Abstract
There is a significant and necessary drive towards reducing CO2 emissions produced by iron and steelmaking industries. Binding Solutions Ltd has developed a technology to contribute to the reduction of CO2 in the short and long term, by introducing a cold-bonded iron ore pellet to the furnace burden. Binding Solutions’ cold-agglomerated pellets, manufactured from magnetite and hematite iron ore, were tested in accordance with ISO standard tests alongside a standard market pellet. ISO standards were developed to assess the quality of lump ore, sinter and hot-bonded or alternatively known as indurated pellets, and there can be anomalies when testing cold-agglomerated pellets by the same methods. Additional test methodology, outside the scope of international standards may be utilised to give a fuller picture of pellet characteristics. The HOSIM test method was used to assess the properties of cold-bonded pellets of different minerologies. Results found that the time needed to reach a set point of reduction for a cold-agglomerated pellet is shorter than for an indurated control pellet. The data also showed that for the same iron ore source, alternate cold agglomeration methods can have a significant impact on the balance of pellet properties.
Introduction
Iron ore pellets were developed as an efficient way of utilising iron ore within a blast furnace burden and are used in both the blast furnace and direct reduction routes of iron production. The conventional process for producing an iron ore pellet is via an induration process and global production is in the region of over 400 Mt per annum. 2 The process to produce an indurated iron ore pellet is carbon intensive, requiring drying and firing with fossil fuels to provide the thermal inputs to create ceramic bonds. 3
Binding Solutions has developed a disruptive technology to produce cold-agglomerated iron ore pellets with technical properties that compare directly with those of an equivalent indurated pellet. A typical Binding Solutions Ltd (BSL) cold-agglomerated pellet (CAP) product is calculated on a whole lifecycle basis to contribute a significantly lower carbon intensity to that of an equivalent indurated product.4,5
The achievable carbon savings between the different pellet types are shown in Table 1.
Quantity in the Mt of CO2 release per Mt of pellet produced. 6

When introducing a new material type to a high volume and complex operation such as a blast furnace, an operator will require assurance that no unacceptable disruption will occur.
There are many common test standards for iron ore pellets in the format of International Standards Organisation (ISO), ASTM, JIS, etc. These are valid for assessing iron ore pellets produced by the induration method and provide for benchmarking to compare different producers and sources.
For BSL CAP technology, a test regime that is outside the scope of those standards which are commonly used may be more appropriate to de-risk an operation. ISO methods were developed in order to compare lump ore, sinter and hot-bonded (indurated) pellets. The BSL CAP is neither lump ore nor hot-bonded and these tests may not produce data which reflect the potential true performance of the product. In BSL CAPs the binders used for agglomeration will be pellet constituents and have the potential to affect the reduction behaviour of the pellets.
Another major difference with BSL CAPs is that as no induration heating has taken place, the original iron ore of the material will be unchanged in terms of oxidative state. In cases where the iron ore is magnetite it means all the reduction tests will be carried out on magnetite and not hematite. The difference between magnetite and hematite reduction is discussed further in this article.
The testing required to assess a ferrous burden may be broadly split into two categories:
Physical Properties – The robustness of an iron ore pellet to withstand transportation and handling Metallurgical and Chemical Properties – To measure the robustness of the pellet during reductive phase changes and the rate at which they occur in various transit zones in the blast furnace.
7
The test data is used by the blast furnace operator, in conjunction with other feed materials, to manage the correct charging rates for acceptable furnace performance.
One consideration is that during the firing stage of producing indurated pellets, the minerology is altered to hematite, α-Fe2O38,9 in almost its entirety regardless of the original feed composition. One characteristic of using magnetite ores towards making indurated pellets is that this ore will oxidise during the induration process resulting in a lower iron content.
In the case of a cold-agglomerated pellet, the original minerology is the same as the source material at the point of feed, which may for example be Fe3O4, α-Fe2O3, FeO(OH), or in mixed combinations. This factor can create unexpected outcomes, as the standard test methods were developed around the indurated pellet type. A literature search indicates low levels of reported work carried out on cold-agglomerated pellet type, due to the novel technology. Unknowns can include reduction rate, disintegration under different conditions and propensity for swelling.
This paper reports a study into the metallurgical reduction behaviour of BSL CAPs under simulated blast furnace conditions by various known ISO standards common in this field of work. A non-ISO standard test method was carried out – the HOSIM test, which is non-isothermal and has a variable reducing gas composition as the test progresses.
The HOSIM test was devised at Ijmuiden Steelworks in the 1980s to better simulate the gas composition and temperature regime experienced by the burden as it descends the blast furnace shaft. 7
Materials selection and characterisation
Cold-agglomerated iron ore pellets were produced by Binding Solutions at their Technical Centre in Teesside, UK and are described in the table below. Production methods use chemical bonding and mechanical pressure, details of which are trade secrets. The dimensions of the BSL CAP product are more ordered than an indurated pellet, though production techniques are used to give a similarity in appearance.
An indurated pellet from the open market was tested alongside the BSL CAPs, as a control reference. Indurated pellets are formed by a pan-pelletisation method before being subjected to heat and this produces a natural variance in size and shape in comparison to a pressure-formed product such as the BSL CAP. The reference numbers as well as a brief description of each sample is shown in Table 2.
Reference number and description of the different pellet type samples studied.
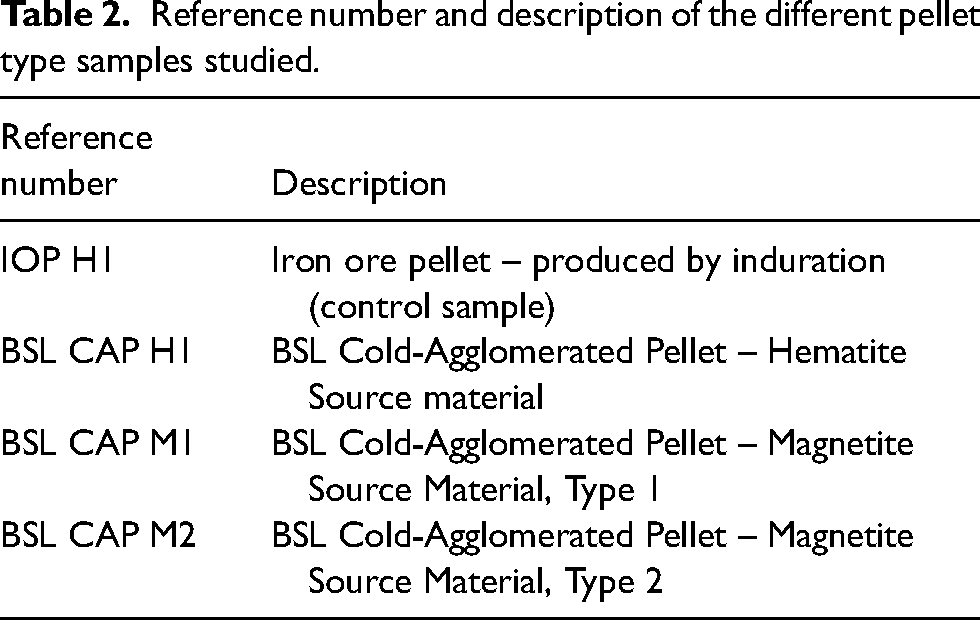
Characterisation of mineralogical type by XRD
X-ray diffraction (XRD) on powdered samples was carried out on a PANalytical Empyrean Series 2 Diffractometer operating Cu Ka1 (λ = 1.54056 Å) radiation; using an automatic slit at a step size of 0.026°, a net time per step of 304.2 s, power settings of 40 kV and 30 mA with a scanning 2θ range from 5 to 100°. To combat the problem of fluorescence from iron-based samples the proportional height detection, PHD was changed to 45%. This acts to better identify and match the peaks within the experimental diffractogram to those from reference phases.
