Abstract
High mortality in bobwhite quail chicks (Colinus virginianus) (35%–85%) was reported from a grower flock in Iowa during July and August of 2022. Two diagnostic submissions of dead, 3-day-old quail chicks were received. Postmortem examination revealed multifocal, pinpoint, pale tan foci in the liver of all birds. Histologic examination revealed moderate to severe, acute, multifocal, random necrotizing hepatitis with multinucleated cells and dystrophic mineralization. Metagenomic sequencing of liver detected orthoreovirus. A high level of avian reovirus (ARV) RNA was identified by real-time reverse transcriptase-PCR. ARV was successfully isolated from liver and lung on the Leghorn male hepatoma cell line. In addition, electron microscopy revealed orthoreovirus viral particles and virus factories in the formalin-fixed livers and viral-infected cell culture. This case highlights ARV as a potential cause of hepatitis in quail chicks and should be considered in the differential diagnosis.
Keywords
Avian reovirus (ARV) belongs to the family Reoviridae and genus Orthoreovirus. The virus is nonenveloped with a double-shell icosahedral capsid and contains a double-stranded RNA genome comprised of 10 segments. ARV is ubiquitous among the poultry population and can be detected in the gastrointestinal and respiratory tracts of healthy birds. Pathogenic strains are associated with arthritis and tenosynovitis in broiler chickens and turkeys. 18 Other forms of the disease have also been described, and are characterized by myocarditis, 1 enteritis, 7 and hepatitis.10,13 The mortality is typically low with moderate to high morbidity. Although it is not often associated with disease in wild birds, there are reports of high mortality outbreaks in the American woodcocks (Scolopax minor), 3 enteric disease in pheasants (Phasianus colchicus), 14 and neuropathy in hooded crows (Corvus cornix). 8 This report describes significant mortality and hepatitis in young bobwhite quail (C. virginianus) infected with ARV.
The Iowa State University Veterinary Diagnostic Laboratory received 2 submissions of 3-day-old bobwhite quail (C. virginianus) in Iowa with a history of sudden death. In July 2022, the grower hatched ~8000 chicks sourced from a breeder flock in Georgia (80% hatching rate) with an 85% mortality rate within the first week after hatching. Seven dead birds were submitted for postmortem examination. In August 2022, the same grower hatched another set of birds with ~35% mortality in the first week after hatching. A second submission of 6 dead chicks was submitted to the diagnostic laboratory. The gross changes in all birds were similar in the first submission (7/7). There were no remarkable lesions externally. The thigh muscles were uniformly dry, indicative of dehydration. The crops were empty of content. The spleens were diffusely pale. Notably, there were multifocal to coalescing, pale pinpoint foci on the liver surface and parenchyma (Fig. 1a). On the second submission, similar gross changes were noted in the livers as previously described in 5/6 chicks. The crops were empty (5/6), and the lungs were congested (4/6). There were no remarkable lesions observed externally. Microscopically, both submissions had multifocal-random, acute necrotizing hepatitis affecting 30% to 70% of the liver parenchyma (Fig. 1b-d). The necrotic areas were characterized by the loss of hepatic cords and replacement with karyorrhectic debris, fibrin, dystrophic mineralization, and scattered heterophils and histiocytes. At the periphery of the necrotic areas, there was hepatocyte dissociation, scattered individual cell necrosis, and syncytial cell formation. The remaining parenchyma contained perivascular mononuclear inflammatory infiltrates and variable degree of bile duct hyperplasia. Routine aerobic and anaerobic bacterial cultures were performed on the pooled liver and intestines of both cases. 2 Only Escherichia coli was isolated from both submissions’ intestines (moderate-to-high growth) and the liver of the second submission (low growth).
