Abstract
Facing the consumption of high-grade iron ore, high-alumina iron ore emerges as a potential alternative for steel companies aiming to cut costs and address resource shortages. This study systematically investigated the impact of increasing the proportion of high-alumina iron ore on sintering performance through sinter pot tests. The detailed microstructural and morphological analyses of the produced sinter phase, combined with the results of thermodynamic calculation on liquid phase formation, elucidated the transformation mechanism of the sinter properties in varying alumina content. The results show that the yield and quality of sinter undergoes a crucial shift at 2.35% content of Al2O3, where yield, drum index, and low-temperature reduction degradation index reach their lowest, indicating a severe degradation. Al2O3 content up to 2.53% proves beneficial for improving sinter reduction index. The major phases found in all resultant sinters by X-ray diffraction are hematite, magnetite, silicate, spinel, and silico-ferrite of calcium and aluminium (SFCA). Increasing Al2O3 content transforms SFCA from acicular to columnar, while porosity exhibits an increasing and then decreasing trend. It is found that the shape of SFCA and porosity are the main factors affecting the quality of sinter. The research findings are helpful to promote the efficient application of high-alumina iron ores in blast furnace ironmaking.
Introduction
As high-grade iron ore resources deplete, turning to low-grade and hard-to-handle iron ore provides an alternative to address this resource scarcity. Currently, high-alumina iron ore (Al2O3 > 2.0 wt.%), which has been readily available in the market, is crucial for steel plants to cut costs and enhance market competitiveness.1–5 The sinter, as a major proportion of blast furnace burden, is therefore expected to have a significant impact on operation, and economic indicators of blast furnace.6–8 The chemical composition and original mineral phase of iron ore play crucial roles in the formation of liquid phase during the sintering process, leading to variations in sinter structure and quality.9,10 Therefore, the utilisation of low-grade iron ores with high-alumina content presents challenges for the sintering process and sinter quality.
The silico-ferrite of calcium and aluminium, known as SFCA phases, are considered the most favourable bonding phases in iron ore sintering due to their excellent characteristics, including high reducibility, stronger mechanical strength, and minimal reduction degradation. 11 These properties play an important role in influencing the productivity and efficiency of the blast furnace. The Al2O3 content significantly influences the formation and morphology of SFCA primarily by participating in its generation. Yang 12 reported that an increase in Al2O3 content enhances SFCA generation, promoting the formation of acicular calcium ferrite characterised by high strength and favourable reduction performance, ultimately improving the sinter quality. However, excessive Al2O3 content within the sinter can alter the SFCA structure from acicular to massive and dendritic. This structural change increases the viscosity of the liquid phase, resulting in decreasing SFCA fluidity and reducing sinter strength. In addition, the aluminium saturated in SFCA is converging towards the glass phase, which increases the content of the glass phase with the worst strength of sinter. Li et al. 13 found that the increase of Al2O3 content increased the viscosity of the sintering liquid phase, and more irregular holes appeared in the sinter, which reduced the strength of the sinter. Umadevi et al. 14 found that when the Al2O3 was 2.00–5.46 wt.%, with the increase of Al2O3 content, the contents of hematite and SFCA in sinter increased, while the contents of magnetite and silicate decreased. Hessien et al. 15 found that the stability of SFCA in sinter depends to a certain extent on the content of Al2O3. When the content of Al2O3 is less than 1.5 wt.%, SFCA will decompose into magnetite and silicate at high temperature; however, when the content of Al2O3 is over 2.5 wt.%, SFCA will exist stably. It is hence very crucial to keep an appropriate blending ratio of high-alumina iron ore to prepare high quality sinter by controlling the quantity and quality of the liquid phase.
The calcium ferrite has yielded two distinct types based on the varying content composition and microstructure. One is high iron and low silicon composite calcium ferrite (SFCA-I), the other is high silicon and high alumina composite calcium ferrite (SFCA).16,17 SFCA-I is formed by the reaction of C2F(2CaO·Fe2O3) with a certain content of Al3+ in solid solution with SiO2 and Fe2O3, and the microstructure is usually dendritic and columnar. The generated glass phase decreases the strength of sinter. 18 Yu et al. 19 pointed out that the increase of Al2O3 content would lead to an insufficient supply of heat in the sintering process, and the formation reaction of calcium ferrite could not fully develop in the direction of equilibrium. In addition, the increase of liquid viscosity and insufficient expansion of liquid phase would lead to the increase of unreacted cores without mineralisation and the decrease of sinter strength.
The increasing Al2O3 content of sinter has brought impacts on the reduction index (RI) of sinter.20–22 In a lower range of Al2O3 content, the increase of alumina fosters the formation of acicular calcium ferrite, which exhibits better reducibility compared to columnar SFCA. Conversely, excessive Al2O3 leads to a shift in calcium ferrite structure from acicular to columnar. In addition, during the reduction of sinter, larger porosity is conducive to promoting the internal diffusion of reducing gas, increasing the gas–solid reaction area, and promoting the reducibility of sinter. The porosity of the sinter is also affected by the Al2O3 content in the sinter.23,24 With the increase of the Al2O3 content, the proportion of low-quality iron ore in the sintering raw materials increases, so the burning loss increases. The more pores generated by the burning loss; the better reducibility of the sinter. As the Al2O3 content continues to increase, the bonding liquid phase formed in the sintering process increases and the viscosity increases, resulting in increased pore closure of the sinter and decreased porosity of the sinter and further inhibition of sinter reduction.25,26
As the Al2O3 content increases, the low-temperature reduction degradation index (RDI) of the sinter significantly deteriorates. This phenomenon is primarily attributed to the resisting stress changes in hematite and alterations in the bonding phase strength. The higher Al2O3 content leads to an enrichment of alumina in the glass phase, causing reduced toughness and, ultimately, the lower fracture toughness of the glass phase. Resisting stress changes proves challenging, and the expansion stress resulting from crystal transformation of hematite during low-temperature reduction concentrates within the glass phase. This concentrated stress leads to the formation and expansion of numerous cracks, significantly exacerbating the degradation of RDI.21–23 Guo et al. 27 observed that increasing Al2O3 content promotes the formation of columnar crystal composite calcium ferrite, which exhibits low toughness and is prone to fracture, resulting in a poor RDI of the sinter. Inazumi et al. 28 believed that the phenomenon causing poor RDI is related to the micromorphology of hematite. Studies have pointed out that the low-temperature reduction of sinter and the regeneration of hematite are the main factors leading to the degradation, and the regeneration of hematite often contains certain solid-solution inclusions and silicate impurities. Many metallurgists attribute the microstructure of regenerated hematite as the main reason for the low temperature reduction degradation of sinter.29,30
The physicochemical and metallurgical properties of sinter with high-alumina content have a significant impact on the stable operation of blast furnace. However, it is still not clear that the transformation rules of the structures and properties, the liquid phase amounts, as well as the morphologies of the bonding phase in sinter with varying Al2O3 contents aroused by increasing the proportion of high-alumina iron ores. Therefore, this study systematically investigates the impact of increasing the proportion of high-alumina iron ores on Al2O3 contents and metallurgical properties in the produced sinters via sinter pot tests. The changes of phases in the sinters and morphologies of SFCA are also analyzed by X-ray diffraction (XRD) and scanning electron microscopy (SEM). The liquid phase amounts are further calculated using FactSage software. The findings contribute fundamental insights to enhance cost-effective blast furnace ironmaking with high-alumina iron ores.
Materials and methods
Raw materials
The chemical compositions of raw materials, including seven types of iron ores, return fines, blast furnace dust, fluxes (i.e. limestone), and coke breeze, are presented in Table 1. Iron ores A, D and F are sourced from Australia, while ores B and C are imported from Brazil. Ores E and G constitute domestic iron ores, with G being utilised sparingly at the production site due to their inferior quality. Ores A–F are identified as the mainstream iron ores for sintering, and the sequence of Al2O3 content from highest to lowest is A > D > F > C > B > E. The ores from Australia (A, D, F) powder exhibits the high Al2O3 content among the imported ones with over 2 wt.%. Specifically, ore A served as the primary ore powder in this study to elevate the Al2O3 content in sinter. Fluxes are adapted to adjust the basicity of the sinter and Al2O3/SiO2 mass ratio. Coke breeze acts as the only solid fuel to provide sufficient heat for sintering.