XRD analysis was used to determine the composition of the pellet samples. The phases identified and the quantity of the phases by semi-quantitative analysis are shown in Table 3. The XRD diffractograms with the main peaks of the minor phases labelled are shown in Figure 1.
XRD diffractograms of showing IOP H1, CAP H1 as hematite and CAP M1 and CAP M2 as magnetite ore. Main peaks of secondary phases, CaMg(Si2O6) (#), SiO2 (*) and quartz (x) labelled.
Major and minor phase determined for each sample using XRD.

Test method
ISO 4700:2015 – Determination of crushing strength Determines average compressive strength until breaking point of 60 individual pellets in a batch. Reported as Decanewton (daN). The strength measurement of the BSL CAP is carried out in a radial direction, which gives lower values than an axial direction and has correlation to an indurated pellet.
ISO 3271:2015 – Determination of tumble and abrasion values Determines the attrition of a batch of pellets under mechanical agitation
ISO 7215: Determination of reducibility by final rate of reduction Determines the percentile loss of available oxygen from iron oxide after 180 minutes, under reductive gas conditions: 30% Carbon Monoxide/70% Nitrogen at a constant 900 °C
ISO 4696-2: Determination of low temperature reduction-disintegration indices by static method Under reactive gas conditions: 30% Carbon Monoxide/70% Nitrogen at a constant 550 °C
ISO 4698: Determination of the free-swelling index Under reactive gas conditions at 950 °C, to assess the potential for gas flow restriction in a Blast Furnace
Figure 2 relates to the experimental setup used to carry out the ISO tests of 7215 and 4696-2. This allows the pellets to be heated in reducing atmospheres at a controlled temperature. The inert and reduction gases which are used during the ISO tests are injected via the bottom of the reactor at controlled flow rates. The gases are passed through both ceramic beads and a porous mesh to evenly disperse the flow as well as reduce the chances of the flowing upward gas affecting the BSL CAPs. The mass of the BSL CAPs changing during the ISO tests are measured via a balance positioned under the furnace system. The balance is tared to zero so only the change in mass of the BSL CAPs are being monitored.
Determining rate of reducibility and durability in BF conditions using HOSIM testing
HOSIM (HOogovens. Simulative Model) 7 testing is used to study performance of burden components in simulated BF conditions. This test uses a gas composition that alters based on the mass change throughout the test, whilst test temperature increases over time, from 400 to 900 °C simulating a realistic BF environment. The gas composition is made up of a mixture containing CO, CO2, N2 and H2.
Test chamber to simulate BF conditions for the reduction of pellets.
HOSIM is designed to measure the change in the atomic ratio of oxygen over iron, O/Fe, as oxygen is removed from the iron ore from reduction. The reduction of a sample is considered complete when the O/Fe value reaches 0.52. To do this the iron ore present within the sample, either hematite or magnetite, as well as their quantities are needed. In the ISO tests the equation for determining reducibility are based on all the iron oxide being hematite. Although this works well for hot-bonded pellets where the vast majority if not all the iron oxide would be hematite. In the case of BSL CAPs the iron oxide type could heavily vary depending on the iron ore used.
Testing was completed at the Steel and Metals Institute (SaMI) on a bespoke automated HOSIM test programme on equipment supplied by RB Automazione.
HOSIM tests require 500 g samples of pre-dried (at 105 °C) pelletised material to be placed into a testing rig. Typical pellet sizes range between 10 and 13 mm; however, the BSL CAPs were generally larger than this, with samples at diameters of 12 to 16 mm. Once the programme was complete, the retort was left to cool in an inert atmosphere, prior to the removal of the specimens.
To determine the disintegration, ergo strength of the specimens after reduction, the specimens were placed in a small tumble drum and tumbled for 300 revolutions at 30 rpm. A sieve analysis was performed to determine the percentage by mass passing 3.15 mm.
The reducibility of a sample is measured from the time taken to reach the molecular O/Fe ratio of 0.52, which equates to the end of the indirect reduction phase in the blast furnace. This test data varies from that produced from ISO 7215, which gives the amount of reduction achieved for a set time/ gas composition and temperature.
Optical and elemental analysis using SEM-EDX
Scanning electron microscopy with energy-dispersive X-ray (EDX) spectroscopy, SEM-EDX, was used to study particle morphology and elemental identification on particle surfaces. SEM-EDX was carried out using a Zeiss Sigma 300 VP Scanning Electron Microscope operating at 20 kV with two detectors, a secondary electron detector and a backscatter detector. Elemental composition was carried out by EDX spectroscopy using an Oxford Instruments 170 Ultim Max detector.
In this study, SEM-EDX was employed as an optical investigation technique for pellets pre- and post-heating. SEM methods would be used to study any visual/elemental changes occurring on both the surface and bulk of the pellet. Before analysis pellets were cold mounted into a resin medium and mechanically polished to obtain a flat even surface.
Results
Physical properties of the pellets
For the purposes of benchmarking and comparison, the physical properties of all pellets before being reduced were measured. The physical properties presented in Table 4 are those of the cold compressive strength (CCS), tumble index (TI), and abrasion index (AI). The CCS values were determined as the overall average of 60 BSL CAP samples, with the TI and AI determined from 15 kg batches of BSL CAP samples.
Cold compressive strength, tumble index and AI results for all samples.

In the case of the TI and AI values the BSL samples of CAP H1 and CAP M2 are comparable to that of IOP H1, representing the industrial standard of indurated pellets. This shows that BSL CAPs can match the indurated pellets for stability during movement and without abrasion causing mass disintegration. In the case of CCS all BSL CAPs are within the recommended range stated by Prabhu et al. of >200 daN. 10 In the case of CAP M2 the CCS value reported is much higher than the other BSL CAPs. CAP M1 and M2 are prepared using the same iron ore source and differ only in binder formulation. The cause for the difference in CCS can only be attributed to the iron ore to binder interactions and future studies on this will be required.
For blast furnace, BF applications the presented physical properties show BSL CAPs to be suitable products along with indurated pellets. For use in a BF, pellets would ideally have TI > 90%, AI < 10% and CCS values between >200 daN.10,11 The reason for this is that BSL CAPs will need to disintegrate and release fines as little as possible during both the movement in production and within the blast furnace shaft itself. If BSL CAPs release high amounts of fines from physical tumble, abrasion and force the fines can causes blockages within the BF shaft and hinder the reduction process significantly. 11
Metallurgical properties
Three tests were selected to assess:
Stability/fines generation in reducing conditions in the mid zone of the Blast Furnace – ISO 4696-2 Comparative time taken to reduce – ISO 7215 Volume increases due to reduction – ISO 4698
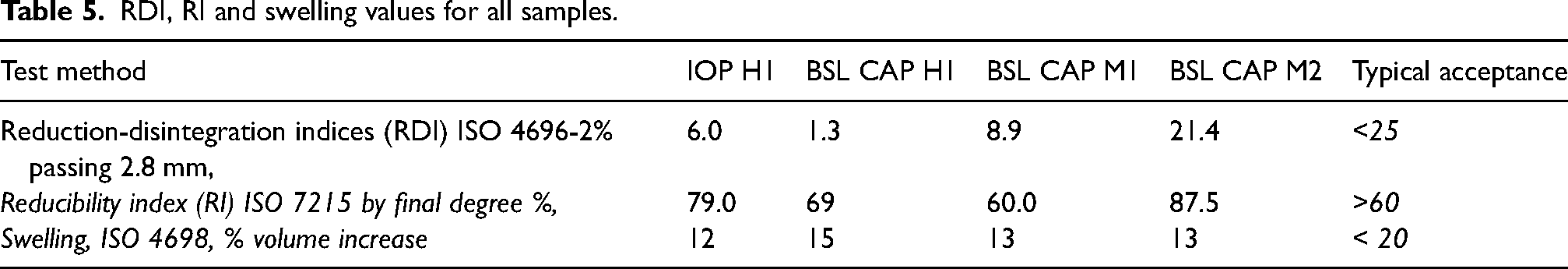
Table 5 shows the results from these tests for each sample.
