Abstract
Objective:
Comparative evaluation of antibacterial and anti-adherent properties of surface-modified stainless steel (SS) orthodontic brackets against Streptococcus mutans (S. mutans).
Materials and Methods:
The study was conducted on 120 SS orthodontic McLaughlin, Bennett, Trevisi (MBT) 0.022″ slot by Leone, Italy. Orthodontic brackets that constituted the sample size were divided into 6 groups, consisting of 20 samples each in two control (non-surface coated) and four experimental groups. The experimental group’s surface coatings were photocatalytic zinc oxide (ZnO) and photocatalytic titanium oxide (TiO2), which were carried out by radiofrequency (RF) magnetron sputtering method for surface modification. Brackets were subjected to microbiological tests against S. mutans. For anti-adherence, weight change, pre- and postexposure, was evaluated to gauge the adherence of bacteria and colony-forming units/milliliter (CFU/mL) count measuring the survival rate of bacterial cells for antibacterial activity.
Results:
The TiO2-coated group showed statistically significant anti-adherence (P-value < .05) against S. mutans than control and ZnO groups. The CFU count of TiO2 group was lower than control as well as ZnO group.
Conclusion:
TiO2 is superior to ZnO and should be continued to be considered for surface modification of orthodontic brackets against White Spot Lesions (WSLs) and gingivitis.
Introduction
Orthodontics has its share of iatrogenic damage potential. 1 Such challenges could be either static or dynamic; latter being the ones that follow delivery of orthodontic forces, 2 while former ones, like white spot lesions 3 (WSL) and gingivitis, 4 relate to integrity or health of the crown having orthodontic attachments and its surrounding gingival tissue. 5 These static issues result from long-term effects of dental plaque that thrives around various components of orthodontic appliances. 6
Routine fixed appliance treatment extends over months involving stainless steel (SS) attachments like brackets and tubes. Archwires and auxiliaries in and around such attachments create a serious challenge to plaque elimination. Material property of SS attachments, through higher critical surface tension, 7 leads to increased plaque accumulation, in turn, increasing Streptococcus mutans (S. mutans) colonies 8 responsible for dental caries as well as increased gingival inflammation.4, 9 Although plaque accumulation and increased bacterial colonies reverse, following appliance removal, the aforementioned static damage caused during treatment may be irreversible. 10
Methods to control increased plaque accumulation vary from plaque elimination through either chemical or mechanical means and incorporation of fluorides in adhesives. 11 Chemical methods utilizing mouthwashes have a corrosive effect on SS by altering its mechanical properties and increasing its surface roughness. This increases friction at the bracket–archwire interface compromising appliance efficacy. 12 Mechanical methods like brushing depend upon patient compliance for their ability of plaque elimination.
The author opines that the overall outcome of orthodontic treatment could be augmented by reducing the onus on patient compliance by modifying certain appliance components like brackets.
Photocatalytic action has shown an enhancement of antimicrobial activity in previous studies with oxides of titanium.13–15 Though titanium dioxide (TiO2) is efficacious, its cytotoxic effect, especially in rutile phase, has triggered concerns. 15 The passivating film of TiO2 is beneficial in countering allergenic properties of nickel retaining its relevance, despite its higher cost. 12
Zinc, an essential mineral for human physiologic functioning, in its oxide form (ZnO), has exhibited antibacterial activity against selective microorganisms.16, 17 ZnO finds wide acceptance for intraoral use in several branches of dentistry. 18 Surface modification of SS brackets with ZnO could be a viable option to test its photocatalytic activity against S. mutans, an aspect yet to be assessed. Moreover, a comparative evaluation of photocatalytic properties of ZnO and TiO2 is conspicuous by its absence.
The present, prospective, in-vitro study was conducted to evaluate and compare antibacterial and anti- adherent effects of photocatalytic ZnO and TiO2 when used as surface modificants for orthodontic SS brackets in reducing proliferation of S. mutans.
