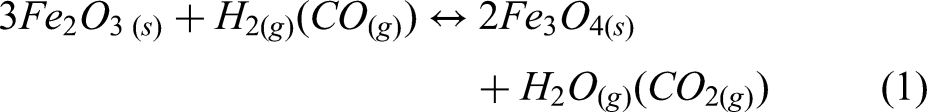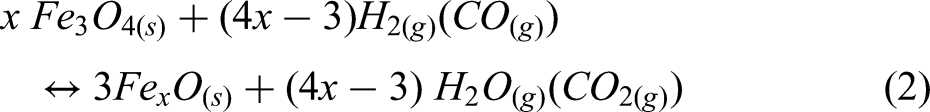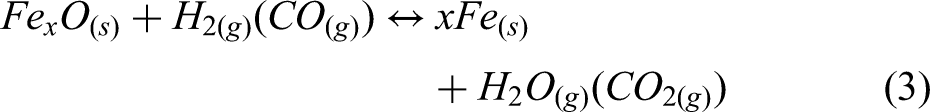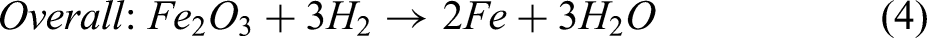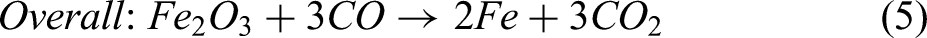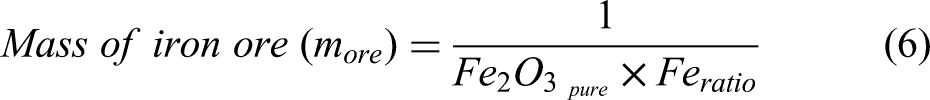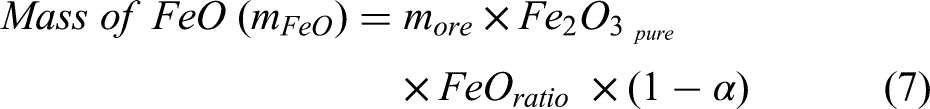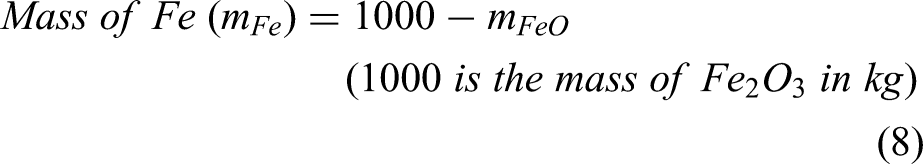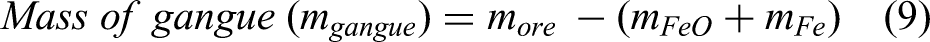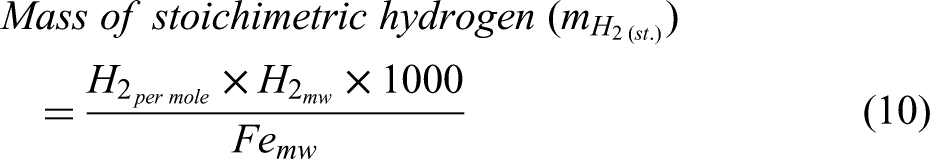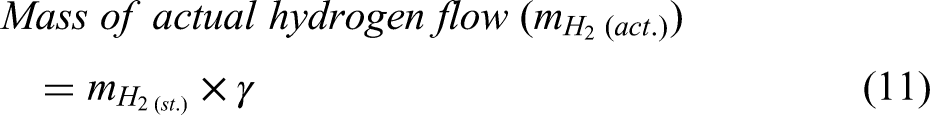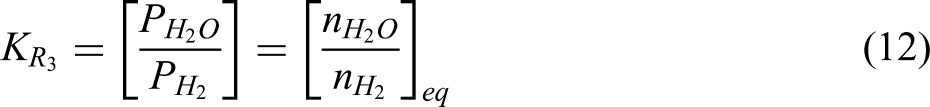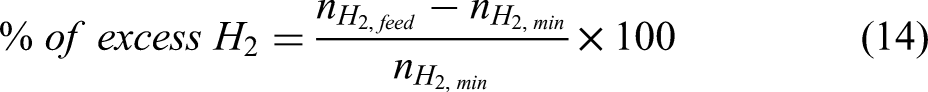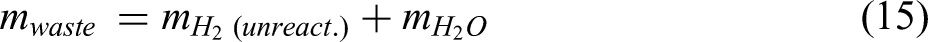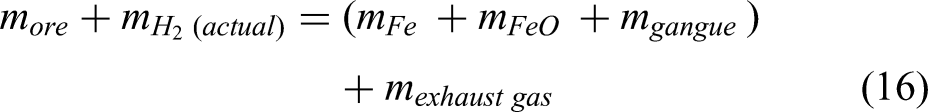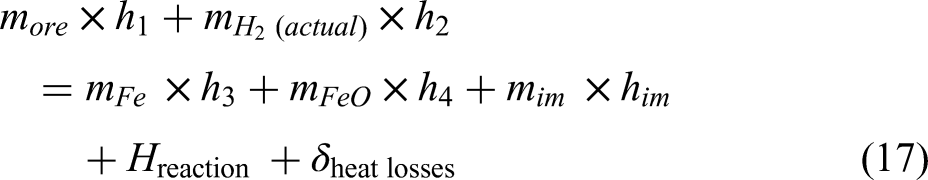Abstract
This process modelling study explored the behaviour of hydrogen-based direct reduced iron (DRI) manufacturing in a shaft furnace. Various performance parameters such as metallisation ratio (MR), consumption of hydrogen per tonne of DRI, production of by-products, reactor energy demand and total energy demands for the process have been analysed with respect to temperature, ore grade (gangue content), and reactant conditions. The HSC Chemistry (H: enthalpy, S: entropy and C: heat capacity) SIM (simulation) module was employed for the modelling coupled with the Gibbs energy minimisation calculation. The shaft furnace was divided into three zones to model the three-step reduction of iron ore in a counterflow arrangement. Results show that temperature and hydrogen supply have a significant effect on the metallisation of DRI. Increasing temperature and hydrogen flow rate were predicted to increase the MR or reducibility. A full metallisation can be achieved with hydrogen supply of 130, 110, 100 and 90 kg/tDRI at 700, 800, 900 and 1000°C, respectively, using the best-grade ore (Fe 69 wt.%, gangue 5.2 wt.%). However, the hydrogen consumption in full metallisation was calculated to be 54 kg/tDRI (tonne of DRI). At full MR, the reactor energy consumption (supplementary electrical energy) was calculated to be 0.56 to 0.59 MWh/t Fefeed using the reactor temperature from 700 to 1000°C. Ore grade or gangue content has a significant impact on reactor energy demand. For example, at 900°C, the top-grade ore was calculated to consume 0.69 MWh/tDRI compared to 0.88 MWh/tDRI using the lowest grade ore. A certain percentage of CO (i.e. 15%) blended with hydrogen was predicted to be beneficial for metallisation, hydrogen consumption, and overall energy demand. However, increasing CO would increase CO2 emissions significantly.
Introduction
The iron and steel industry is a major emitter of global CO2 emissions, responsible for approximately 7 to 9%.1–3 Conventional blast furnace-basic oxygen furnace (BF-BOF) steelmaking process (73.2% share) emits about two tonnes of CO2 per tonne of steelmaking due to the use of 800 kg of coal/t steel.4,5 Direct reduced iron (DRI) followed by electric arc furnace (EAF)/BOF is the most mature technology to combat CO2 emission capable of reducing CO2 emission by over 90% from the conventional BF-BOF route.1,6,7 DRI is a process of converting iron from iron ore/pellet directly by reducing it with reformed natural gas, hydrogen or syngas without converting it into molten metal. 8
Currently, the DRI-EAF route produces just over 5% of steel worldwide.9,10 However, currently produced DRI is primarily based on natural gas (76%) or syngas derived from coal and, therefore, is not emission-free.11–13 Hydrogen is an alternative to natural gas and is a good reducing agent capable of substituting natural gas fully or partially and has recently attracted significant investment. 14 A pilot scale trial from Midrex showed that a 90% reduction in CO2 is possible by substituting 70% of natural gas with hydrogen. 15 The natural gas-based shaft furnace technology manufacturer Energiron has tested the use of over 90% hydrogen at the Hysla, Monterrey facility achieving high metallisation of 94 to 96% and reported that its shaft furnace could be optimised to 100% hydrogen.16,17
According to IEA (International Energy Agency) estimates, about 15% of global steel will be produced from hydrogen-based DRI (H2DRI) by 2050. 18 Several pilot and commercial scale projects are underway worldwide, including HYBRIT project in Sweden, 19 H2Future in Europe, 20 tkH2Steel by ThyssenKrupp in Germany,20,21 Hamburg22,23 and HyDeal Espana 24 projects by ArcelorMittal, SALCOS by Salzgitter AG, 21 and MIDREX in the USA.17,25,26 A number of technologies are used to produce DRI, such as fluidised beds, rotary kilns, rotary hearth furnaces and shaft furnaces. However, over 70% of the DRI is produced using a shaft furnace (MIDREX, Energiron), 10 which is used in the well-known HYBRIT project. 6 Some key challenges with H2DRI steelmaking include the availability of affordable green hydrogen, reactor heat balance and unsuitability of using high gangue iron ores. 27 Also, there is a concern with producing zero carbon hydrogen DRI regarding transportation and melting subsequent EAFs.27,28 However, the scope of the study is limited to primarily dealing with hydrogen and energy requirements for making DRI concerning ore grade/gangue content, temperature and reductant conditions.
Temperature is an important parameter for the production of DRI, and generally, increasing temperature increases the reduction rate. However, Chen et al. 29 reported that the reduction rate decreases at a temperature over 900°C. Ranzani da Costa 30 claimed that 800°C is the ideal temperature for the hydrogen-based reduction of iron ore based on laboratory kinetic experiment. That work also studied the effect of hydrogen and natural gas-derived syngas on the reduction of haematite. Results showed that the reduction rate using hydrogen is much faster than that of syngas. Zou et al. 31 reported that the higher diffusivity of hydrogen gas helps reduce iron ore faster than syngas or CO.