RDI, RI and swelling values for all samples.
Under a reducing atmosphere of CO:N2 (30:70%) the reducibility index (RI) for all samples has been determined. Hematite and magnetite will have different initial reduction reactions under blast furnace conditions when carbon monoxide, CO is used as the reducing agent at 900 °C. A representation of the differences in the reduction behaviour is shown via the thermodynamics of these initial reduction reactions.
9
This shows the initial reduction for hematite to be exothermic, whilst that for magnetite would be endothermic. The reduction of hematite to magnetite also has a lower dependency on CO concentration, when compared to the reduction of magnetite which requires much higher CO concentrations.
11
These factors need to be considered, along with others when comparing the reducibility behaviour of BSL CAPs from hematite and magnetite ores.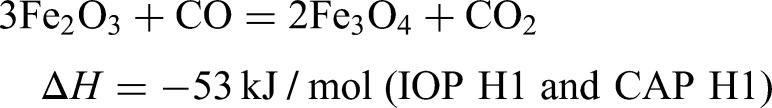
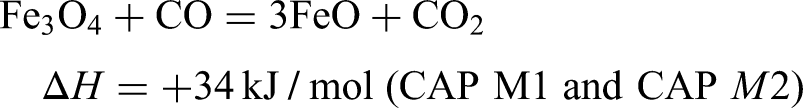
In the case of BSL CAP M1 and M2, both pellet batches use a magnetite iron ore but differ in their binder formulations. BSL CAP M1 formulation shows the best structural stability with an RDI of 8.9% compared to that of CAP M2 at 21.4%. However, in the case of reducibility performance BSL CAP M2 has an improved higher RI of 87.5%. The reason for the differences can be attributed to the formulations with the binders, with those in BSL CAP M1 favouring RDI stability at the cost of reducibility efficiency. In the case of BSL CAP M2 the binders do not have the same level of thermal stability as those used in M1 and result in higher RDI values. However, this can lead to higher reduction gas permeability of the BSL CAPs which favours better reducibility towards iron metal. Acting as a further example of the inverse relationship of reducibility to reduction-disintegration.12,13
In the case of swelling both show the same value of 13% indicating that different formulations can give the same result.
These initial results show how cold-agglomerated technology can produce BSL CAPs that can be used in a blast furnace as a constituent of the burden. All these tests are published by the International Standards Organisation (ISO) and are by nature relevant to the existing technology of indurated iron ore pellets, and the static test conditions are relative to this product type. Further testing was undertaken to give additional confidence for the cold-agglomerated pellet technology.
Optical imaging of reduced cold-agglomerated pellet
The BSL CAP H1 samples, post-ISO 7215 reduction were cut and mounted in epoxy resin for analysis via SEM-EDX analysis. This was used to obtain microscopic images and showed that metallic Fe was present throughout the entirety of the pellets from the surface to the centre. This also shows the efficiency of the CO molecules to move through the body of the cold-agglomerated pellets and achieve reduction. Figure 3, SEM images of BSL CAP H1 the surface, quarter depth, and centre of the pellet.

SEM-EDX images at 200× magnification of the diameter of a BSL CAP H1 sample showing intensity of dark grey FeOx particles and light grey coloured metallic Fe particles.
A polynomial graph of the elemental intensity ratio between Fe/O atoms, Figure 4, reinforces the statement that reduction has occurred right to the core of the BSL CAP. The Fe/O intensities are shown to be the most intense at the opposing sides of the BSL CAP but also show that reduction has occurred within the BSL CAP core.
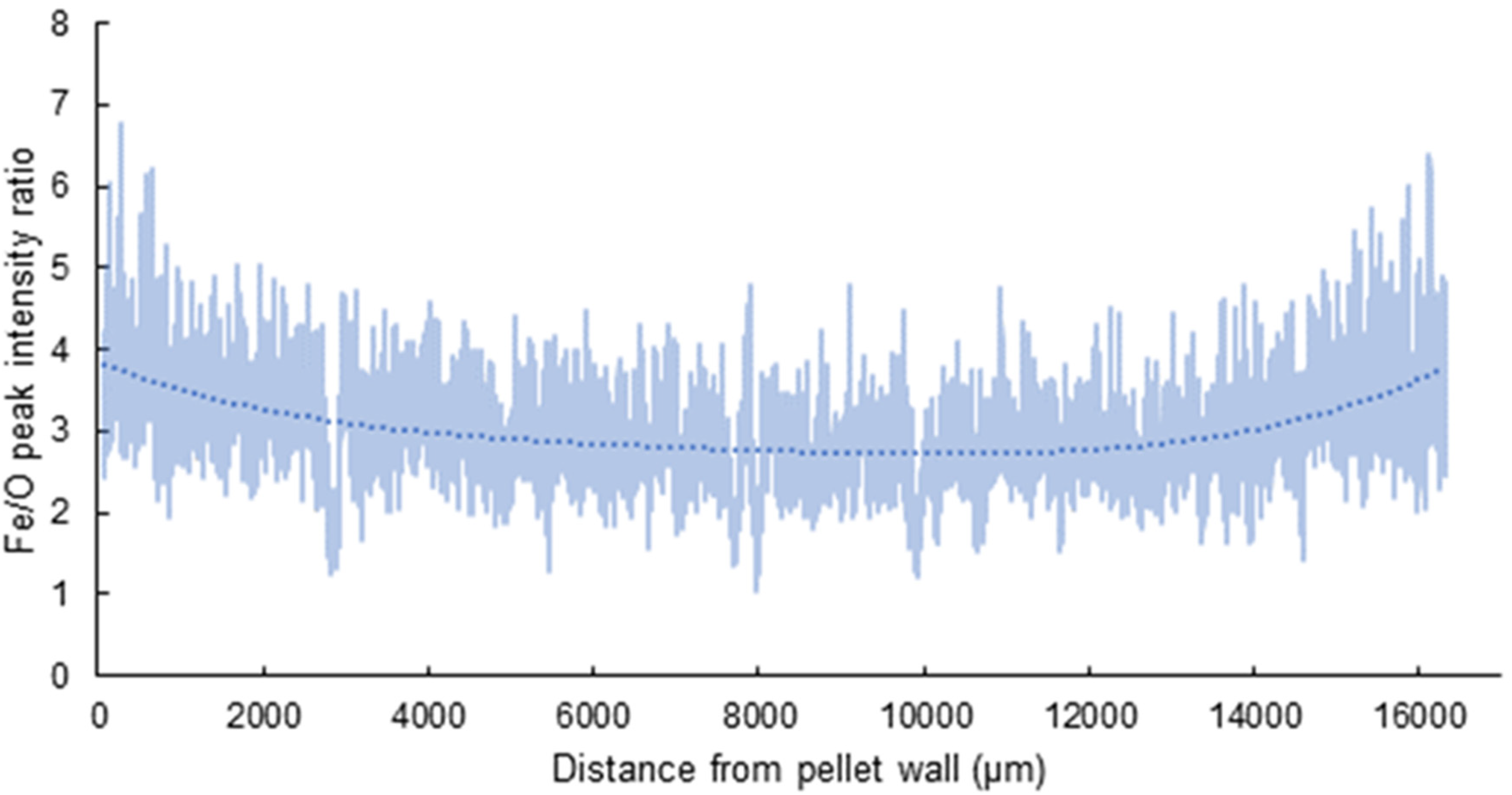
Fourth order polynomial curve of Fe/O intensity ratio versus distance from one CAP surface through the centre to the opposing side. Curve shows that going through the pellet from surface-to-surface reduction has taken place throughout.