Chemical compositions and loss on ignition of the adopted raw materials (wt.%).
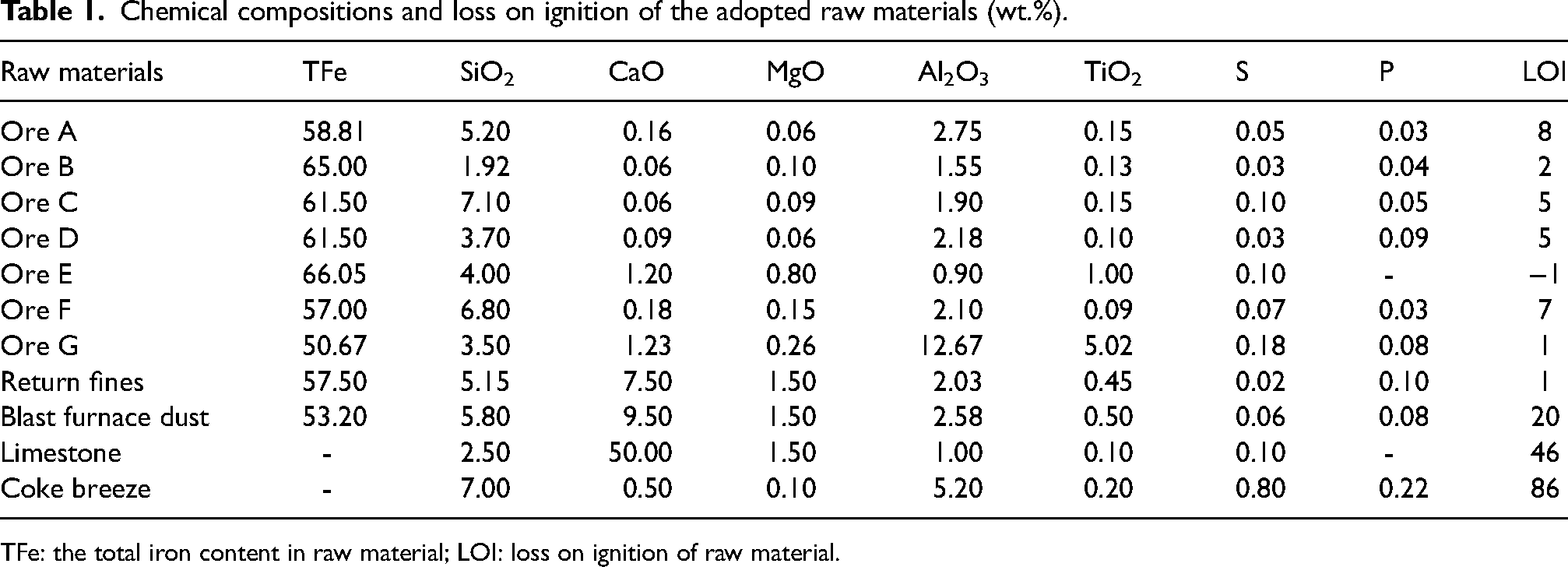
TFe: the total iron content in raw material; LOI: loss on ignition of raw material.
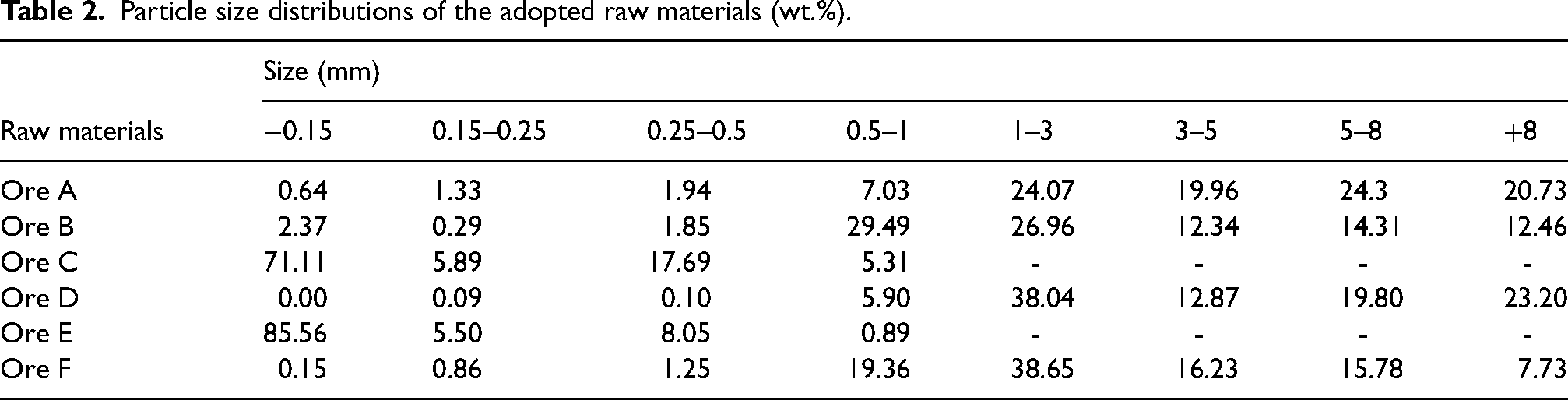
The particle size distributions of the main iron ores are shown in Table 2
Particle size distributions of the adopted raw materials (wt.%).
Sinter pot test and performance testing of the produced sinter
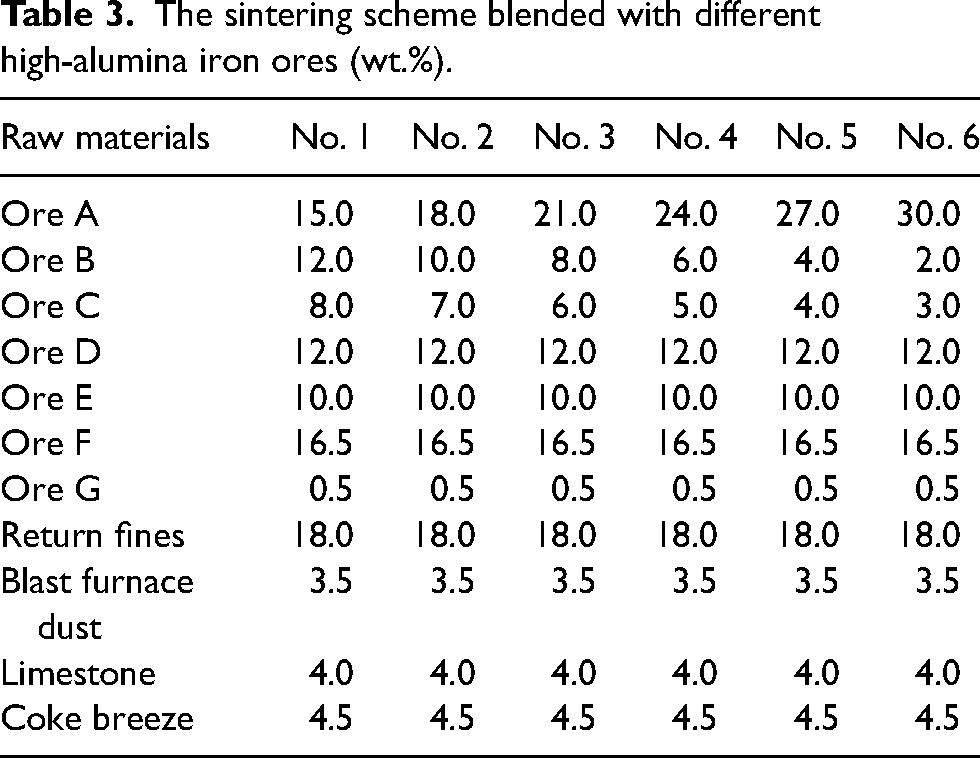
Sintering pot tests were carried out in a pilot scale pot with 300 mm in diameter and 700 mm in height. In the experiment, the test scheme adopts the material ratio employed in on-site sintering at a steel enterprise as the base case to investigate the impact of Al2O3 content on sintering behaviour and performance. The sintering scheme and detailed sintering process parameters are shown in Tables 3 and 4. By increasing the amount of iron ore A, and reducing the proportion of ores B and C. Fluxes are used as a binder to improve the granulation of sinter mixture and to adjust the basicity and Al2O3/SiO2 ratio of sinter mixture to remain around 1.9 and 0.44, respectively. In Table 3, the total percentage of the iron-bearing materials (ores A–G, return fines, blast furnace dust) and coke breeze as solid fuel is equal to 100%, while the limestone as fluxes is added by way of an additional 4%.
