Abstract
Objective:
To evaluate the biofilm adhesion to 4 types of ‘as retrieved’ archwires during orthodontic treatment. To compare biofilm formation by viable cell counting (colony-forming units [CFU]) of microbes (Staphylococcus aureus, Streptococcus mutans and Candida albicans).
Methods:
The study was conducted on 40 patients who were divided into 4 equal groups based on the type of archwire of same cross-section (0.019" × 0.025"): group 1—copper-nickel titanium (Cu-NiTi); group 2—titanium molybdenum alloy (TMA); group 3—stainless steel (SS); and group 4—aesthetic coated SS. After 1 month of fixed orthodontic therapy, the archwires were removed carefully from the oral cavity.
Results:
Biofilm absorbance and biofilm concentration were significant (P ≤ .05) in all the 4 groups.
Conclusion:
The group 2 wire showed the maximum affinity for biofilm absorbance and biofilm concentration, whereas the group 4 wire showed the least affinity. The group 4 wire promoted less retention of oral bacteria compared with the wires of the other groups, whereas the group 1 wire showed maximum retention. In the present study, all types of wire showed maximum affinity for S mutans and the least affinity for C albicans, except for the group 1 wire, which showed maximum affinity for S aureus. Biofilm adhesion could be a significant determining factor in the selection of orthodontic archwire for patients primarily at risk of dental caries or periodontal diseases, or both.
Introduction
Orthodontic materials like archwires, brackets, bands, and various fixed components impede oral hygiene, leading to plaque accumulation, and are therefore highly responsible for microbial adhesion, which in turn disposes the wearer to an increased burden and possibility of subsequent infection. 1 The formation of dental microbial biofilm is facilitated by areas where the initially loosely adhering bacteria are protected from removal forces. 2 The archwires used in orthodontic practice provide a conducive environment for oral microorganisms to grow, enough to cause dental diseases such as caries and periodontal diseases.3, 4
An acquired pellicle is formed from selective adsorption of the specific salivary biopolymers on the surface of orthodontic materials. 5 This initial coating forms a connection or interface between the surface (orthodontic archwires) and the colonizing oral microbes. 6 Initial bacterial colonization on the acquired salivary pellicle is a precondition for the development of mature biofilm.
The orthodontic archwire is a mainstay of orthodontic appliances. The orthodontic archwire material has progressed from stainless steel (SS) and the cobalt-chromium-nickel (Co-Cr-Ni) alloy to the nickel-titanium (NiTi) alloy.7, 8 These material changes have led to increased super elasticity, thermal shape memory, corrosion resistance, and biocompatibility.
9
The growing number of adult orthodontic patients has given rise to a demand for more aesthetic orthodontic appliances,
10
as seen in the development of polymer-based aesthetic wires, metallic archwires coated with polymer materials,11, 12 and rhodium-ion-implanted and gold-plated wires. However, coatings on orthodontic metal wires may influence surface characteristics, such as roughness and hardness, as well as mechanical properties, such as bending behavior. Orthodontic archwires serve as a medium for plaque accumulation, thereby increasing the level of microorganisms in the oral cavity.
13
Various surface characteristics of orthodontic archwires, such as surface roughness, surface free energy, corrosion susceptibility, etc, have been found to affect the adhesion of biofilm and microbial colonization, which has been documented. Although the mechanical and physical properties of orthodontic archwires have been extensively studied, very little in vivo research has been done to evaluate and compare the biofilm adhesion of different archwire materials. Hence, the formation of biofilm on orthodontic archwires of various types, such as SS, Cu-NiTi, titanium molybdenum alloy (TMA) and aesthetic coated SS, was analyzed in this study. Thus, this clinical study was designed:
To evaluate the biofilm adhesion to 4 types of ‘as retrieved’ archwires during orthodontic treatment. To compare biofilm formation by viable cell counting of microbes (Streptococcus mutans, Staphylococcus aureus, and Candida albicans) on different archwire surfaces.
Methods
Sample Design
The present study was planned to consist of 40 randomly selected patients undergoing orthodontic treatment. It was done to assess biofilm formation on 4 types of archwire material and microbial colonization on their surface. Four different archwire materials were used, with 10 wires in each group (Table 1).
Experimental Population
Only those individuals satisfying the inclusion and exclusion criteria were selected for the study. Inclusion criteria included patients with good oral hygiene and with no signs of redness, edema or bleeding during brushing. No professional teeth-cleaning measures except plain-water rinses were used during the period of the study.
