Abstract
Lesions associated with perianesthetic death (PAD) postmortem submissions are infrequently reported in the literature, with no studies comparing findings between general and referral practices (RPs). This study compared PAD postmortem submissions in cats from a referral teaching hospital (referral practice, RP) and general practices (GP) in Saskatchewan. In the RP, death was most commonly due to euthanasia (15/23, 65%), with most cases having severe underlying disease. In GP, most deaths were unassisted (37/45, 82%), and most animals (33/37, 89%) had an undiagnosed condition or an unknown cause of death. The American Society of Anesthesiologists (ASA) physical status classification was high (ASA III-V) in 16/23 (70%) of RP cases and low (ASA I-II) in 38/45 (85%) of GP cases. Cats with limited medical history accounted for 5/23 (22%) of the RP submissions and 17/45 (38%) of the GP submissions. Reporting of gross examination findings and tissues collected for histologic examination were inconsistent. For example, although the presence of negative pressure within the thoracic cavity is evaluated routinely during a complete postmortem examination, its presence (or absence) was only reported in 4/45 (9%) of cases where the animal died unassisted. No significant difference was found in determining the cause of death between RP and GP when euthanized cases were excluded (P = .445). A standardized perianesthetic postmortem checklist is proposed to enhance reporting and improve diagnostic consistency.
Keywords
The death of a pet around the time of an anesthetic procedure, though infrequent, can warrant submission to a veterinary pathologist for postmortem examination. Over the past 30+ years, numerous studies have investigated the incidence and risk factors of perianesthetic deaths (PADs) in various species.3,7,19,23,36 Across studies, cats consistently have a higher reported incidence of PAD (0.06%–5.1%) when compared to dogs (0.05%–1.49%).3,13,21,22,26,33,38 The variation in reported PAD incidence is influenced by several factors including differences in study definitions and the population studied. Some research includes all deaths that occurred during or around the time of an anesthetic event while others only include deaths solely attributed to anesthesia.3,22 In addition, the follow-up period varies across studies, ranging from immediately after anesthesia to up to 2 weeks later.2,3,36 The population studied also affects the reported incidence of PAD. As an example, referral practices (RPs), including private clinics and academic teaching hospitals, typically report higher PAD rates than general practice (GP) clinics. For cats, the incidence of PAD at RPs ranges from 0.43% to 5.1% while the incidence in GPs is 0.10% to 0.29%.8,16,17,21,26
Despite numerous clinical studies investigating PAD, few have used postmortem examination to characterize the pathologic lesions.12,18,35 A notable finding in pathology studies of PAD in cats is that 34% to 63% lack significant lesions to explain the cause of death.12,18 None of these studies have specifically compared the postmortem findings between GP and RP submissions. Previous studies have provided recommendations on the information required in a PAD submission form and the procedures for conducting a PAD postmortem examination.11,12 However, no standardized checklist for pathologists has been proposed, and recommendations for histologic sample collection remain broad.11,12
The objectives of this study were to (1) compare the postmortem findings of PAD submissions in cats between GPs and a referral teaching hospital (RP) in Saskatchewan (Canada); (2) assess current reporting practices in cases of PAD to identify inconsistencies and areas requiring improvement; and (3) develop a standardized PAD postmortem checklist to guide pathologists during their investigation. We hypothesize that cats dying within 1 week of an anesthetic procedure at an RP and submitted for postmortem examination are more likely to have lesions that confirm or are suspected to be associated with cause of death compared to cats submitted from GPs within the same province.
Materials and Methods
Data were collected retrospectively from searches of two computerized databases, with the search limited to the period between January 2015 and December 2021. Inclusion criteria were cats submitted for gross necropsy to Prairie Diagnostic Services, Inc. that died within 7 days of a general anesthesia. Data were obtained from the Veterinary Medical Center (RP), the teaching hospital associated with the University of Saskatchewan’s Western College of Veterinary Medicine, and from GPs across the province of Saskatchewan. The first database was the Western College of Veterinary Medicine’s VetNet database, which contained all anesthetic procedures performed on cats. The second database, Prairie Diagnostic Services, Inc. Casebook, was searched using the following terms: “anesthes*,” “anaesthes*,” “surgery,” and “dental.” Data from the GPs also contributed to the study published by Rose et al, 35 which used information from pathology reports to categorize the cause of death, evaluate completion of the submission forms, and identify the most significant lesions.
Data collected from the postmortem reports included sex, neuter status, age, facility where the procedure was performed (GP vs. RP), history provided by the submitting practitioner, mechanism of death (died unassisted vs. euthanized), whether CPR was performed and details of CPR, gross and histologic lesions, and the pathologist’s comments. Animals with limited clinical history including rescue cats, barn cats, outdoor cats, or stray cats were designated as “cat with limited history” for the purpose of this study. Each animal was assigned an American Society of Anesthesiologists (ASA) physical status classification either based on the provided history (GPs) or on the anesthetic record (RP). The ASA classification is used in both human and veterinary medicine to assess a patient’s pre-anesthetic physical condition and estimate the perianesthetic risk. Higher ASA classification has been correlated with increased anesthesia-related mortality in small animals. 21 Cats that did not have enough information within the clinical history to assign an ASA classification were classified as not available. The cause of death in cats was classified as either confirmed/uncertain or unknown. This categorization was based on the definitions used in a previous study. 35 A confirmed cause of death was assigned when the pathologist determined that the gross findings, histologic lesions, or clinical history (individually or in combination) explained the animal’s death. In contrast, an uncertain cause of death was assigned when lesions (or pertinent clinical history) were identified but their role in the death of the animal was uncertain. Since both these categories involved identifiable lesions (or important clinical history information), they were grouped into a single category (confirmed/uncertain). Cause of death within the confirmed/uncertain group was then classified into 1 of 4 categories: 24 (1) death due to an anesthetic complication, (2) death due to a surgical complication, (3) death due to a pre-existing disease, or (4) death due to an undiagnosed condition. A fifth category was used for cases where significant lesions (or pertinent clinical history) explaining or contributing to death were not identified, that is, and (5) unknown cause of death.