The sintering scheme blended with different high-alumina iron ores (wt.%).
Sintering process parameters in sinter pot test.

The study analyzed various sintering indexes, including sinter speed, sinter yield, tumbler index, and productivity. These indexes were assessed based on the Chinese national standards and metallurgical industry standards (YB/T 421-2014, GB/T 24531, and ISO 3271-2007). The metallurgical performances including RDI and RI are detected according to the international standards of GB/T 13242-2017 and GB/T 13241-2017.
Characterisation
The phase identification and relative content of the samples were investigated by an X-ray diffractometer (D/max2500, Rigaku), coupled with a monochromator and a copper Kα X-ray source. 40 kV accelerating voltage and 150 mA accelerating current were used. The scanning range was 10°–90° with 8°/min scanning speed. The microstructure of the produced sinter samples was demonstrated by means of the optical microscope (OM, Primostar 3, Zeiss) and SEM (JSM-7800F, JEOL). The SEM instrument was equipped with an energy disperse spectroscopy detector (EDS, 80 mm2 X-MaxN Silicon Drift), and 15 kV accelerating voltage was selected for the microanalyzer operation.
Liquid and alumina containing phases calculation
According to the main composition of sinter, the amount of liquid and alumina containing phases, and the liquid phase viscosity of the 10.95%CaO-5.76%SiO2-X Al2O3-9.00%FeO-(71.39-X)Fe2O3 system (basicity(
Results and discussion
Chemical composition and phase transformation of the produced sinter
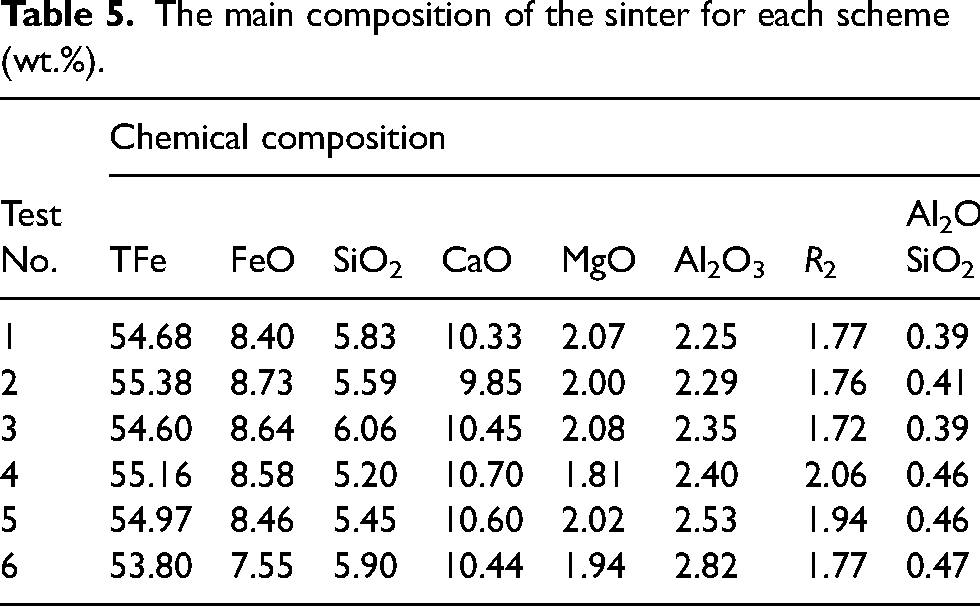
Table 5 shows the main composition of the sinter for each scheme. It can be seen that the chemical composition of the produced sinter has changed with an increasing proportion of high-alumina ore A, especially in Al2O3 content, binary basicity (
The main composition of the sinter for each scheme (wt.%).
The distinct phases exert noticeable effects on the sintering process and properties of the sinter. XRD analysis of the produced sinter with varying Al2O3 contents is depicted in Figure 1. Predominant phases in the sinter comprise hematite, magnetite, silicate, spinel, and SFCA, with SFCA primarily existing in the form of hedenbergite. No new diffraction peaks emerge with increasing alumina content, signifying the absence of new phases. As the Al2O3 content increases to 2.40%, the peak intensity in the sinter remains relatively stable. Maximum X-ray diffraction peak intensity occurs at an Al2O3 content of 2.53%, reflecting the varied intensity primarily due to changes in the proportion of each phase in the sinter.
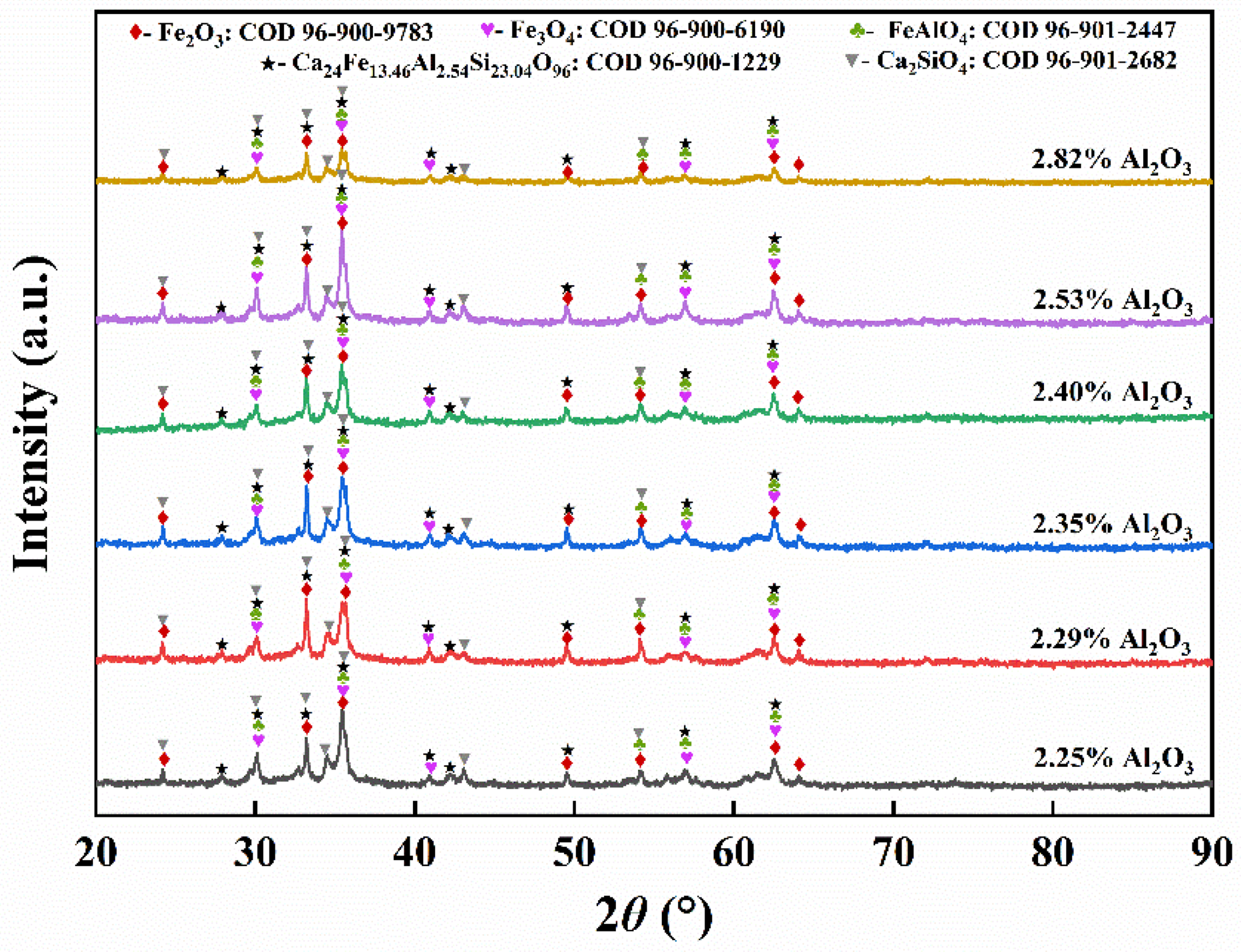
XRD patterns and mineral phases of sinters with different Al2O3 contents.