A pilot scale trial from Midrex which produces the world's 80% DRI 10 shows that the shaft furnace requires 550 Nm³H2/tDRI (49.0 kg/tDRI) for reduction and another 250 Nm³H2/tDRI (22.3 kg/tDRI) as a heat source. 32 The shaft furnace technology from HYL/Energiron also tested different percentages of hydrogen in its furnace. The study reported that using 100% hydrogen in ENERGIRON-ZR technology requires 2.0 GJ/tDRI less energy input than natural gas as reforming of natural gas is not required here. 33 A study by Vogl et al. 34 showed that per tonne of DRI requires about 2.75 MWh (9.94 GJ) energy, which includes electrolysis and ore heating.
Numerous studies35–40 have reported the reduction behaviour of iron ore using shaft furnaces. However, they have generally investigated the process using a mixture of hydrogen and CO as a reducing agent. Some pilot-scale trials have recently used pure hydrogen to reduce iron ore.26,41,42 However, the specific energy consumption in the literature varies markedly, ranging from 3.5 to 5.5 MWh/tls (which corresponds to 12.6 to 19.8 GJ/tls).6,17,30,34,43 Also, there is a limited process model in the literature on emerging hydrogen-based DRI manufacturing, 12 including limited attention to the shaft furnace. 6 Hence, this study systematically investigates the effect of hydrogen supply on metallisation, consumption of hydrogen and energy per tonne of DRI, the effect of gangue content on specific energy consumption, and finally, the effect of blending H2 with CO on the performance of DRI production.
In this article followed by introduction in the first section, the second part describes the material and energy balance of H2DRI-based steelmaking. Sections Methodology and Model validation discuss the methodology and validation respectively. The results are discussed in section Results and discussion, which include the reduction behaviour of iron ore using pure hydrogen, followed by the effect of gangue content and co-reductant (H2 + CO) on the production of DRI, before overall conclusions are presented.
Material and energy balance in hydrogen-based DRI production
This study implements a model of the reduction of iron ore in a shaft furnace using hydrogen, via a simplified three-zone formulation. The reactor is assumed to be fed with cold hydrogen and ore, and any required supplementary heating is assumed to be via electrical heating. The reduction of iron ore involves the reduction of haematite (Fe2O3) to magnetite (Fe3O4) as the first step; magnetite to wustite FeO/Fe(1−x)O as the second step; and finally, wustite to pure iron (Fe) in the third and final step.30,44 Reactions involving these three-step reductions of haematite are shown in Equations 1–3, while the overall reactions using H2 and CO are shown in Equations 4 and 5.
Step 1:
The term metallisation ratio (MR) (α) is defined as the share of metallic iron obtained after reduction in a reduction furnace (i.e. shaft furnace). The DRI process produces metallic iron and wustite. The amount of metallic iron and wustite at the exit of the shaft furnace can be calculated from Equations 7 and 8.
In practice, excess hydrogen is needed to promote the forward reaction in the third step of the haematite reduction, as shown in Equation 3. From the thermodynamic perspective, this reduction of wustite to metallic iron is limited by chemical equilibrium and can be reverted above a certain critical ratio of water vapour to Hydrogen. 46 This equilibrium ratio of H2O/H2 of the wustite reduction by hydrogen is quite low at lower temperatures. The endothermic nature of this reaction also further encourages reversion, that is when the reaction temperature is lowered (not maintained), the reaction system can enter a condition that promotes reversion. From the kinetics perspective, the use of pellet ores in the shaft reactor will likely be limited by diffusion processes. Hence, a very high H2 utilisation is a challenge. Considering all of these, excess hydrogen is needed in the reactor to prevent the reverse reaction and facilitate complete reduction. This can be done by recirculating (recycling) unutilised hydrogen from the off gas to the reactor as is the practice in industry.
The equilibrium constant derived from Equation 3 is as follows:
Mass balance for the DRI furnace
A conceptual diagram for the mass and energy balance for the shaft furnace is shown in Figure 1, while Equation 16 shows the mass balance equation of the system for the shaft furnace.
17
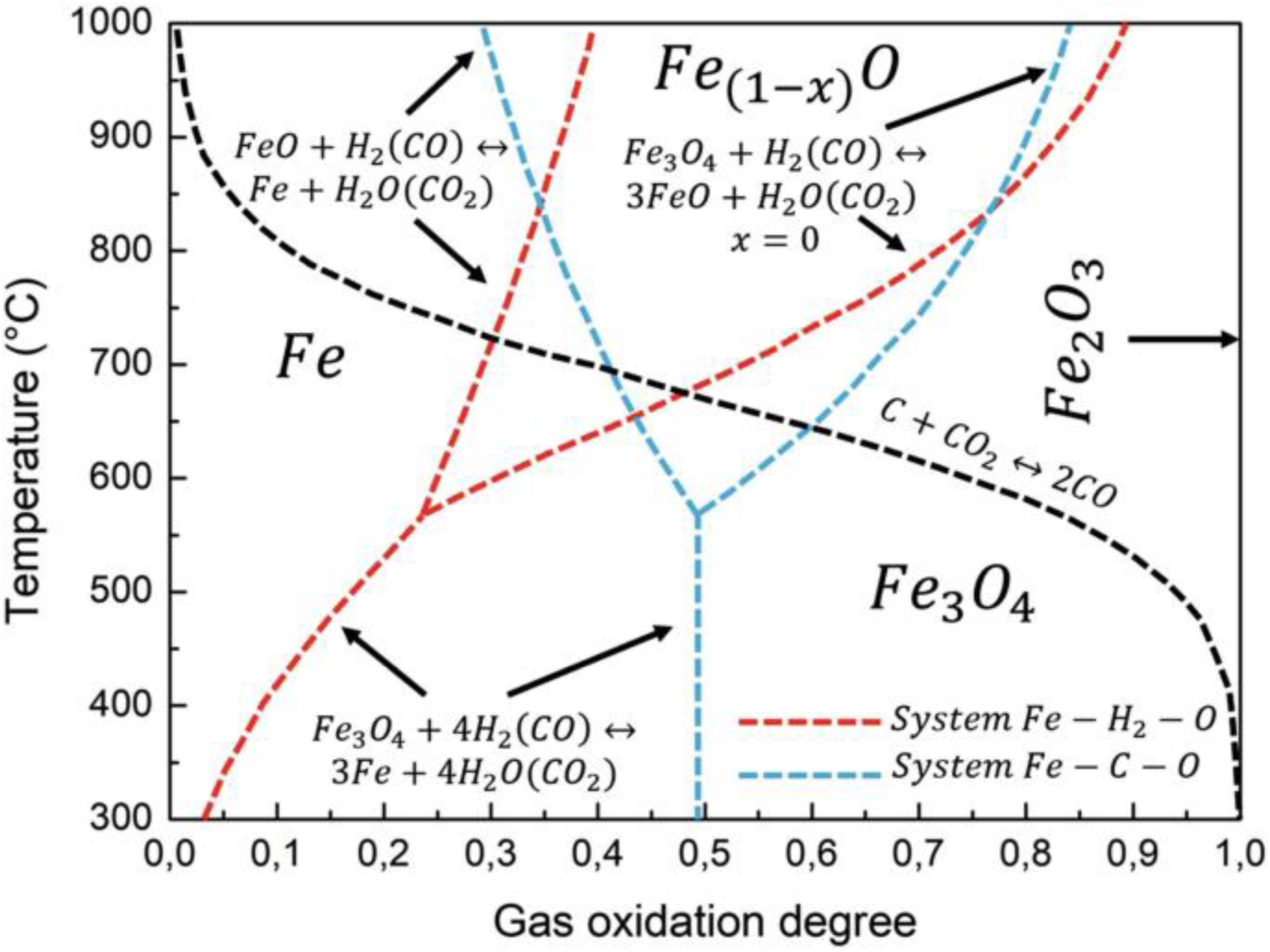
Reduction of iron oxides concerning gas oxidation degree and temperature using H2 and CO as reductants. 47
Energy balance for the DRI furnace
The energy balance equation of the system for the shaft furnace is shown in the Equation 17 below.