Most results are presented descriptively, as the data were drawn from two different populations, and the total number of anesthetic procedures (denominator) and anesthetic deaths (numerator) in the GP population were not known. Statistical analyses were performed using commercial software (GraphPad Prism 10.0.0 for Windows, GraphPad Software, Boston, Massachusetts USA). Fisher’s exact test (2-sided) was used to investigate the effect of type of center (GP vs RP) on determining the cause of death. A P < .05 was considered statistically significant. Results were expressed as odds ratio and the 95% confidence intervals (CIs) calculated using the Baptista-Pike method. The data analyzed in this study are available in the Supplemental Materials.
Results
Within the RP VetNet database, a total of 1343 anesthetic procedures were identified (3 animals were excluded for not meeting the inclusion criteria). Of these, 1275/1343 cats (94.9%) were alive within the 7 days post-anesthesia. A total of 68/1343 cats (5.1%) died or were euthanized within 7 days of anesthesia. Of the 68 cats, 24 died unassisted (1.8% of 1343 anesthetized) and 7 of these animals underwent a postmortem examination (0.5% of 1343 anesthetized). The remaining 44 cats were euthanized (3.3% of 1343 cats anesthetized) with 12 undergoing postmortem examination (0.89% of 1343 anesthetized).
From the Prairie Diagnostic Services, Inc. Casebook, 49 cases of death occurring within 7 days of an anesthesia were identified. There were 4 cats that overlapped with the VetNet database. Data from these cases were only included once for analysis. Combining the Prairie Diagnostic Services, Inc. Casebook and VetNet data, a total of 45 cases were submitted from GPs in Saskatchewan, and 23 cases were submitted from the RP. Table 1 summarizes the key findings.
Demographic data for cats with perianesthetic death that were submitted to Prairie Diagnostic Services from general practices (GP) and a referral practice (RP) in Saskatchewan, Canada.
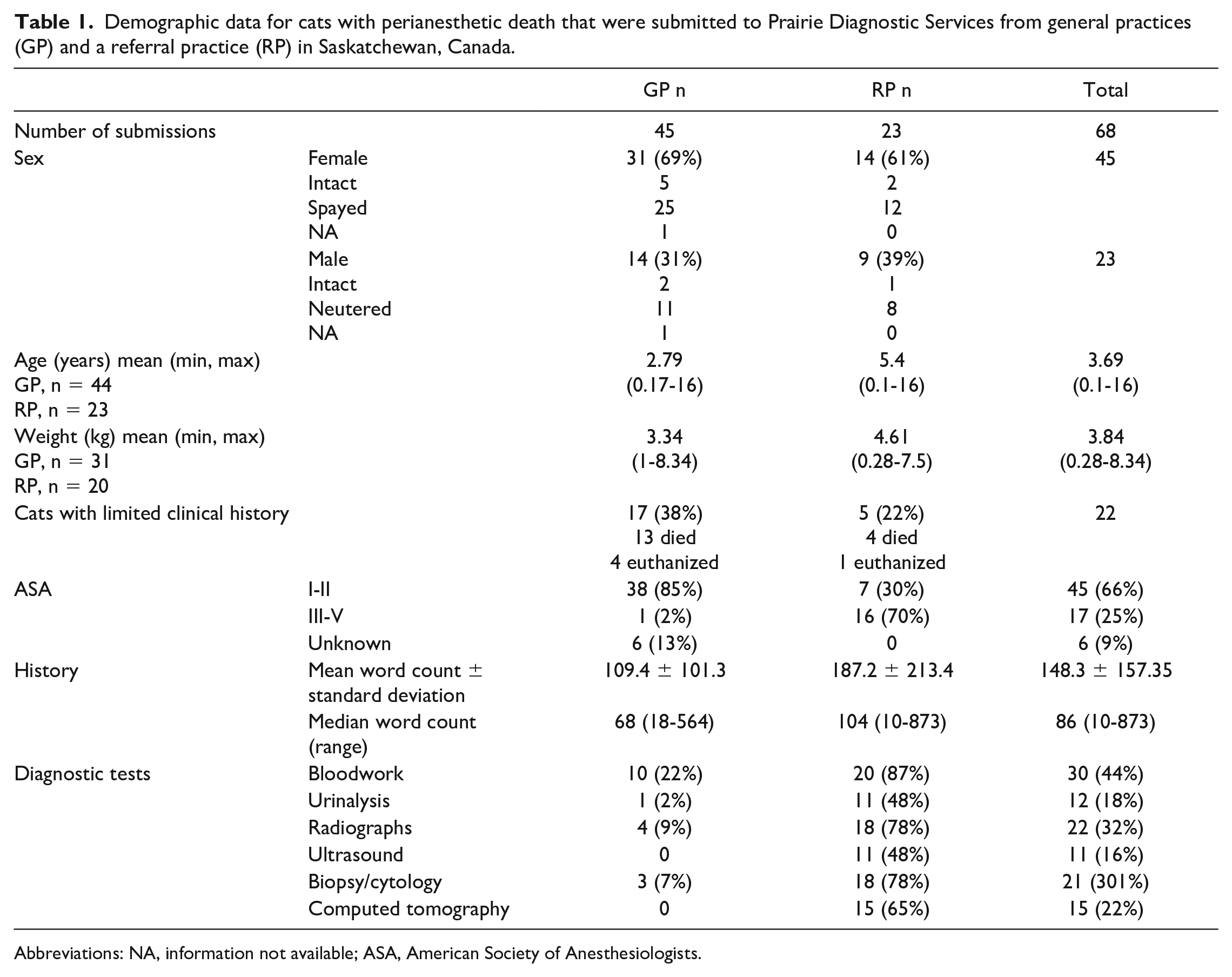
Abbreviations: NA, information not available; ASA, American Society of Anesthesiologists.