Exclusion criteria included patients having any systemic diseases or undergoing antibiotic therapy and those with any preexisting periodontal conditions.
Infection control included carrying a clinical examination using aseptic precautions. Other armamentaria included a distal-end cutter, spectrophotometer, and digital counting unit.
After 1 month of fixed orthodontic therapy, the archwires were removed carefully to avoid iatrogenic biofilm dislodgement, rinsed with an air/water spray to loosen the debris, and then air-dried. For samples of all types, a 20 mm piece was cut with a distal-end cutter from the end of ‘as retrieved’ archwires. Each piece of archwire was dipped in 2 mL of BHI (brain heart infusion) broth meant to be used as a microbial environment and transportation medium. It was placed in a self-closing sterilized plastic bag. On the outer surface of each bag, the name of the patient, date of insertion, date of removal, and type of archwire were recorded. The bags were stored until the time of investigation.
Three different agar plates were prepared in the laboratory:
Mitis salivarius (MS) agar media for S mutans; Mannitol salt agar (MSA) media for S aureus; and Potato dextrose agar (PDA) media for C albicans. The positive observations for the suspected microbial species were inferred as follows: On MS Agar media: Pinpoint dark blue/black convex colonies represented S mutans. On MSA media: Formation of a yellow zone around the convex colonies on the pink background of media described the presence of S aureus. On PDA media: Large, convex, creamy bacteria-like colonies with a peculiar smell on the fungal culture medium described the presence of C albicans.
Scan Spectrum Curve
Preparation of the Calibration Curve of the Dye
3D Spectra of Preparation of the Calibration Curve of the Dye
Quantitative Biofilm Measurement
The wires were washed carefully with phosphate-buffered saline (PBS), after which they were stained with 0.5% crystal violet solution for 30 min, washed 5 times with distilled water, and left to dry at room temperature for 30 min. The crystal violet was then solubilized by the addition of 95% ethanol. The absorbance and concentration of the biofilm were determined by spectrophotometry using a spectrophotometer. The absorbance was determined at 568 nm (Figures 1–3). The optical density (OD) 568 threshold value over which strains were considered to be significant biofilm formers was 0.51.
Statistical Analysis of the Result
All the data were recorded on an Excel sheet and subjected to statistical analysis using the SPSS software (Statistical Package for the Social Sciences, version 22.0, Chicago, IL).
Results
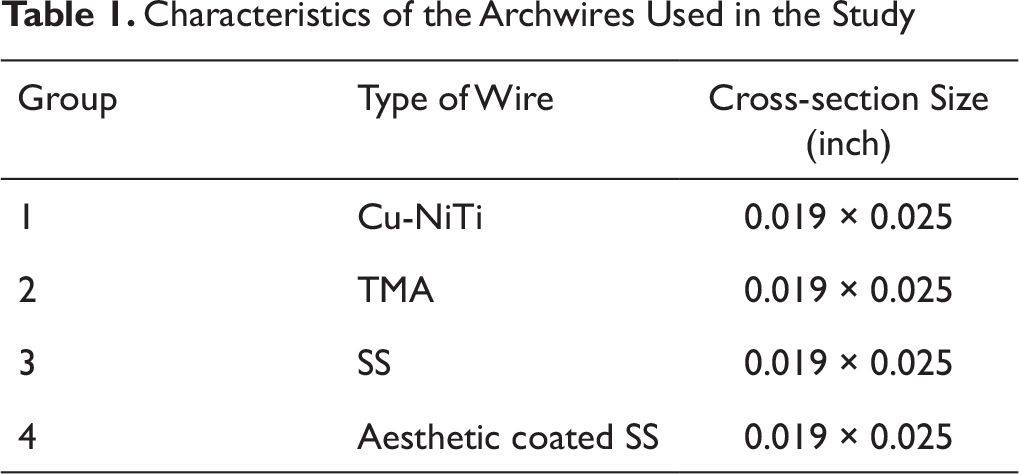
Table 1 shows the characteristics of the 4 types of orthodontic archwires investigated in our study. Group 1 comprised Cu-NiTi archwires, group 2 TMA archwires, group 3 SS archwires, and group 4 aesthetic coated SS archwires. All 4 types of archwires were of the same cross-sectional dimensions (0.019" × 0.025").