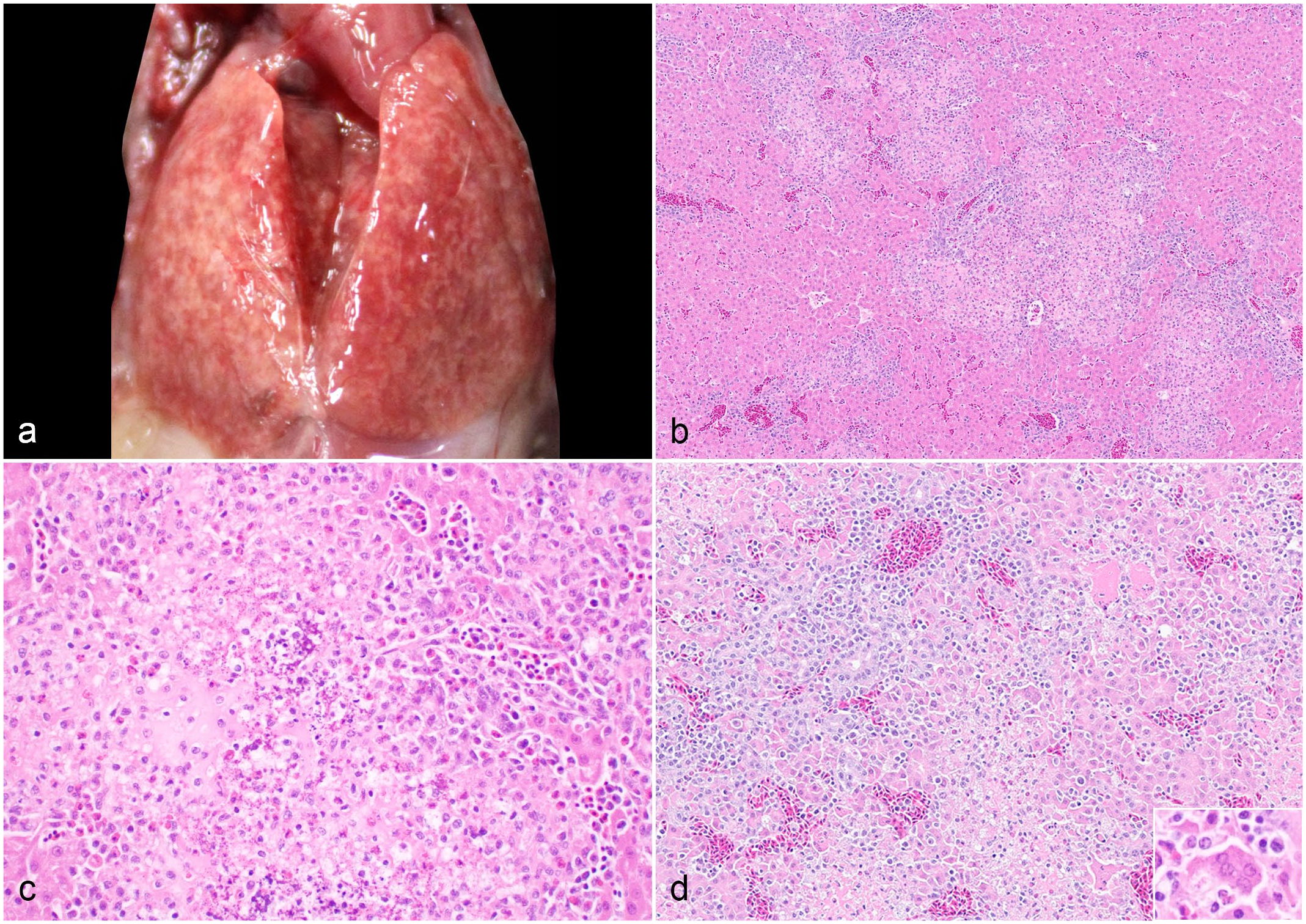
Reoviral hepatitis, liver, quail. (a) The liver has multifocal to coalescing pale tan discoloration. (b) There are multifocal random areas of acute hepatic necrosis with disruption of hepatic cords. There are increased biliary profiles in the portal areas. Hematoxylin and eosin (HE). (c) Dystrophic mineralization is present in the necrotic focus with degenerated heterophilic and histiocytic infiltrates. HE. (d) The hepatocytes are dissociated, vacuolated, and hypereosinophilic. There are aggregates of karyolytic and karyorrhectic cells and syncytial cell formation (inset) in the necrotic areas. The blood vessels are circumscribed by mononuclear inflammatory cells. HE.
