Abstract
Large-scale retrospective studies allow for identification of disease trends, such as predisposing factors, typical clinical signs, and range of histologic lesions, which cannot be determined in individual case reports. Lesions of the endocrine pancreas of ferrets are extensively reported; however, there are no in-depth investigations of lesions in the exocrine pancreas. This retrospective analysis presents the histologic features, clinical signs, and concurrent diseases of lesions in the exocrine pancreas of ferrets. Seventy-seven lesions were reported and included acinar cell hyperplasia (n = 32), chronic pancreatitis (n = 16), acute pancreatitis (n = 13), acinar cell adenoma (n = 5), acinar cell carcinoma (n = 4), acinar cell atrophy (n = 3), presumptive acinar cell hypoplasia (n = 2), and lymphoma (n = 2). Our results demonstrate that acinar cell hyperplasia and chronic pancreatitis can both cause grossly visible pancreatic nodules. Hyperplasia was not associated with neoplastic transformation. In addition, acinar cell adenoma was slightly more common than carcinoma, which is contrary to most reports of neoplasia in ferrets. Our findings also suggest that acute pancreatitis can be a sequela to pancreatic biopsy and that there may be an association between chronic pancreatitis and diabetes mellitus in ferrets. Finally, zinc toxicosis was found to be an unlikely cause of pancreatitis in these ferrets based on zinc tissue concentration testing in a subset of cases.
Pancreatic lesions in ferrets are common and overwhelmingly described in the endocrine pancreas.2–4,7,18,20,21,25,32 Insulinomas (beta cell tumors) account for up to 22% of all neoplasms in ferrets3,18 and cause unregulated insulin secretion with episodic hypoglycemia, lethargy, ataxia, collapse, and weight loss.2,15,20,21,25,29,32,36 Ferrets may have multiple insulinomas but metastasis is rare.2,20,29,36 Other lesions in the endocrine pancreas of ferrets are uncommon and include vacuolation of islet cells (secondary to diabetes mellitus) and pancreatic polypeptidoma.7,21,25,29,32
Spontaneous lesions of the exocrine pancreas in ferrets are less commonly described. The only naturally occurring lesions reported in the exocrine pancreas of ferrets are exocrine tumors and pancreatitis.2–4,10,12,13,17,18,21,25,28,37,38,39 Exocrine pancreatic neoplasms occur less frequently than insulinomas. In a study of 574 neoplasms in ferrets, exocrine pancreatic tumors were the seventh most common neoplasm with 8 total cases, while 139 were insulinomas. 18 In a separate study of 856 neoplasms in ferrets, there were 10 exocrine pancreatic tumors and 190 islet cell tumors. 3 While, in the authors’ experience, exocrine hyperplasia is a common finding in ferrets, pancreatic exocrine adenomas and hyperplasia are less commonly seen than insulinomas and are typically considered incidental findings. 10
Reports of pancreatitis in ferrets are rare. Pancreatitis may occur spontaneously in obese ferrets, after pancreatic biopsy (i.e. insulinoma excision), or as a sequela to diabetes mellitus.11,25,32,39 Ferrets with systemic coronavirus infection may also develop pyogranulomatous pancreatitis, although inflammation is more common in other organs such as the small intestines, kidneys, liver, and spleen.11,16 Acute pancreatitis in ferrets has also been associated with zinc toxicosis. Feeding over 3000 ppm zinc produced acute pancreatitis in 2 of 7 ferrets and acute renal tubular injury and anemia in all 7 animals. 31 Natural zinc toxicosis was documented in a group of ferrets housed in and fed through steam-treated galvanized steel enclosures, though histopathology of the pancreas was not reported in these cases. 30 In other domestic species, chronic zinc toxicosis can lead to diffuse acinar necrosis and atrophy with prominent interstitial fibrosis.8,15 Rarely, zinc deficiency has been associated with pancreatitis in animals. 35
To the author’s knowledge, a large-scale study of lesions in the exocrine pancreas of ferrets has not been conducted. The aims of this study are to better identify the spectrum of histologic lesions within the ferret exocrine pancreas and commonalities in signalment, clinical signs, gross findings, and comorbidities for each type of lesion, and to investigate if zinc toxicosis may be a factor in the development of pancreatitis in this species.
Materials and Methods
The Northwest ZooPath (Monroe, WA) archival database was searched for biopsy and necropsy specimens from domestic ferrets with lesions in the exocrine pancreas. All cases were submitted from primary veterinarians, fixed for >24 hours in neutral-buffered formalin, processed routinely for histology, sectioned at a 5-µm-thickness, and stained with hematoxylin and eosin.
All cases were reviewed by one board-certified pathologist (EEBL) to standardize nomenclature. Diagnoses were categorized according to established definitions and included atrophy, hypoplasia, pancreatitis, hyperplasia, adenoma, and adenocarcinoma. 15 Patient age, sex, animal type (zoo animal or pet), clinical signs, concurrent diseases, concurrent histologic diagnoses, and cause of death (necropsy submissions only) were gathered from finalized histology reports. Cases were categorized according to exocrine pancreatic lesions to identify commonalities in other data points. Follow-up data, including survival times and causes of death, were gathered from successive biopsy reports within the Northwest ZooPath archival database.