Null Hypothesis
There is no change in the anti-adherent and antibacterial effect of the bracket’s surface modified with TiO2, ZnO, and their photocatalytic effect against S. mutans.
Alternative hypothesis for acceptance is as follows:
There is a change in the anti-adherent and antibacterial effect of the bracket’s surface modified with TiO2, ZnO and their photocatalytic effect against S. mutans.
Materials and Methods
After a pilot study on 6 brackets, ensuring propriety and accuracy for all methodological steps of the proposed study; subsequent to obtaining prior approval from the Institutional Ethics Committee, the present study was conducted on 120 samples of SS mandibular incisor orthodontic brackets of preadjusted edgewise appliance (PEA), 0.022″ × 0.028″ slot size; McLaughlin, Bennett, Trevisi (MBT) prescription, Leone, Italy. As advised by a qualified biostatistician, these 120 brackets, while configuring a 10% possibility of bracket sample rejection resulting from imperfect coating, constituted the sample size for the present study.
Segregation of Brackets
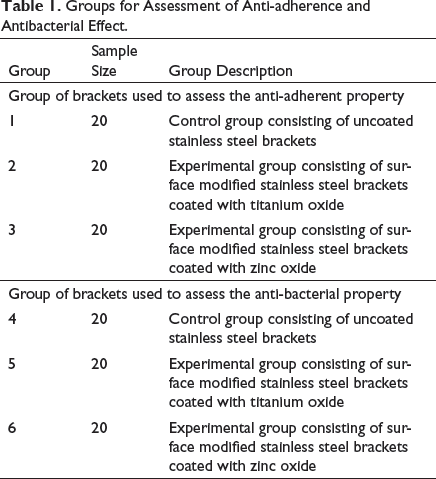
Groups for Assessment of Anti-adherence and Antibacterial Effect.
Surface Modification of Brackets
Brackets from experimental groups were surface modified at Tata Institute of Fundamental Research (TIFR), Colaba, Mumbai, by radiofrequency (RF) magnetron sputtering method. The sputtering process removed surface atoms from a solid cathode by bombarding it with positive ion discharge from an inert gas—Argon. Surface atoms removed from the cathode were deposited on the substrate to form a thin film coated on its entire surface. 19 In the present study, orthodontic brackets from each of the four experimental groups were the “substrate,” while the “target” was either TiO2 or ZnO. Thinness and uniformity of the sputtering was carefully evaluated under scanning electron microscope (SEM) during the pilot study. Subsequently, all parameters that ensured the desired quality of sputtering were carefully replicated during the entire study.
The substrate was placed in a vacuum chamber and pumped down to a prescribed process pressure. The aforementioned sputtering (Figure 7), lasting 20 min, was carried out in batches of 20 brackets, while maintaining a constant distance of 7 cm between substrate and target. Brackets were then oxidized in an ambient environment of an open-air furnace at 500°C for 5 h, rendering them ready for further testing.
Prepping for Microbiological Testing
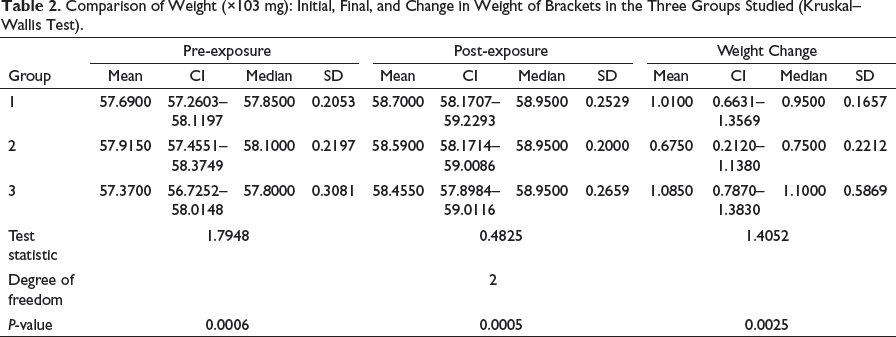
Comparison of Weight (×103 mg): Initial, Final, and Change in Weight of Brackets in the Three Groups Studied (Kruskal–Wallis Test).
