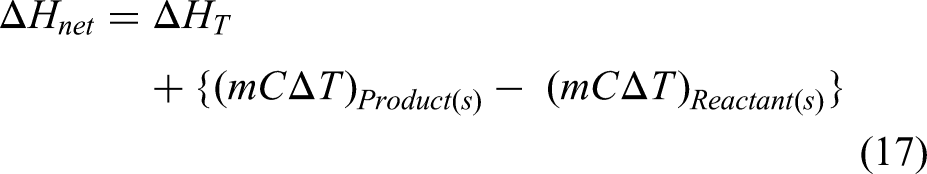
Post 1373 K, Fe further reaches its melting point which will require heat to raise the temperature, followed by requirement of latent heat and superheating. As melting point of steel is affected by amount of carbon present in it, the amount of carbon was assumed to be same as that obtained after tapping. The melting point for this composition was obtained from iron-carbon equilibrium diagram. The thermodynamic data utilised for this model are outlined in Table 10. 39
Thermodynamic data input for the energy requirements model.

The heat required for melting and attaining tapping temperature was calculated using the following formula.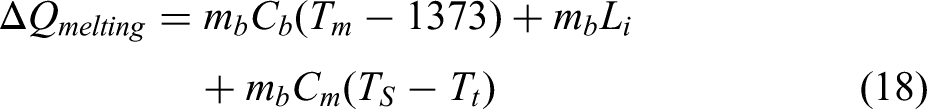
where,
Calculation of energy requirement for route 3.
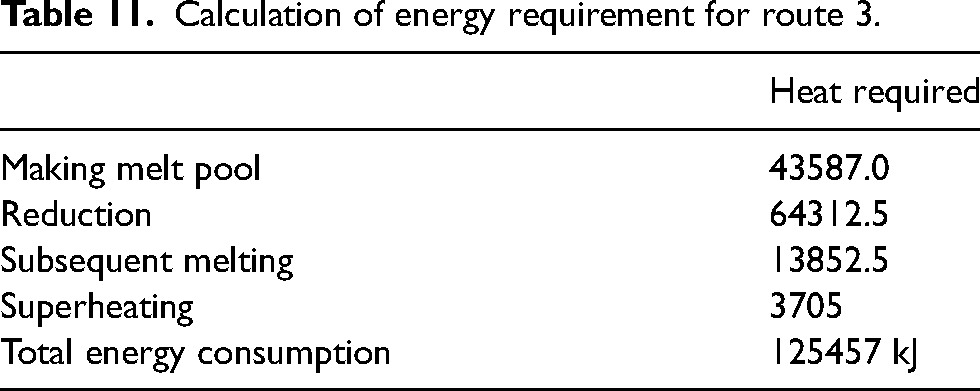
The total energy consumption calculated using above model 125457 kJ which is approximately equal to 35 kWh of electrical energy. It can be understood from above analysis that out of total energy consumed for reduction of briquettes taking place at temperature below 1373 K consumes the largest share of energy required for melting of briquettes. Figure 10 presents a comprehensive distribution of energy consumption in various processes of recycling of briquettes.