To assess the possibility of zinc toxicosis or deficiency in the pathogenesis of pancreatic injury, a subset of cases was selected for trace mineral analysis. Formalin-fixed, paraffin-embedded tissue blocks of 5 cases of acute pancreatitis, 1 case of chronic pancreatitis, and 1 case of lymphoma were submitted to Michigan State University Veterinary Diagnostic Laboratory (commercial laboratory test). Dry weight zinc concentrations were determined in the pancreas using inductively coupled plasma mass spectrometry. Dry weight concentrations were converted to wet weight concentrations using a presumed pancreatic water content of 70.53%. 24
Results
Northwest ZooPath archives contained 4412 ferret biopsy and necropsy reports, 69 of which had lesions reported in the exocrine pancreas (Supplemental Table S1). Forty-seven cases were submitted as biopsies, and 19 cases were necropsies. The sampling method was not reported in 3 cases. Diagnoses included acinar cell hyperplasia (n = 32, 42%), chronic pancreatitis (n = 16, 21%), acute pancreatitis (n = 13, 17%), acinar cell adenoma (n = 5, 6%), acinar cell carcinoma (n = 4, 5%), atrophy (n = 3, 4%), hypoplasia (n = 2, 3%), and lymphoma (n = 2, 3%) (Table 1). Eight cases had multiple diagnoses in the exocrine pancreas, including 5 cases (6%) with concurrent chronic pancreatitis and acinar cell hyperplasia, 1 case with concurrent acute pancreatitis and acinar cell hyperplasia, 1 case with concurrent acute pancreatitis and acinar cell atrophy, and 1 case with concurrent chronic pancreatitis and acinar cell atrophy. Follow-up data were available for 7 of 49 biopsy cases.
Summary of total number of cases, sampling method, average age, and concurrent lesions in 69 ferrets with exocrine pancreatic lesions.
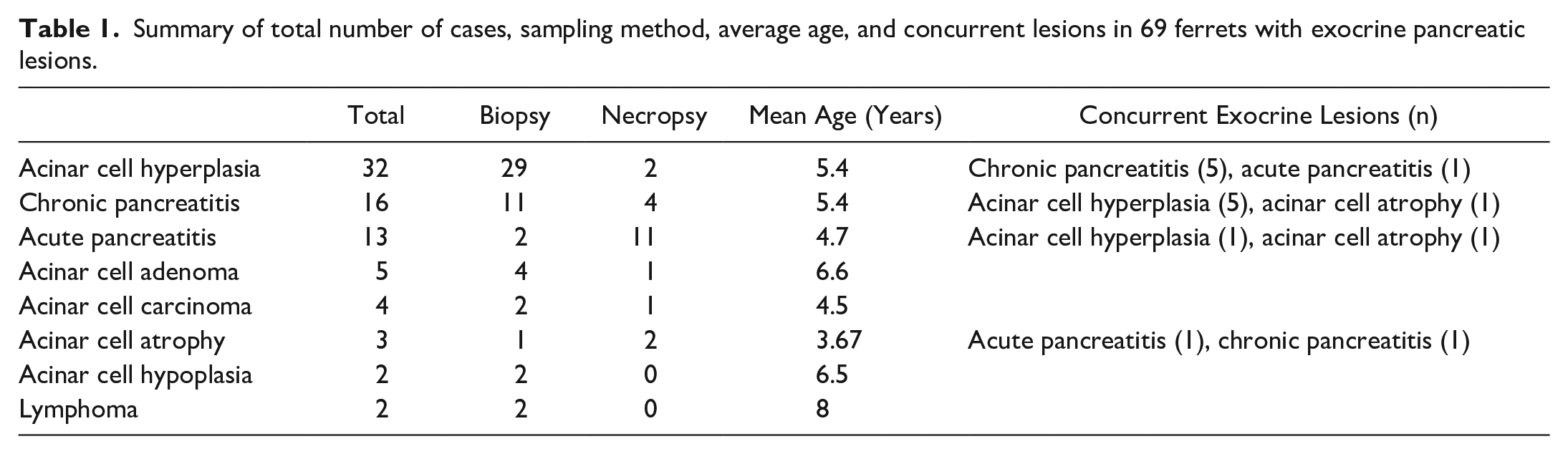
Acinar cell hyperplasia was identified via biopsy (n = 29), necropsy (n = 2), or following an unspecified procedure (n = 1) in 28 pets and 4 zoo animals, including 18 males, 10 females, and 4 animals of undetermined sex. The animals ranged from 3 to 7 years old (mean = 5.4 years). None of the reported clinical signs were shared by 2 or more animals. Concurrent diseases present in more than one animal included adrenocortical neoplasia or hyperplasia (n = 14), islet cell neoplasia (n = 12), and inflammatory bowel disease (n = 7). At surgery or necropsy, pancreatic nodules were identified in 21 animals. Histologically, the pancreas was expanded by variably well-demarcated, roughly circular nodules of acinar cells, 1 to 8 mm in greatest dimension (mean = 2.9 mm), minimally compressing the surrounding parenchyma without any capsule formation, which differentiated them from acinar cell adenomas. Acinar cells were supported by scant fibrovascular tissue and admixed with rare ductules. Zymogen granule density was variable and slightly less or slightly more than those in acinar cells in the rest of the pancreas. Nuclei were either the same size or slightly larger than acinar cells in the surrounding tissue. Mitotic counts ranged from 0 to 5 per 2.37 mm2 (mean = 0.5) (Fig. 1A).