Microbial Type Cell Culture and Gene Bank (MTCC), Chandigarh, was the source for strain of S. mutans, MTCC 497, used in the present study and was inoculated into 5 mL of Brain Heart Infusion (BHI) Broth. This BHI Broth was incubated for 24 h at 37°C for it to be used as a “reservoir” for all further testing detailed as follows:
Bacterial Adhesion Testing of Brackets
From the “reservoir,” 10% BHI Broth was micropipetted into a sterile beaker containing 100 mL of BHI broth. It was then incubated for 24 h to create a suspension of S. mutans. Based on periodic visual check of the turbidity level of this suspension, a “final concentration level” of 10% was determined for further testing. A sample of 1 mL of this suspension was pipetted into each Eppendorf tube belonging to groups 1, 2, and 3 to immerse the brackets. These immersed brackets were incubated at 37°C for 24 h under illumination of ultraviolet-A (UV-A) black light in the incubator (Figure 8).
Brackets were then carefully removed with a fine tweezer and immersed into a solution of 10% formaldehyde, contained in a petri dish, for 30 min to immobilize the cells, while ensuring that adjacent brackets did not touch. Using a dropper, a gentle wash with distilled water for every bracket was followed. Brackets were then dried in a desiccator for 24 h. Weight of every bracket was obtained with an analytical balance and recorded as postexposure weight. When compared with its pre-exposure weight, the weight change of brackets was an index of quantifying bacterial adhesion.
Antibacterial Activity Assay of Brackets
For the strain of MTCC 497 of S. mutans, culture broth from the aforementioned “reservoir” was diluted with BHI broth to achieve an optical density of 4.0, using a standard microbiological instrument, DensiCHEK. Then, 1 mL of this stock culture was diluted in 100 mL of sterile saline. Of this diluted bacterial suspension, 10 mL was transferred into petri dishes containing uncoated SS brackets, TiO2-coated brackets and ZnO-coated brackets, which are depicted as groups 4, 5, and 6, respectively (Table 1).
These petri dishes were illuminated with UV-A black light inside the Biosafety Cabinet Class II for 60 min. After illumination, 100 µL of the bacterial suspension was serially diluted from 10−1 to 10−4 and plated onto BHI agar plates for 48 h at 37°C. For the purpose of determining bacterial colony count of S. mutans for individual petri dishes, a standard microbiological instrument, Digital Colony Counter, was used. Antibacterial activity was described as the survival rate of bacteria based on the colony-forming units (CFU) observed (Figure 9). The same is depicted in Table 4A.
Statistical Methods
All data were entered into a Microsoft Office Excel (version 2013) in a spreadsheet, which was prepared and validated for the data form. Data were entered and checked for errors and discrepancies. Data analysis was carried out using Windows-based “MedCalc Statistical Software” Version 17.8.2 (MedCalc Software bvba, Ostend, Belgium;
All testing was carried out using one-sided tests at alpha = 0.05.
Thus, criteria for rejecting the null hypothesis were “P”-value = <.05.
Results
For ease of description, the results are segregated under the following heads.
Bacterial Adhesion Testing of Brackets
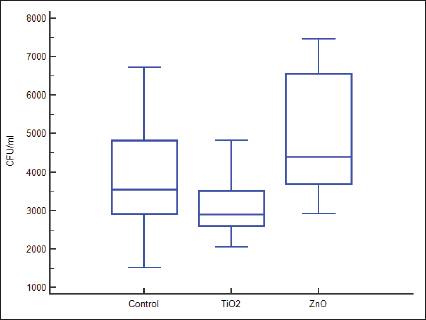
For assessment of adhesion of S. mutans to the surface of orthodontic brackets, the “net” gain in weight resulting from adherence had to be determined. As the actual weight change was miniscule, for data depiction and drawing meaningful inference, this was subjected to a standard statistical conversion. The post-conversion values in milligrams are presented in Table 2. Distribution of scores of weights is similar in shape (Figure 2).