Table 2 summarizes causes of death based on whether cats died unassisted or were euthanized. A higher percentage of animals from the RP were reported to have been euthanized (15/23, 65%) compared to those from GPs (8/45, 18%). Among the 15 euthanized animals from the RP, all had a confirmed/suspected cause of euthanasia and 10/15 (67%) were classified as ASA III-IV. Reasons for euthanasia in the RP included 6 cats with unresectable neoplasms diagnosed during the procedure (exploratory surgery or imaging), 2 cats with respiratory tract neoplasms that deteriorated post-anesthesia, 6 animals in poor condition before anesthesia with continued decline in health status postanesthesia leading to euthanasia (e.g., urethral obstruction), and 1 apparently healthy cat with limited history based on clinical examination before anesthesia that declined after the procedure and was diagnosed with feline parvovirus infection (panleukopenia) on postmortem examination. In contrast, 4 cats from GP were euthanized due to secondary complications from anesthesia or surgery, while 4 other cats were euthanized after deteriorating postanesthesia, with no cause of death identified on postmortem. Surgical and anesthetic complications included repeated self-trauma to the surgical site, closed pop-off valve, septic peritonitis following abdominal surgery, and significant postspay hemorrhage. All euthanized GP cats with a known ASA classifications (n = 7) were classified as low risk (ASA I-II).
Cats from general practice (GP) and referral practice (RP) categorized as died or euthanized and placed in 1 of the 5 major categories for cause of death: anesthesia complication, surgery complication, pre-existing disease, undiagnosed condition, and unknown.
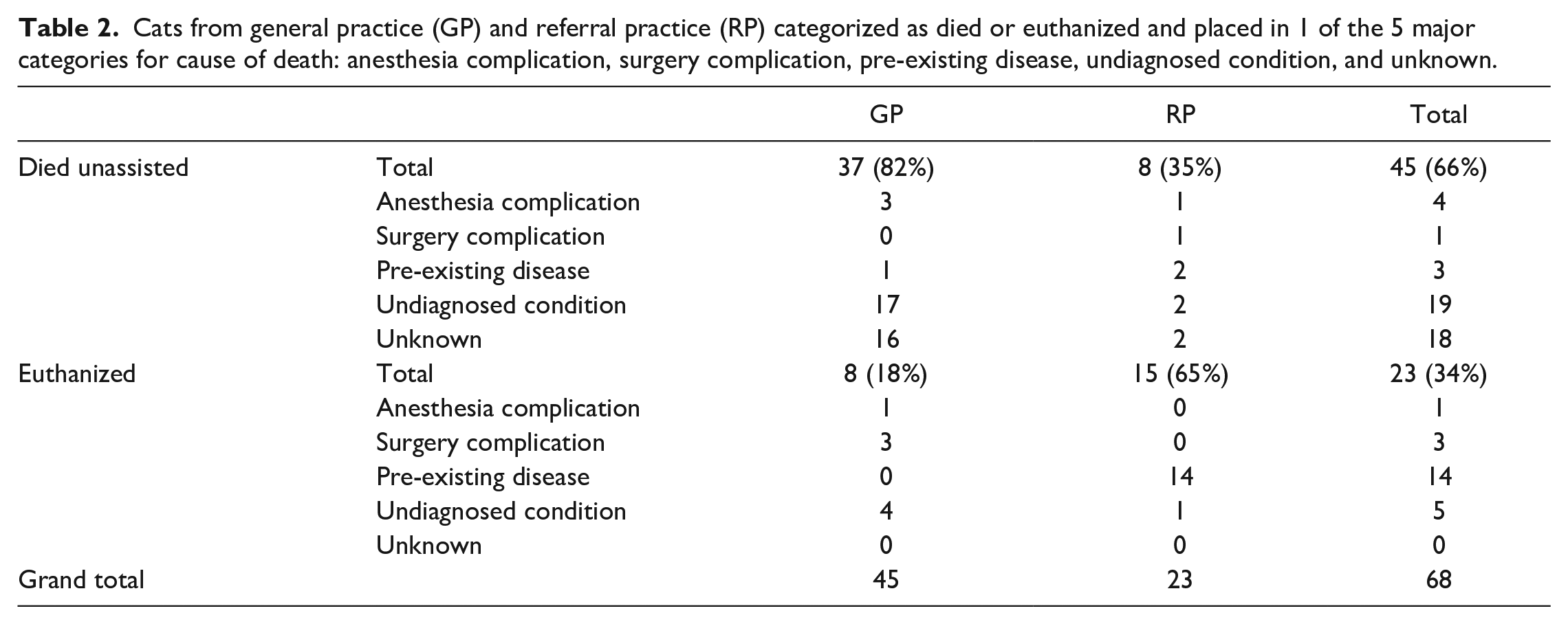
A total of 8/23 (35%) of RP cats died unassisted compared to 37/45 (82%) of GP cats (Table 2). At the RP, in cases with an unknown cause of death, one ASA I-II kitten with a limited history died during fracture repair, and one ASA PS III-V cat, who had an esophageal tube placed, recovered from anesthesia but died later that day. In GPs, where the cause of death was unknown, all cats with an assigned ASA classification (n = 14) were classified as I or II and were undergoing elective procedures.