The Baur-Glässner diagram, shown in Figure 1, provides a useful visual representation of the thermodynamics involved in the reduction of iron oxides. This diagram illustrates the stability regions for various phases of iron oxide, which are influenced by temperature and the gas oxidation degree (GOD). The GOD refers to the proportion of oxidised gas components relative to the combined total of oxidised and oxidisable gas components. By assessing the GOD value of a gas composition, one can gauge its reduction driving force, with a lower GOD indicating a higher reduction force for the gas mixture. 8
According to the diagram, carrying out reduction using hydrogen at the highest attainable temperature is thermodynamically favourable. This is because the stability region of iron formation by hydrogen reduction expands as the temperature rises. As a result, there is an enhanced theoretical utilisation of gas and a greater thermodynamic driving force for the reduction process. The complete reduction process using hydrogen is characterised as endothermic, whereas reduction with carbon monoxide is exothermic. 48 This clearly illustrated in Figure 2 developed using FactSage software that shows the overall enthalpy of reaction for a complete reduction of Fe2O3 at different temperatures. Consequently, when employing hydrogen for reduction, additional energy needs to be supplied to maintain a consistent reduction temperature. Depending on the specific industrial procedure, this requirement can present challenges since energy must be introduced into the system. This can be achieved by preheating the input material or through specialised process design. 8
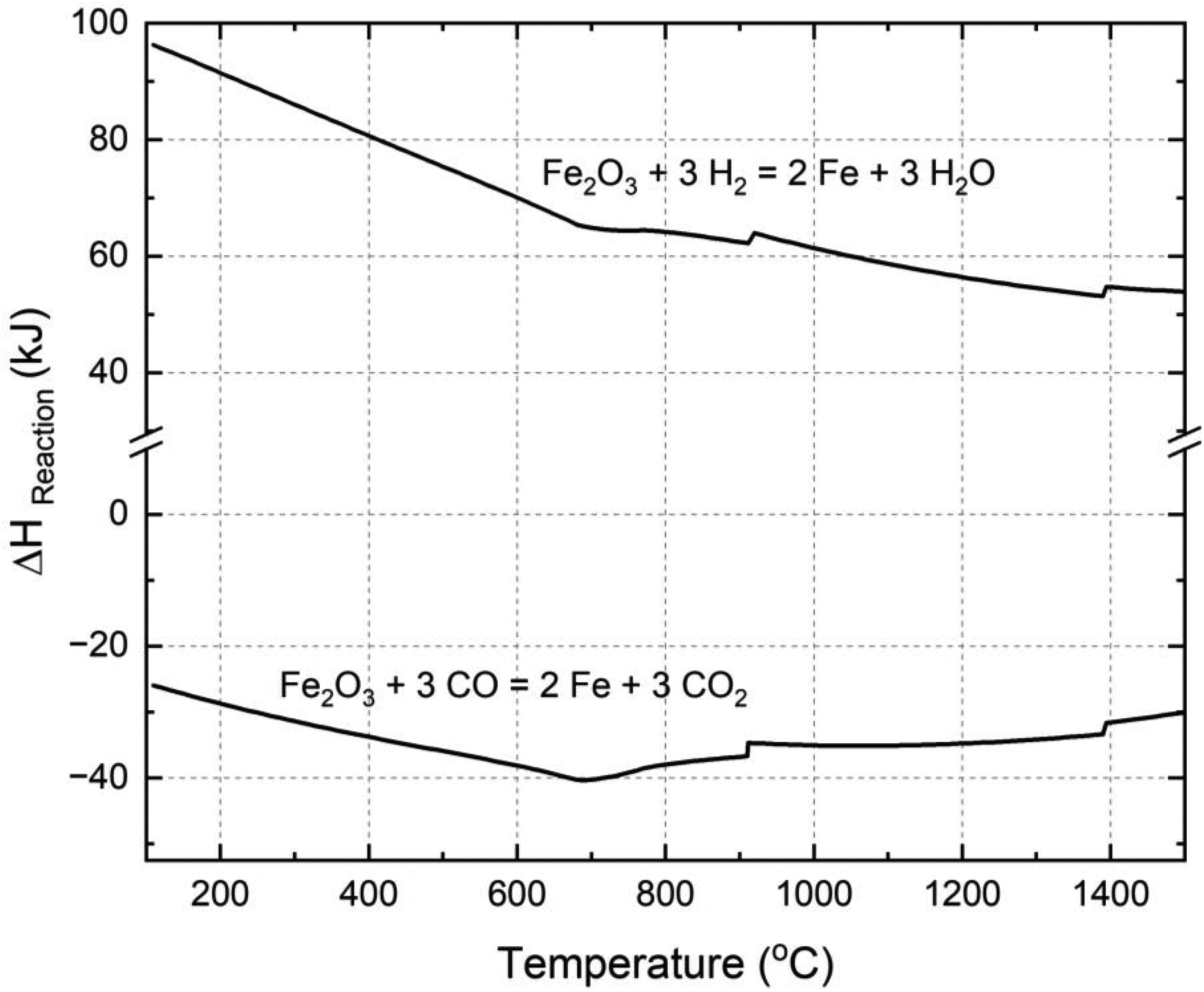
Enthalpy of reduction reactions of Fe2O3 by H2 and CO.
Methodology
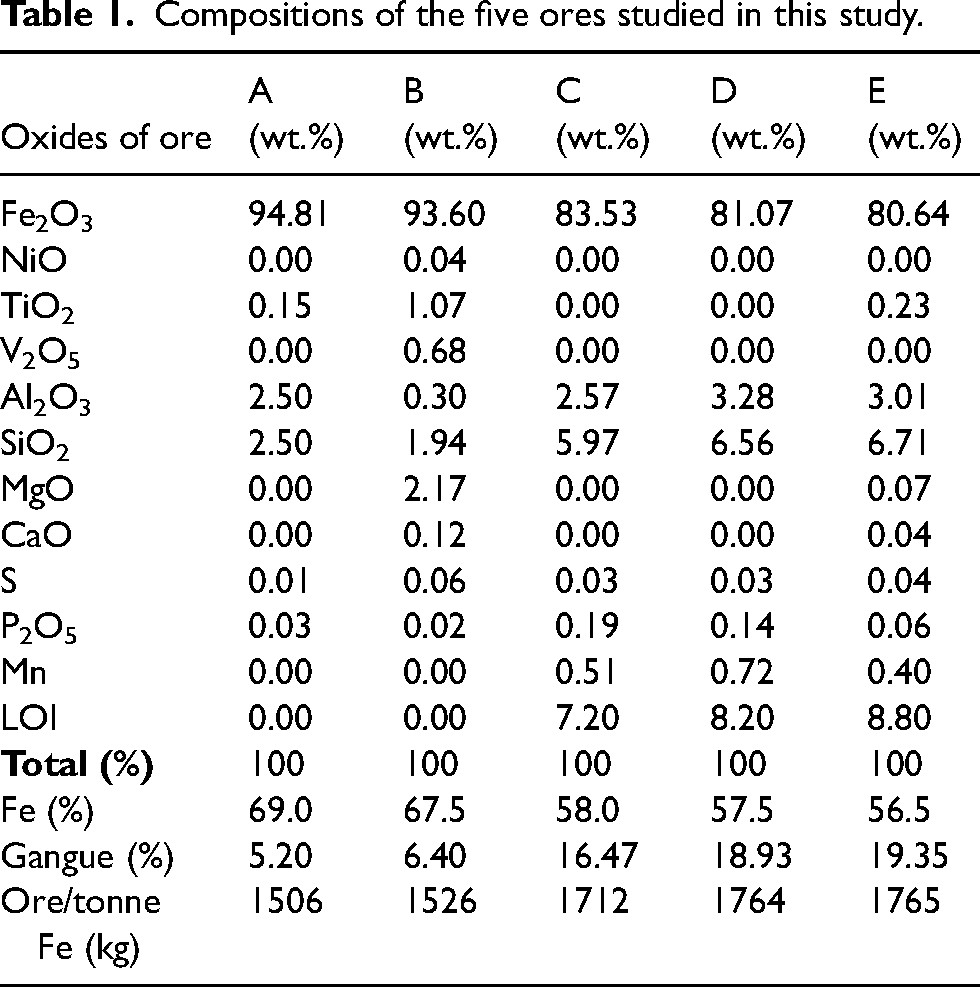
The lump ores examined in this research were chosen to encompass a diverse range of Fe/gangue contents and were sourced from various iron ore companies in Australia. According to the Fe content, the selected ores listed in Table 1 can be broadly categorised into two types. Ore A and B are haematite (Fe2O3), while the other three are goethite/limonite (FeO(OH)/FeO(OH).nH2O). The gangue content in ores A and B ranges between 5 and 6 wt.%, while the other three ores (goethite/limonite) contain a gangue of 16 to 19 wt.%. Moreover, the loss on ignition (i.e. H2O) in goethite/limonite ore varies over 7 to 8%. Over 90% of gangue is composed of Al2O3 and SiO2 except for ore B. Ore B contains Al2O3 and SiO2 of total 35%, while the other the rest include MgO (35%), TiO2 (16%) and V2O5 (11%). The ore quality presented in Table 1 is in the order of highest to lowest concerning ‘
Compositions of the five ores studied in this study.
The conceptual process flow diagram of the model is shown in Figure 3. Three distinct zones were considered to make a counterflow arrangement of the shaft furnace (e.g. MIDREX type reactor). Upper zone was considered at the top of the shaft furnace, while middle and bottom zones were the middle and bottom parts of the shaft reactor. Iron ore drops from the top, while reductants (H2/CO) flow from the bottom upwards. Although the modelling carried out in this study is a thermodynamic/equilibrium-based, the division in the reactor with three zones was necessary to model the practical counterflow arrangement of the shaft reactor including three-step reduction described in Section Material and energy balance in hydrogen-based DRI production.
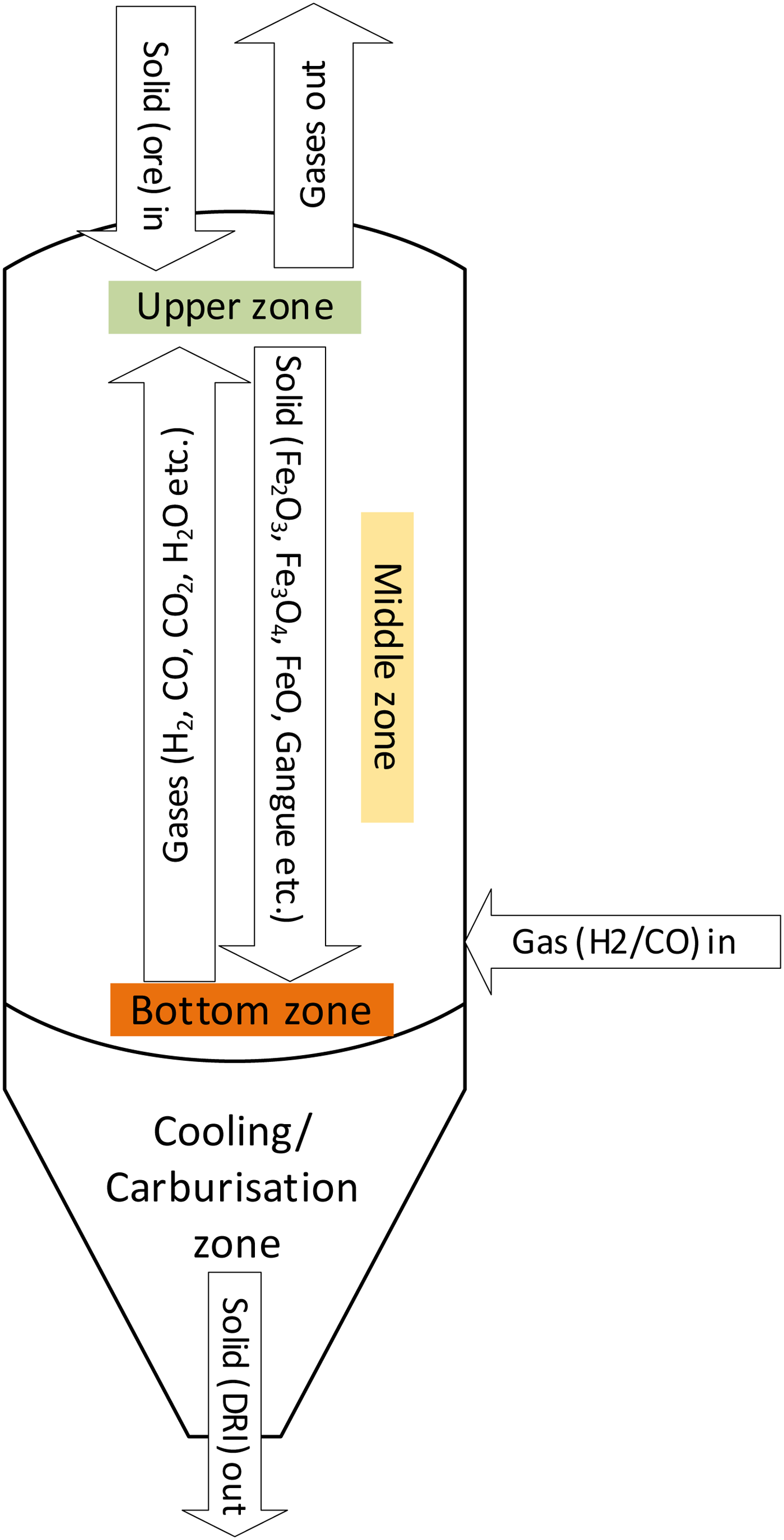
Conceptual diagram of the process model.