After studying the reducibility properties of cold-bonded pellets via ISO 7215 methods, more in-depth studies were carried out. This involved studying the reducibility performance in simulated blast furnace conditions via HOSIM testing.
HOSIM testing
Batches of BSLCAP H1 and BSL CAP M1 were produced using the iron ore types of hematite and magnetite from different sources. This was to investigate how the change in minerology would affect the overall reduction behaviour of a BSL CAPs.
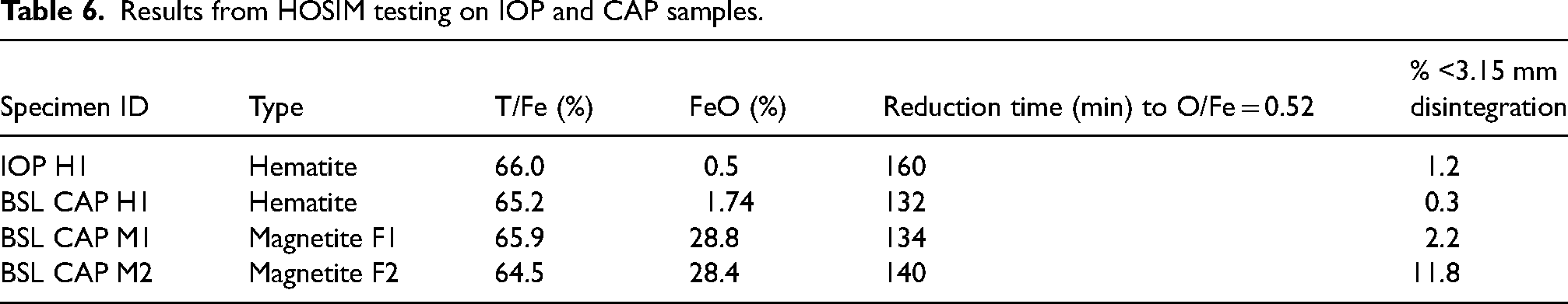
Two formulations were utilised to produce BSL CAPs using magnetite ore, batch BSL CAP M1 and M2. These were produced to investigate the effects of different formulations on the same iron ore had on reducibility. Results shown in Table 6 report reduction times were similar for the hematite and magnetite BSL CAPs with a difference of approximately two minutes. A lower level of disintegration was obtained from BSL CAP H1, with less than 0.3% lost. BSL CAPs M1 and M2 also exhibit relatively low disintegration, with less than 2.5% loss. Typically, where the HOSIM test is used as part of a burden assessment at an iron production site, values <5% are acceptable.14–16
The reduction time for the two magnetite formulations differs by only 6 minutes, indicating similar rates of reduction. Batch BSL CAP M2 exhibited a disintegration higher than 10%.
HOSIM testing involves measuring the reduction of a sample until the atomic ratio of O/Fe reaches 0.52. To do this the total iron content within the sample (T/Fe%) and the FeO (%) is required to determine the initial O/Fe value. The FeO (%) is the amount of Fe2+–O groups within the sample. This is important to quantify as this can be used to determine the quantity of magnetite within a sample to obtain an accurate reduction time. Hematite will only have Fe3+–O so any Fe2+–O present will be for magnetite, wustite would not be found naturally in iron ore as it is unstable below 570 °C. 17 Results from the HOSIM tests on the samples are presented in Table 6.
Results from HOSIM testing on IOP and CAP samples.
HOSIM testing begins at 400 °C, when the reducing atmosphere is introduced, and the balance is zeroed. During testing it was observed that the BSL CAPs show signs of mass loss during the heating phase, without any reduction gases present. These mass losses under what is considered inert conditions show a loss of up to 2% total mass of the CAP samples. This value is greater than the quantity of any material within the CAPs that undergo pyrolysis and, as mentioned previously all samples were pre-dried to remove any moisture. It is proposed that the pyrolysis of the BSL binders is causing an internal reducing atmosphere, that is, the release of gaseous CO towards reducing the CAPs. As the gas is released is reduces the iron oxide through removing oxygen with the CO gas converting to CO2. Figures 5 to 8 show the HOSIM results for all samples which compare mass loss and mass loss rate over time and temperature. Based on the mass losses, the points where the samples have undergone reduction reactions towards the next iron oxide phase leading to metallic iron has been identified on the mass loss curve. This is to represent the decrease in oxygen over the reaction and how the mass loss was used to identify the atomic ratio of O/Fe during the overall reduction progress. These points have also taken into consideration the excess loss of mass the samples underwent during the initial inert conditions. The mass loss curve also shows the point at which the balanced would be zeroed at 400 °C. This identifies the mass lose occurring under just the inert conditions which was used to determine how much potential early reduction is occurring.
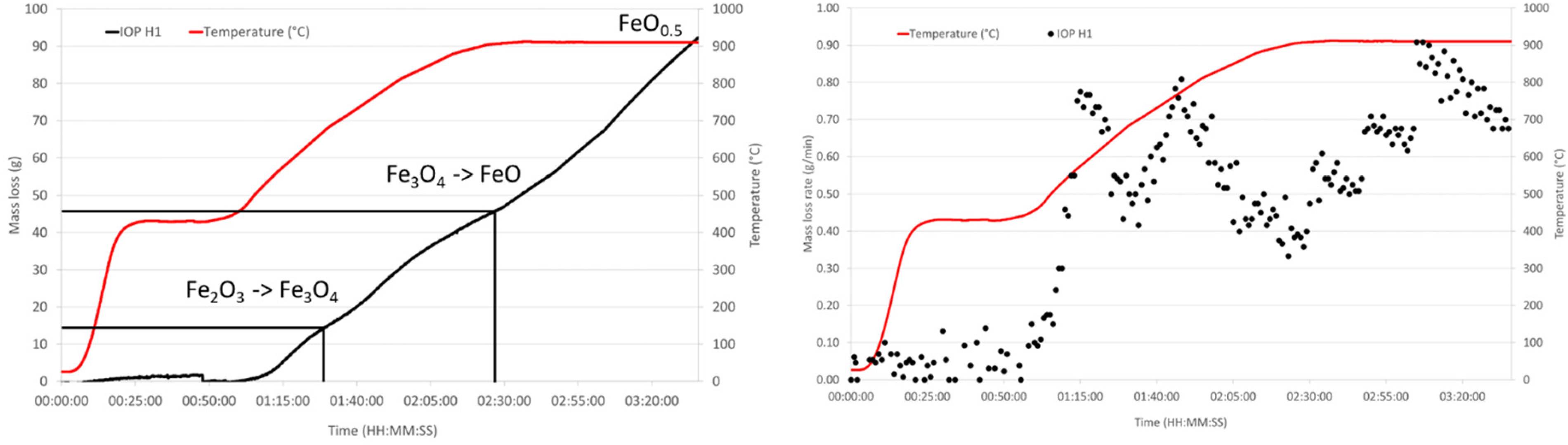
HOSIM graphs showing the loss of mass and mass loss rate over time for IOP H1. Also, shown on the mass loss curve are the points where the reduction reactions have been completed.
