Abstract
Rapid palatal expansion has been associated with significant short and long term unwanted effects such as dentoalveolar tipping and detrimental periodontal effects when carried out in skeletally mature individuals. Mini implant assisted rapid palatal expansion (MARPE) has proven to reduce the undesirable effects seen in tooth supported expanders and allows expansion even in adults. A variety of designs have been suggested with their pros and cons. Biomechanically, it is important that the TADs attachment of the expansion appliance should be as close to the palatal vault as possible. This prevents bending of TADs and allows for a better split by keeping forces closer to the bone. According to the T zone concept given by Benedict Wilmes, the bone in the canine-premolar region is much superior for TAD insertion and stability as compared to the bone in the molar region. Considering all the above features a new MARPE appliance was designed which is versatile, anatomically compatible and easy to use. FAVEX is a treatment-centric appliance so that the treatment need not be appliance centric.
Introduction
Maxillary transverse deficiency is one of the most commonly undiagnosed or ignored problem. 1 It is a finding that is reported in 21% of children and 10% of adult population. 2 Proffit and White estimated that 30% of adult patients have transverse discrepancy. 3
Mini implant assisted rapid palatal expansion (MARPE) has proven to reduce the undesirable effects seen in tooth supported expanders and allows expansion even in adults, unlike conventional RPE designs.4, 5 A variety of designs have been suggested in scholarly literature. The initial designs were modified hyrax screws with attachments soldered to receive the temporary anchorage devices (TADs), with or without connection with molar bands.6, 7 These designs work well but laboratory fabrication stage of the appliance is cumbersome. The procedure of MARPE took a new turn and rapidly grew in popularity with Won Moon introducing the MSE expander. 8 A prefabricated device with inbuilt TAD attachment sites and phenomenal amount of documented results have made it a very popular appliance.
There are two schools of thought as far as appliance design is concerned. Ludwig and others state that a completely bone supported design, without any attachments to teeth, allows skeletal expansion without dental side effects. 7 In contrast certain researchers opine that hybrid appliances produce greater skeletal expansion, ensuring minimal alveolar/dental tipping as compared to only a bone supported design.9-12
In clinical practice, both the bone-bone and bone-tooth anchored designs have their applications. In adult cases where diagnostic findings indicate a difficult palatal split, more TADs can be added, taking the number of TADs to 3 on either side, ie, 6 in total with an aim of increasing the anchorage. 7
The T zone concept introduced by Benedict Wilmes, states that the bone in the canine- premolar region is much superior for TAD insertion and stability as compared to the bone in the molar region. 13 Studies have also emphasized that the palatal bone thins out rapidly antero-posteriorly. 14 Thus for better stability, resistance against tipping and dragging the TADs in the palatal bone under expansion loads, it is preferable to keep TADs in the anterior palate. 9 The anterior palate also allows the clinician better access during TAD insertion.
When a bone-bone type of MARPE design is used, the support wires connecting the appliance to the first molars enhance the stability of the appliance during insertion of the mini screws and subsequent use. It is preferable to keep the molar band connected wires, at least in the initial stage of expansion till the suture splits. This would prevent excessive loading of the TADs when the resistance to expansion is high. Post the appearance of midline diastema, if desired so, the molar support arms can be cut off. This would prevent any further tipping of the molars and allow some relapse if needed, whereas the expansion continues with much less resistance and loading of the TADs. This also allows the appliance to be used as a viable adjunct with aligners.
Biomechanically, it is important that the TADs attachment of the expansion appliance should be as close to the palatal vault as possible. This prevents bending of TADs and allows for a better split by keeping forces closer to the bone. In many designs of pre-fabricated appliances, with rigid holes to insert TADs, it is not always possible as the expander screw body has to be seated either too posteriorly for this or there has to be a compromise on the expander size. Analysis of the literature suggests there is no perfect expander design fit for all cases.
An introduction to a new design of the MARPE appliance is made here. With FAVEX, it is possible to combine desirable design features which can be modified as per the case. The advantage of mass production of this appliance is to reduce the laboratory steps and cost.
The most desirable features of MARPE can be summarized as:
Design which can be easily customized as per the requirements of a given case with minimal efforts. Sturdy design to avoid bending of the expander screw while activation. Minimal interference with the palatal soft tissues in expander placement. Minimal pressure points on the palatal mucosa and avoidance of food traps. Straight line easy access to the expansion screw for self-activation. A ratchet type opening key for easy engagement while activation. Option of using as a bone-bone supported or bone-tooth supported expander. Option of using with bands, acrylic splint or 3D printed metal splint. TADs placement in T zone. Adaptable extensions for TADs to get a close fit with palatal anatomy. Option of 2, 4, or 6 TADs as per need. Use of TADs anterior or posterior to the expander as per bone availability. A mass produced appliance to minimize the cost.
Design of FAVEX
Considering all the above features a new MARPE appliance was designed which is versatile, anatomically compatible, and easy to use. FAVEX is a treatment centric appliance so that the treatment need not be appliance centric.