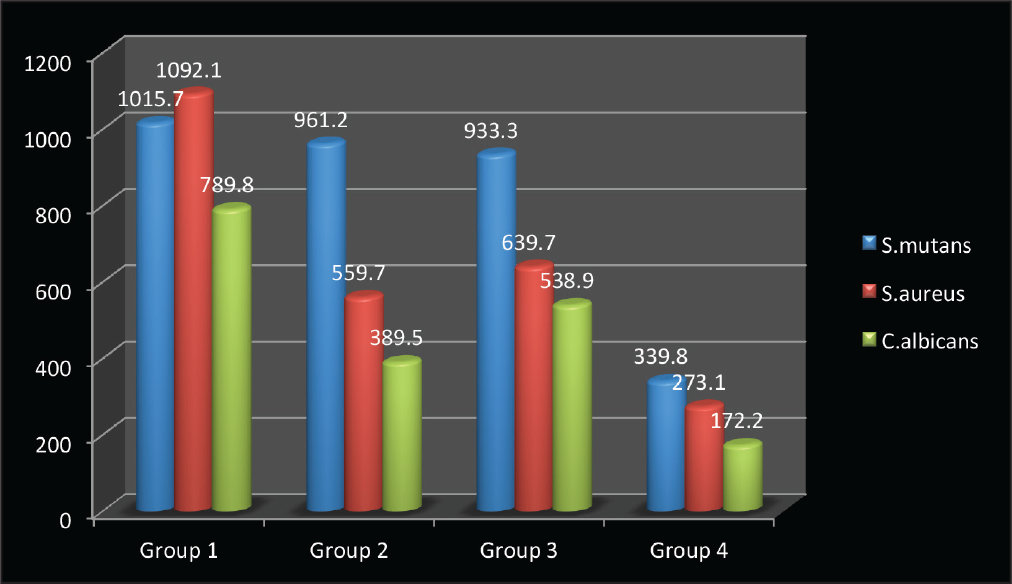
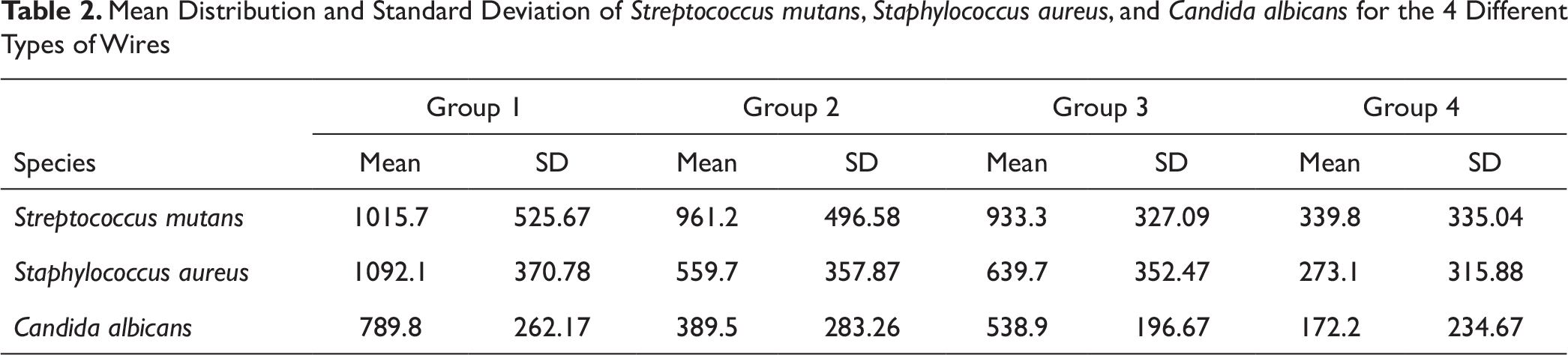
Table 2 and Figure 4 show the mean distribution and standard deviation of S mutans, S aureus, and C albicans on the 4 different types of wires.
Table 3 shows the results of a 1-way ANOVA, performed to compare the mean values of the colony-forming unit (CFU) counts of the 3 microbes—S mutans, S aureus, and C albicans—for all the 4 groups of wires. All the parameters showed a statistically significant difference between the Cu-NiTi, TMA, SS, and aesthetic coated SS wires. The mean standard deviations of the adhesion of S mutans to the 4 types of archwires—group 1, group 2, group 3, and group 4—were 1015.7 (±525.67), 961.2 (±496.58), 933.3 (±327.09), and 339.8 (±335.04), respectively (P ≤ .05; F = 5.41).