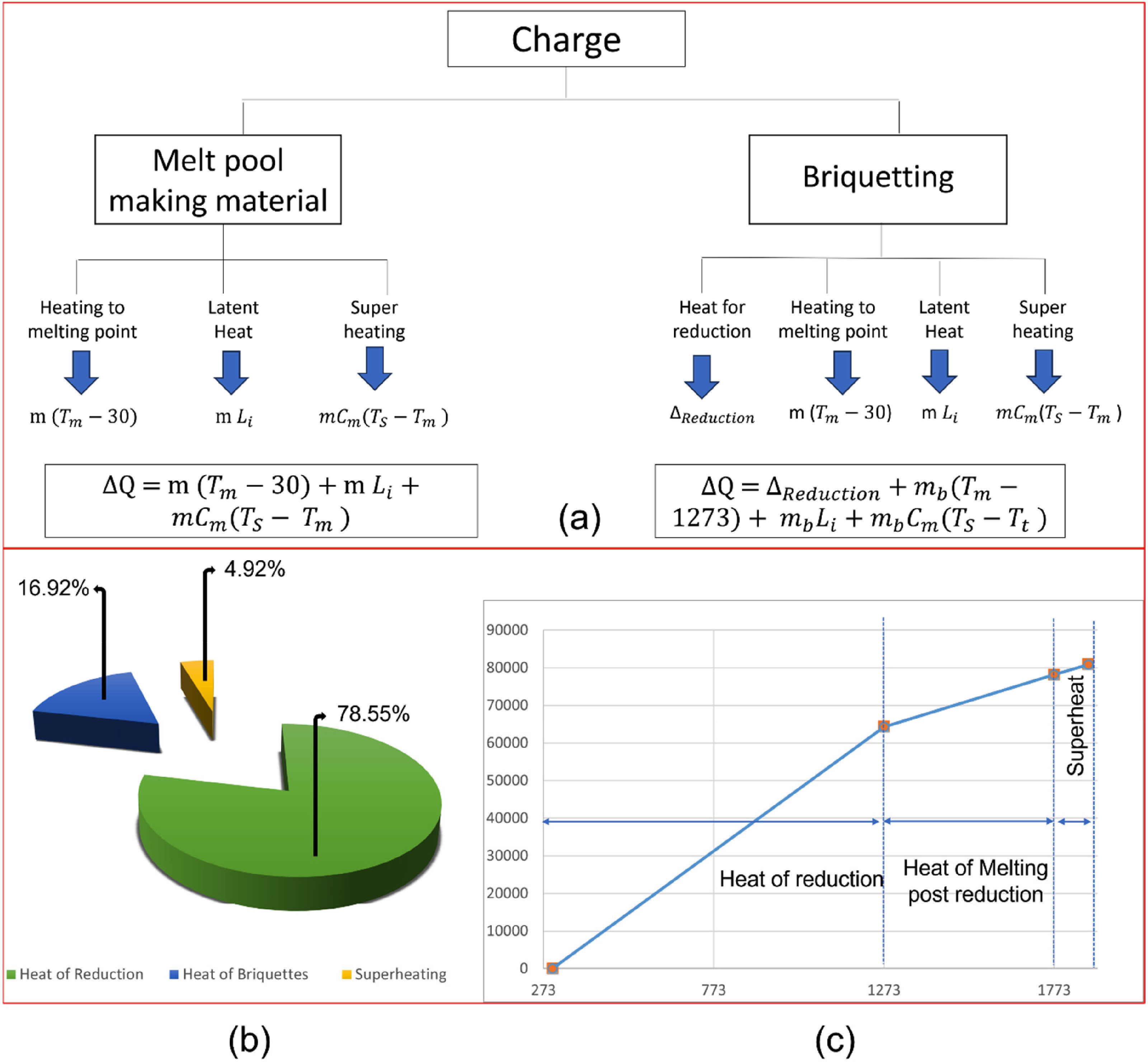
(a) Various energies involved during melting of briquettes in IF; (b) Distribution of energy consumption during recycling of briquettes in route 3 and (c) temperature versus heat consumption for recycling of briquettes in route 3. IF: induction furnaces.
It can be observed that out of total heat consumed for melting briquettes, 78.55% of it goes for reduction, whereas rest of the heat goes to raise temperature of briquette, melting and superheat.
Conclusions
The current work presents a technical study for recycling of mill scale obtained from various steel production processes. The main findings and conclusions of this study are as follows:
Stoichiometric calculations established that 10% to 12% carbon is required for reduction of oxides present in the mill scale which was also backed by TGA analysis. TGA further confirms that the reduction of Fe₂O₃ to metallic iron occurred most efficiently above 1273 K. Induction furnace trials using different routes established that the optimal metal-to-metal recovery was achieved when ratio of mill scale to the material used for creating the melt pool in the IF maintained near unity. The use of MS plates for creating the melt pool proved to be more effective in terms of metal recovery compared to using PI. The recovery of iron, nickel and molybdenum increased by 24.44%, 14.45% and 47.47%, respectively, when using MS plate-based melt pool compared to Pig Iron-based melt pool. The slag generated in every route was studied in terms of composition and phases using XRF and XRD. Dominated by FeO and SiO₂, the slag also contains traces of other elements like Mn, Ni and Mo, indicating metal loss during melting. Heat calculation using a simplified model revealed that, while melting of briquettes a significant portion of energy (∼79%) is consumed for reduction of briquettes.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