Liver samples pooled from the second submission were subjected to metagenomic next-generation sequencing (Illumina MiSeq, San Diego, CA) to further determine the etiological agent. 6 This analysis revealed sequencing reads belonging to orthoreovirus (SRA: PRJNA1040964). Genomes of other microorganisms were not detected. Phylogenetic analysis was performed on full σC nucleotide sequences using the MEGA 11 software. The analysis included 37 reference ARV sequences derived from chickens and turkeys. The σC sequence demonstrated a close phylogenetic identity to turkey ARV strains (Fig. 2). Subsequently, all 7 livers, lungs, and brains from the first submission pooled by the sample type were tested using an ARV real-time reverse transcriptase-PCR targeting the conserved region of the M1 gene, 20 yielding positive detections with cycle threshold values of 14.3, 20.0, and 23.8, respectively. Next, virus isolation was attempted on the liver and lung tissue homogenates from the first submission using the Leghorn male hepatoma cell line (ATCC CRL-2117). 11 Large bloom-like syncytial cells were observed in both the liver- and lung-inoculated cells. Immunofluorescence using an FITC-conjugated ARV antibody (USDA-NVSL, Ames, Iowa) revealed positive labeling in the cells inoculated with liver or lung tissue homogenates. 11 In addition, ARV real-time reverse transcriptase-PCR was performed to confirm that reovirus was successfully isolated from the liver and lung homogenates (Supplemental Table S1). 20 Long dsRNA (>30 bp) was detected in the areas of hepatic necrosis via immunohistochemistry (anti-double strand RNA [rJ2] antibody, Sigma-Aldrich, USA) (Supplemental Figure S1, Supplementary Materials). Transmission electron microscopy (ThermoFisher FEI Tecnai G2 BioTWIN, Hillsboro, OR) was performed on selected areas of necrotic liver lifted from the formalin-fixed paraffin-embedded blocks and viral infected cell culture fixed in 2.5% glutaraldehyde in 0.1M cacodylate buffer (Fig. 3a) (Supplementary Materials). The ultrastructural findings revealed mature reovirus viral particles and virus factories (VF) in the viral-infected cells’ cytoplasm (Fig. 3b).