When comparing cases where a lesion was identified that was confirmed/uncertain to be associated with death versus unknown cause of death, cats within the RP were more likely to have a cause of death confirmed/uncertain than cats in GP (P = .021) with an odds ratio of 5.79 (95% CI: 1.24-27.05). However, when animals that were euthanized were removed from the analysis, there was no difference between the confirm/suspected and unknown cause of death categories in animals that died unassisted (P = .445).
The gross pathology reports from cats that died unassisted in both the GPs and the RP were compared (Fig. 1). Overall, reporting was similar between GPs and the RP with the most and least frequently recorded variables being body condition and negative thoracic pressure, respectively. Histologically, the frequency of tissue sampling in all deaths and lesions identified are summarized in Table 3. The lungs and the heart were the most frequently collected (98% each), while trachea was infrequently examined histologically (4%) (Table 3).
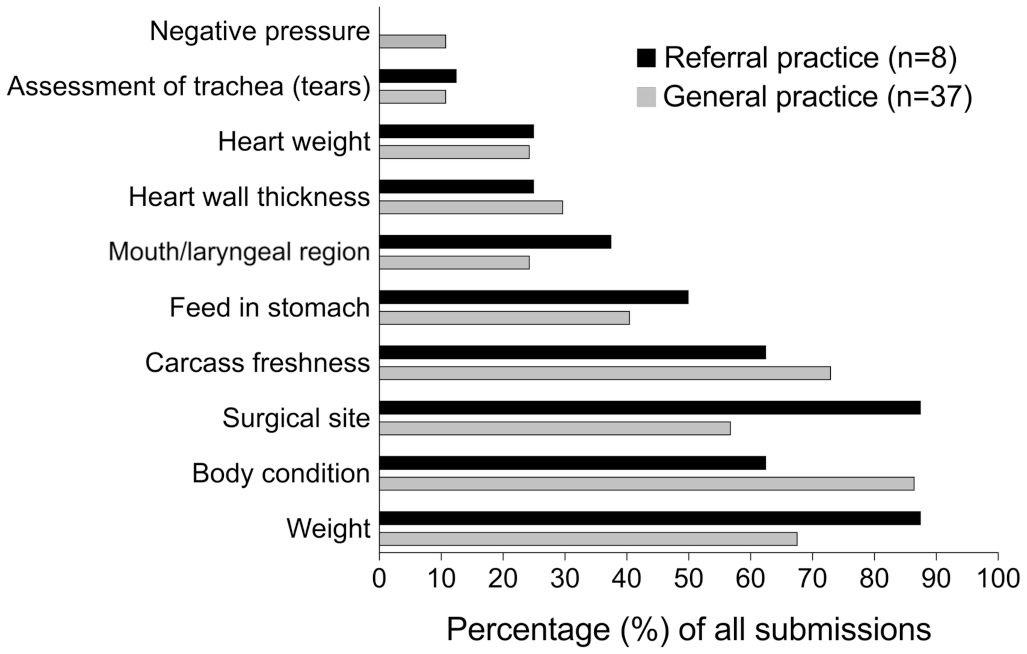
Frequency of reported findings within the gross postmortem reports from cats submitted from a general practice and referral practice that had died within 1 week of anesthesia.
Frequency of histologic examination of organs from cats submitted from a general practice (GP) and referral practice (RP) that had died within 1 week of an anesthesia and the major lesions identified.