Pancreatic lesions in domestic ferrets. Hematoxylin and eosin. (a) Acinar cell hyperplasia. The pancreas is expanded by well-demarcated nodules of acinar cells with minimal compression of the surrounding parenchyma and no capsule formation. (b) Chronic pancreatitis. Moderate numbers of lymphocytes and few plasma cells infiltrate the interstitium and acini with mild ductular ectasia and hyperplasia. (c) Acute pancreatitis. Regions of lytic necrosis replace acinar cells and contain abundant variably fragmented neutrophils, karyorrhectic cellular debris, edema, and a small amount of fibrin. (d) Acinar cell carcinoma. Neoplastic cells form acini and tubules and invade the muscularis externa of the duodenum (arrow). The tumor is associated with a marked scirrhous response. Inset: Higher magnification of neoplastic cells.
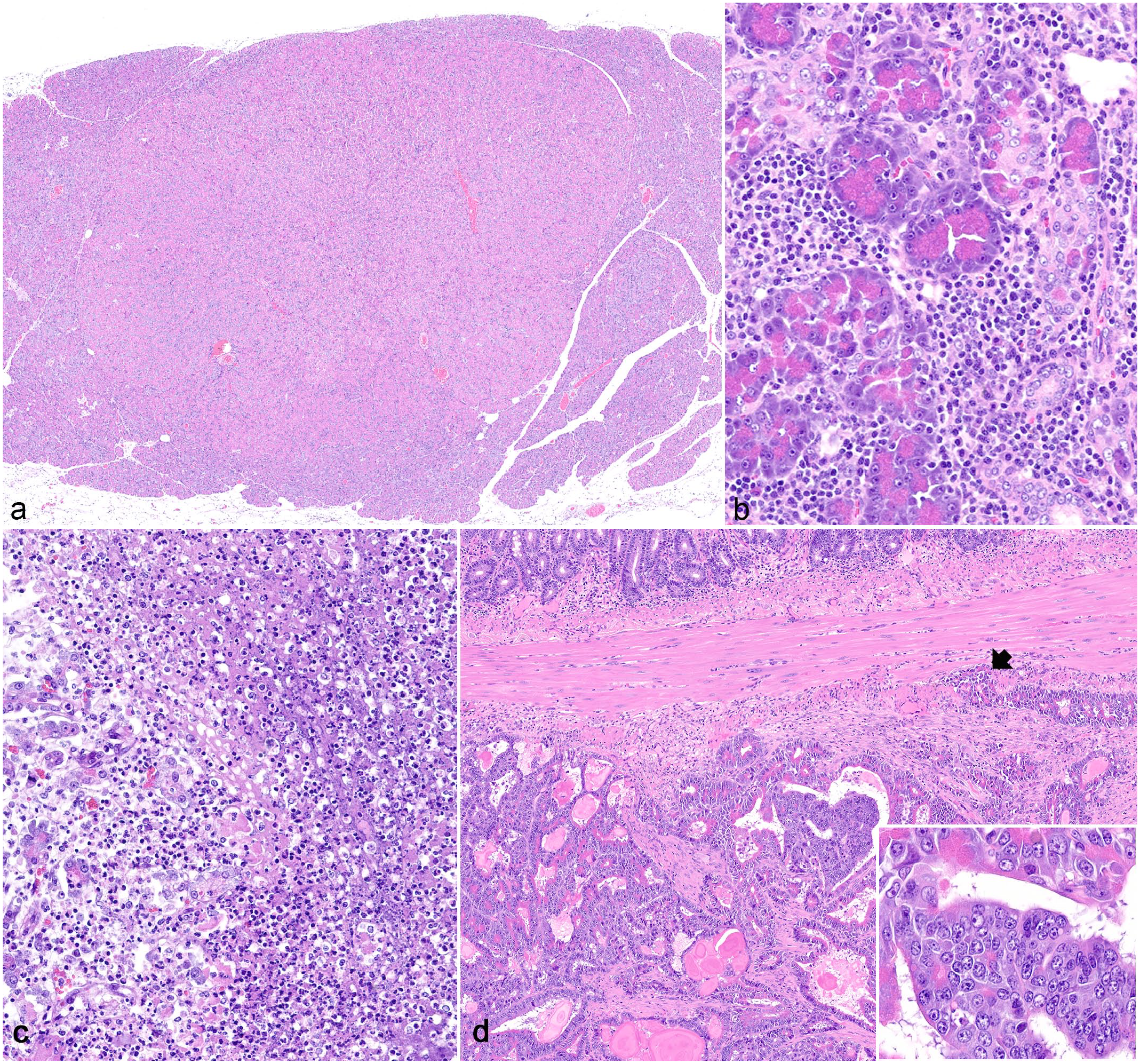
Chronic pancreatitis was identified via biopsy (n = 11), necropsy (n = 4), or unknown sampling methods (n = 1) in 15 pets and 1 zoo animal, including 8 males and 8 females ranging from 4 to 8 years old (mean = 5.4 years). Concurrent diseases present in more than one study animal included inflammatory bowel disease (n = 8), islet cell neoplasia (n = 8), adrenocortical neoplasia (n = 7), and diabetes mellitus (n = 3). Clinical signs reported in more than one study animal include hyperglycemia (n = 3, all cases with diabetes mellitus), hypoglycemia (n = 3, 2 of which had islet cell neoplasia), and high lipase and globulins (n = 2). At surgery or necropsy, nodules were seen in the pancreas of 8 animals, 5 of which had insulinomas in addition to chronic pancreatitis and 3 of which had no other lesions in the pancreas, suggesting the nodules seen grossly were due to chronic pancreatitis. Histologically, clusters and individual lymphocytes and rare plasma cells infiltrated the interstitium and acini, and there were variable amounts of interstitial fibrosis and ductular hyperplasia (Fig. 1B); scattered neutrophils were also present in 3 cases. Cases were classified as mild (less than 5% of acini infiltrated by lymphocytes or clusters of less than 50 lymphocytes; n = 9), moderate (5%-20% of acini infiltrated by lymphocytes or clusters of more than 50 lymphocytes, n = 5), or severe (affected over 20% of acini with or without accompanying fibrosis, n = 2).