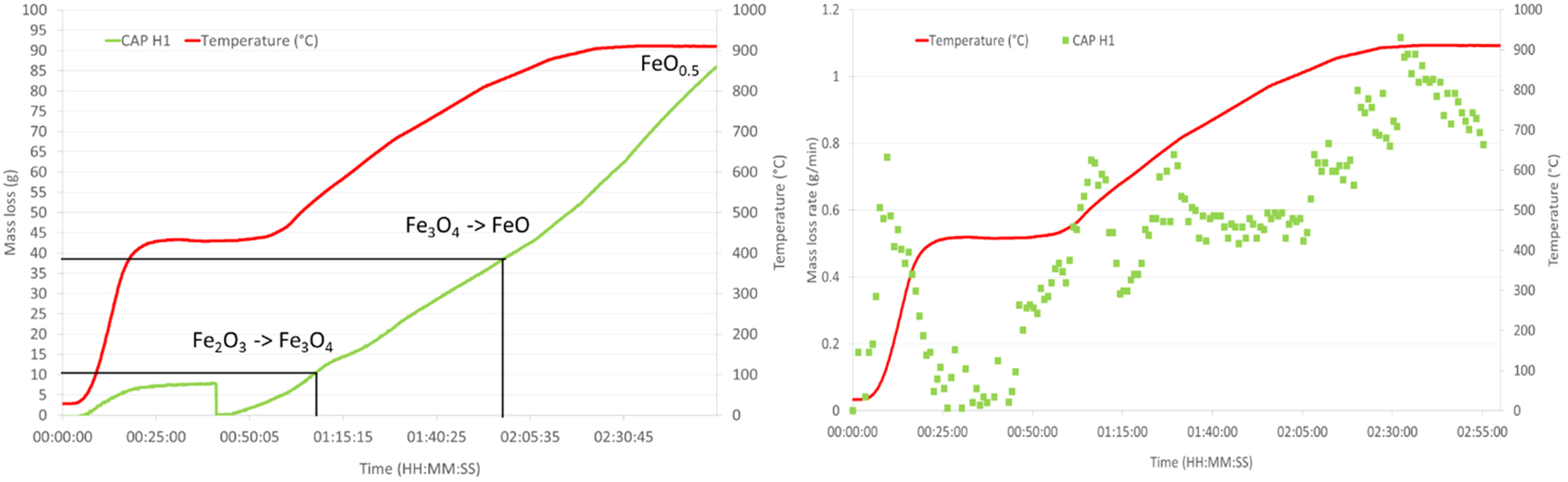
HOSIM graphs showing the loss of mass and mass loss rate over time for CAP H1. Also, shown on the mass loss curve are the points where the reduction reactions have been completed.
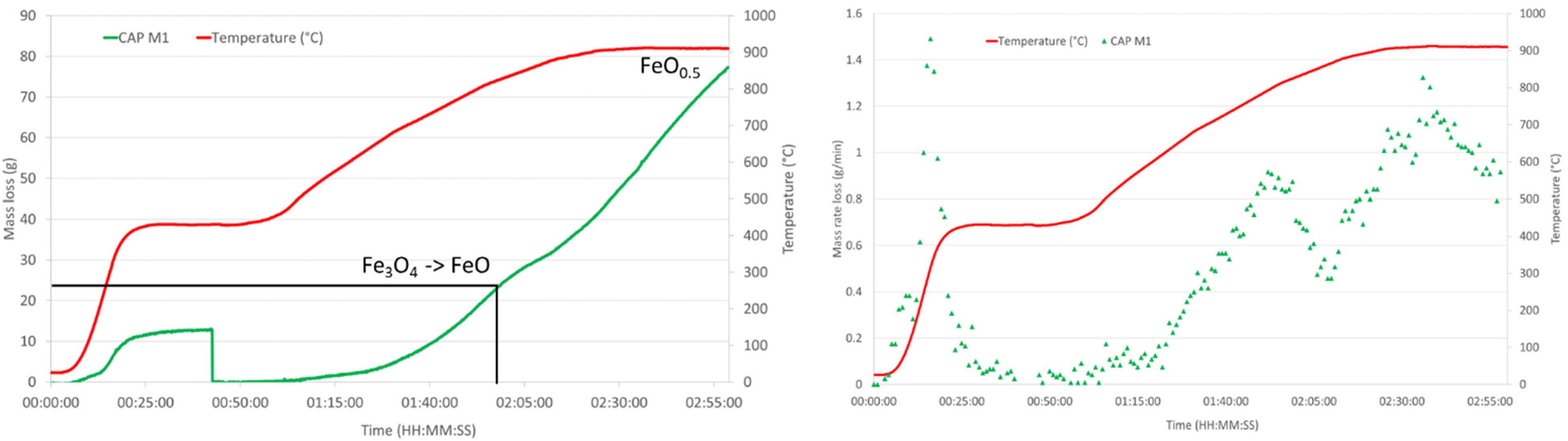
HOSIM graphs showing the loss of mass and mass loss rate over time for CAP M1. Also, shown on the mass loss curve are the points where the reduction reactions have been completed.
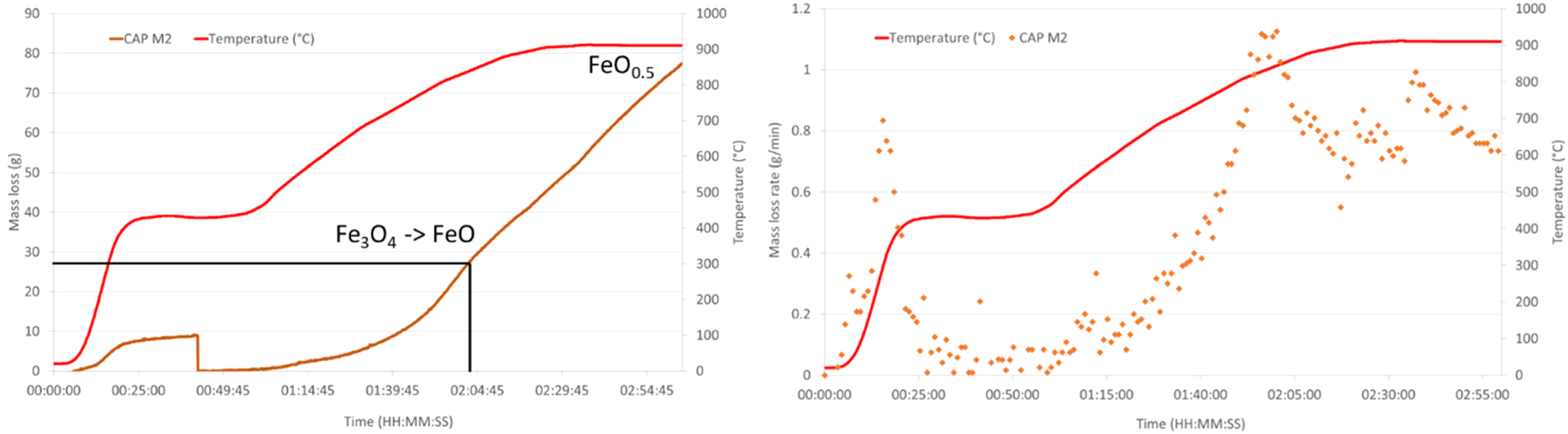
HOSIM graphs showing the loss of mass and mass loss rate over time for CAP M2. Also, shown on the mass loss curve are the points where the reduction reactions have been completed.
The comparative plots of the mass loss and mass loss rates over time for the different sample types are shown in Figures 9 and 10.