Mean Distribution and Standard Deviation of Streptococcus mutans, Staphylococcus aureus, and Candida albicans to the Different Types of Wires
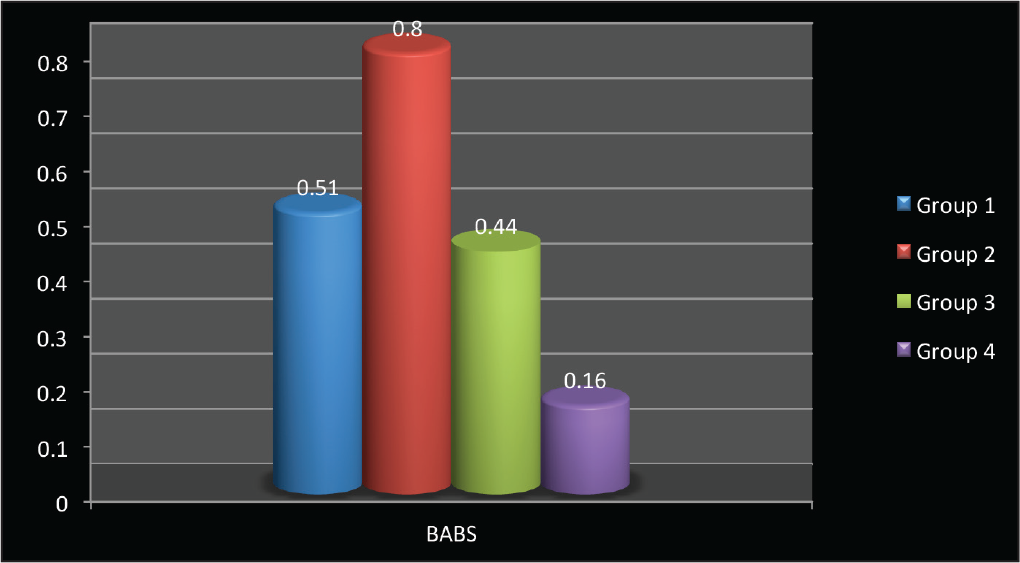
Mean Distribution and Standard Deviation of Biofilm Absorbance on the Different Types of Wires
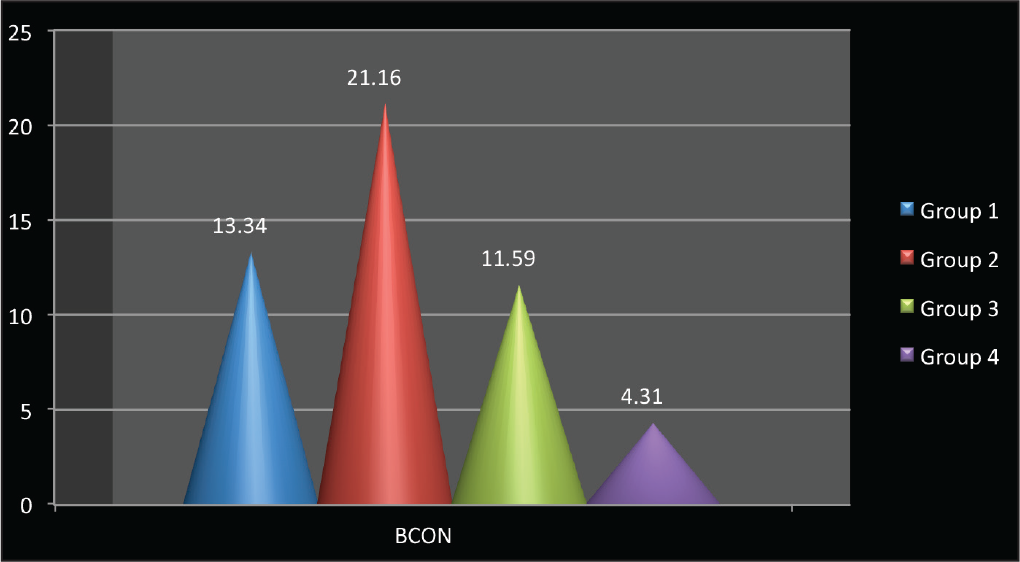
Mean Distribution and Standard Deviation of Biofilm Concentration on the Different Types of Wires
Characteristics of the Archwires Used in the Study
The mean standard deviations of the adhesion of S aureus to the 4 types of archwires—group 1, group 2, group 3, and group 4—were 1092.1 (±370.78), 559.7 (±357.87), 639.7 (±352.47), and 273.1 (±315.88), respectively (P ≤ .05; F = 9.40).