FAVEX has two extension arms to attach the screw to the molar bands or the acrylic splint as per need. In case a bone-bone expander has been planned, the arms can be adapted on the occlusal surface of the molars and temporarily bonded with composite resin to stabilize the appliance during TAD insertion and cut off afterwards.
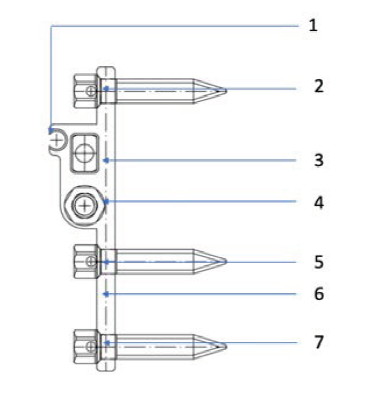
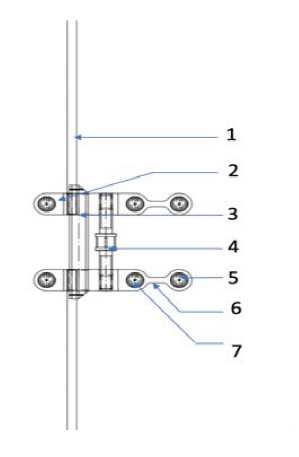
The TAD attachments (Figures 1 and 2 nos 2, 5, 7) are designed to receive 2 mm diameter TADs. (2) is posteriorly placed and (5) and (7) are anteriorly placed and connected by a bendable connector (6). With this design, TAD locations (5) and (7) are always in the T zone, even with necessary posterior positioning of the expander screw. The TAD arms can be bent at the connector (6) to conform to the anterior palatal curvature.
FAVEX design has a distinct feature of a rectangular beam shaped support bar (3). As the principles of engineering show, a rectangular cross sectional beam is more resistant to bending stresses when compared to a round one. In adults, the sutural resistance can be much higher. Some of the existing designs with round support bars have been reported to bend under pressure. The rectangular bar of FAVEX can resist deformation under much higher forces which might be generated during expansion in adult patients. The single rectangular support bar positioned distal to the threaded activation screw has few more important advantages. Absence of interference from the anterior support bar, allows anterior palatal placement. The tapered ends of the bar further aid in this.
The activation hex (Figure 2, no 4) has a clear straight access for visualization and self-activation by the patient.
Schematic Drawing of Favex: Side View.
Schematic Drawing of Favex: Top View.
FAVEX allows the orthodontist to choose the appliance design and modify the appliance with less laboratory steps. The following designs of FAVEX can be used:
Bone-bone anchored device (Figure 3). In adolescent patients, side arms soldered onto the molar bands and only 2 anterior bicortical TADs, just next to the expander body may be sufficient (Figure 4). 2 TADs placed anteriorly, just abutting the expander, and 2 posterior TADs (Figure 5). 4 TADs placed anterior to the expander screw (Figure 6). In adults, a design of the appliance utilizing all 6 TADs, bands on the molars, and corticotomy to assist the suture opening may allow expansion in most difficult cases (Figure 7).
Bone-bone anchored design: The support arms can be temporarily bonded with composite to stabilize the expander for TAD insertion and cut off afterwards.
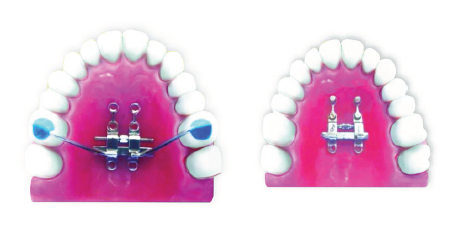
Bone-Tooth Anchored Design: Only 2 Anterior TADs and Support of Molars.
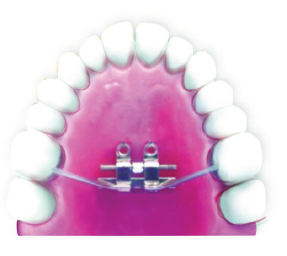
Bone-Tooth Anchored Design: 2 Anterior TADs, 2 Posterior TADs, and Support of Molars.
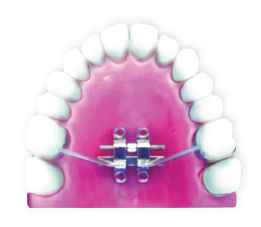
Bone-Tooth Anchored Design: 4 Anterior TADs and Support of Molars.
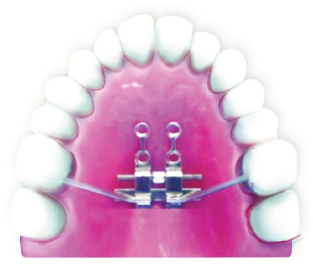
Bone-Tooth Anchored Design: 4 Anterior TADs, 2 Posterior TADs, and Support of Molars.