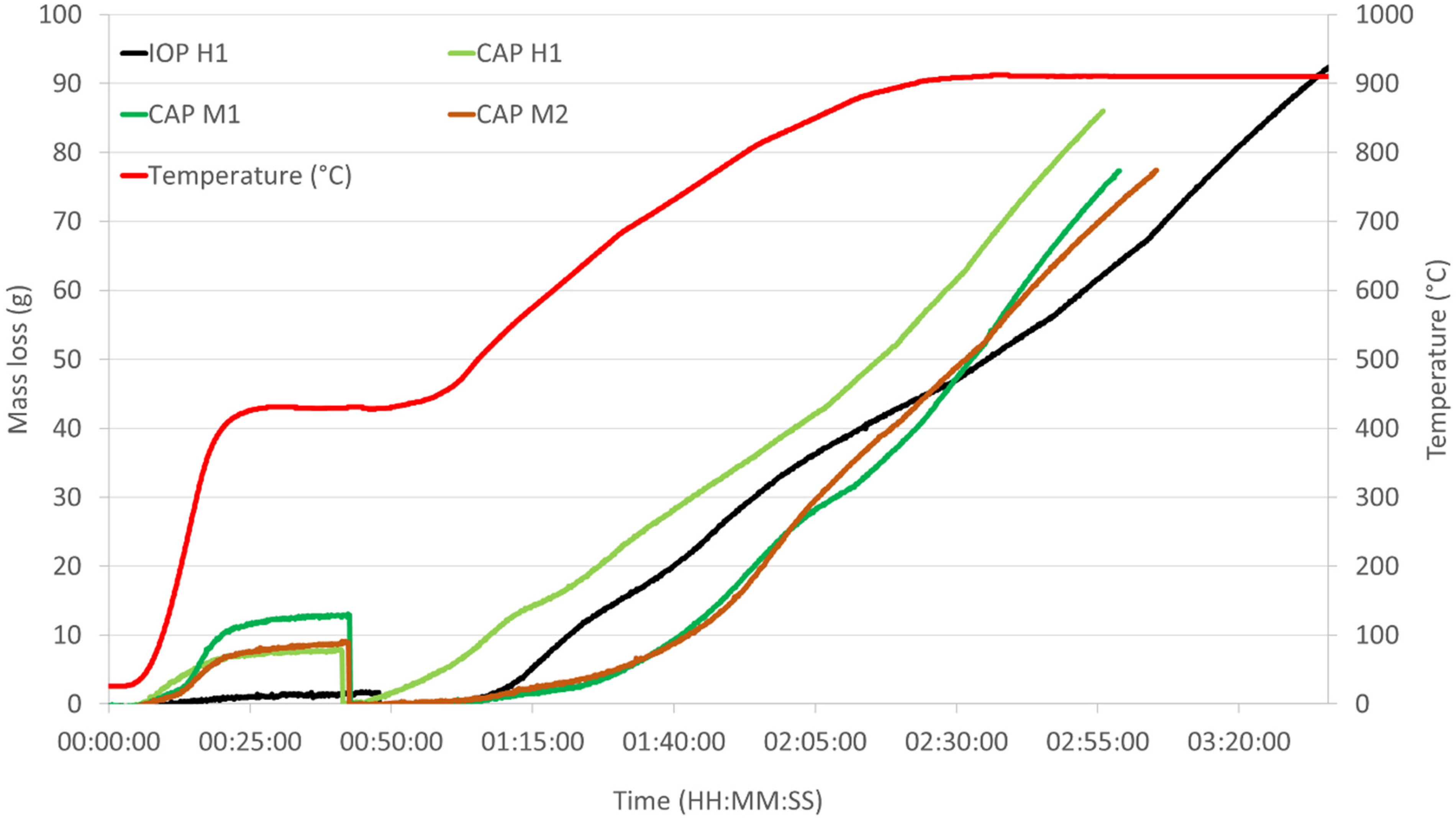
HOSIM curve for mass loss over time for control IOP and all CAP samples.
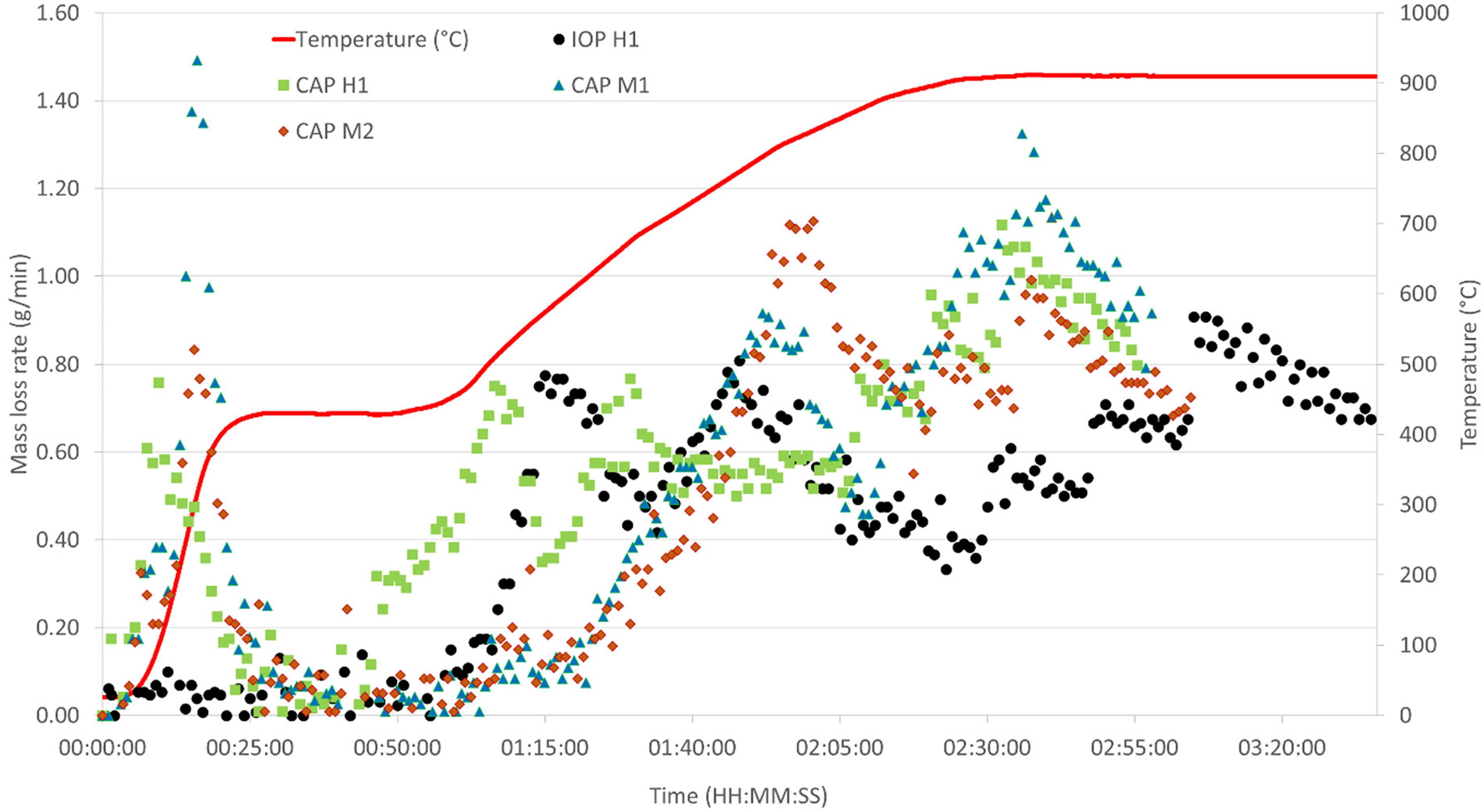
HOSIM curve for rate of mass loss over time for control IOP and all CAP samples.