As per data exhibited in Table 2, bracket samples from group 1 had an initial median weight of 57.8150 mg and final median weight of 58.9500 mg, the increase in weight of 0.9500 mg being statistically insignificant. Likewise, bracket samples in group 3 had an initial median weight of 57.8000 mg and final median weight of 58.9500 mg, the increase in weight of 1.1000 mg being statistically insignificant.
However, bracket samples in group 2 exhibited an initial median weight of 58.1000 mg and final median weight of 58.9500 mg. This increase in weight of 0.7500 mg was statistically significant at P = .0005 (Table 2).
Thus, brackets from group 2, which were surface modified with TiO2, showed relatively reduced “increase in weight,” which is a function of lesser adhesion of bacteria (Table 3 and Figures 1 and 2).
Multiple Comparison Graph.
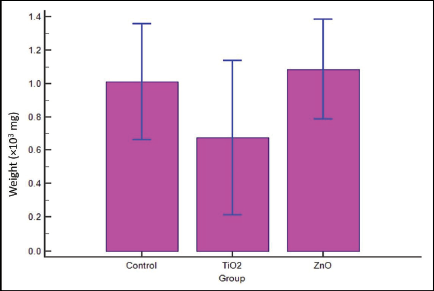
Multiple Variables Box Plot for Anti-adhesion (Kruskal–Wallis Test).
Antibacterial Activity Assay of Brackets
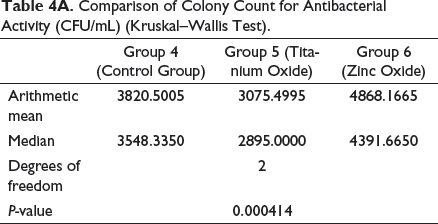
Table 1 details the groups that were used for the present assessment. Table 4A presents the survival rate of S. mutans for bracket samples in groups 4, 5, and 6, which represent control group, group with TiO2-coated brackets, and Group with ZnO-coated brackets, respectively. Distribution of scores is similar in shape (Figure 4).
Survival rate of S. mutans was 3548.3350 CFU/mL in group 4, 2895.0000 CFU/mL in Group 5, and 4391.6650 CFU/mL in Group 6. Thus, as compared to groups 4 and 6, antibacterial activity in group 5 is higher and statistically significant at 95% confidence limit (Figures 3A and 4).
Multiple Comparison Graph.
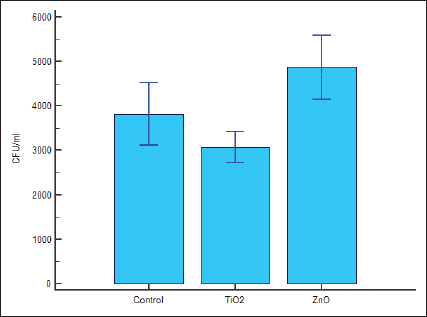
Multiple Comparison Graph Log Transformed.
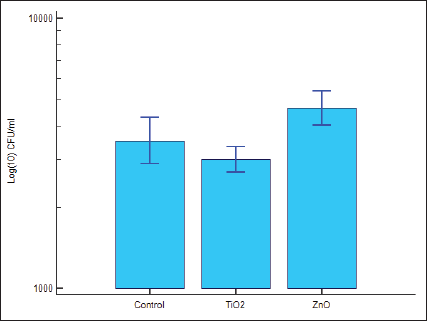
Multiple Variables Box Plot for Antibacterial Test (Kruskal–Wallis Test).
Logarithm of colony count (log CFU) detailed in Table 4B, showed the relevant values for Groups 4, 5, and 6 to be 3541.6114, 2894.8614, and 4381.9691, respectively. Thus, antibacterial activity is higher as well as statistically significant for group 5 as compared to groups 4 and 6, with P = .000414 (Figure 3B).