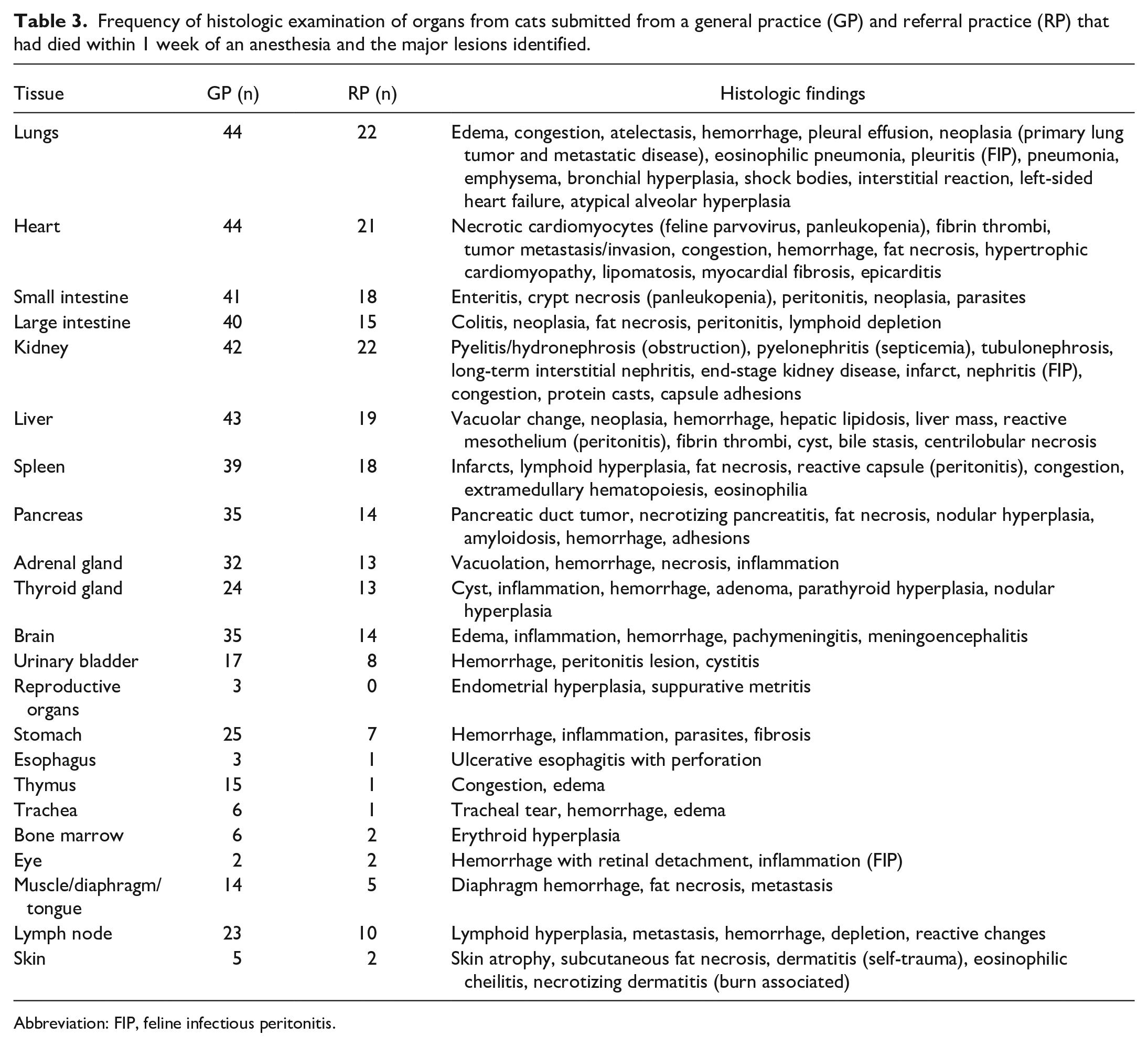
Abbreviation: FIP, feline infectious peritonitis.
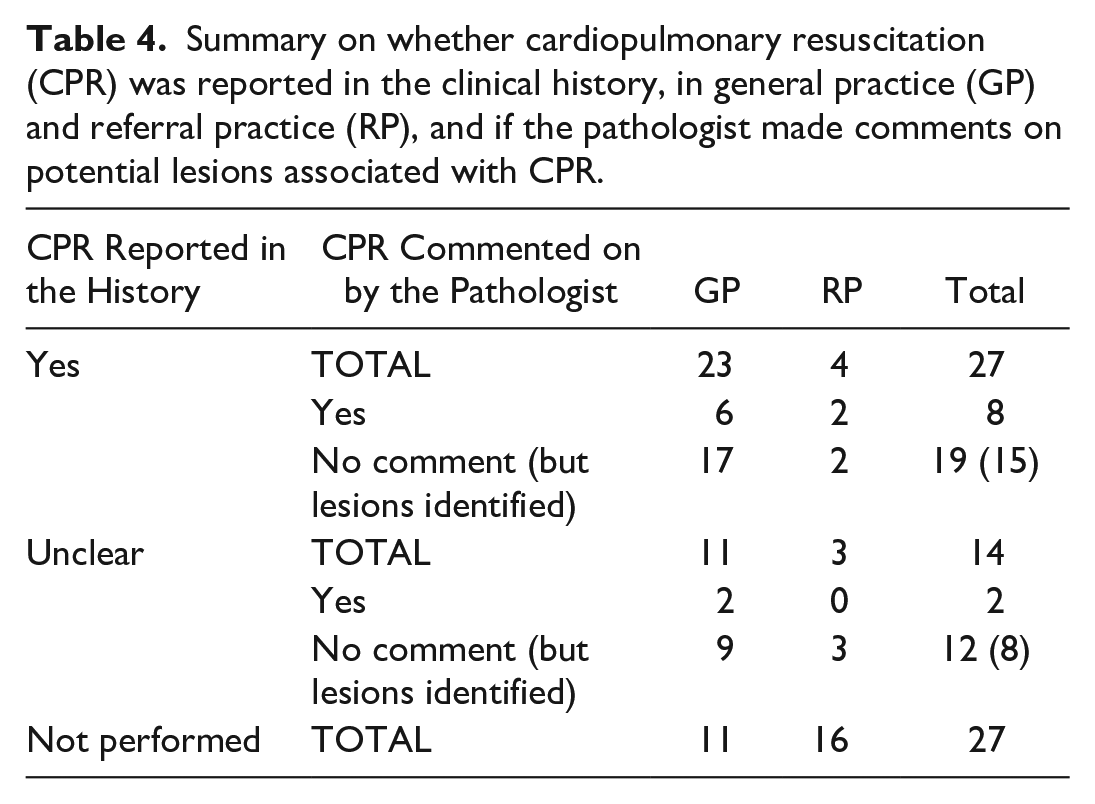
According to submission forms, CPR was performed in 27/68 (40%) cases, not performed in 27/68 (40%) cases, and unclear if performed in 14/68 (20%) cases (Table 4). Lesions that were potentially associated with CPR were pulmonary atelectasis, lung contusions/hemorrhage, lung congestion/edema, cardiac hemorrhage, pleural effusion, and pulmonary emphysema. Potential CPR-associated lesions were identified in 8/27 (30%) CPR-performed cases and 2/14 (14%) CPR-unclear cases. Additional gross or histologic lesions potentially related to CPR but not noted by the pathologists were found in 15 CPR-performed and 8 CPR-unclear cases. Including these, 23/27 (85%) CPR-performed and 10/14 (71%) CPR-unclear cases had potential lesions associated with CPR (Table 4).