Acute pancreatitis was identified via biopsy (n = 2) or necropsy (n = 11) in 12 pets and 1 zoo animal, including 8 males, 2 females, and 3 animals of undetermined sex, and ranged in age from 11 months to 7 years (mean = 4.7 years). Of the 11 necropsy cases, necrotizing pancreatitis was identified as the primary (n = 8) or one of several (n = 3) contributors to mortality. Clinical history was available in 9 cases, 4 of which had abdominal surgery with pancreatic biopsy for pancreatic masses (insulinomas) 8 to 72 hours prior to death. In all cases with prior pancreatic biopsy, neither gross nor histologic lesions of pancreatitis were reported in the initial biopsy. Clinical signs reported in >1 animals in the cohort included lethargy (n = 4) and diarrhea (n = 3). Concurrent diseases present in more than one animal include inflammatory bowel disease (n = 4), islet cell neoplasia (n = 2), adrenocortical neoplasia (n = 2), and membranoproliferative glomerulopathy (n = 2). Gross findings of the pancreas were available in 7 cases and were described as fibrinous or purulent (n = 4), reddened (n = 2), or thickened (n = 1). Histologically, exocrine tissue contained multifocal single-cell necrosis or lytic necrosis characterized by abundant, variably fragmented neutrophils, karyorrhectic cellular debris, edema, and a small amount of hemorrhage and fibrin (Fig. 1C). Regions of lytic necrosis and inflammation extended into the surrounding mesenteric adipose tissue in 8 of 13 cases, 4 of which also underwent saponification characterized by maintenance of adipocyte shape with loss of nuclei, replacement of the cytoplasm by hazy, pale basophilic stippling, and circumferential neutrophils and streams of nuclear debris.
Acinar cell adenoma was identified via biopsy (n = 4) or necropsy (n = 1) in 4 pets and 1 zoo animal, including 2 males, 2 females, and 1 animal of undetermined sex that ranged from 5.5 to 7 years old (mean = 6.6 years). Gross findings were available in 3 cases, all of which described a nodule or mass in the pancreas. There were no reported clinical signs or concurrent diseases shared by 2 or more animals. Histologically, adenomas ranged from 3 to 10 mm in greatest dimension (mean = 6.6 mm) and were composed of uninodular (n = 3) or multinodular (n = 2) masses of exocrine tissue separated from the remaining compressed pancreas by thin fibrous connective tissue capsules, which differentiated them from acinar hyperplasia. Neoplastic cells had variable amounts of zymogen granules, sometimes fewer and sometimes more than the surrounding pancreas. Neoplastic cells had oval nuclei with stippled chromatin and one, small prominent nucleolus. Anisocytosis and anisokaryosis were mild to moderate, with up to 50% variation in cellular and nuclear size. Mitotic count ranged from 2 to 21 per 2.37 mm2 (mean = 7). Few interstitial lymphocytes were seen in one case. Central edema and cystic formation were present in one case.