Microstructure and morphology of the produced sinter
Figure 2 shows the optical microscopic image and porosity of sinter with different Al2O3 contents. It can be observed that the sinter has a large pore structure at a high Al2O3 content, and the porosity of the sinter increases first and then decreases with the increase of Al2O3 content. When the Al2O3 content is 2.83 wt.%, the proportion of a mineral powder in the raw material of the sample increases to 30%, the crystalline water content of the sintered mixture increases, and the LOI increases from 10.9% to 11.7%. The porosity should have continued to increase, but the increase of Al2O3 content will lead to an increase in the viscosity of the liquid phase of the sinter, and the liquid phase flow is difficult, blocking the holes generated by the LOI, resulting in a lower porosity.

Optical microscopic image and porosity of sinter with different Al2O3 contents.
The main mineral phase component of sinter is calcium ferrite, magnetite, hematite and silicate, which contribute for 80–90% of the total mineral content of sinter. The microstructure of sinter SFCA with varying Al2O3 contents is depicted in Figure 3. At an Al2O3 content of 2.25 wt.% (Figure 3(a)), SFCA predominantly exhibits a needle-shaped structure, with bright white hematite present in proximity to the holes and cracks. When the Al2O3 content is 2.29 wt.% (Figure 3(b)), the predominant formation is dendritic SFCA, accompanied by a few of needle-like SFCA and silicate phases appearing in the intersecting gaps of the dendrites. At an Al2O3 content of 2.35 wt.% (Figure 3(c)), dendritic SFCA and a substantial amount of magnetite intertwine, and some SFCA takes on a sheet-like form, enveloping the magnetite. With further increase in Al2O3 content to 2.40 wt.% (Figure 3(d)), the micromorphology of SFCA becomes noticeably coarse, transitioning into columnar SFCA. The gaps within these columnar crystals are filled with silicate phases, characterised by low reduction properties, which are not conducive to the RDI. Upon reaching an Al2O3 content of 2.53 wt.% (Figure 3(e)), SFCA and magnetite exhibit a columnar shape, with partial fusion resulting in the appearance of sheet-like SFCA. Finally, at an Al2O3 content of 2.82 wt.% (Figure 3(f)), the columnar SFCA and a significant amount of magnetite interact and degrade each other. The gaps between the columnar SFCA show a reduced presence of the silicate phase compared to Figure 3(d), indicating a continued growth of columnar crystals and an increase in magnetite.
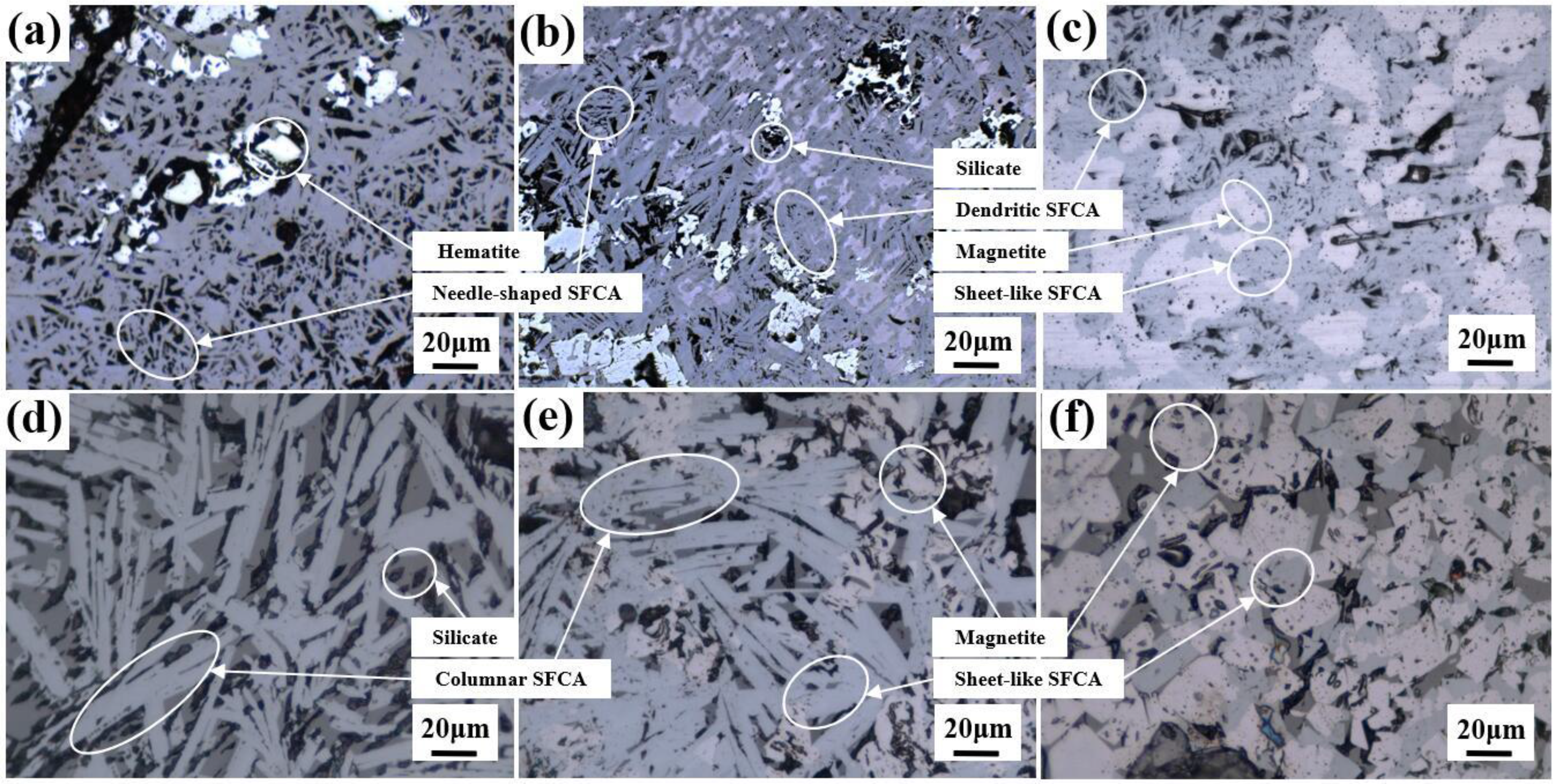
Microstructure of SFCA in resultant sinter with different Al2O3 content by SEM analysis.
SEM-EDS analysis was employed to investigate the microstructure of sintered samples with varying Al2O3 contents, and the findings are presented in Figure 4. The light grey silicates (P3, P5) are characterised by higher alumina content when compared to SFCA. As the Al2O3 content increases, there is a gradual rise in the alumina content within the SFCA, leading to a coarser structure in these SFCA formations. Notably, at an Al2O3 content of 2.53 wt.%, the microstructure of the sinter becomes highly intricate, posing challenges in distinguishing distinct phase regions.