A thermochemical package HSC Chemistry 10.0 was used for the development of mass and energy balance; and thermodynamic equilibrium modelling using the Gibbs energy minimisation technique.49,50 The process flow diagram of the thermodynamic equilibrium model is shown in Figure 4. The HSC chemistry database contains physical, chemical and thermodynamic properties for the chemical elements/compounds involved. The equilibrium model assumes that the reactor is a close system, i.e. isothermal and that there is no pressure loss. 51 The residence time in the shaft reactor is sufficiently long for all reactions to reach equilibrium. 52 To account for heat loss, the model assumes a temperature difference of 50°C between middle and bottom zone based on the plant data.53,54 The feed H2 was supplied at 930°C as it is suggested that the temperature of the feed gas should be as high as possible below softening temperature of the DRI (≤ 956°C). 6 The typical operating conditions of the shaft furnace are reported in Table 2. The model also assumes that the gangue contents do not participate in the chemical reaction due to the low temperature of the shaft reactor. The melting temperature of the gangue contents is higher than 1000°C.
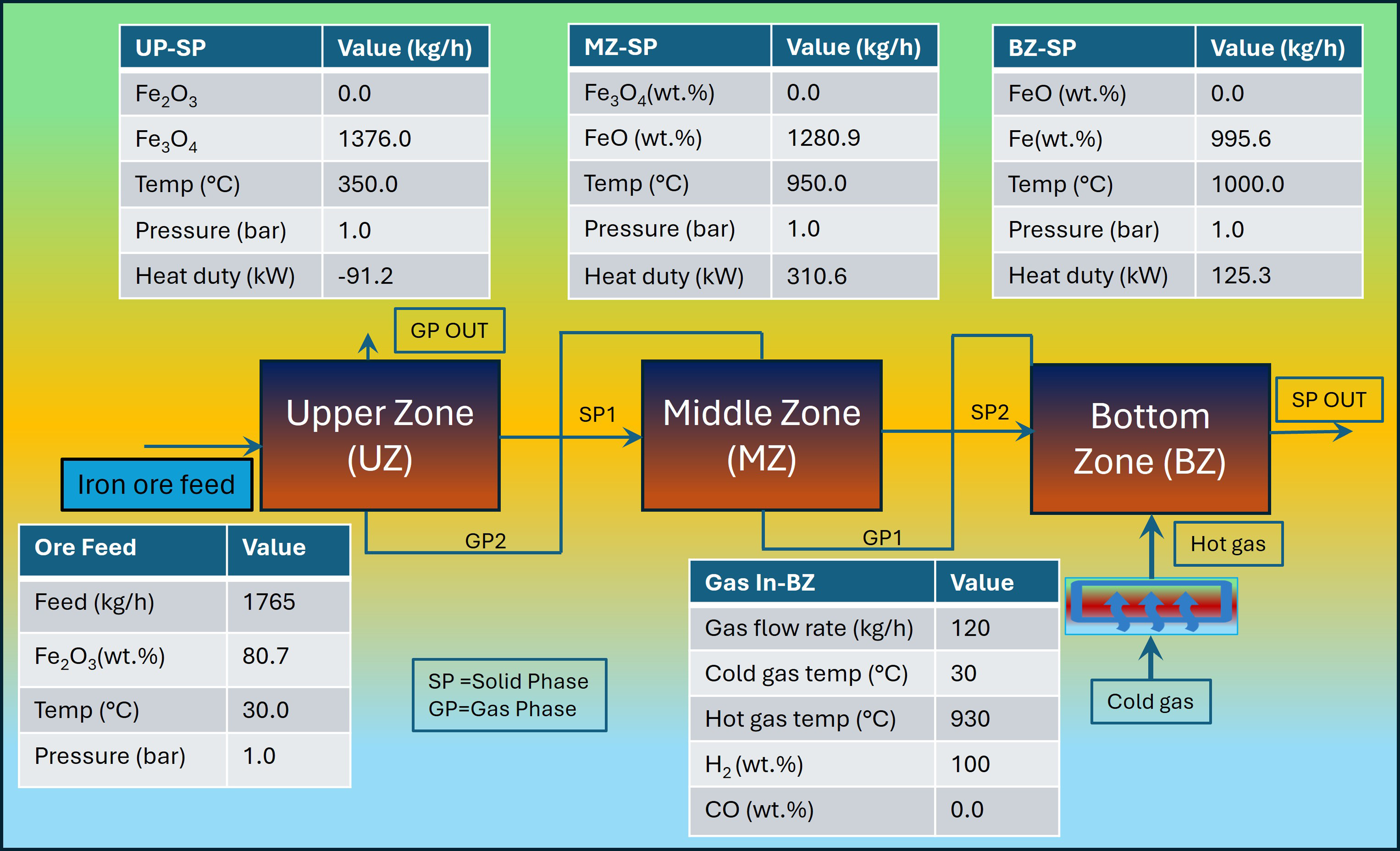
Process flow diagram of the thermodynamic equilibrium model with mass and energy parameters for different zones of the shaft reactor.
Typical operating conditions of the shaft furnace.

Model validation
The model was validated with published industrial data on Gilmore Steel Corporation (Portland, OR, USA) plant operated using a shaft furnace. The details of the operating conditions collected from literature37,40,53 are shown in Figure 5.
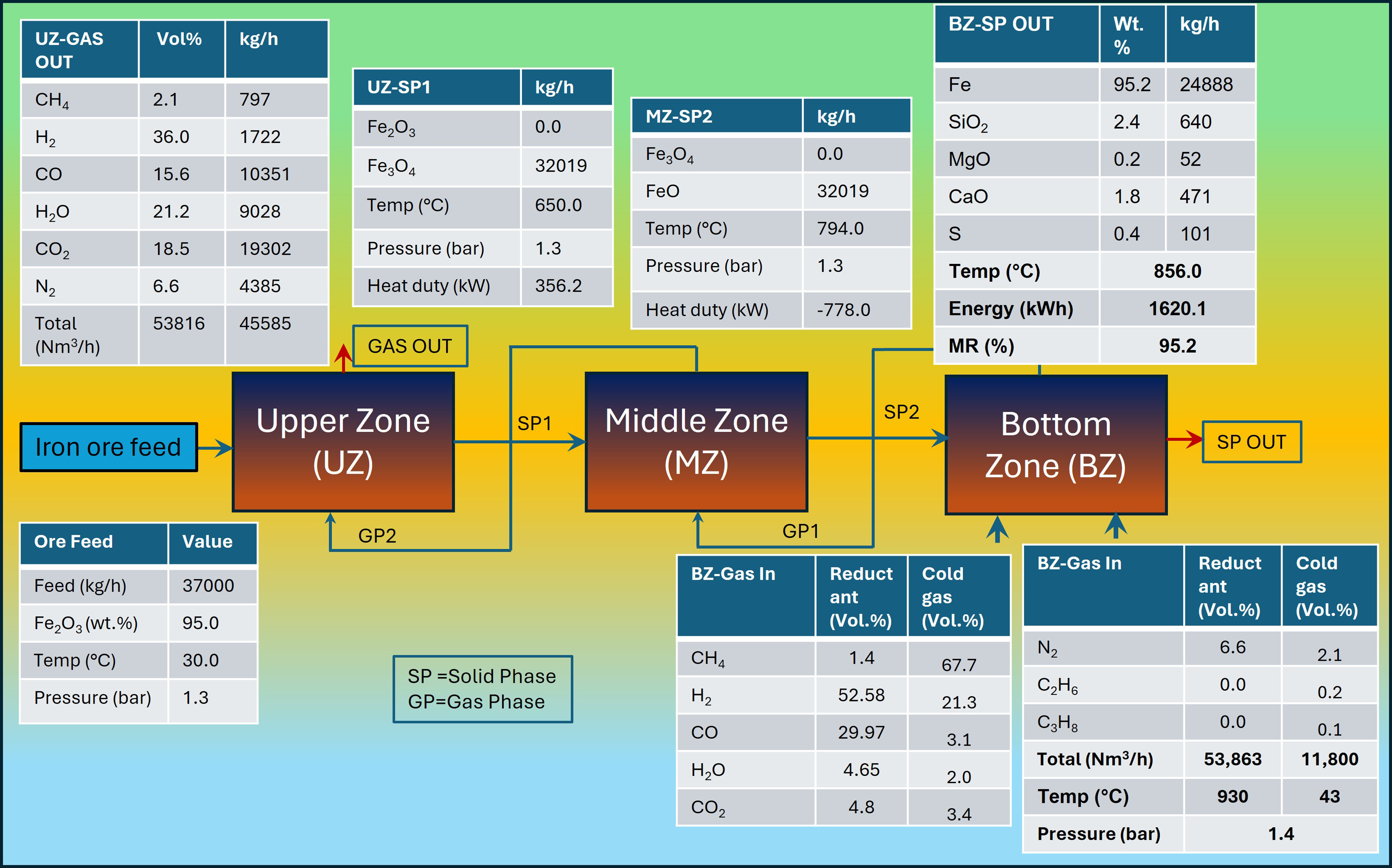
Operating conditions of the Gilmore plant (Portland, OR, USA).