The mean standard deviations of the adhesion of C albicans to the 4 types of archwires—group 1, group 2, group 3, and Group 4—were 789.8 (±262.17), 389.5 (±283.26), 538.9 (±196.67), and 172.2 (±234.67), respectively (P ≤ .05; F = 11.10).
Tukey’s honestly significant difference (HSD) tests were conducted on all possible pairwise contrasts. Groups 1 to 4 were compared on the basis of adhesion of S mutans, S aureus, and C albicans to all 4 types of wires, which showed significant differences (P ≤ .05) as shown in Table 4.
For S mutans, group 4 showed a significant difference with group 1 (P = .006), group 2 (P = .014), and group 3 (P = .020).
For S aureus, group 1 showed a significant difference with group 2 (P = .009), a significant difference with group 3 (P = .031), and a highly significant difference with group 4 (P = .000).
For C albicans, group 1 showed a significant difference with group 2 (P = .005) and a highly significant difference with group 4 (P = .000), and group 3 showed a significant difference with group 4 (P = .010).
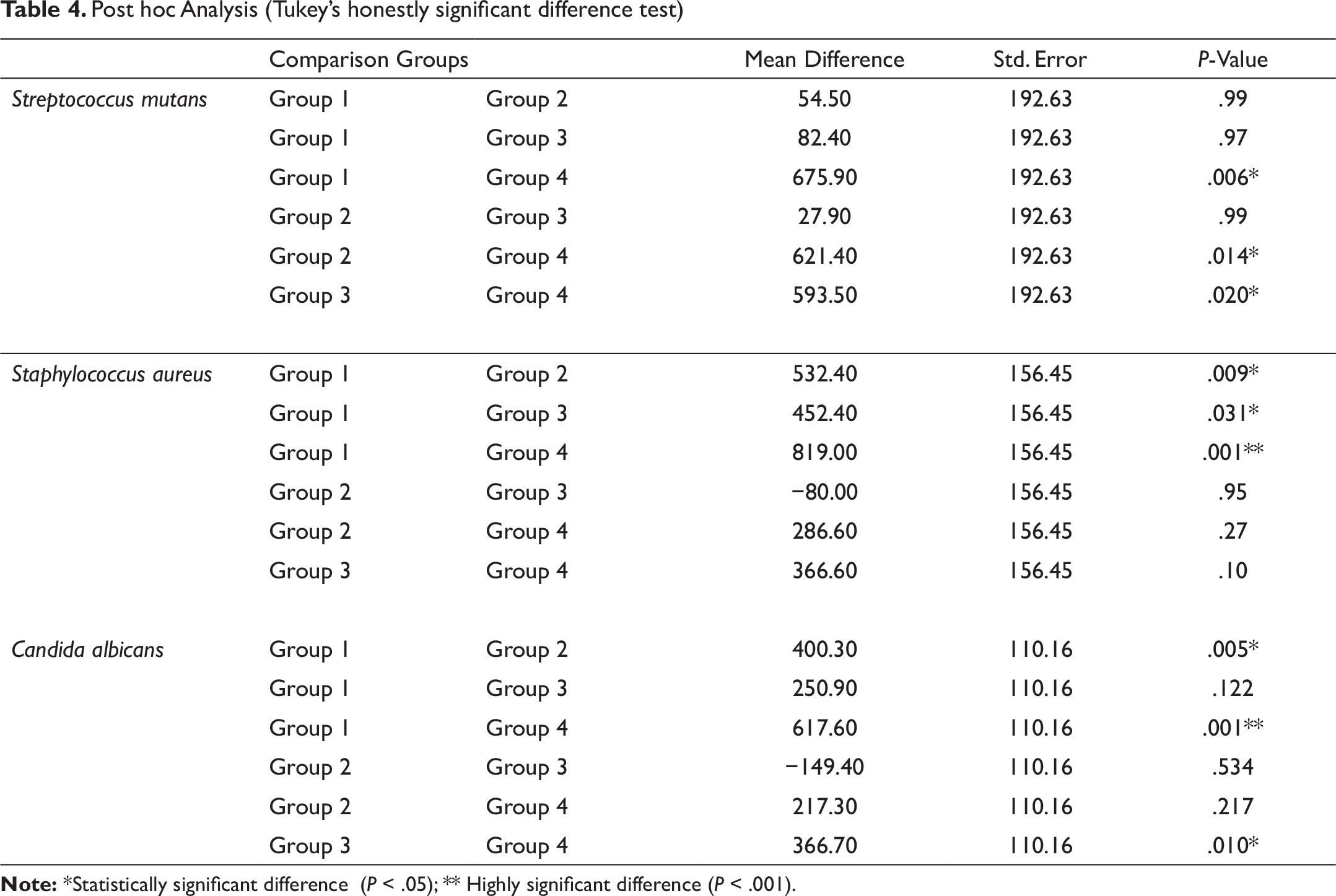
Table 5 and Figures 5 and 6 show the mean distribution of biofilm absorbance and concentration on all the 4 groups of wires.