Acinar cell carcinoma was identified via biopsy (n = 2), necropsy (n = 1), or unknown methods (n = 1) in 4 pets, including 1 male and 3 females that ranged from 2 to 7 years old (mean = 4.5 years). Clinical history was available in 2 cases, neither of which reported signs referable to the neoplasm. Acinar cell carcinoma was identified as the primary contributor to mortality in the one necropsy case. Gross findings were available in 3 cases, 2 of which described a mass in the pancreas and 1 of which described multiple masses in the mesentery. No trends in concurrent diseases were observed. Histologically, carcinomas were composed of invasive moderately cellular neoplasms associated with a dense scirrhous response (Fig. 1D). Carcinomas ranged from 0.8 to 2.1 cm in greatest dimension on histologic section (mean = 1.4 cm). Neoplastic cells formed tubules, acini, and rarely cystic spaces. Neoplastic cells were polygonal with variable amounts of zymogen granules (often less than the surrounding pancreas). Nuclei were oval with stippled chromatin and one, small prominent nucleolus. Anisocytosis and anisokaryosis were mild to marked, with up to a 100% variation in cellular and nuclear size. Mitotic counts ranged from 10 to 53 mitotic figures per 2.37 mm2 (mean = 22.8). Inflammation was minimal to absent. Three cases had hemorrhage and necrosis in the neoplasm. Metastases were identified in 3 cases; 2 cases had mesenteric metastases, one of which had miliary nodules of carcinoma, consistent with carcinomatosis, and 1 case had hepatic metastasis.
The only nonpancreatic neoplasm identified in the pancreas was lymphoma. No other metastatic neoplasms were identified. Lymphoma was identified via biopsy (n = 2) in 2 pets, including an 8-year-old female and 1 animal of unknown sex and age. Lymphoma was present in other sites in both animals, specifically in the lymph nodes (n = 2). There were no reported clinical signs shared by the 2 animals. Histologically, neoplastic lymphocytes were small (nuclei 1-1.5 times the diameter of a red blood cell) in both cases.
Exocrine pancreatic atrophy was identified via biopsy (n = 1) or necropsy (n = 2) in 2 pets and 1 zoo animal, including 2 females and 1 male ranging from 2 to 6 years old (mean = 3.67 years). All 3 animals had a history of weight loss. At surgery or necropsy, one animal had a small pancreas, one had a pancreas covered in fibrin (this animal also had necrosuppurative pancreatitis), and one had no gross lesions reported in the pancreas. Concurrent diseases present in more than one animal include hepatic lipidosis (n = 3) and inflammatory bowel disease (n = 2); one animal also had diabetes mellitus. Histologically, atrophic exocrine cells had reduced size with fewer zymogen granules and cytoplasmic basophilia, and there was variable loss of organized acinar architecture (Fig. 2A). Exocrine cells adjacent to pancreatic islets were less affected.
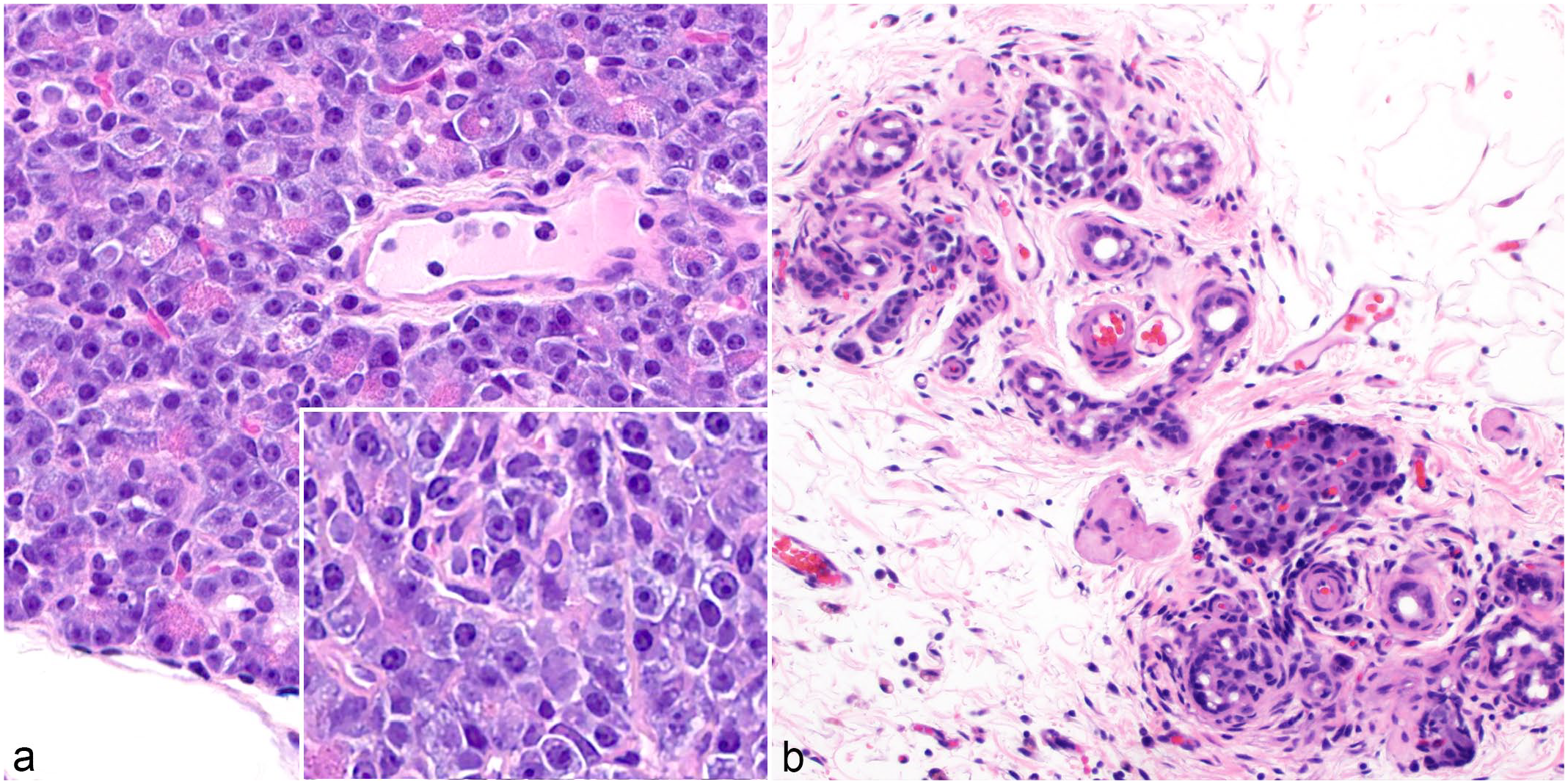
Pancreatic lesions in domestic ferrets. Hematoxylin and eosin. (a) Pancreatic atrophy. Exocrine cells have diminished numbers of zymogen granules. Inset: Higher magnification of atrophic cells. (b) Pancreatic hypoplasia. Acinar cells are absent, with only ductules, interstitial tissue, and a pancreatic islet remaining.
Presumptive pancreatic exocrine hypoplasia was identified via biopsy (n = 2) in 2 pets, both males ranging from 6 to 7 years old (mean = 6.5 years). Both animals had a history of hypoglycemia and islet cell carcinomas (insulinomas) identified at biopsy. Histologically, both cases had pancreatic lobes with complete loss of acinar cells with only ductules and interstitial tissues remaining (Fig. 2B). Scattered exocrine cells were present in adjacent pancreatic lobes, and there was no evidence of fibrosis, inflammation, or ductular ectasia or hyperplasia suggestive of a primary lesion causing acinar loss.