Phylogenetic tree of the avian reovirus (ARV) σC nucleotide sequence detected in the quail liver with hepatitis (red arrow) compared with 37 reference ARV σC sequences from chickens and turkeys retrieved from the GenBank. The analysis was performed on MEGA 11 software. The phylogenetic tree was constructed from the neighbor-joining method using the Kimura 2-parameter model. The percentage of replicate trees in which the associated sequences clustered together in the bootstrap test (1000 replicates) are shown next to the branches. The scale bar indicates nucleotide substitutions per site.
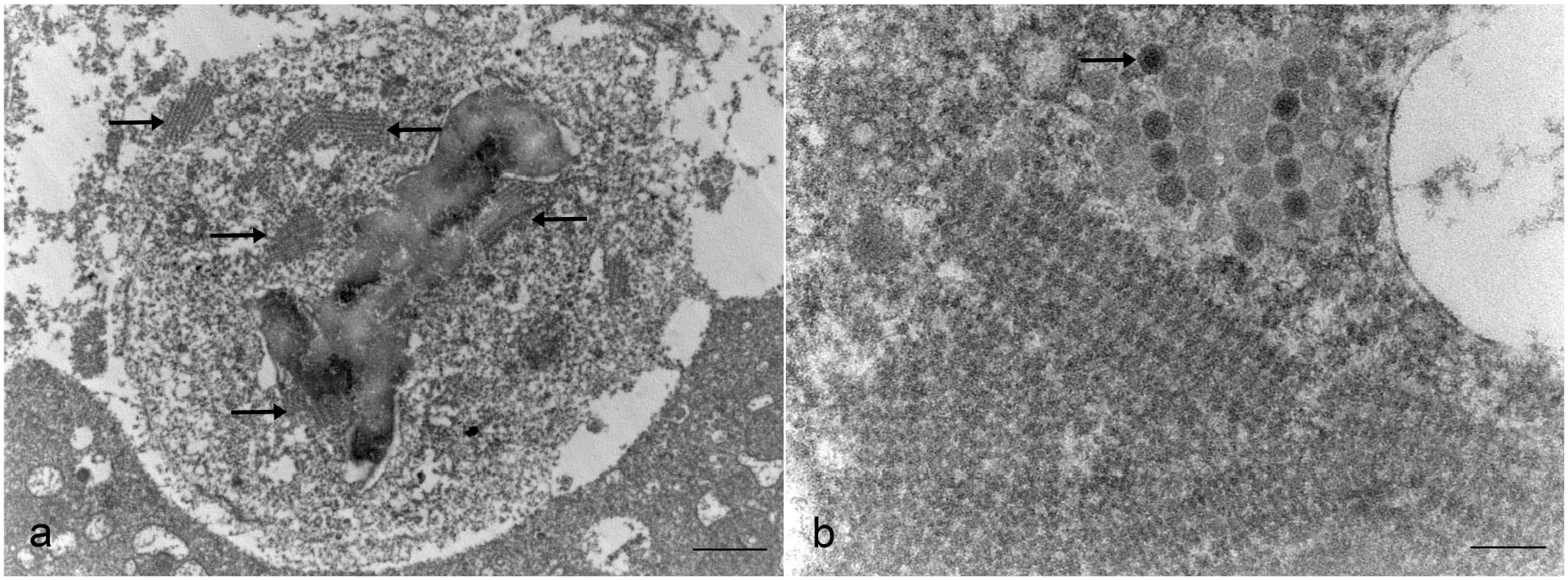
Transmission electron microscopic images of the reoviral infection. (a) Liver, quail. There are rows of viral factories within the cytoplasm of infected cells (arrows). Scale bar = 1 μm. (b) Leghorn male hepatoma cell. Reoviral virions (arrow) are 70 to 75 nm in diameter with an inner electron-dense core surrounded by an inner shell. Scale bar = 200 nm.
The common differential causes of hepatitis in young quail in North America include systemic bacterial infection (e.g., E. coli, Salmonella spp) and aviadenovirus. Manifestations of systemic bacterial infection in the liver of quail are characterized by areas of hemorrhage, fibrin, hepatic necrosis, marked heterophilic and/or histiocytic inflammation, heterophilic granuloma, and the presence of bacterial colonies. Hepatic lesions are often accompanied by other changes such as airsacculitis, pericarditis, and coelomitis. Due to the ubiquitous nature of E. coli and the inconsistent detection from both submissions, the bacteria isolated from the liver is of equivocal significance. Based on the microscopic findings and the detection of long double strand RNA, we hypothesized that the liver changes were due to an acute viral infection. However, additional diagnostic testing is necessary to confirm a viral infection. Metagenomic sequencing can serve as a hypothesis-free diagnostic tool capable of detecting novel or undetermined pathogens. 9 It can nonselectively detect the genomic component of organisms in clinical samples and provides the genome sequence of the pathogen of interest. Utilizing metagenomic next-generation sequencing, we identified ARV in the liver and further confirmed the infection by the high levels of viral RNA detected by reverse transcriptase-PCR, virus isolation, and visualization of viral-infected cells through electron microscopy. The laboratory findings, along with the liver lesions, provide compelling evidence that the cause of hepatitis and high mortality in these quail was due to an ARV infection.