Summary on whether cardiopulmonary resuscitation (CPR) was reported in the clinical history, in general practice (GP) and referral practice (RP), and if the pathologist made comments on potential lesions associated with CPR.
Discussion
Overall, animals submitted from the RP were significantly more likely to yield a confirmed or uncertain lesion compared to those submitted from GPs. Several factors likely contribute to this difference. First, animals from the RP more frequently underwent diagnostic tests prior to anesthesia (Table 1). Although certain advanced procedures such as computed tomography scans are more accessible at referral centers, even basic diagnostics such as bloodwork were performed in more than 75% of RP cases compared to fewer than 25% of GP cases. Increased pre-anesthetic testing may have provided a more comprehensive assessment of the patient’s health status, aiding in the identification of underlying diseases prior to anesthesia. In addition, the clinical histories submitted with RP cases tended to be more detailed, as reflected by a higher median word count, potentially improving the pathologist’s ability to accurately identify significant lesions at postmortem examination. A greater proportion of RP animals also had pre-existing illnesses, as indicated by higher ASA classification scores, further increasing the likelihood of detecting gross and/or histologic lesions.
Differences in logistical practices and motivations for postmortem submission between RP and GP settings may have also influenced the postmortem results. At the RP, postmortem examinations are routinely offered for all deaths, irrespective of whether anesthesia was involved, leading to submission of a broader range of cases, including those with known or suspected pre-existing diseases. In contrast, postmortem submissions from GPs are likely more selective, often limited to unexpected deaths where the cause was unclear and owners were motivated to seek an explanation. As a result, a submission bias may be playing a role in the results seen between the 2 practice types.
Despite an initially significant difference in the identification of lesions that were confirmed/uncertain contributors to death between RP and GP submissions, this distinction was no longer statistically significant once euthanized animals (many of which had known underlying conditions) were excluded from the analysis. This finding suggests that, irrespective of the practice type, determining the cause of death in animals that die unassisted during anesthesia remains challenging and is likely multifactorial. Factors that may contribute to a low diagnostic yield include acute anesthetic or patient-related complications that either do not produce lesions (e.g., electrolyte imbalances) or occur too rapidly to allow lesions to develop (e.g., hypoxia), as well as the potential for lesions that could be overlooked during postmortem examination. In animals that died unassisted, most GP cases were categorized as either due to an unknown disease or an unknown cause, while the RP had deaths in all 5 categories (Table 2). This may reflect the more detailed clinical histories and diagnostic workups typically available in referral settings, which could help to more accurately categorize these deaths. Overall, the absence of identifiable lesions in many cases across both practice types supports the limitations of relying solely on postmortem examination to determine the cause of PAD.
An interesting finding was that cats with limited clinical histories (e.g., rescue cats, barn cats, outdoor cats, or stray cats) represented approximately 20% (RP) to 33% (GP) of postmortem submissions. The significance of this finding remains unclear. These animals may more likely be submitted for postmortem examination because of the lack of need of permission from an owner to submit these cases. Another possibility is that these animals often have limited veterinary history or pre-anesthetic examinations, potentially leading to missed underlying morbidities and an increased risk of anesthetic complications. A study by Gerdin et al 18 specifically examined shelter cats and found that 33% had underlying conditions, such as Aerostrongylus abstrusus or hypertrophic cardiomyopathy; however, most of the cases (63%) had no significant lesions to explain the cause of death. Therefore, more research into this study population needs to be conducted.
Cardiopulmonary resuscitation encompasses a range of procedures including chest compressions (sometimes direct cardiac massage), endotracheal intubation, positive pressure ventilation, intravenous and intracardiac injections of pharmacologic agents, tracheostomy tube placement, thoracocentesis, and defibrillation.10,32 Lesions identified as associated with CPR in previous studies include pulmonary hemorrhage, pulmonary congestion and/or edema, pulmonary atelectasis, pneumothorax, cardiac hemorrhage and/or edema, myocardial necrosis, hemopericardium, hemothorax, hemomediastinum, diaphragmatic hemorrhage, intercostal hemorrhage, thymic hemorrhage, rib fracture, gastric bloat, ascites, hemoabdomen, hepatic laceration or hematoma, gall bladder hemorrhage, retroperitoneal hemorrhage, and/or multi-organ congestion or hemorrhage.12,27,32 Therefore, the submitting clinician needs to provide as much detail as possible if this procedure was performed. In this study, pathologists commented on CPR-related lesions in just under one quarter of the cases where CPR was confirmed or it was unclear if it was performed. Interestingly, when reviewing the gross and histologic reports in both the CPR-performed and CPR-unclear groups, additional potential CPR-related lesions were found. Other studies have found an incidence between 34% and 58.3% for CPR-related lesions.12,27,32 The previous study that identified a higher incidence of CPR-related lesions noted that their higher incidence is likely related to a single pathologist reviewing histology slides and considering even mild lesions such as hemorrhage to be associated with CPR. 32 This study and previous reports support the lack of standardization in defining what qualifies as a lesion associated with CPR and that more studies need to better characterize these lesions.