Dry weight zinc concentrations in 4 cases of acute pancreatitis (cases 44, 45, 49, and 55) ranged from 85.56 μg/g (25.21 μg/g wet weight) to 373.61 μg/g (110.10 μg/g wet weight) with an average of 187.12 μg/g (55.14 μg/g wet weight). In one case each of chronic pancreatitis (case 39) and lymphoma (case 69), dry weight zinc concentrations were 25.61 ug/g (7.55 μg/g wet weight) and 158.14 μg/g (46.60 μg/g wet weight), respectively.
Discussion
In this study, acinar cell hyperplasia was the most common histologic finding in the exocrine pancreas. Hyperplastic nodules are frequently removed from patients undergoing lumpectomy or partial pancreatectomy for insulinoma removal, and hyperplastic nodules can be mistaken for islet cell tumors at surgery. Acinar cell hyperplasia is well documented in dogs, cats, and cattle. 34 In one survey-based study regarding neoplasms of pet ferrets in Japan, 5 of 6 nodules identified in the exocrine pancreas were due to acinar cell hyperplasia. 22
Across domestic species, pancreatic acinar cell hyperplasia may be due to consumption of diets rich in protein or energy, 15 regeneration after a previous insult,15,23,34 or incidental aging changes. 10 A prospective study evaluating the pancreas of 101 dogs at necropsy found that acinar cell hyperplasia was correlated with increased age but not with a history of pancreatitis. 23 In our study, cases were limited to archived cases submitted for pancreatic disease and histologically confirmed exocrine lesions, so the prevalence and age distribution of acinar cell hyperplasia across the broader population of ferrets are areas for further research. Five of 16 chronic pancreatitis cases had concurrent acinar cell hyperplasia, suggesting compensatory regeneration after inflammatory insults or necrosis. There were no cases of concurrent acinar cell hyperplasia and exocrine neoplasia (adenoma or carcinoma), which is consistent with the finding in other species that acinar cell hyperplasia is not associated with neoplastic transformation. 23
Chronic and acute pancreatitis were the next most commonly identified lesions in this study. Pancreatitis occurs when there is premature activation of pancreatic pro-enzymes leading to parenchymal and vascular damage and subsequent ischemic necrosis. 15 The most potent of these enzymes is trypsin, which activates the kinin system, coagulation cascade, and complement systems. 15 Severe acute pancreatitis may result in a systemic inflammatory response and multiple organ dysfunction syndrome.15,34 Chronic pancreatitis often follows repeated bouts of acute pancreatitis and is characterized by variable lymphoplasmacytic inflammation with replacement fibrosis. 34
In domestic animal species, acute pancreatitis can be incited by a variety of causes, including pancreatic duct obstruction, abnormal transport of pancreatic enzymes, direct injury to acinar cells (i.e. abdominal trauma, surgical manipulation, or toxins), systemic viral infections, or hypoperfusion. 34 In dogs, acute pancreatitis may be caused by dietary indiscretion or high fat meals, and overweight middle-aged dogs are predisposed. 34 Acute pancreatitis in ferrets has been reported in association with obesity, pancreatic surgery, diabetes mellitus, and systemic coronavirus infection.11,25,32,39 In our study, 4 of 9 (44%) acute pancreatitis cases with clinical histories available had a history of pancreatic biopsy within 3 days of death. None of the biopsy cases reported gross or histologic evidence of pancreatitis at the initial surgery. This suggests that acute pancreatitis is a possible sequela to pancreatic biopsy in ferrets.