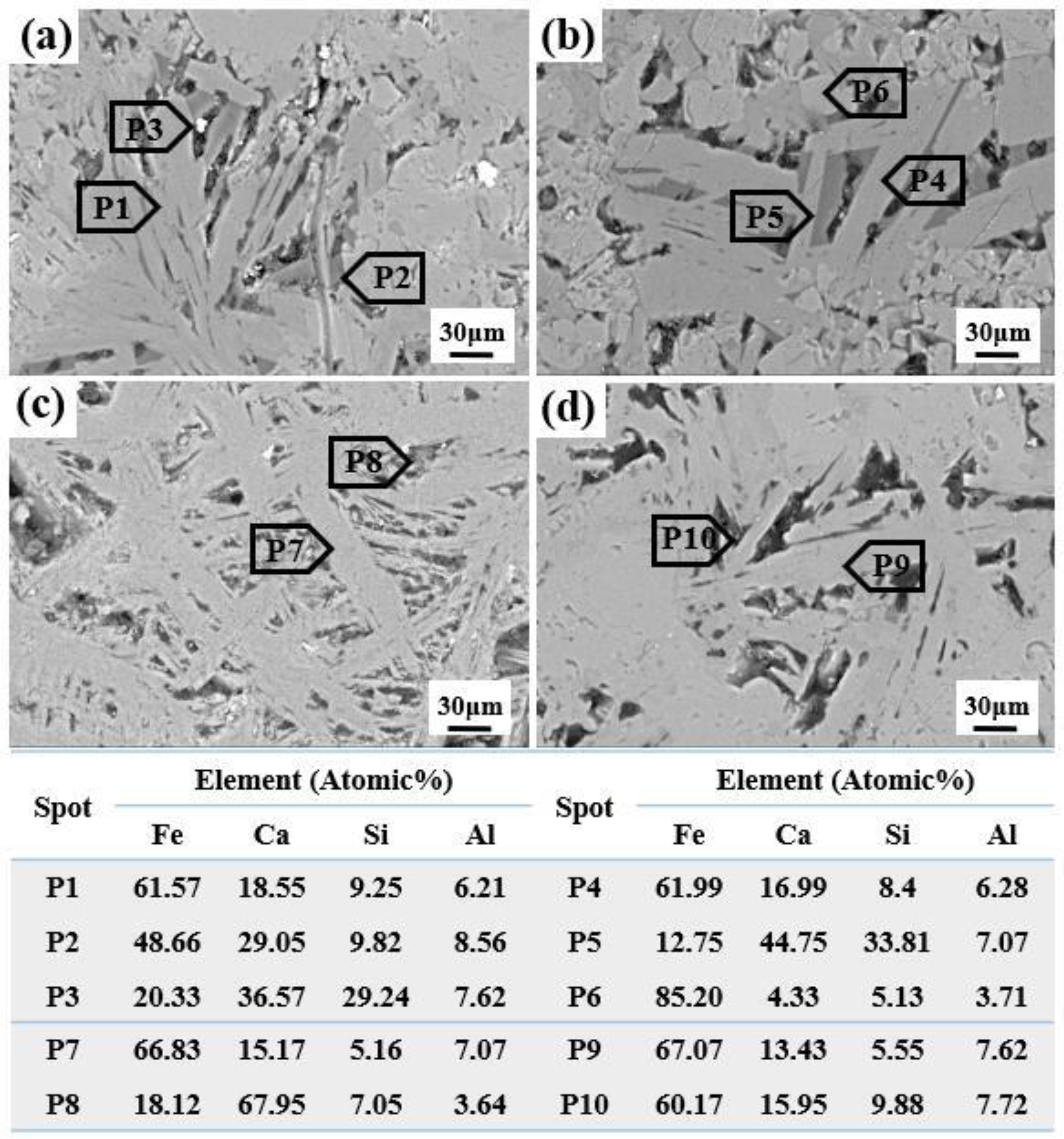
Microstructure of sinter with different Al2O3 contents obtained by SEM-EDS analysis.
According to the results above, the increase of Al2O3 content leads to a simultaneous increase in SFCA and hematite content, while maintaining a relatively high porosity of the sinter, increasing the reducibility of the sinter. However, when the Al2O3 content is over 2.53 wt.%, the reducibility of the sintered ore starts to decline. This can be attributed to a decrease in the content of hematite and porosity within the sintered ore. In general, SFCA with higher iron contributes to enhanced quality in the resulting sinter product, leading to increased strength and reducibility.17,31 Porosity is also a crucial factor in the reduction of the sintered ore. 32 In addition, the larger grain size of the magnetite makes it more challenging to reduce. Moreover, the reduced content of hematite in the sintered ore contributes to a decrease in its reducibility. The content of aluminium-containing spinel, silicates, and hematite phases increases, which are known for having lower strength in the sinter. Notably, silicates, which expand by approximately 10% in volume at 657 °C, and hematite, being a primary phase in the sinter with RDI. Therefore, the increased content of spinel, silicates, and hematite is a significant factor exacerbating the phenomenon of low-temperature reduction degradation in the sinter.
Effect of increasing the proportion of high-alumina iron ore on sintering indexes
The particle size distribution and average particle size of the produced sinters with variable Al2O3 contents are given in Figure 5. As the Al2O3 content increases from 2.25 to 2.35 wt.%, there is a gradual decrease in the average particle size, primarily attributed to the decrease in the proportion of sinter with a particle size of more than 40 mm. However, once the Al2O3 content exceeds 2.35 wt.%, an upward trend in the average particle size is observed. Figures 1 and 2 addressed the impact of Al2O3 content on the phase content and porosity of the sinter. At an Al2O3 content of 2.35 wt.%, both silicate and alumina spinel reach their maximum values. Simultaneously, the sinter demonstrates its maximum porosity. These factors collectively contribute to a reduction in its strength and a decrease in the proportion of particle sizes exceeding 40 mm. Figure 6 illustrates the variation of tumble index, yield and vertical sintering speed with the increasing Al2O3 content in sinter. It can be seen that when the Al2O3 content increases to 2.35 wt.%, the drum index and yield of sinter deteriorate sharply; after 2.35%, the drum index and yield increase and remain around 68.5% and 82.5%, respectively. While the vertical sintering speed shows an opposite trend.
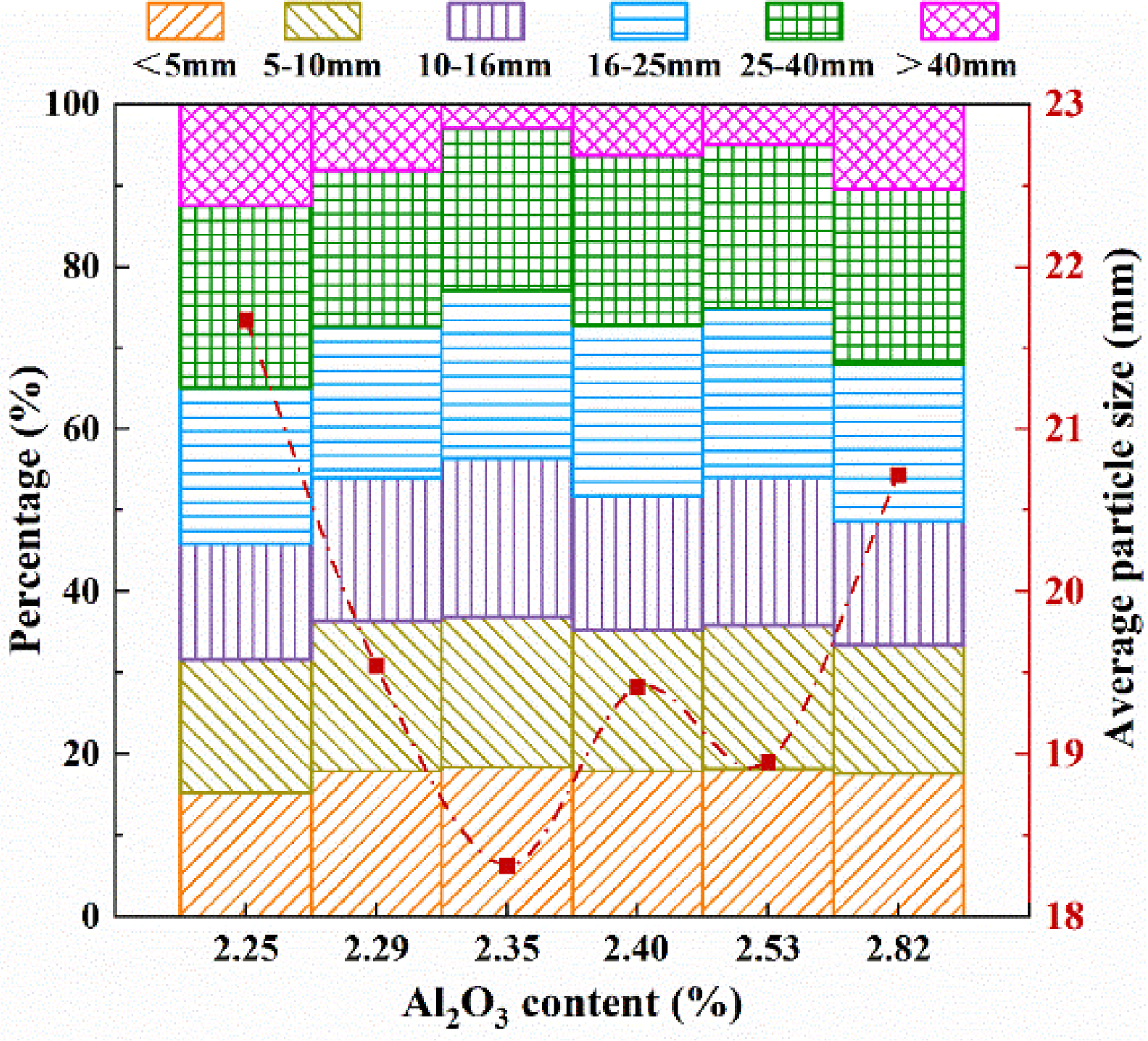
The particle size distribution and average particle size of the produced sinter with variable Al2O3 contents.