The postmortem reports revealed inconsistencies in the documenting of gross findings and incompleteness in tissue collection for histology. Based on the authors’ experience, many undocumented procedures were likely routinely performed, as pathology training often emphasizes reporting only abnormal findings. However, this current way of documenting PAD postmortems may warrant re-evaluation, particularly when there is potential for the case to become medico-legal. Delay11,12 has made evidence-based recommendations for the standard postmortem examination in cases of PAD submissions. Using these recommendations, the authors of this study have developed a proposed checklist and using data from this study highlight the value of collecting certain tissue samples for histology (Supplemental Table S1). The following paragraphs summarize important points of the postmortem checklist.
Improving objective data collection during the examination could provide more consistency in the postmortem report. Notably, body weight was not consistently recorded in postmortem reports, despite its importance for assessing organ size. Body weight can also be of value in assessing the hydration status of the animal. Clinical studies have identified excessive fluid administration as a risk factor for PAD in cats, likely due to historically high fluid rates. 4 Comparisons of pre-anesthetic body weights from the clinic and postmortem body weights could support fluid overload as being a factor in the animal’s death as studies have shown that increases greater than 10% of the body weight are suggestive of fluid overload. 5 The proposed checklist therefore recommends recording both pre-anesthetic and postmortem weights to assess for evidence of fluid accumulation. In addition, objective scoring systems for external nostril narrowing and soft palate thickening, particularly in brachycephalic breeds, have been developed and could be applied to reports to more accurately assess airway obstruction.9,25 Incorporating standardized objective measures into postmortem protocols can help support the significance of a lesion and improve reporting quality.
In addition to recording all gross findings as per the proposed checklist (including normal findings), consistent sampling of tissues beyond the major organ systems (e.g., heart and lungs) can help not only identify potential clues that contributed to the animal’s death but also information on the overall health of the patient.
The brain was one of the more consistently sampled tissues, and although it is highly susceptible to hypoxic injury (particularly in regions such as the hippocampus, cerebral cortex, basal nuclei, thalamus, and cerebellar cortex) no cases in this study exhibited such lesions. This is likely due to the acute nature of the fatal events, as histologic lesions of hypoxia typically require 4 to 12 hours to become detectable by light microscopy following the insult.28,30 Nevertheless, other significant lesions were identified, including inflammation consistent with feline infectious peritonitis, highlighting the importance of brain collection for ruling out infectious causes. 34 Similarly, although ocular tissues were rarely collected, they also provided valuable diagnostic information, with lesions associated with neoplasia, feline infectious peritonitis, and bleeding disorders identified. Both the brain and eye should be routinely collected during postmortem examinations, as they can reveal systemic diseases that may only manifest lesions at these sites.
The most common endocrine organs collected were the pancreas, adrenal glands, and thyroid glands, and regular collection of these organs is important given their ability to cause physiologic changes in the patient that could increase their risk of anesthetic complications. In this study, although most thyroid lesions identified were likely incidental findings, lesions such as small adenomas or nodular hyperplasia could potentially be of clinical significance. In cats, even small functional thyroid masses can result in hyperthyroidism, a condition associated with metabolic disturbances including tachycardia, arrhythmias, and hypertension. If not well managed prior to anesthesia, these abnormalities can be exacerbated by anesthetic agents and may contribute to fatal anesthetic complications. 31 Therefore, postmortem thyroid lesions should be interpreted alongside the clinical history, including whether the cat was known to be hyperthyroid and whether the condition was well regulated. Similarly, the adrenal glands, another endocrine organ frequently sampled, rarely exhibited specific lesions in this study; however, their evaluation remains important. Endocrine disorders such as hypoadrenocorticism or hyperadrenocorticism, particularly in dogs, can lead to significant electrolyte imbalances, cardiovascular instability, and altered stress responses that may complicate anesthesia. 39
Although the gastrointestinal tract may not seem valuable in animals without a history of digestive disease, its evaluation can still provide important information. Gross examination should include a description of gastric contents. The presence of ingesta in the stomach during postmortem examination raises concerns for complications such as regurgitation or aspiration, which can lead to esophageal and tracheal damage and potentially aspiration pneumonia.1,29 In this study, one cat exhibited ulcerative esophagitis with esophageal rupture, resulting in fatal pleural effusion. The esophageal damage was suspected to result from long-term gastroesophageal reflux, but the history did not report this as a clinical problem in this patient. In other cases, gastrointestinal findings such as parasitic infestations or depletion of Peyer’s patches can suggest need for better deworming protocols or suggest some sort of systemic illness. These observations demonstrate that gastrointestinal tract evaluation can uncover subtle or incidental findings that may inform the overall health status of the animal.
The urogenital system should be carefully evaluated particularly in cases involving abdominal surgery or abdominal complications. The content of the urinary bladder and the ability to express the bladder should be evaluated to ensure no trauma was caused to these tissues by the surgery. In one case, a spay surgery caused accidental ligation of one of the ureters leading to damage to the associated kidney. In addition, while reproductive tissues were collected in a minority of cases, significant lesions such as cystic endometrial hyperplasia and endometritis were identified. Although relatively rare, reproductive tract pathology can have systemic implications and should be evaluated when present.