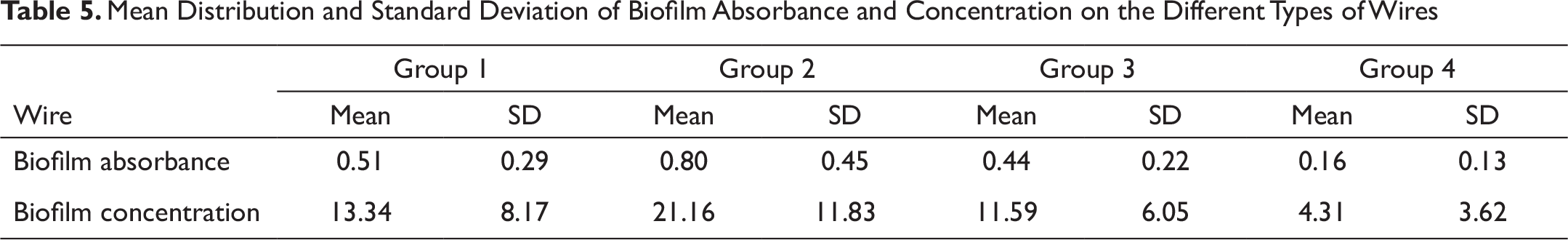
Table 6 shows the results of the ANOVA that was performed to compare the mean values of biofilm absorbance and concentration for all the 4 groups of wires. All the parameters showed a statistically significant difference between the Cu-NiTi, TMA, SS, and aesthetic coated SS wires (P ≤ .05).
The mean standard deviations of biofilm absorbance on the 4 types of archwires—group 1, group 2, group 3, and group 4—were 0.51 (±0.29), 0.80 (±0.45), 0.44 (±0.22), and 0.16 (±0.13), respectively (P ≤ .05; F = 3.836).
The mean standard deviations of biofilm concentration on the 4 types of archwires—group 1, group 2, group 3, and group 4—were 13.34 (±8.17), 21.16 (±11.83), 11.59 (±6.05), and 4.31 (±3.62), respectively (P ≤ .05; F = 3.613).
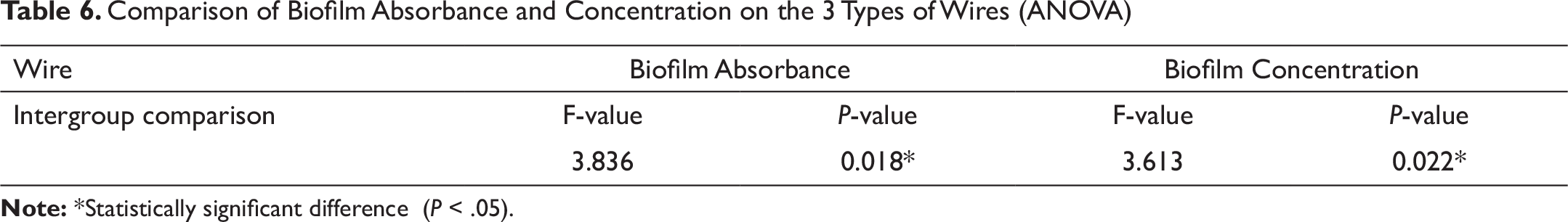
Specifically, Tukey’s honestly significant difference tests were conducted on all possible pairwise contrasts. Groups 1 to 4 were compared on the basis of absorbance and concentration of biofilm on them, which showed significant differences (P ≤ .05 ) as shown in Table 7.
For biofilm absorbance, group 1 showed a significant difference with group 4 (P = .018).