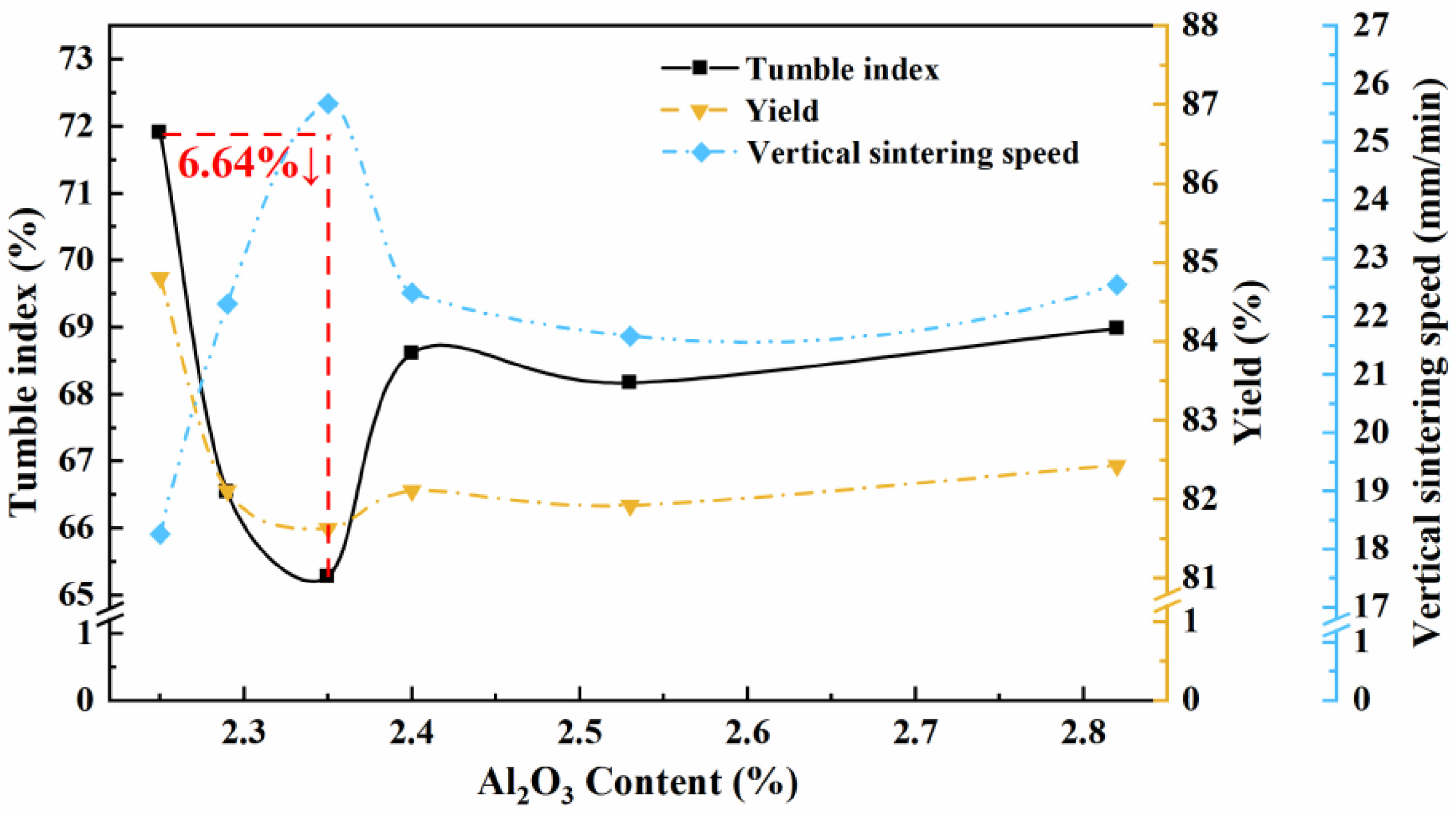
Variation of tumble index, yield and vertical sintering speed with Al2O3 content of sinter.
Effect of increasing the proportion of high-alumina iron ore on metallurgical property
The metallurgical properties (RI and RDI) of sinter at different Al2O3 content are presented in Figure 7. As the Al2O3 content increases, the RI of the sinter varies within the range of 89.1% to 93.4%, demonstrating an initial increase followed by a subsequent decrease. The maximum reduction degree occurs when the Al2O3 content is at 2.53 wt.%. The increase in Al2O3 content leads to an increase in SFCA and hematite content, concurrently maintaining a higher porosity in the sinter, resulting in a continuous rise in RI. However, the RI of the sinter decreases after the Al2O3 content is over 2.53%. On one hand, this can be attributed to the decrease in the content of hematite and porosity in the sinter. On the other hand, the results of the phase analysis (Figure 4) of the sinter explain that the magnetite crystal grains are larger and connected end-to-end, complicate the reduction of larger magnetite particles or limit it to superficial reduction. For the RDI, there is a significant decrease when the Al2O3 content reaches 2.35 wt.%. Combined with changes in the phase content of the sinter (Figure 1(b)), the content of spinel, silicate and hematite increases in the sinter. Spinel and silicate have low strength in the sinter and are prone to areas where cracks propagate (the volume expansion of silicate is about 10% at 657 °C). Hematite is the main factor of RDI. When hematite is reduced to magnetite, the structure changes from the trigonal to the equiaxed. Stress resulting from this expansion exacerbates the degradation of the sinter. Hence, the increased content of spinel, silicate, and hematite significantly contributes to the worsening of the RDI of the sinter. However, after the Al2O3 content surpasses 2.40 wt.%, the sinter RDI begins to improve and eventually stabilises.