Although skin was infrequently collected, it identified potentially significant findings in a few cases. Lesions included coagulative necrosis consistent with thermal injury and changes suggestive of underlying endocrinopathy. Historically, thermal burns have been associated with the use of older heating devices, such as hot water bottles, which can cause direct burns in anesthetic patients who are unable to move away from the heated devices and also have different perfusion of the skin.6,15 Newer technologies such as the Bair Hugger (3M, St. Paul, Minnesota, United States of America) have reduced the incidence of such injuries. Identifying these lesions could draw attention to a practice not transitioning to these better practice standards. 37 In addition, dermatologic findings may indicate undiagnosed endocrine disorders, such as diabetes mellitus or hyperthyroidism, which, as previously discussed, can potentially lead to anesthetic complications if not well regulated. 20 Again, these few cases highlight the value of regularly sampling the skin in PAD cases.
Other listed tissues occasionally revealed significant findings, including metastatic neoplasia in muscle, erythroid hyperplasia in bone marrow, amyloidosis in the pancreas, and lymphoid depletion in the spleen and lymph nodes. Ideally, the proposed list of tissues for sampling should be evaluated in future studies to identify the most diagnostically valuable specimens while minimizing unnecessary collection. Streamlining tissue collection in this manner would be particularly beneficial in diagnostic laboratories managing high case volumes.
In human medicine, postmortem pathology reports in cases of PAD are not interpreted in isolation; instead, they form part of a multidisciplinary investigation that often includes anesthesiologists and surgeons. 24 This collaborative approach recognizes that pathology alone cannot fully determine the sequence of events leading to death. In contrast, veterinary medicine has not widely adopted this practice. Clinicians often submit PAD cases for necropsy with the expectation that an underlying disease will be identified, as demonstrated by Rose et al, 35 where 88.7% of clinicians suspected that the animal had a pre-existing condition that may have predisposed it to an anesthetic complication. Although pathologists may identify significant lesions, determining whether the lesion was the sole cause of death is difficult without detailed clinical histories and consultation with an anesthesiologist.
Human studies of PAD have also identified that many “anesthetic incidents” (including death) frequently involve human error. 24 Given that veterinary anesthesia is often administered by practitioners with less formal anesthesia training compared to human medicine, it is reasonable to assume that human error may similarly contribute to veterinary PAD. For instance, if a patient developed severe laryngospasm during intubation, a pathologist might identify swelling of pharyngeal tissues postmortem. However, without anesthetic records reviewed by an anesthesiologist, important contributing factors may be overlooked. For example, both insufficient anesthetic depth and absence of preoxygenation have been shown to increase the risk of laryngospasm complications, and without identifying these deficiencies, opportunities for improving patient safety and preventing future incidents may be missed. 14
It should be noted that even with standardized postmortem protocols and checklists, determining the cause of death in PAD cases will likely remain challenging without collaboration with veterinary anesthesiologists. Many anesthetic complications occur acutely and leave no discernible lesions, while some identified lesions may be incidental and unrelated to death. Accurate interpretation requires integrating the pathology findings with detailed clinical histories and consultation with a veterinary anesthesiologist.
This study evaluated postmortem findings in cats that died within 1 week of anesthesia. The study identified that regardless of submission source, animals without significant pre-existing disease were less likely to have identifiable lesions explaining their deaths and that currently the reporting of postmortem PAD cases is not standardized. In addition, a proposed standardized postmortem checklist was developed, which provides a starting point for improving consistency, though future validation studies are needed.
This study had several limitations. Cases were drawn from different databases, and not all referral submissions were suspected PAD cases at the time of necropsy, limiting direct comparisons. ASA classifications were assigned differently between groups, and the sample size, particularly from GP was small. Future studies should aim for a prospective design with a standardized data collection and larger cohorts.
In conclusion, while pathology remains an important part of a PAD investigation, accurate determination of cause of death requires a multidisciplinary approach that combines pathology, clinical history, and anesthetic evaluation.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858251361134 – Supplemental material for Trends in feline perianesthetic death postmortem submissions from a referral teaching hospital and general practices in Saskatchewan (Canada) and a proposed postmortem checklist
Supplemental material, sj-pdf-1-vet-10.1177_03009858251361134 for Trends in feline perianesthetic death postmortem submissions from a referral teaching hospital and general practices in Saskatchewan (Canada) and a proposed postmortem checklist by Nicole Rose, Bruce K. Wobeser, Jennifer L. Davies and Daniel J. Pang in Veterinary Pathology
Footnotes
Acknowledgements
The authors extend their gratitude to the pathologists at Prairie Diagnostic Services Inc. for providing postmortem reports that contributed to this dataset. Also thank you for Drs. David Brodbelt, Enrique Aburto, and Michael Zabrodski for their feedback on the manuscript. They also sincerely thank the clinical veterinarians from the Veterinary Medical Center at the University of Saskatchewan’s Western College of Veterinary Medicine, as well as those from general practices across Saskatchewan, for their dedication in caring for these animals and their willingness to submit cases of anesthetic-related deaths for postmortem examination. They would also like to thank Dr. Sarah Parker (Western College of Veterinary Medicine) for her assistance in statistical analysis.
Author Contributions
NR reviewed all the pathology reports and entered the data into an Excel spreadsheet. American Society of Anesthesiologist physical status classification was performed by NR and DP. Statistical analysis was performed by NR and DP. The manuscript was written by NR and edited by DP, BW, and JD.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