The mass loss rates show different trends in the curves of the hematite-based CAP H1 pellets compared the magnetite-based CAP M1 and M2. CAP H1 shows a distinctive increase in mass loss rate at the point where the reductive atmosphere is introduced, and the temperature rises. Two distinctive peaks of increased rate loss are observed before another one is seen at the point the temperature stabilises. In the case of the magnetite pellets, they show distinctive rate loss peaks late into the HOSIM test. The reason for the different trends is due to the difference in reducibility properties of the different ores.
The difference in minerology will have a significant impact on reducibility behaviour. This needs to be considered when comparing the BSL CAPs, as hematite reduction will be faster and more favoured than magnetite, especially at higher temperatures. This is due to the lower diffusivity of oxides within the crystal structure of magnetite caused its denser and more stable crystal structure. 18 During the HOSIM test the CAP samples are subjected to ramping temperatures which makes comparing their thermal behaviour from phase changes complex. Further studies will be carried out to understand the behaviour of CAPs based on their different minerology.
HOSIM tests are designed to be completed once the O/Fe ratio reaches 0.52 that is determined from the loss of mass in oxygen. Figures 8 and 9 and Table 6 show that all the CAP pellets were 20–30 min faster than the indurated pellets to reach the end point of the test.
Photographs (Figures 11 to 14) of each sample batch before and after HOSIM testing were taken to show the deterioration caused during the testing. From simple visual analysis all samples appear to have undergone reduction resulting in what appears to be sponge iron. This is from the change in colour towards a metallic grey for all samples, regardless of iron ore precursor. More importantly each sample show signs of swelling which is a result of the reduction reactions from the original iron ore towards sponge iron. The presence of disintegration is also shown from these figures and agrees with the values reported in Table 6 with CAP H1 showing the lowest values, and CAP M2 the highest.

Photograph of IOP H1 before and after HOSIM testing.

Photographs of CAP-H1 before and after HOSIM testing.

Photograph of batch CAP-M1 before and after HOSIM testing.

Photograph of batch CAP-M2 before and after HOSIM testing.
SEM-EDX testing was carried out on HOSIM tested and untested CAP batches (Table 7 and Figures 15 to 17). This type of analysis is a semi-qualitative approach which is used to complement and reinforce the results shown from the HOSIM curves through elemental analysis on the surface of the particles which are located at the centre of the BSL CAPs themselves. The particle surface elemental composition results show the effects of reduction from HOSIM on the CAPs with the changes in the elemental composition (Table 7). This is done through comparing the molecular O/Fe ratio values on the particle surfaces at the centre of the CAPs for pre- and post-reduction. All CAP samples show a reduction in oxygen and an increase in iron on the surface of the particles within the CAP. It also shows reduction regardless of iron ore or agglomeration formulation used.
SEM results showing surface elemental composition of particles within the CAP samples before and after HOSIM testing along with changes in molecular O/Fe ratio.
Overlay images from SEM testing for BSL CAP H1 sample. Left is before HOSIM test and right is after.

Overlay images from SEM testing from CAP M1 samples. Left is before HOSIM test and right is after.

Overlay images from SEM testing from CAP M2 samples. Left is before HOSIM test and right is after.
Studying mass loss from heating cold-bonded pellets/briquettes under pure N2
The HOSIM results showed signs of mass loss for all CAP samples whilst still under inert conditions. To understand the mass loss, a mass loss profile study under an inert N2 atmosphere was carried out.
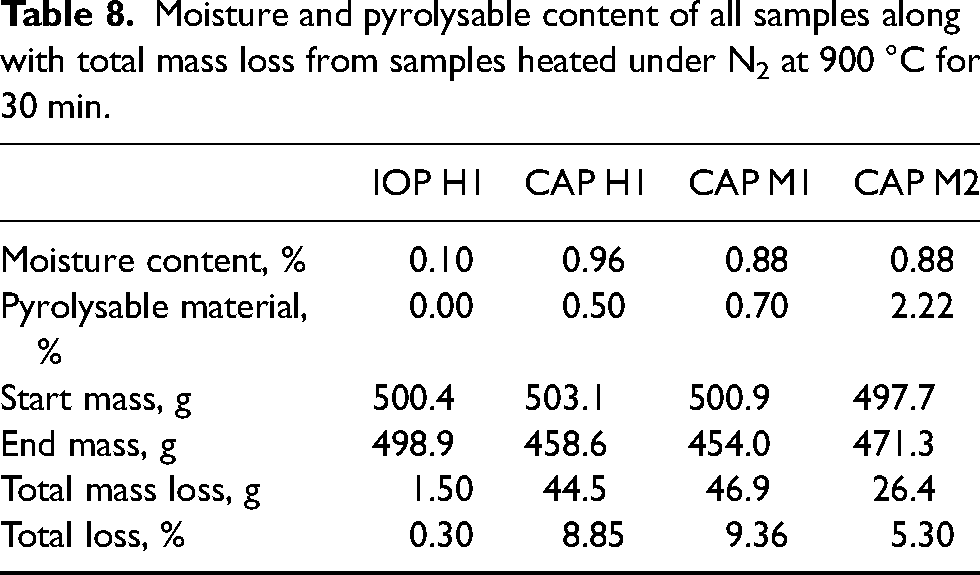
The mass loss from heating under N2 at 900 °C for 30 min was reported (Tables 8 and 9) and showed all CAP samples had a reduction in mass greater than the combined mass of moisture and material that will burn and be removed in atmospheres absent of oxygen.
Moisture and pyrolysable content of all samples along with total mass loss from samples heated under N2 at 900 °C for 30 min.
Theoretical, overall, and additional mass loss % for all samples from N2 at 900 °C for 30 min.
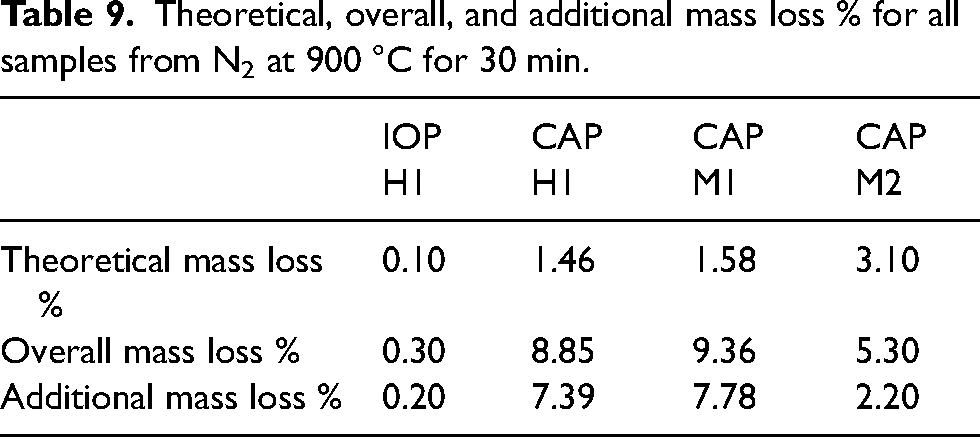
The control IOP H1 sample shows negligible mass loss from heating which is to be expected with no reductive gas present. In the case of the CAP samples, mass losses greater than those theoretically calculated were observed with additional mass losses varying from 3 to 8%. Even if all pyrolysable material had disintegrated this would not account for the overall mass losses observed.
These results indicate that the additional mass loss was not caused from the removal of chemical binder or from the HOSIM testing but because of the CAPs themselves. Initially it was proposed that the additional mass losses were the result of internal reactions from the pyrolysis of the binders in the BSL formulations used to agglomerate the CAPs. The proposed reactions are the internal release of CO that will interact with the iron oxide causing reduction reactions to occur and will result in CO2 being released.