Another reported cause of acute pancreatitis in ferrets is ferret systemic coronavirus, which resembles the granulomatous (dry) form of feline infectious peritonitis. The ferret disease is characterized by systemic, often vasocentric, granulomatous inflammation on the surface and within the parenchyma of multiple organs, including the liver, kidney, spleen, lung, brain, and pancreas.11,16,20 This progressive, rapidly fatal disease generally affects animals under 1 year of age.11,20 In an initial case series describing ferret systemic coronavirus-related lesions, 3 of 23 reported cases featured pancreatitis. 11 In another report of a 1-year-old female spayed ferret, the primary organ affected was the pancreas, with acute suppurative pancreatitis, peripancreatic fibrosis, and necrotizing mesenteric steatitis. 39 We did not identify any cases consistent with ferret systemic coronavirus-associated pancreatitis in our study.
In our cases of chronic pancreatitis, several common comorbidities were identified, including insulinoma, inflammatory bowel disease, adrenocortical tumor, and diabetes mellitus. Insulinomas and adrenocortical neoplasms are extremely common in ferrets, together accounting for 38% and 60% of all ferret neoplasms in 2 large studies of ferrets.3,18 Given the high incidence of these lesions and the fact that insulinoma excision is a common reason for submission of pancreatic tissues for histologic examination, the authors cannot speculate on the correlation between these common comorbidities in this study.
Similarly, inflammatory bowel disease is a common lesion in ferrets, with one study demonstrating an incidence of 92% in all ferret necropsy specimens. 19 Inflammatory bowel disease is characterized by chronic lymphoplasmacytic inflammation in the stomach and intestine and is often considered a diagnosis of exclusion after other enteric diseases have been ruled out, such as food intolerance or infectious diseases (i.e. Helicobacter mustelae).19,33 In our study, 8 of 16 chronic pancreatitis cases also had inflammatory bowel disease. Cats are also prone to developing both inflammatory bowel disease and pancreatitis. 6 A relatively short duodenum and common duodenal entrance for the bile and pancreatic ducts is thought to predispose cats to duct reflux, ascending bacterial infections, and resultant pancreatitis.6,34 Ferrets share these anatomic features, so a link between inflammatory bowel disease and pancreatitis may exist in this species as well.9,26
In our study, 3 ferrets with chronic pancreatitis had concurrent diabetes mellitus. Diabetes mellitus is uncommonly diagnosed in ferrets and is usually a transient condition secondary to pancreatectomy. 25 To the authors’ knowledge, concurrent pancreatitis and diabetes mellitus in the ferret has only been reported in 2 individual case reports. Acute pancreatitis was reported in an obese, 5.5-year-old, female spayed ferret with concurrent diabetic ketoacidosis, pancreatic insulinoma, and hyperadrenocorticism. 25 A separate case report described diabetes mellitus in a 6-year-old, neutered male ferret with chronic pancreatitis and hyperadrenocorticism. 5 In these reports, it was not possible to determine if diabetes mellitus caused the pancreatitis through the toxic effect of hyperlipidemia, if pancreatitis-mediated loss of islet cells caused the diabetes mellitus, or if hyperadrenocorticism caused steroid-induced insulin resistance and subsequent diabetes mellitus.5,25 In our study, only one ferret with diabetes mellitus had a reported adrenal lesion (not further characterized); the remaining 2 cases may indicate an association between pancreatitis and diabetes mellitus in ferrets.
Acinar cell adenoma was diagnosed in only 5 of the 69 (7%) ferrets with exocrine pancreatic lesions in this study. In all species, acinar cell adenomas are considered rare incidental findings, which is consistent with our study.10,34 In ferrets, acinar cell carcinomas are considered more common than adenomas.3,4,10 In our study, however, adenomas (n = 5) were slightly more common than carcinomas (n = 4). A similar finding was found in a study of 574 neoplasms of ferrets, where 6 of 10 exocrine pancreatic neoplasms were adenomas. 18 Adenomas may be underrepresented in previous literature because differentiation from hyperplasia requires evaluating the relationship to surrounding tissue (i.e. presence or lack of surrounding connective tissue capsule), which may not be apparent if the neoplasm is narrowly or incompletely excised. 15 In addition, carcinomas may be more commonly sampled due to rapid spread and disease progression, which trigger surgical biopsy or necropsy.