Mean Distribution and Standard Deviation of Streptococcus mutans, Staphylococcus aureus, and Candida albicans for the 4 Different Types of Wires
Comparison of Adhesion of Streptococcus mutans, Staphylococcus aureus, and Candida albicans to the 3 Types of Wires (ANOVA)

Post hoc Analysis (Tukey’s honestly significant difference test)
Mean Distribution and Standard Deviation of Biofilm Absorbance and Concentration on the Different Types of Wires
Comparison of Biofilm Absorbance and Concentration on the 3 Types of Wires (ANOVA)
Post hoc Analysis (Tukey’s honestly significant difference test)
Discussion
Orthodontic archwires play a significant role in plaque accumulation. As archwires can be removed and studied during a regular patient treatment visit, they represent an ideal model for the in vivo study of material alterations. Insertion of orthodontic wires creates new surfaces available for biofilm formation; therefore, awareness of the bacterial adhesion tendency of new orthodontic archwire materials should be developed in order to select an archwire type that attracts less biofilm. Also, knowledge regarding growth and adhesions of cariogenic bacteria on orthodontic material will help better prevent white spot lesions. 14
Traditional orthodontics may be described as variable-cross-section orthodontics, where small wires are used for light forces and large wires for heavier ones. Variable-modulus orthodontics (Burstone), 15 on the other hand, takes advantage of different materials while maintaining the same or similar cross-sections. As is seen, there are definite advantages in using wires of varying materials in optimizing control of tooth movement. Orthodontic arch wires differ in their surface characteristics, such as surface roughness, surface free energy, corrosion susceptibility, etc, which are found to affect biofilm adhesion and colonization over them. Thus, it is necessary to examine the biofilm adhesion to and colonization over all 4 types of orthodontic archwires discussed in this article. 16
The scope of the present study was to quantitatively evaluate the adhesion of biofilm to 4 types of orthodontic archwires of the same cross-sectional dimensions (0.019" × 0.025"), and also to measure microbial colonization over them. Along with biofilm quantification, comparison of biofilm formation on all 4 types of wires was done by measuring the CFU count of S mutans, S aureus, and C albicans after 1 month of archwire placement.
Three organisms were investigated through their CFUs. The bacterial culturing method 17 was used in the present study to assess the composition of biofilm due to its capacity to detect multiple bacterial species simultaneously and the possibility to obtain absolute and relative counts of the bacterial species. S mutans was studied because of its well-documented role in the pathogenesis of caries. High levels of acidogenic bacteria are present in plaque, notably S mutans. Lundstrom and Krasse 18 reported an increase in the level of S mutans after the insertion of an orthodontic appliance. Orthodontic archwires provide a good habitat for oral microorganisms, sufficient to cause periodontal diseases and dental caries. S aureus is the major causative agent of the major human disease. In the oral tract, S aureus has been associated with dentoalveolar infections and oral mucosal lesions, and its colonization on mucosal surfaces, the tongue, saliva, the periodontal pocket, and supragingival tooth surfaces has been demonstrated. 19 The most common Candida species isolated in orthodontic patients is C albicans. 20 It has 2 roles in the oral environment: producing biofilms on dental surfaces and co-aggregating with oral bacteria. 21 Although no healthy individual develops a Candida infection from orthodontic appliance use, non-Candida carriers can become Candida carriers via orthodontic therapy. 22 The results of our study indicated that in vivo biofilm detection was positive on all types of wires and that the affinity for the adhesion of biofilm differed among the different types of wires. When we compared different types of wires for biofilm absorbance and concentration, the results showed significant differences among the different types of wires (P ≤ .05). The group 2 (TMA) wires showed the maximum affinity for biofilm absorbance and concentration, followed by the group 1 (Cu-NiTi) wires, whereas the group 4 (aesthetic coated SS) wires showed the least affinity. Among groups 2, 3, and 4, the most abundantly found organism in the biofilm was S mutans, while the least was C albicans. On the other hand, in group 1 (Cu-NiTi), the most abundantly found organism in the biofilm was S aureus. The results of our study (Table 4) indicated that for S mutans, the group 1 (Cu-NiTi), group 2 (TMA), and group 3 (SS) wires showed a significant difference with the group 4 (aesthetic coated SS) wires (P ≤ .05). These were in accordance with the results of a study done by Kim et al, 23 who investigated 6 archwires, 4 coated and 2 uncoated (NiTi and SS), and measured the surface free energy and surface roughness using dynamic contact angle analysis and profilometry, respectively.