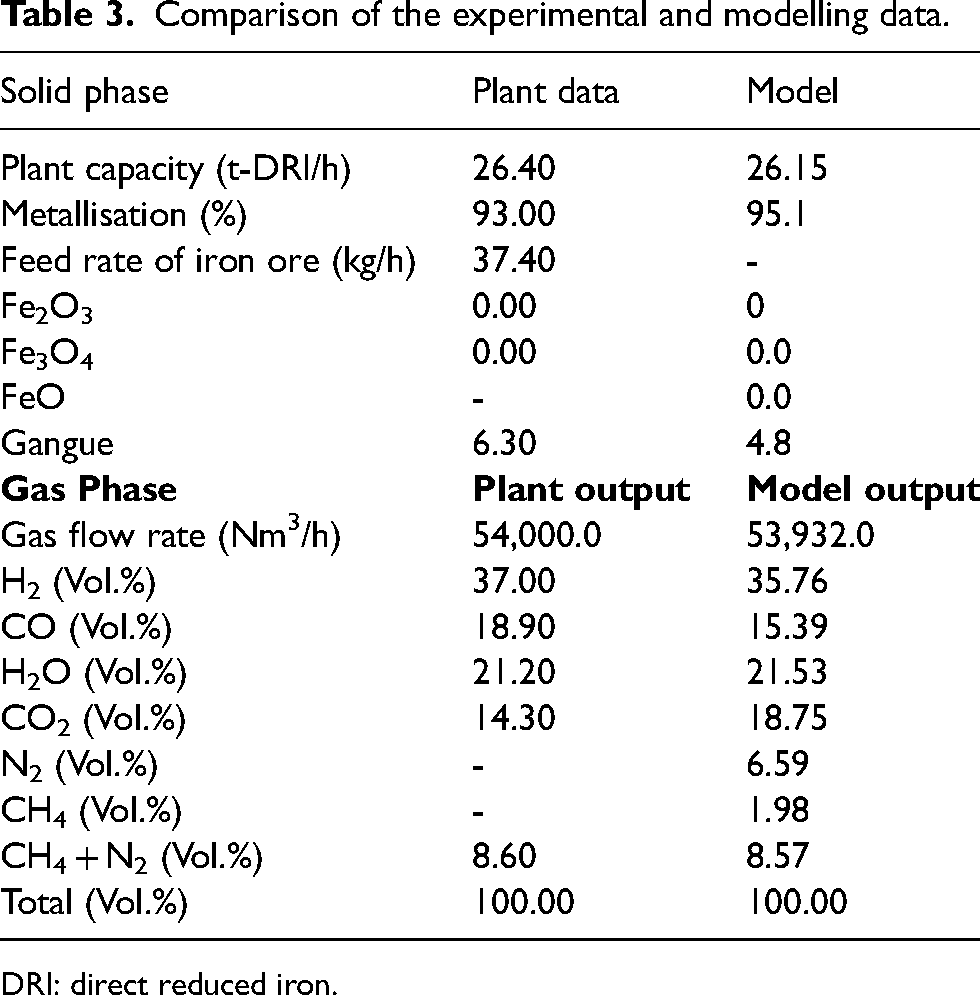
A comparison between the experimental and modelling data is shown in Table 3. The solid phase and gas phase modelling data matches well with the experimental results.30,37,40,53 As can be seen, our model predicts MR of 95.1%, just over 2% higher than the 93% MR reported from the operational plant. After validation of the model, a range of sensitivity studies was carried out using various operating conditions described in the following sections. The gas phase data also matches reasonably well with relatively high RPD for CO and CO2, which is due to the assumption in reactor temperature, especially in upper zone.
Comparison of the experimental and modelling data.
DRI: direct reduced iron.
Results and discussion
Production of DRI using pure hydrogen
The effect of hydrogen flow rate on the MR under different reactor temperatures of 700 to 1000°C (bottom zone) is shown in Figure 6(a) using ore A. As an obvious fact increasing the hydrogen flow rate increases the MR or reducibility. Similar effects were observed concerning increasing temperature. However, the degree of increased metallisation is substantially higher at higher flow rates and temperatures. Although, in this process modelling, increasing temperature consistently increases the MR, some studies, for example, Chen et al., 29 showed that at temperatures over 900°C, the reduction rate decreases due to the sintering and formation of the diffusion boundary layer. As seen in Figure 6(a), the metallisation ranges over 19 to 100% at a temperature of 700°C, while this is 56 to 100% using the temperature of 1000°C under the hydrogen flow rate of 50 to 130 kg/h (H2/Fe: 0.05–0.13) for the highest grade ore studied (ore A).
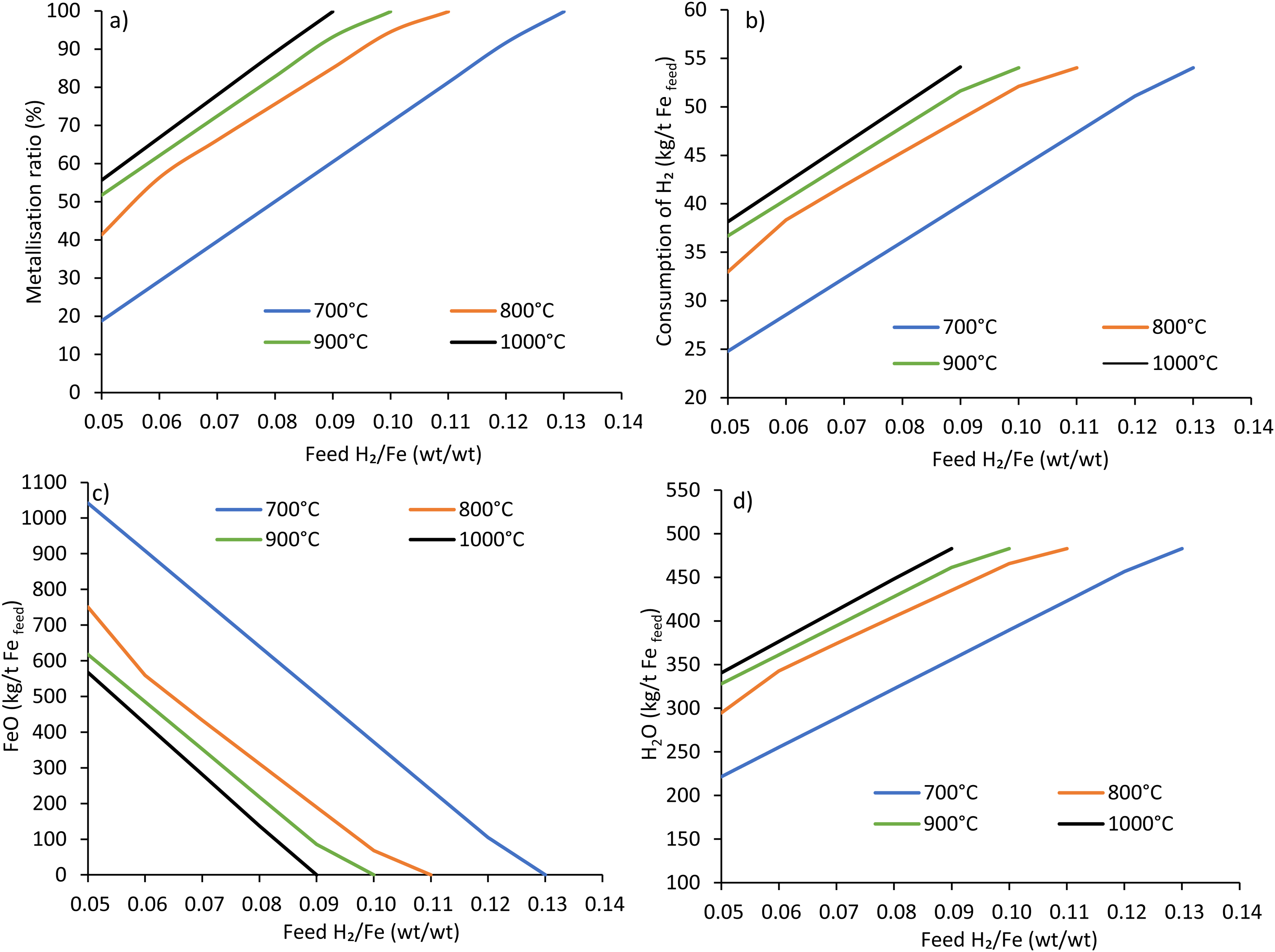
Effect of H2/Fe ratio (wt./wt.) on (a) metallisation ratio, (b) consumption of H2, (c) FeO, and (d) H20 at different temperatures using ore A (Fe 69 wt.%, gangue 5.2 wt.%).
It is worth mentioning that the input ore quantity from different ores was varied so that the output can provide one tonne of iron if 100% metallisation is reached. Considering other ores (B-E), the MR, the consumption of hydrogen and the degree of changes concerning temperature and reductant supply rate also followed the same trend as ore A, because the gangue contents were not assumed to participate in chemical reactions at such a low temperature. However, there was significant variation regarding reactor power consumption and by-products, such as water, concerning ore types.
The effect of the H2/Fe ratio on hydrogen consumption is shown in Figure 6(b), which follows almost a linear trend. As seen in Figure 6(b), at 1000°C and a hydrogen flow rate of 90 kg (H2/Fe:0.09), hydrogen consumption was calculated to be 54 kg to reduce one tonne of Fe2O3 into metallic iron. The results indicate that about 1.66 times the stoichiometric hydrogen supply is necessary to reach full metallisation at 1000°C. As shown in Equation 3, the final step of the reaction is the formation of metallic iron and water vapour from the reaction of wustite and hydrogen, which is significantly limited by chemical equilibrium. 46 This reaction produces a large amount of water vapour, pushing the reaction in the reverse direction. Hence, excess hydrogen from the reactant side is needed to reach equilibrium.