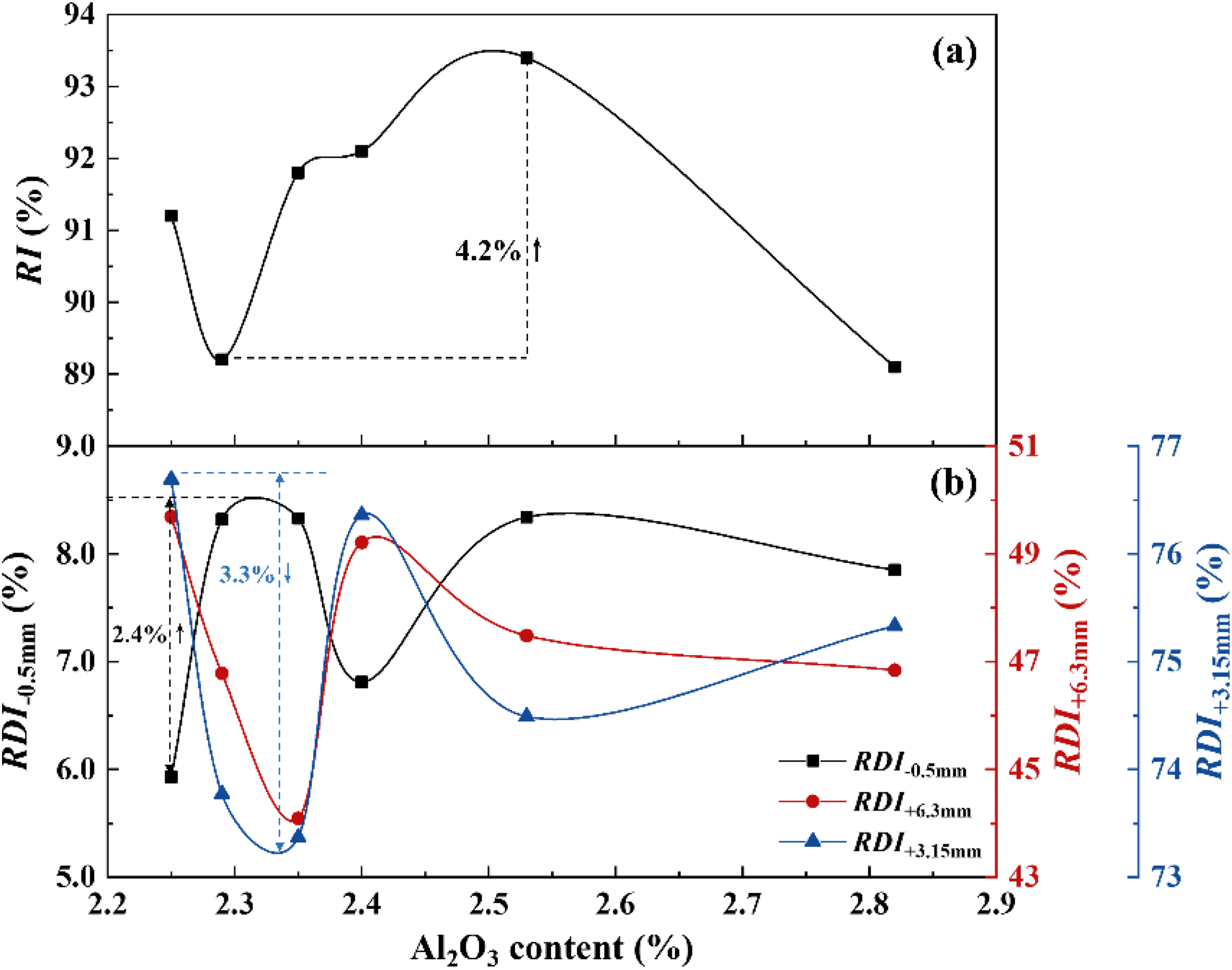
Variation of RI and RDI of the produced sinter with different Al2O3 contents.
Effect of Al2O3 content on liquid phase formation of iron ore blending in high temperatures
In the sintering process of iron ore, the formation of liquid phases is the basis of agglomeration, which affects the mineral phase composition and microstructure of the produced sinter. Al2O3 as the key composition of liquid phases, influences structure and properties of sinter. Thus, the effect of Al2O3 content on the liquid phase amount and its viscosity is calculated by FactSage software, shown in Figure 8.
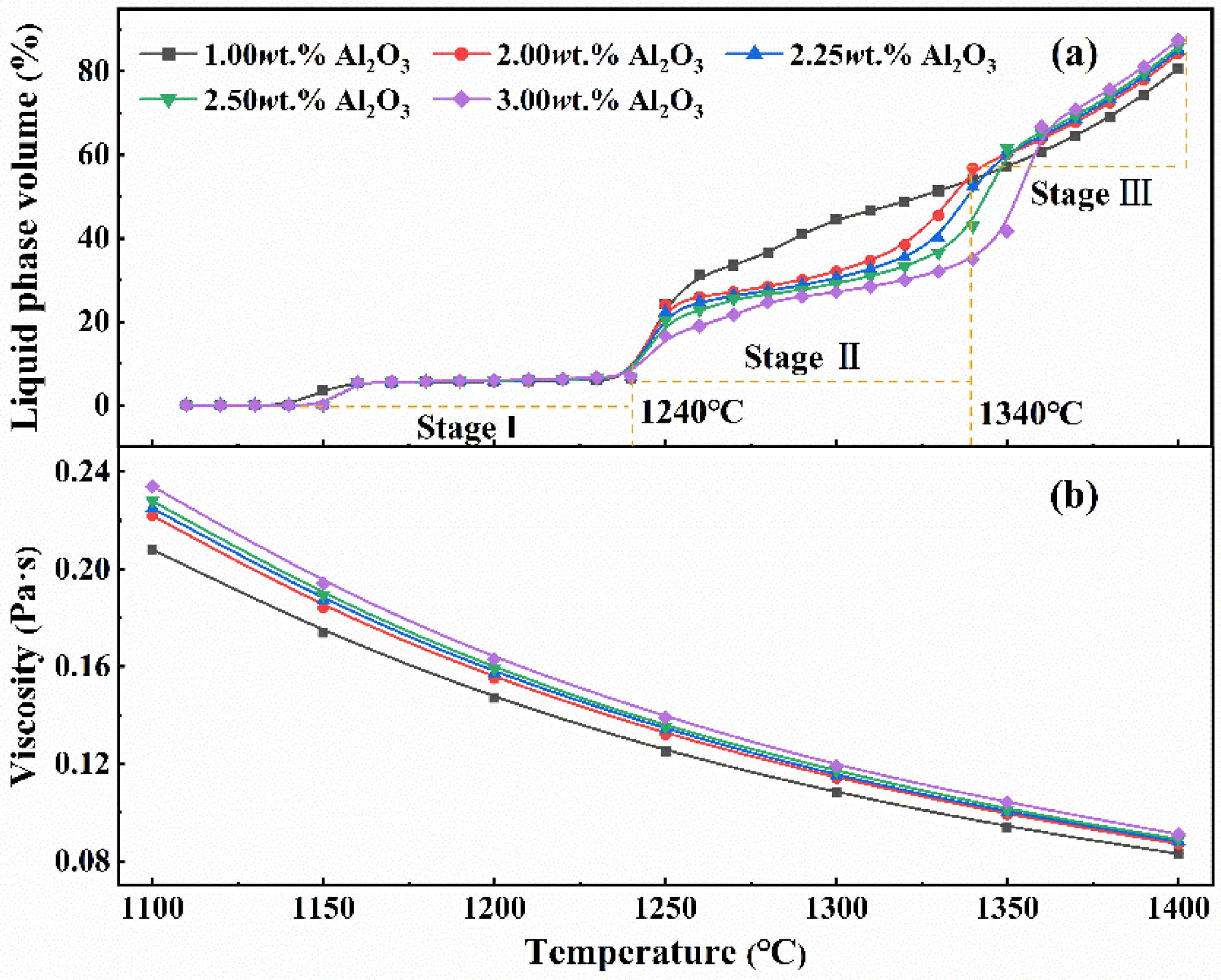
Effects of Al2O3 content on the volume and viscosity of liquid phase in sintering process.
From Figure 8(a) it can be seen that with the increase of temperature, the change of liquid phase volume in sinter is divided into three stages. In the first stage with temperature ranging from 1140 °C to 1240 °C, no significant difference is observed in the amount of liquid phase. In the second stage, as the temperature ranges from 1240 °C to 1340 °C, the increase in Al2O3 causes a decrease in the amount of liquid phase. In particular, the liquid phase is remarkable reduced when Al2O3 increases from 1% to 2%. When the temperature exceeds 1340 °C, stage III, the increase in Al2O3 causes an increase in the amount of liquid phase, although not markedly significant. It is well known that the sintering temperature of iron ore is the same as the second stage ranging from 1240 °C to 1340 °C in the actual sintering process, the maximum of liquid phase volume is around 50%, which is in agreement with that given in the literature. 33 In addition, the liquid volume increases with increasing temperature. Therefore, in the process of blending sintering using high-alumina iron ore, the proper increase of sintering temperature is favourable to promote the formation of liquid phase.
From Figure 8(b) we can further find that the liquid viscosity increases with the increase in Al2O3 content. This is attributed to Al2O3 in the slag usually acts as a network former, it increases the degree of structural polymerisation of the slag, increasing the viscosity of the liquid phase.34,35 A reduction in liquid phase fluidity hampers pore filling between raw materials. After solidification, the sinter displays weak bonds and irregular holes, compromising its strength and RDI.