Group 5, containing TiO2-coated brackets, showed statistically significant decrease in the survival rate of S. mutans as compared to brackets from control group and the group containing brackets surface modified with ZnO.
On the basis of the aforementioned results, the null hypothesis was rejected, and the alternate hypothesis was accepted.
Titanium Oxide-Coated Stainless Steel Orthodontic Brackets.

Zinc Oxide-Coated Stainless Steel Orthodontic Brackets.

Substrate Inside Sputtering Unit During Sputtering Procedure.

Orthodontic Brackets Under Exposure to UV-A Back Light.

Colonies of S. Mutans Observed under Digital Colony Counter.

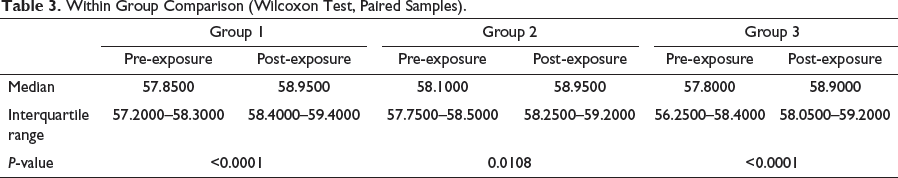
Within Group Comparison (Wilcoxon Test, Paired Samples).
Comparison of Colony Count for Antibacterial Activity (CFU/mL) (Kruskal–Wallis Test).
Back-transformed After Log Transformation (Kruskal–Wallis Test Log Transformed).

Discussion
WSLs present themselves as a twin problem, an aesthetic problem, and another in breaching integrity of the tooth structure through demineralization of enamel surface. The latter leads to carious lesions around orthodontic attachments. The acidogenic property reflects an obvious relation between S. mutans, formation of carious lesions, and enamel dissolution. Ensuring that the environment around fixed orthodontic appliances is devoid of S. mutans and suchlike microbes, additional help from antibacterial property exhibited by the appliance components may ward off long-term ill effects.
Photocatalytic property of TiO2 is responsible for its anti-adherent and antibacterial effect. Photocatalytic activity 20 is a chemical reaction in which a catalyst hastens the reaction in combination with light, be it sunlight or UV light. This property has far-reaching applications in industrial water purification and as semiconductors to purify air.
For a material to exhibit antibacterial property, the antibacterial substance must be incorporated in components that make up the material, or it can be used in the form of a coating of nanoparticles over the existing surface. TiO2 and ZnO have been used as a coating of nanoparticles on orthodontic brackets, and their mode of action has also been studied. Photocatalysis is due to its oxidizing capacity and expresses itself when illuminated with UV light of a wavelength less than 385 nm.