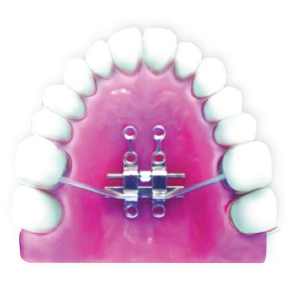
Figures 8-18 show the use of FAVEX in different cases where mid palatal suture split has been achieved in adult patients. Bone-bone or tooth-bone anchored design for MARPE has been used for skeletal expansion.
Pretreatment Intraoral View Showing Severe Transverse and Anteroposterior Jaw Discrepancy.
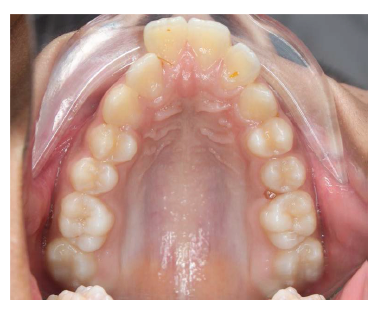
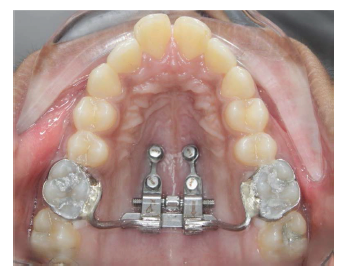
MARPE Inserted.
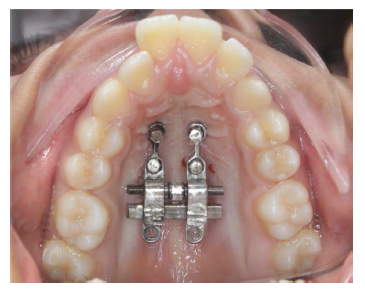
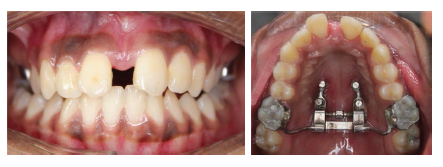
Post Expansion.
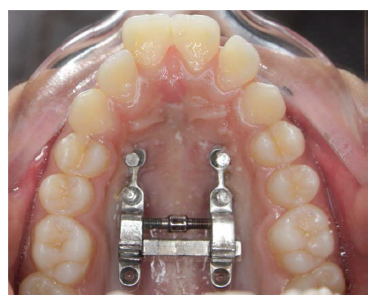
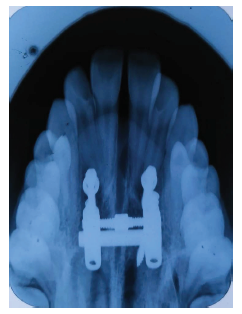
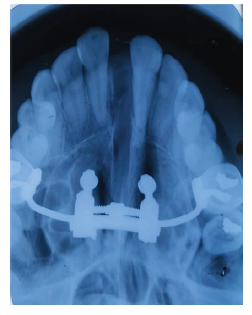
Post Expansion Occlusal Radiograph Showing Palatal Split.
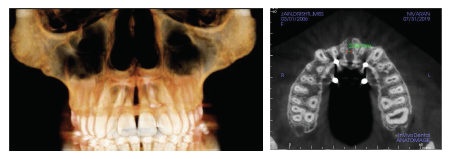
Post Expansion CBCT.
Pretreatment Intraoral View Showing Severe Transverse and Anteroposterior Jaw Discrepancy.
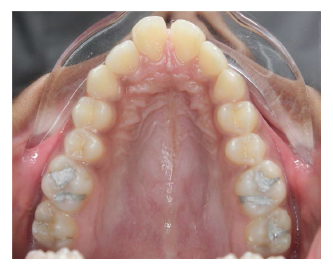
MARPE Inserted.
Post Expansion Intraoral Photographs.
Post Expansion Occlusal Radiograph Showing Palatal Split.
Post Expansion Orthopantomograph.
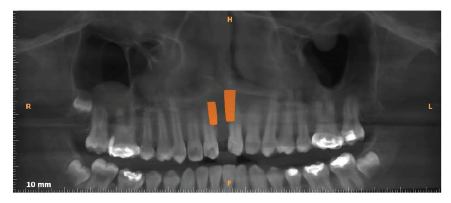
Post Expansion CBCT.
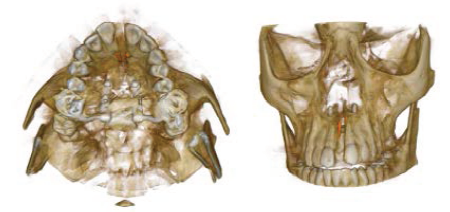
Conclusion
There are two main advantages of the FAVEX expander. It can effectively be used as a bone-bone anchored or tooth-bone anchored expander due to versatility of its design. The activation hex has a ratchet type opening key which makes the FAVEX expander very easy for self-activation by the patient thus increasing the compliance.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