The variation in the percentage of aluminium-containing phases at different Al2O3 content with temperature is illustrated in Figure 9. The content of Ca2Al2SiO7 in feldspar phases (Figure 9(a)) shows a decreasing trend with increasing temperature, while Fe2Ca2AlO7 in feldspar phases (Figure 9(b)) initially increases and then decreases. Notably, an observable change occurred at 1240 °C, especially for the sinter with an Al2O3 content of 1.00%, the content of Ca2Al2SiO7 and Fe2Ca2AlO7 decreased rapidly. When the Al2O3 content is over 1.00%, the decline is relatively gradual. Moreover, as the Al2O3 content increases, the rate of decrease moderates. The content of FeCa2Al2O7 in the feldspar phase (Figure 9(c)) increases with the rise in Al2O3 content. The attenuated decrease in Fe2Ca2AlO7 content suggests that Al2O3 inhibits the involvement of Ca2Al2SiO7 and Fe2Ca2AlO7 in the formation of the liquid phase, thereby stabilising their presence.
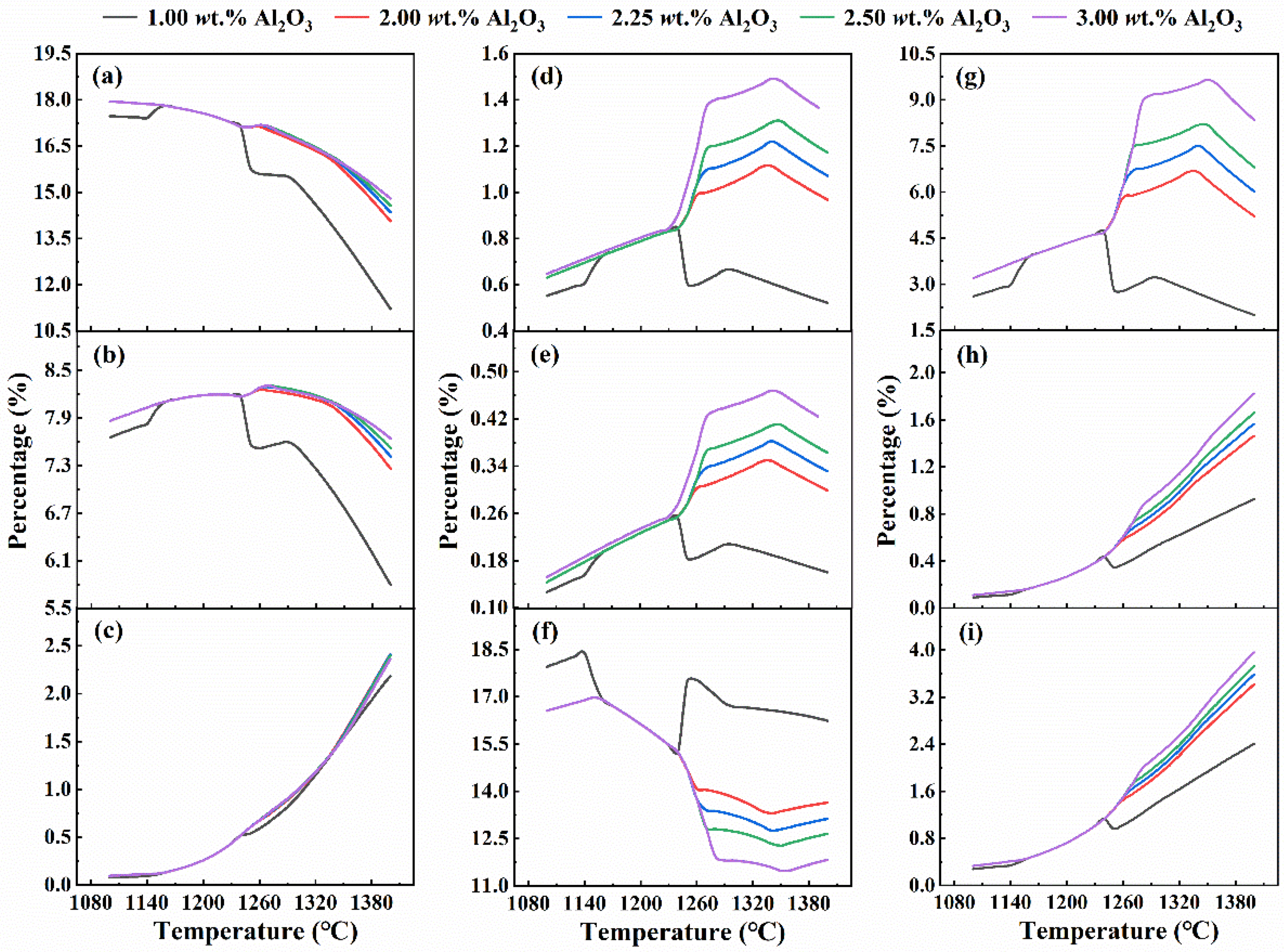
The aluminium-containing phase content changes with temperature under different Al2O3 contents. (a) Ca2Al2SiO7, (b) Fe2Ca2AlO7, (c) FeCa2Al2O7, (d) MgAl2O4, (e) Fe2AlO4, (f) FeAl2O4, (g) CaAl2SiO6, (h) CaAlSi2O6, and (i) FeCaAlSiO6.
Among MgAl2O4 (Figure 9(d)), Fe2AlO4 (Figure 9(e)), and FeAl2O4 (Figure 9(f)), the proportion of FeAl2O4 undergoes the most substantial transformation. As the Al2O3 content in the sinter increases, the spinel phases MgAl2O4 and Fe2AlO4 experience an escalation during the sintering process, whereas the proportion of FeAl2O4 diminishes. This indicates that Al2O3 plays a pivotal role in promoting the engagement of FeAl2O4 in the formation of novel low-melting-point liquid phases.
The figures depict the variations in content for three pyroxene phases: CaAl2SiO6 (Figure 9(g)), CaAlSi2O6 (Figure 9(h)), and FeCaAlSiO6 (Figure 9(i)). The increase in Al2O3 content inhibits the decreasing trend of CaAl2SiO6 while promoting the formation of CaAlSi2O6 and FeCaAlSiO6. As a result, CaAl2SiO6, which should participate in the solid phase reaction to form a liquid phase, exist more stably during the sintering process, thus reducing the amount of liquid phase.
Based on the above results of liquid phase calculation and microstructure characterisation, as well as sinter properties measurements, it will be inferred that the morphology of SFCA in liquid phase and pore structure are the main factors affecting the properties of sinter, instead of the amount of liquid phase.
Conclusions
The effect of different alumina content on sintering property was systematically explored through sinter pot tests. The pertinent mechanisms were elucidated through mineralogical analyses. The influence of Al2O3 on the production quality indicators and metallurgical property of sinter undergoes a significant shift when Al2O3 content is at 2.35 wt.%. Yield, drum index, and RDI are at their lowest at an Al2O3 content of 2.35 wt.%, indicating a severe degradation of sinter quality. Al2O3 content within 2.53 wt.% is advantageous for enhancing sinter RI, showing an overall decrease and then increasing trend. Characterisation analysis of microstructure and morphology reveals that major phases in sinter include hematite, magnetite, silicate, spinel, and SFCA. The increase in Al2O3 content prompts the development of composite SFCA from needle-like to columnar in the sintered microstructure, while porosity exhibits an increasing and then decreasing trend. With an increase in Al2O3 content, the proportion of aluminium in SFCA gradually increases, and the content of SFCA increases as well. The content of SFCA reaches its maximum when Al2O3 content reaches 2.40 wt.%, coinciding with the maximum percentage of hematite. However, the silicate and spinel reached their maximum at an Al2O3 content of 2.35 wt.%, leading to a deterioration in sinter strength and RDI. The morphology of SFCA in liquid phase and pore structure have a significant impact on the properties of sinter rather than liquid phase volume.
Footnotes
Acknowledgments
I would also like to thank Handan Iron & Steel Group Co., Ltd for providing iron ore for the research.
Data availability
Data will be made available on request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China and Chongqing Natural Science Foundation Innovation and Development Joint Fund (Grant Nos. U2003215 and 2023NSCQ-LZX0150).