The MR was also found to be a function of temperature. The degree of metallisation using 90 kg hydrogen was determined to be 60, 85, 93 and 100% at a temperature of 700, 800, 900 and 1000°C (Figure 6(a)), while the consumption was 40, 49, 52 and 54 kg, respectively (Figure 6(b)). The results indicate a relationship of
Vogl et al. 34 reported a total H2 flow rate of 824 Nm3/tDRI (∼ 74 kg/tDRI) and a consumption of 527 Nm3/tDRI (∼ 47 kg/tDRI) at 800°C with MR of 94%, meaning the degree of utilisation is 64%. However, the current study shows that, to reach 94% MR at 800°C, the H2 supply rate should be 100 kg/tDRI, from which the consumption would be 52 kg/tDRI, which is consistent thermodynamically. Ranzani da Costa et al. 30 reported that 100% metallisation can be achieved at a temperature of 800°C with a hydrogen flow rate of 1609 Nm3/t-pellet, representing a utilisation ratio of around 38%. Spitzer et al. 55 reported a hydrogen utilisation rate of about 55% at 900°C, which is 57% in the current study with an MR of 93%. Shao et al. 6 reported a hydrogen utilisation ratio of less than 25% and recommended feeding H2 with higher temperatures and increasing furnace length. It is important to keep the H2 flow rate as low as possible, balancing with MR, because a higher H2 flow rate leads to the requirement of large reactor, gas handling and heat exchanger, thus increases energy consumption and capital cost. 6 The results from the current study are supported by the literature that is feeding of a much higher amount of hydrogen against the requirement/consumption to satisfy the thermodynamic equilibrium. Hence, top gas recirculation in the furnace is recommended. 40
The effect of feed H2/Fe ratio (wt./wt.) and temperature on the production of FeO are shown in Figure 6(c) using ore A. Increasing feed H2/Fe ratio and temperature decreases FeO due to the increased MR. In contrast, increasing the hydrogen supply rate and temperature increases the production of H2O, as shown in Figure 6(d). Again, the degree of change concerning hydrogen supply rate and temperature is higher at higher hydrogen supply rates and temperatures. Stoichiometrically, reducing one tonne of Fe2O3 using pure hydrogen produces 487 kg of water. As the degree of metallisation increases due to the increased hydrogen supply rate and temperature, the formation of water increases.
The results indicate that water production from ore A at full metallisation is 483 kg/t Fefeed at 1000°C with a feed H2/Fe ratio of 0.09. The production of water is also a function of temperature. Considering other ores, water production and the degree of changes concerning temperature and reductant supply rate also follow the same trend as ore A. However, the output water quantity varies primarily due to the variation in inherent water content known as LOI in the studied ores (later reported in the effect of gangue in section Effect of gangue content on the production of DRI).
The energy consumption due to the consumption of H2 concerning the hydrogen feed rate is shown in Figure 7(a). As seen, increasing the temperature increases hydrogen consumption and, thereby, the energy from hydrogen. At an H2/Fe ratio of 0.09 (wt./wt.), the consumption of energy from hydrogen is recorded to be 4.8, 5.8, 6.2 and 6.5 GJ/t Fefeed considering the temperature from 700 to 1000°C. The energy consumption at 900°C is reported to be 6.3 GJ/tDRI at full metallisation in the literature, 56 which is nearly the same as current study of 6.48 GJ/tDRI. Using electrolyser efficiency of 60%, Shao et al. 6 reported an energy consumption of 9.8 GJ/tDRI, which is 71% of the total energy consumption (13.9 GJ/t Fefeed). In contrast, considering the electrolyser efficiency of 73%, Bhaskar et al. 17 reported an energy consumption of 9.64 GJ/t Fe (2680 kWh/t Fe). A numerical study by Ranzani da Costa 30 reported an electrolyser energy consumption of 10.6 GJ/t Fe. Considering the same electrolyser efficiency, the current study result closely matches with literature data.6,17,30 As a major source of energy consumption, the efficiency of the electrolyser plays a decisive role in developing HDRI-based steelmaking using shaft furnaces.
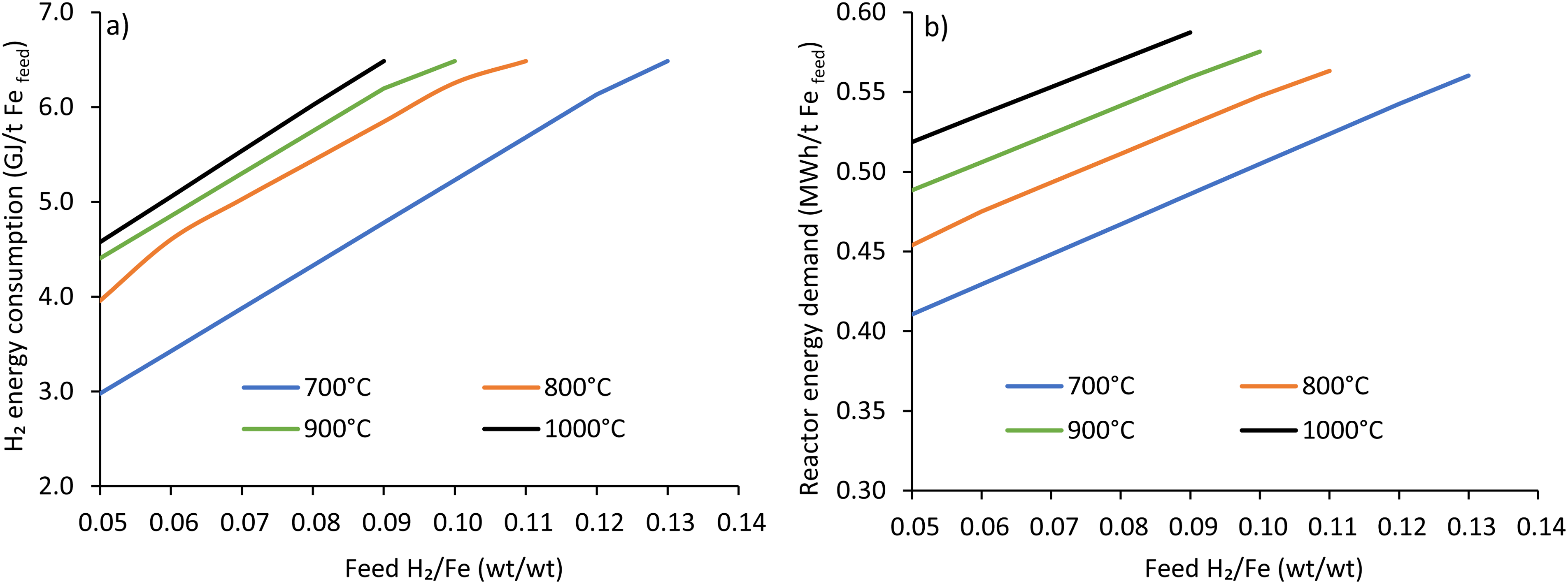
Effect of H2/Fe ratio (wt./wt.) on (a) energy from H2 and (b) reactor energy consumption at different temperatures using ore A (Fe 69 wt.%, gangue 5.2 wt.%).
The effect of feed H2/Fe ratio (wt./wt.) on reactor energy demand is shown in Figure 7(b) using ore A. As an obvious fact, increasing reactor temperature and H2 flow increases the energy demand. Increasing the hydrogen supply rate increases energy demand due to the endothermic reaction of the haematite reduction and higher water content in the system. At an H2/Fe ratio of 0.90 (wt./wt.), energy consumption was determined to be 0.49, 0.53, 0.56 and 0.59 MWh/t Fefeed, using the reactor temperature from 700 to 1000°C, which corresponds to the energy requirement of 1.75, 1.9, 2.0 and 2.12 GJ/t Fefeed. The reactor energy in the current study is the sum of energy for hydrogen heating and energy required to maintain reactor temperature. Other numerical studies in the literature reported that hydrogen heating requires 3.6 to 3.8 GJ/tDRI.6,30 This high energy demand from those studies was due to the high hydrogen supply of over 210 kg/tDRI (2363 Nm3/tDRI), out of which only 48 kg/tDRI was consumed, while the rest 162 kg/tDRI (1821 Nm3/tDRI) was recycled. 6 While compared with the hydrogen supply rate of 90 kg/tDRI, the energy consumption from the current study is somewhat similar to the literature.
The total energy consumption is the summation of energy consumption due to the consumption of hydrogen and energy needed for the reactor to achieve the desired temperature. As t H2/Fe: 0.09 (wt./wt.), the total energy consumption is 6.5, 7.7, 8.2 and 8.6 GJ/t Fe feed for the reactor temperature of 700, 800, 900 and 1000°C, which corresponds to the MR of 60, 85, 93 and 100%, respectively. Based on the MIDREX natural gas-based DRI plant, the energy requirement for about 10 GJ with an MR of 93% is similar to the MR achieved at 900°C in this study. 56 Results depict that about 75% of the energy is consumed due to the production/consumption H2 out of total energy at full MR.
Effect of gangue content on the production of DRI
The effect of gangue content has almost no effect on the MR due to the consideration of no interaction of gangue content during reduction at such a low temperature under thermodynamic modelling. However, kinetic modelling and plant data is needed to verify the prediction.
The ore quality seems to play an important role in energy consumption. As seen in Figure 8, the higher the ore quality, the lower the energy consumption. The gangue content in a particular ore is marked as the dotted lines. The ore A to E corresponds to the gangue content of 5.20, 6.40, 16.47, 18.93 and 19.35%. The results show that increasing the gangue content in the ore increases the energy demand in the reactor linearly. For example, at 900°C, the reactor energy consumption using different ore is recorded as 0.69, 0.70, 0.84, 0.87 and 0.88 MWh/t Fe feed – a 27% increase in energy due to the increase in gangue content by 14%.