Reports of reovirus infection in quail are rare. In 1990, a report of respiratory disease and high mortality was described in young bobwhite quail associated with reoviral infection. 12 Lesions of acute necrotizing hepatitis and airsacculitis were observed in young bobwhite quail with reovirus and adenovirus isolated from the liver and the tracheal samples. Replication of the disease and hepatic changes were demonstrated with experimental inoculation of the isolated reovirus. In addition, ARV association with enteric disease in conjunction with intestinal cryptosporidiosis was reported in bobwhite quail. 17 However, genomic characterization was not described in either report. The microscopic findings of acute necrotizing hepatitis and syncytial cell formation in ARV infection have been described in turkey poults and experimentally infected newborn chickens.10,13 Other causes of viral hepatitis reported in gallinaceous birds include aviadenovirus and picornavirus (turkey viral hepatitis).4,5 Microscopic changes of aviadenovirus infection include distinct intranuclear inclusion bodies, 4 and viral particles can be observed in the nucleus by electron microscopy. Interestingly, the microscopic changes in the liver of turkey viral hepatitis exhibit similar syncytial cell formation as ARV infection. 5 The pancreatic change is inconsistently observed with turkey viral hepatitis. Thus, differentiating ARV and turkey viral hepatitis infections requires ancillary testing.
ARV viral particles are icosahedral, nonencapsulated, and approximately 70 to 80 nm in diameter (Fig. 3b). A mature ARV virion contains an inner electron-dense core surrounded by outer shells. Upon infection, orthoreovirus forms VF that are globular or filamentous structures dependent on the viral strains. 19 Ultrastructurally, we observed rows of organized VF with adjacent assembled viral particles in the viral-infected cells (Fig. 3). The VF is the site within the host cell where viral replication, particle assembly, and genome packaging occur. 15 The previously reported globular and filamentous VF structures were not observed, suggesting a potentially different genomic packaging mechanism in this ARV variant. ARV VF can be observed as intracytoplasmic viral inclusion bodies under light microscopy. This is most prominent in hematoxylin and eosin-stained viral-infected cell cultures and is not typically observed in vivo. 13
Diagnosis of ARV infection is challenging due to the difficulty in distinguishing nonpathogenic from pathogenic strains. The identification of ARV via PCR, metagenomic next-generation sequencing, and virus isolation methods does not provide sufficient evidence to establish a correlation with a disease process. Direct visualization of the virus by electron microscopy can further confirm infection at a cellular level. In addition, targeted direct tissue detection assays such as immunohistochemistry and in situ hybridization are useful for confirmation of infection. However, these assays are not widely available for ARV or are currently cost-prohibitive for routine diagnosis of poultry diseases. Correlations of ARV genotype and pathogenicity have been explored using the principal antigenic protein σC. 16 Although it is a useful target for phylogenetic comparison, it lacks association with pathogenicity. Whole-genome sequencing is by far the optimal path to demonstrate the pathotypes of ARV on a molecular level. 16
It is unclear how the quail chicks reported here were exposed to the ARV. Vertical transmission from the breeder stock is suspected based on the age of the affected quail and the consecutive outbreaks. Other less probable transmission sources include horizontal transmission at the hatchery, from nearby poultry facilities, or from wild birds. Additional viral sequence analysis and epidemiological investigation may help determine the source of the viral infection. Finally, the observed mortality rate associated with ARV infection was notably high. This could be attributed to several factors such as the naïve immune status of the flock, high viral load, high pathogenicity of the virus strain, or a combination of these factors. Future pathogenicity studies on viral isolates from these quails can be performed to confirm the virulence.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241302842 – Supplemental material for High mortality associated with avian reoviral hepatitis in young quail (Colinus virginianus)
Supplemental material, sj-pdf-1-vet-10.1177_03009858241302842 for High mortality associated with avian reoviral hepatitis in young quail (Colinus virginianus) by Panchan Sitthicharoenchai, Jianqiang Zhang, Liying Tian, Judith Stasko, Amro Hashish, Cheng-Shun Hsueh, Yuko Sato, Benjamin Hause and Mohamed El-Gazzar in Veterinary Pathology
Footnotes
Acknowledgements
I thank the necropsy and histology technicians at Iowa State University Veterinary Diagnostic Laboratory that assisted with the tissues sample collection and histology slide preparations.
Author Contributions
PS and ME are the diagnostic case coordinators and performed postmortem examination and microscopic evaluation, JZ and LT facilitated and performed the viral isolation, JS prepared and performed electron microscopy, BH and AH performed viral sequencing and phylogenetic analysis, CH and YS facilitated and performed the immunohistochemical assay.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