Photocatalytic property is retained even if TiO2 nanoparticles are used as surface coating. TiO2 decomposes the organic compounds in a series of oxidation reactions, leading to production of carbon dioxide. Reactive oxygen species (ROS) like hydroxyl radicals are formed during the oxidation reactions and are responsible for bacterial inhibition. Photocatalytic activity of TiO2 is attributed to direct photochemical oxidation of intracellular coenzyme A to its dimeric form. This oxidation, through involvement of cell wall and cell membrane, was the root cause of respiratory activity. Oxidation increases cell permeability and allows free efflux of intracellular contents. A direct attack on the cellular components, made possible by “free” TiO2 gaining access into the membrane-damaged cells, results in alteration of protein structure, leading to acceleration of cellular death. TiO2-treated cells continue to lose their viability when illumination to UV light stops. Once the lethal oxidation reaction is initiated by the photocatalytic reaction of surface-modified TiO2, damage induced by oxidation continues in the dark through the Fenton reaction, a free radical chain reaction of lipid peroxidation. 19
ZnO has demonstrated antimicrobial activity when incorporated into dental cements, mouth rinses, and periodontal dressings. 21 Effectiveness of antimicrobial activity of ZnO nanoparticles against Escherichia coli (E. coli), is better researched17, 22, 23 than against S. mutans. 22 Exact mechanism of antibacterial activity of ZnO is yet to be ascertained. It may be attributed to being a sequel to contact between ZnO nanoparticles and bacteria 22 that creates a disturbance in bacterial growth or nanoparticles gaining entry into bacterial cells. Bactericidal effect observed is due to disruption of bacterial enzymatic system through displacement of magnesium ions. The bactericidal and bacteriostatic actions are due to generation of ROS, like hydrogen peroxide, whose effect may be potentiated by hydroxyl radicals. ROS is responsible for cell wall damage, subsequent to localized ZnO contact, leading to increased membrane permeability and uptake of toxic, dissolved Zinc ions. While this is similar to the mechanism of action described for TiO2, cell growth inhibition and eventual cell death are due to weaker mitochondria, intracellular outflow, and release of gene expression in oxidative stress. 17
While TiO2 and ZnO have been individually researched for the property of anti-adherence and antibacterial activity against bacterial species, a comparative assessment being conspicuous by its absence was the motivation for the present study performed on surface-modified orthodontic brackets against S. mutans. The results of the present study, depicted by tables and graphs, clearly indicate that though both TiO2 and ZnO have displayed the property of anti-adherence and antibacterial activity, TiO2 appears to be superior to ZnO. Results of the present study are in concurrence with the studies by Ramazanzadeh et al, 16 Salehi et al, 24 and Magnusson et al. 25
The author opines that the reason for reduced efficacy of ZnO in the present study may be attributed to the strain of S. mutans used. Further studies with different strain(s) of S. mutans may yield a favorable antimicrobial effect. Though there is formation of ROS, they do not inhibit the growth of S. mutans or having a bactericidal effect on the cells. A probable hypothesis for this could be the proteins, which are involved in the process of biofilm formation and adhesion that may be damaged during the process of oxidative stress. The expression of various gene factors, which are also responsible for surface attachment and biofilm formation are inhibited. Oxidative stress enzymes, superoxide dismutase, and pseudo-catalysis showed slightly increased activity in S. mutans on exposure to nanoparticles of Zinc, which may lead the oxidative stress to minimize formation of streptococcal biofilm. The ROS formed by TiO2 may have a greater ability to damage the cell membrane in contrast to ROS formed by ZnO.
Similar to the results of the present study, findings by Eshed M., Lellouche J., Matalon S., Gedanken A., and Banin E. were that ZnO film did have an inhibitory effect on biofilm formation, though not statistically significant; this observed difference in the activity of TiO2 and ZnO was arguably attributed to the ability of the S. mutans strain to induce antioxidant activity to protect itself. SS brackets and surface modified with TiO2, can be used for significant reduction of adherence of dental plaque. Undesirable sequelae of WSL and dental caries as well as chronic inflammation of gingiva can be reduced in a statistically significant manner due to enhanced anti-adherent and antibacterial effect. Findings of this study may offer a clinician an option of using surface-modified attachments, which create a better oral hygiene status and be a game changer in treatment of cases with periodontally compromised patients.
For permitting clinical utilization of the photocatalytic effect of surface-coated brackets, the criticality of duration of exposure to UV light that the surface-coated brackets need to be subjected to is an area that bears clinical consideration. Further research will be required to assess the duration of efficacy of photocatalytic activity.
Subjecting other strains of S. mutans to parameters of the present study may permit further learning of the photocatalytic effect, especially ZnO-coated brackets, for obvious cost benefit.
Conclusion
TiO2 is superior to ZnO and should be continued to be considered for surface modification of orthodontic brackets against white spot lesions (WSLs) and gingivitis.
Footnotes
Acknowledgments
Prof. Pushan Ayyub, Department Chair, TIFR and Mr. Rudheer Bapat, Scientific Observer, TIFR. Department of Condensed Matter Physics and Materials Science.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