In ferrets, acinar cell carcinomas are aggressive and infiltrative neoplasms that spread through carcinomatosis or, less commonly, via metastasis to lymph nodes, abdominal organs, or the lungs.13,17,32,37,38 Metastasis or local extension typically occurs prior to diagnosis.37,38 These findings were confirmed in our study, in which 3 of 4 cases had metastasized, including to the liver and abdominal cavity (carcinomatosis). Previous case reports in ferrets have described osseous metaplasia, extrahepatic biliary obstruction, or mastocytic peritoneal effusion in acinar cell carcinomas;17,28,37 none of these were observed in our cases.
Lymphoma was the only multicentric neoplasm identified in the pancreas in this study. Lymphoma is the most common neoplasm of the hematopoietic and alimentary systems and the third most common neoplasm overall in ferrets. 10 All of our cases were presumed multicentric with lymph node involvement. Lymphoma of the mesenteric lymph nodes is a commonly reported finding in ferrets. In a study of 29 cases of lymphoma in ferrets, 28 had abdominal visceral involvement, and 25 had mesenteric lymph node involvement. 1
Atrophy and presumptive hypoplasia were the least commonly diagnosed lesions of the exocrine pancreas in this study, with 3 (4%) and 2 (3%) cases, respectively. Negative energy balance, protein deficiency, and nutrient deficiency (i.e. selenium, zinc, or copper) are reported causes of acinar cell atrophy in veterinary species. 15 In our study, all 3 cases of acinar cell atrophy had a history of weight loss and concurrent hepatic lipidosis, indicating negative energy balance in these patients. While negative energy balance could be the cause of atrophy in these cases, it is also possible that pancreatic atrophy caused these clinical signs through exocrine insufficiency and maldigestion. 14 Acinar cell atrophy can also be caused by pancreatic duct obstruction, which is evident histologically by the presence of ductular hyperplasia and retrograde ductal ectasia, and a case report in a ferret described pancreatic adenocarcinoma causing bile duct obstruction, interstitial fibrosis, and acinar cell atrophy.15,28 In our study, one case had concurrent chronic pancreatitis and ductular hyperplasia. In this case, inflammation and fibrosis may have led to ductal obstruction and acinar cell atrophy.
Presumptive exocrine pancreatic hypoplasia was diagnosed in 2 cases. Differentiation of exocrine pancreatic atrophy and primary hypoplasia is characteristically difficult (juvenile pancreatic atrophy in dogs, for instance, was long thought to be due to pancreatic hypoplasia before underlying transient lymphocytic pancreatitis was identified). While we cannot completely rule out a primary cause, our 2 cases lacked histologic features that would indicate acinar destruction and loss during a previous episode of pancreatitis (i.e. significant fibrosis, duct hyperplasia, duct ectasia, or islet loss), thus we categorized these cases as presumed hypoplasia. Exocrine hypoplasia has not been previously reported in ferrets and has rarely been reported in other species.15,27 In calves, pancreatic hypoplasia is limited to the exocrine tissue, with pancreatic ducts and islets unaffected, similar to our cases. 15 The only reported clinical sign in our 2 cases was hypoglycemia, likely due to concurrent insulinomas, suggesting that pancreatic hypoplasia was subclinical.
A normal range for ferret pancreatic zinc concentrations is not available in the published literature; however, a study on dietary zinc tolerance in mink, a related species, demonstrated wet concentrations between 27 and 111 μg/g in animals with no clinical or histologic evidence of pancreatic injury. None of the ferrets in our report had zinc levels above this interval, making it unlikely that zinc excess played a role in the development of pancreatitis in our study. In all but 2 cases, zinc levels were above the lower range of this reference interval, indicative of adequate zinc concentrations. One case of chronic pancreatitis and one case of acute pancreatitis had levels slightly lower than the mink range. In mice and rats, zinc deficiency has been associated with increased inflammation, possibly due to diminished antioxidant activity, and zinc supplementation can be protective against pancreatitis. 24 Whether zinc deficiency played a role in the pathogenesis of pancreatitis in these cases is unknown.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241266943 – Supplemental material for Review of spontaneous lesions in the exocrine pancreas of domestic ferrets (Mustela furo)
Supplemental material, sj-pdf-1-vet-10.1177_03009858241266943 for Review of spontaneous lesions in the exocrine pancreas of domestic ferrets (Mustela furo) by Sarah B. Sulkosky, Michael M. Garner, Mark Burgess, Bruce H. Williams and Elise E. B. LaDouceur in Veterinary Pathology
Footnotes
Acknowledgements
Thank you to Rosy Canfield of the Joint Pathology Center for assistance with an archival search. The authors also thank Roy Brown of Histology Consulting Service for slide preparation, Cathy Minogue and Christie Buie of Northwest ZooPath for archival data retrieval, and Dr. Birgit Puschner of Michigan State University for assistance with zinc testing and interpretation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The views expressed in this abstract are those of the authors and do not necessarily reflect the official policy of the Department of Defense or U.S. Government.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