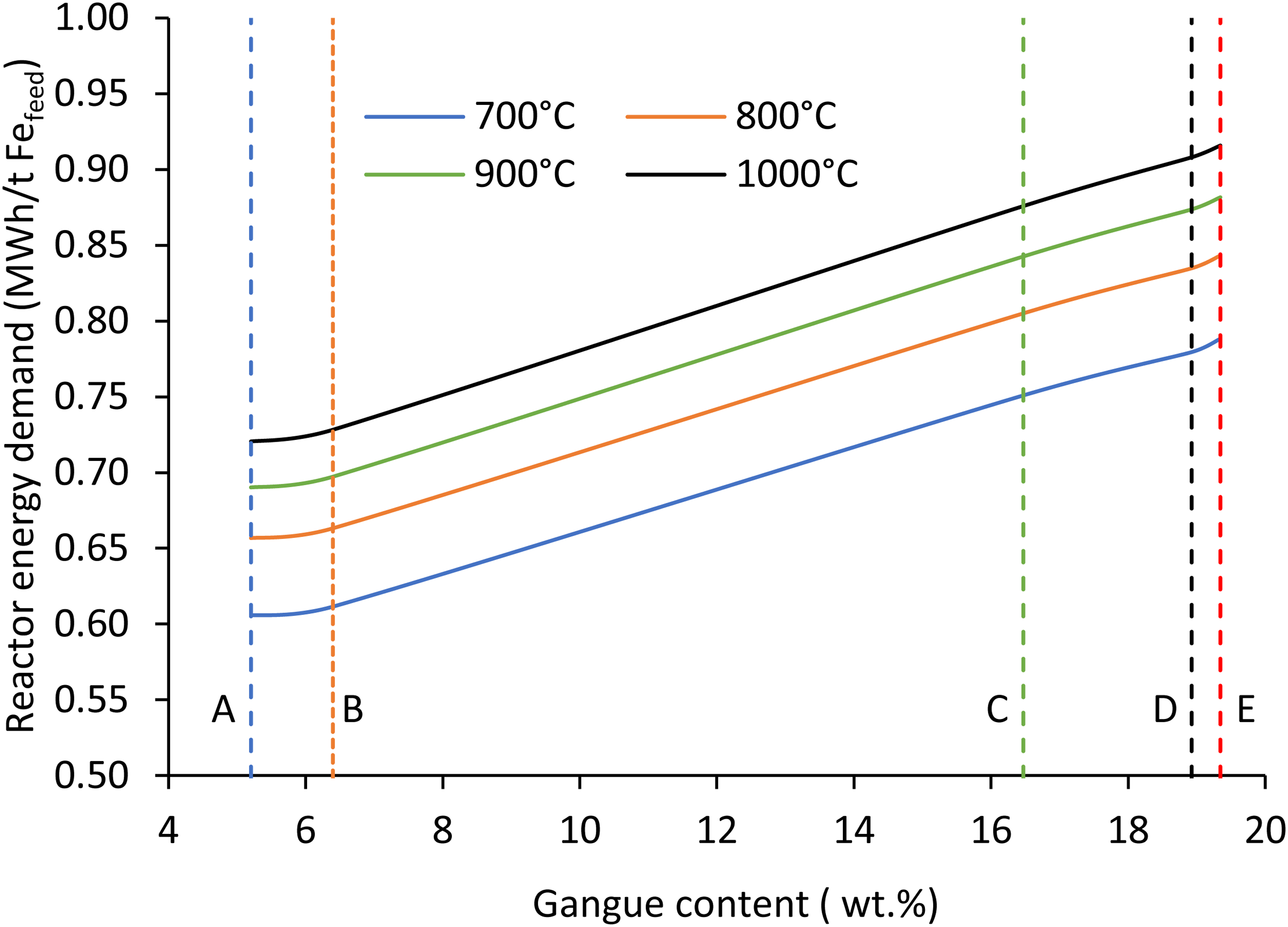
Effect of gangue content on a reactor energy demand at different temperatures using ore A to E and H2/Fe ratio of 0.10 (wt./wt.), (Fe 57–69 wt.%, gangue 5.2–19.3 wt.%).
Effect of co-reductant (H2 +CO) on the production of DRI
The thermodynamic prediction on the performance of the shaft reactor is reported in this section under mixed hydrogen and carbon monoxide reduction atmosphere. Two different flow rates were chosen under two H2/CO ratios (wt./wt.) of 85:15 and 70:30 against the stoichiometric requirement to reach full metallisation. The flow rate was based on the stoichiometric requirement of reductant to reduce one tonne of iron. Stoichiometrically, 54 kg of H2, while 525 kg of CO is required to reduce one tonne of Fe2O3. This study considered the ratio (wt./wt.) based on the stoichiometric requirement. For example, the supply rate of H2 and CO under an H2: CO ratio of 85:15 (wt./wt.) is determined to be 0.85*54: 0.15*525 = 45.9 kg of H2 and 78.8 kg of CO.
The temperature was found to have an insignificant impact on metallisation under blended conditions. Hence, the effect of reductant flow rate and excess reductants are reported in this section. The concentration of H2 and CO was increased proportionally with a multiplying factor (MF) of 1.1 and 1.2 to evaluate their effect on reducing iron ore and reaching full MR.
The effect of MF or the degree of excess reductant on MR is shown in Figure 9(a) at 700°C. As observed, increasing the excess reductant with an MF of 1.2 leads to 100% MR for both cases. The variation in MR between the two flows was negligible (about 1% before reaching full MR). However, in the case of pure hydrogen, a hydrogen flow rate of 130 kg is required to reach full metallisation at 700°C (Figure 6(a)). Therefore, it seems that the addition of a certain percentage of CO is more effective than pure hydrogen thermodynamically.
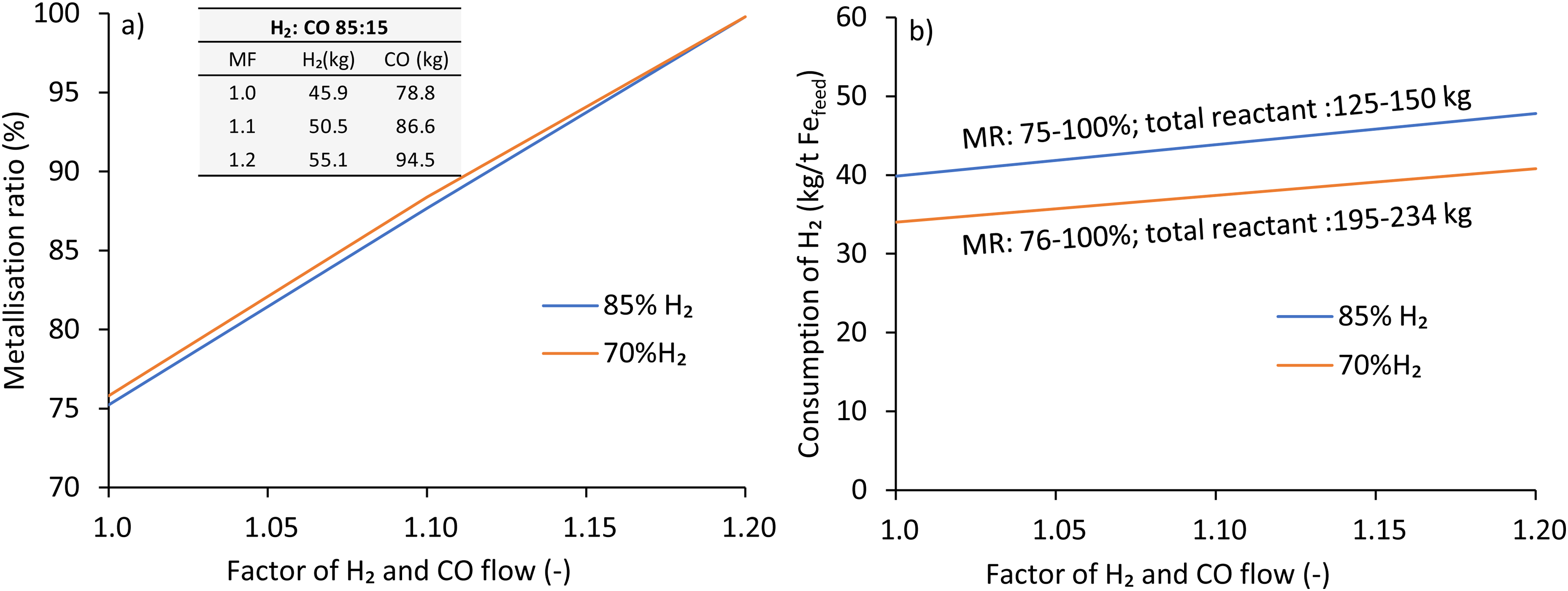
The effect of excess reductants on the (a) metallisation ratio (wt./wt.) and (b) consumption of H2 under H2:CO: 85: 15 and 70: 30 using iron ore A at 700°C (Fe 69 wt.%, gangue 5.2 wt.%).
Increasing the excess reductant increases hydrogen consumption in both cases, with approximately 6.0 to 7.0 kg higher in 85%H2 cases than in 70%H2 cases, as shown in Figure 9(b). The consumption of H2 at 85%H2 cases was calculated to be 39.9, 43.8, 47.8 and 50.9 kg, indicating the consumption of overall 86% against feed H2, whereas the consumption of CO was 100% for all cases. The consumption of H2 at 70%H2 cases is relatively high, with 34.0, 37.4, 40.8 and 43.9 kg representing overall consumption of 90% against feed H2, while the consumption of CO was 100% for all excess reductant conditions. As seen in Figure 9(a), at 85 H2%, a full metallisation is reached using MF of 1.2 with an H2 feed of 55.1 kg and CO feed of 94.5 kg. In contrast, a full metallisation can be achieved with an H2 feed of 45.4 kg and a CO feed of 189 kg under the 70 H2% case.
The current results suggest that CO is the favoured reductant thermodynamically over H2, which can be easily explained by the Gibbs free energy as shown in Figure 10 calculated using FactSage. The Gibbs free energy using CO ranges −610 to −919 kJ compared to −469 to −727 kJ using H2 over the temperature of 700 to 1000°C. Hence, it is clear that the reduction with CO provides higher negative Gibbs free energy, leading to more robust spontaneous reduction compared to the reduction with H2.