This was later proven via the analysis of the samples after heating in an inert atmosphere via XRD (Figure 18). The diffractograms showed no other iron oxide phases alongside hematite in IOP H1, presence of magnetite (10%) with hematite in CAP H1, and the presence of wustite in CAP M1 at (15%) and CAP M2 (10%).
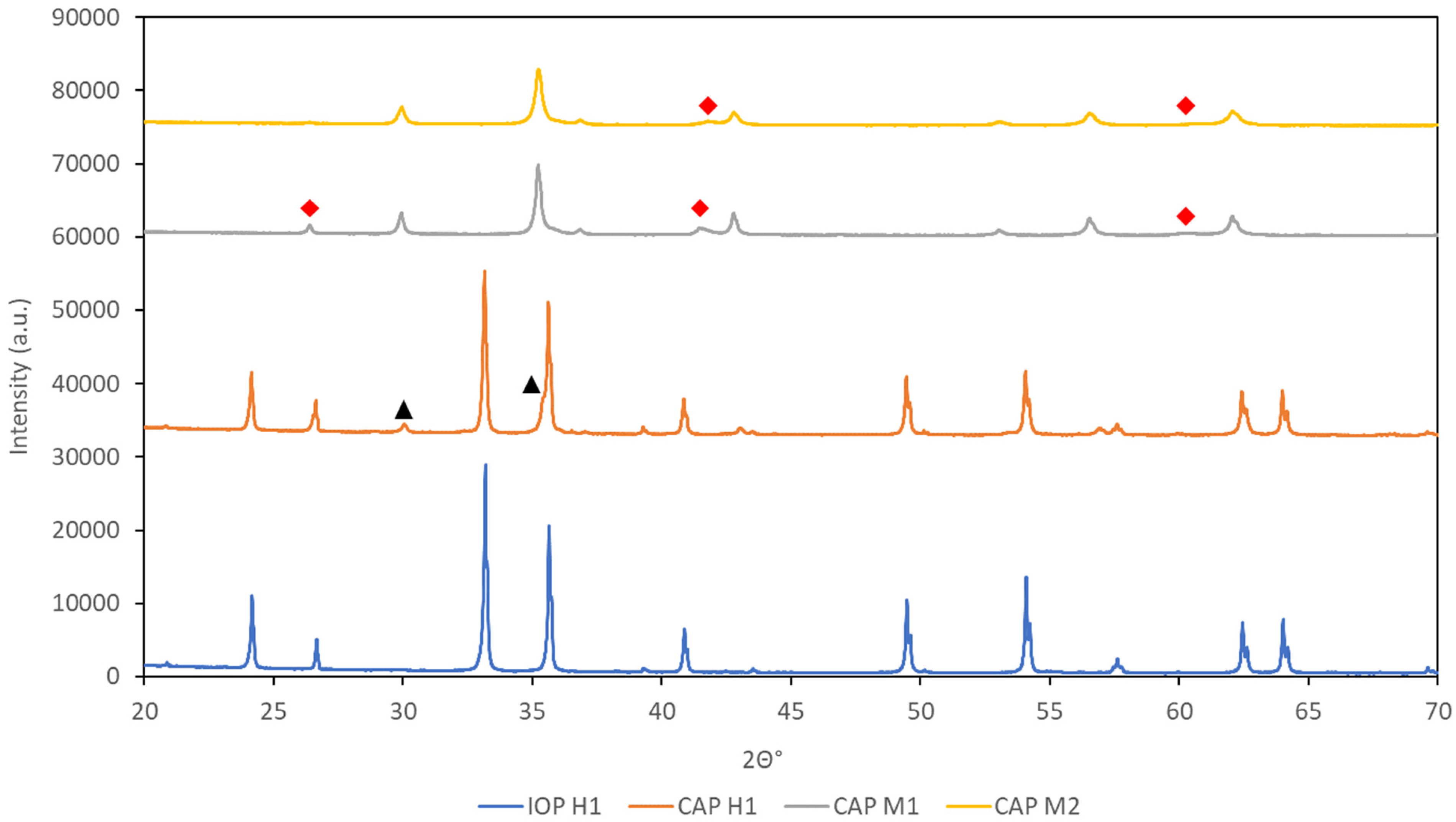
XRD diffractograms of IOP H1, CAP H1, CAP M1 and CAP M2 after heating in N2 at 900 °C for 30 min. The black triangles represent peaks belonging to magnetite, whilst the red diamonds represent peaks belonging to wustitie.
This type of reduction under an inert gaseous atmosphere could only occur with CAPs and not with the IOP samples. This is because the binders added for induration would be disintegrated almost completely during the induration process and not be present for the reduction process. This additional mass loss via HOSIM testing is shown for CAPs and not for IOP samples.
Conclusions
This study provides data which shows that the reduction characteristics of iron ore BSL CAPs, in conditions that simulate the upper to mid zone of a blast furnace. Both hematite and magnetite sourced ores were used to prepare CAPs with the respective reduction behaviour studied. The effects of different binder formulations on reduction of CAPs with the same type of ore was also carried out in the case of the magnetite samples.
Simulation of the mid zone conditions of a blast furnace was initially done through using ISO tests 4696-2 and 7215. In these tests, the CAPs were exposed to the reduction gas compositions and temperatures present in a blast furnace for the initial reduction (ISO 4696-2) and final reduction (ISO 7215).
For the ISO 4696-2 the CAPs were shown to vary in performance to the IOPs. CAP H1 which is a hematite-based like IOP H1 demonstrates a decrease in RDI to 1.3%, compared to 6.0% with the IOP. CAPs M1 and M2 do show an increase of 2.9 and 15.4%, respectively. However, this will be based more on the difference in minerology with magnetite swelling and disintegrating more than hematite. In the case of ISO 7215 the IOP H1 did have a higher RI compared to CAPs H1 and M1, with M2 being higher at 87.5%. The different values for CAP M1 and M2 show that CAPs with the same mineralogy but different binder formulations can result in different levels of disintegration and reduction rate. These results also show the inverse relationship between reduction degradation and reducibility, which may be balanced by using different formulations and processing to meet a particular furnace requirement.
HOSIM testing studies the performance of burden components in simulated BF conditions. Where the gas composition alters based on the mass change, whilst test temperature increases over time. Results from the HOSIM tests showed that all BSL CAP samples reached the atomic O/Fe ratio of 0.52 more quickly than that of the IOP H1 samples, by up to 20–30 min. This demonstrates that CAPs have a potential advantage of faster reduction, which could equate to a productivity increase over indurated pellets. There are many other variables in the blast furnace, so a substantial trial campaign would be necessary to quantify this.
In terms of BSL CAP M1 and M2, the initial rate of reduction was slow in comparison to the IOP H1 and CAP H1 hematite samples though the reaction became faster as the test progressed, such that overall reduction time was of the same magnitude. This shows that if magnetite iron ore is introduced as BSL CAPs, the reduction will be satisfactory.
Comparison of post-HOSIM disintegration showed CAP H1 had lower levels at 0.27% than that of IOP H1 at 1.77%. A lower level of fines generation in a blast furnace is favourable in terms of operational stability.
Of further note was the observed loss in mass under inert nitrogen at 400 °C. For IOP H1 not significant mass loss was observed whereas all BSL CAPs show a mass loss regardless of minerology or binder formulation. A study in mass losses of BSL CAPs under nitrogen compared to indurated pellets, showed CAPs as losing between 3 and 8% more mass than theoretically calculated. This indicates that for BSL CAPs, there are chemical changes resulting from the pyrolysis of binders used for agglomeration that introduce a reductive atmosphere.
With the introduction of technologically advanced cold-bonding systems, the existing test methodologies and test results should be subject to scrutiny and interpretation, with a view to introducing alternative test methods and parameters for assessment.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