The amount of MS (S mutans and Streptococcus sobrinus) adhering to the wires was quantified using the CFU-counting method. The S mutans adhesion results showed a significantly higher adhesion in NiTi alloy wires than in SS wires. Thus, Kim et al concluded that the SS orthodontic wires may be preferable to NiTi wires in terms of reducing S mutans adhesion. Also, the coating of metallic wires was found favorable for decreasing MS adhesion. Similarly, in a study by Raji et al, 24 it was concluded that bacterial plaque accumulation on epoxy- and resin-coated NiTi orthodontic archwires was significantly lower than that in uncoated NiTi wires. This was attributed to the difference between their surface characteristics, such as surface energy and surface roughness, which were more in the uncoated NiTi archwires. The ANOVA test (Table 6) was used to compare the mean values of biofilm absorbance and concentration for all the 4 groups of wires. All the parameters showed a statistically significant difference between the Cu-NiTi, TMA, SS, and aesthetic coated SS archwires (P ≤ .05). These findings were in support of those of Taha et al, 25 who in their study on 3 brands of aesthetic coated NiTi archwires concluded that surface roughness and biofilm adhesion to aesthetic coated wires increased after intraoral use at all-time intervals, with a positive correlation between the adhesion and surface roughness of S mutans, S aureus, and C albicans. The results of our study (Table 4) indicated that for S mutans, group 1 showed no statistically significant difference with group 3 (P = .09) but showed a statistically significant difference for S aureus and C albicans (P ≤ .05). These were similar to the results of a study by Eliades et al, 26 who concluded that the alteration in the composition of surface NiTi archwires after intraoral exposure for 1 to 6 months, due to the occurrence of amorphous precipitates and microcrystalline particles in proteinaceous biofilm, led to the high affinity of S aureus for titanium surfaces. These results were in support of the results of a study by Al-Lami et al, 27 who revealed that in multiple comparisons, the highest adhesion of cariogenic Streptococci was to NiTi and epoxy-coated archwires, while the least adhesion was to SS and Teflon-coated wires. This contradicted the results of a study by Kumar and Konda, 28 who stated that no significant difference in the degree of surface deposits on the 3 test archwires (TMA, NiTi, and SS) was observed. However, the surface deposits on TMA archwires were found to be more compared to those on NiTi archwires, which in turn were more than those on SS archwires. Studies on SS wires have showed that the smoothness of their surface is responsible for the decrease in the count of Streptococcus colonies on them. 29 This contradicted the results of a study conducted by Shin et al 30 using 2 types of SS archwires and 2 types of NiTi archwires. The surface corrosion of the archwires was determined macroscopically, through scanning electron microscopy and spectrophotometry. The deposits on the wires were identified using X-ray diffraction to evaluate and identify the surface deposits on the SS and NiTi archwires. After 12 weeks of immersion, more surface deposits were found on the SS wires than on the NiTi wires. Shin et al 30 concluded that NiTi wires did not present surface morphology alteration with increasing saliva exposure, indicating a higher corrosion resistance than SS wires. Our analysis provides critical information concerning the performance of the materials in the environment in which they are intended to function, and further research is warranted to analyze the roles of various predisposing factors on the formation of surface biofilm. The limitation of this study is the impossibility of standardizing eating habits.
Conclusion
The present study indicated that the affinity for biofilm adhesion differs with different types of wires.
Biofilm absorbance and concentration were significant in all the 4 groups.
The group 2 (TMA) wires showed the maximum affinity for biofilm absorbance and concentration, due to the increased surface roughness of TMA archwires, whereas the group 4 (aesthetic coated SS) wires showed the least affinity for biofilm absorbance and concentration.
The present study indicates that significant differences were found between group 1 (Cu-NiTi) and group 4 (aesthetic coated SS) for all 3 organisms—S mutans, S aureus, and C albicans.
The group 4 (Esthetic coated Stainless Steel) wires promoted less retention of oral bacteria compared with those of groups 1, 2, and 3, and the group 1 (Cu-NiTi) wires showed the maximum retention.
In the present study, all types of wires showed maximum affinity for S mutans and the least affinity for C albicans, except for the group 1 (Cu-NiTi) wires, which showed maximum affinity for S aureus.
Biofilm adhesion could be the significant determining factor in the selection of an orthodontic archwire for patients primarily at risk of dental caries or periodontal diseases, or both.