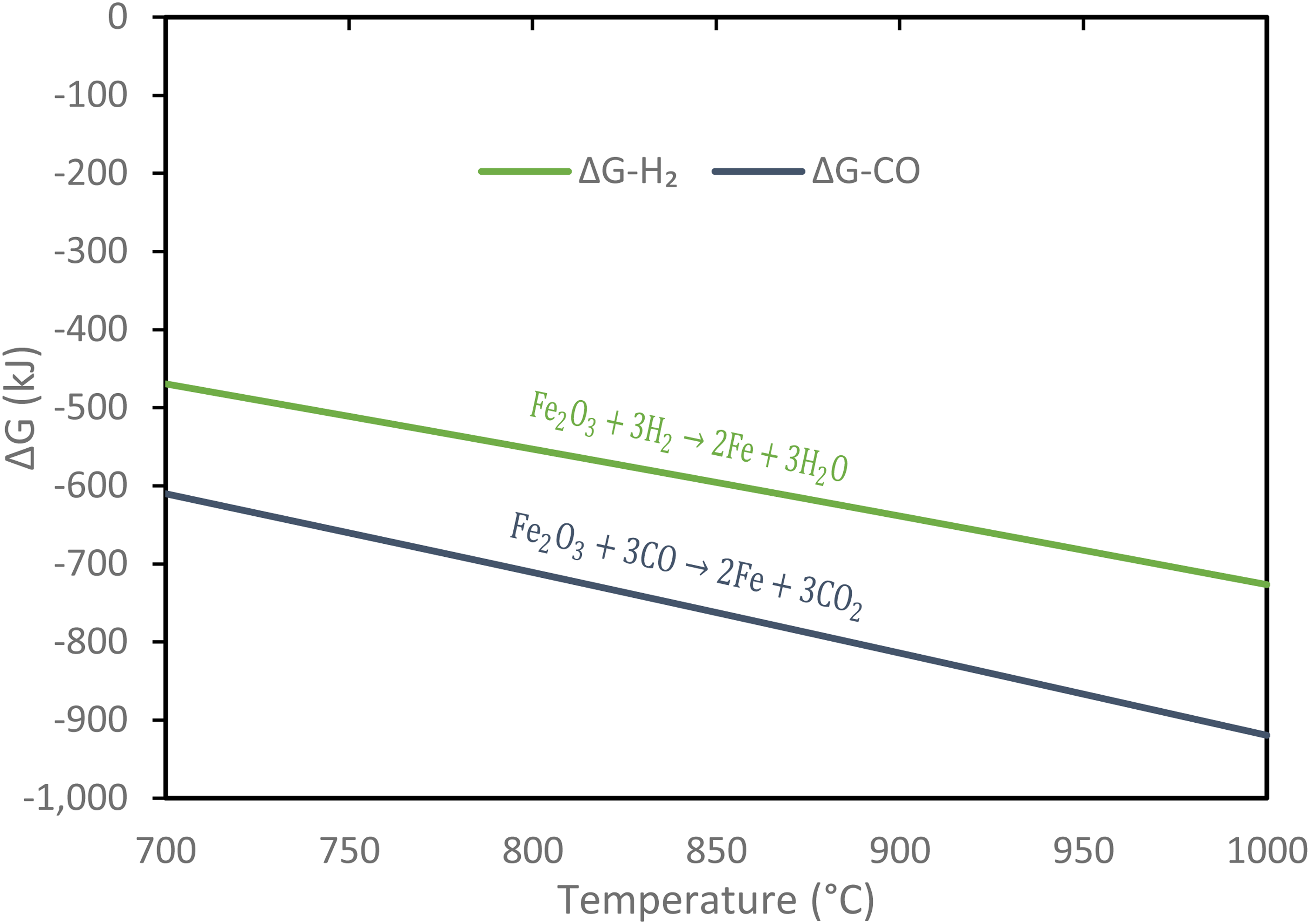
Gibbs free energy with respect to temperature for the reduction of haematite using H2 and CO.
The effect of the excess reductant on energy consumption from H2 and CO is shown in Figure 11. Increasing the excess reductant increases the energy consumption (LHV basis) under both conditions (Figure 11(a)). The energy consumption under the 85%H2 case varies between 5.66 and 6.78 GJ/Fefeed, compared to 5.81–6.97 GJ/Fefeed in the 70%H2 case. These variations in energy consumption under different H2 and CO flows are due to the variation in consumption rate (Figure 9(b)) and the calorific value of H2 and CO gases.
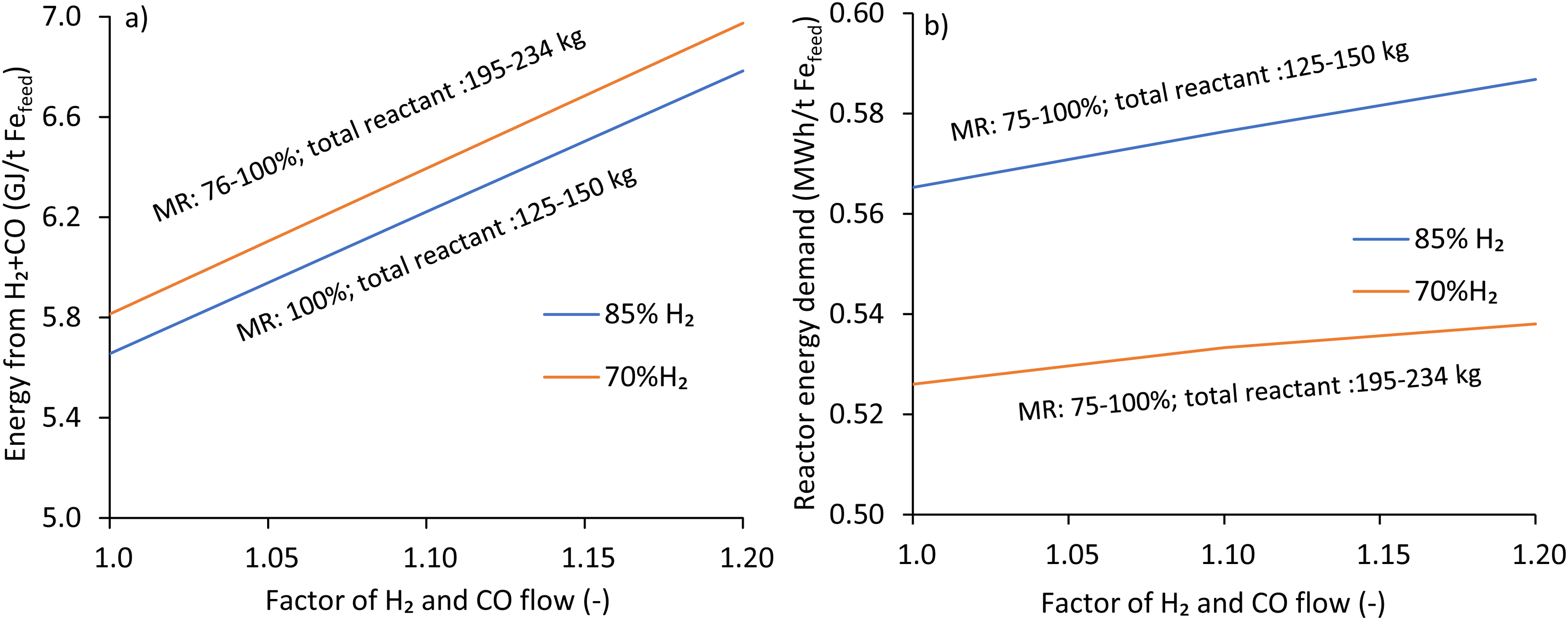
The effect of excess reductants on energy consumption from H2 and CO, H2:CO: 85: 15 and 70: 30 using iron ore A (Fe 69 wt.%, gangue 5.2 wt.%).
The effect of the excess reductant on reactor energy demand is shown in Figure 11(b). Excess reductant increases the reactor energy demand under blended conditions. At full MR (MF: 1.2), the 85% H2 case consumes about 0.59 MWh/t Fe feed in contrast to 0.54 GJ/t Fe feed under 70% H2 cases.
The arguments for introducing CO into the shaft furnace relate mainly to generating a DRI with some carbon, as this can assist with safe handling and melting in subsequent steel making stage but there are also some benefits in terms of the energy requirement for the process, those this has to be balanced against the amount of CO2 generated and energy associated with generating CO, the later issue beyond the scope of this study.
Conclusions
Thermodynamically increasing the hydrogen flow rate and temperature helps increase the reducibility and MR. A full metallisation can be achieved with feed H2 flow of 90 kg/t DRI at 1000°C, which is 100, 110 and 130 kg using 900, 800 and 700°C, respectively. However, 54 kg H2 is consumed to reduce one tonne of haematite if the off gases are recycled.
The ore quality plays an important role in energy consumption, that is the higher the ore quality, the lower the energy consumption. For example, at 900°C, the reactor energy consumption using top-grade ore (Fe: 69 wt.%) was 0.58 MWh/tFefeed compared to 0.73 MWh/tFefeed using lowest grade ore (Fe: 56.5 wt.%).
The reduction behaviour using pure hydrogen significantly differs from that of blended CO and hydrogen atmosphere. Under blended conditions, the variation in performance parameters concerning temperature does not exist. Hence, the concentration of H2 and CO was increased proportionately with a MF of 1.1 and 1.2 to see their effect on reducing iron ore. It is seen that decreasing the concentration of H2 by increasing CO in the blend helps increase the MR. The consumption of H2 using 85% H2 was 90% compared to 86% using 70% H2, while the consumption of CO was 100% in both cases. The results are a repercussion that the reduction of CO is thermodynamically favoured over that of H2.
The results suggest that blending a small percentage of CO with hydrogen is more beneficial than that of pure hydrogen in terms of metallisation, hydrogen consumption and overall energy consumption. For example, only 15% blending of CO with 85% hydrogen results in 100% metallisation requiring a supply of only 46 kg hydrogen in contrast to at least 100 kg at 900°C.
Footnotes
Acknowledgements
Authors would like to acknowledge Heavy Industry Low-carbon Transition Cooperative Research Centre (HILT CRC) with project number HILT.RP1.004 for financial support. Also, Authors would like to acknowledge Australian federal Government and industry partners: HILT, CRC Limited (CRC Entity), Roy Hill Holdings Pty Ltd, FMG Procurement Services Pty Ltd, Grange Resources (Tasmania) Pty Ltd, OneSteel Manufacturing Pty Limited and MHI Australia Pty Ltd
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Heavy Industry Low-carbon Transition Cooperative Research Centre (HILT CRC), (grant number HILT.RP1.004).