Abstract
Alternative therapies that can help achieve complete remission in dogs with lymphoma include total body irradiation and hematopoietic cell transplant, though there are few reports describing successes and pathologic sequelae of these procedures. During a 10-year period, 94 dogs with multicentric lymphoma received a hematopoietic cell transplant following total body irradiation at North Carolina State University College of Veterinary Medicine. Seven of these 94 dogs (7%) died prior to discharge, five (5%) of which presented for postmortem examination. Of these dogs, four received an autologous hematopoietic cell transplant, while one received a haploidentical allogeneic hematopoietic cell transplant. All five dogs had bone marrow depletion with all hematopoietic lines affected. Three had systemic candidiasis, while two had bacterial infections. To the authors’ knowledge, this is the first report to document pathologic findings and development of systemic mycoses in dogs post total-body irradiation therapy and hematopoietic cell transplant.
Lymphoma is the most common hematopoietic malignancy diagnosed in dogs. It typically affects middle-aged, large breeds with an incidence of 25 per 100,000 dogs. 3 Of the various forms of lymphoma, multicentric lymphoma is most common. Multicentric lymphoma denotes disease affecting multiple lymph nodes throughout the body with or without visceral organ involvement. 27 There are a wide variety of chemotherapy treatment options for canine multicentric lymphoma, which can involve single agent or multiagent protocols, such as CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone).23,28 While lymphoma is treatable, complete remission is short-lived or unattainable in many cases, leading to the need and use of multimodal approaches. Total body irradiation (TBI) is a supplemental modality that has been used in dogs to treat hematopoietic malignancies and autoimmune diseases. 12 Half-body and TBI in conjunction with chemotherapy is reported to extend dogs’ lives beyond chemotherapy alone.20,23 However, the efficacy of TBI and chemotherapy is limited by gastrointestinal, pulmonary, and bone marrow toxicities, so other treatments are desired.18,19,26
In human medicine, hematopoietic cell transplant (HCT) is used to treat a wide variety of hematopoietic malignancies, including lymphoma. The three most common types of HCT procedures include autologous peripheral blood HCT (autoHCT), identical allogeneic peripheral blood HCT (alloHCT), and haploidentical alloHCT.2,17 AutoHCT utilizes hematopoietic CD34+ cells isolated from the lymphoma patient, while identical alloHCT utilizes human leukocyte antigen-matched donor CD34+ cells. When a perfect human leukocyte antigen-matched donor cannot be identified, a haploidentical alloHCT may be considered, in which one of the donors MHC I or II genes is mismatched with the recipient. Once the CD34+ hematopoietic progenitor cells are isolated from the donor, they are transplanted into the patient.24,25
Canine HCT has been used as a preclinical model of human HCT, investigating many clinical pitfalls in human transplant medicine, including regimen-related toxicities, post-HCT relapse, and graft-vs-host disease.13,14 Hematopoietic cell transplant has been shown to extend disease-free survival times of dogs with high-grade B- and T-cell lymphoma compared to chemotherapy and TBI alone.1,8,19,24,25 In one study, 9/38 dogs (24%) with multicentric lymphoma that underwent an autoHCT had a median overall survival of 31 months. 1 Another study on alloHCT reported 8/9 dogs (89%) with multicentric B-cell lymphoma that received this treatment had a median survival time greater than 48 months. 9 This is compared to the 8- to 12-month average survival time achieved with multidrug chemotherapy protocols.18,28 Total body irradiation is often implemented before HCT, and has been shown to improve engraftment by eradicating host’s diseased bone marrow before transplantation. 14 Potential post-HCT complications, some of which may be attributed to myeloablative effects of TBI, include sepsis, hemorrhage, engraftment failure, or relapse. 28 While there is veterinary literature describing HCT in dogs, most focus on the clinical aspect, and few reports on the pathologic features are available. This report aims to characterize gross and histologic changes observed in canine lymphoma patients who died or were euthanized post-HCT.
North Carolina State University College of Veterinary Medicine is one of the few veterinary institutions that offers HCT as a therapy to treat hematopoietic malignancies, such as lymphoma, in dogs. Between March 2009 and November 2019, 94 dogs received HCT, with 87 dogs successfully treated and discharged. Seven of the 94 dogs (7%) died naturally or were euthanized prior to discharge. Of those that died, 5 (5/94, 5%) were submitted to the pathology service for postmortem examination.
All 5 dogs were diagnosed with high-grade B-cell multicentric lymphoma and had finished receiving a multiagent CHOP-based chemotherapy protocol about 2 to 3 weeks before HCT. Dogs receiving an autoHCT (4 of the 5 dogs) received high-dose cyclophosphamide (500-750mg/m2) 2 weeks before arrival. Once in hospital, stem cell mobilization was accomplished using twice-daily subcutaneous filgrastim (5 μg/kg, Neupogen, Amgen) injections for 5 days. On day 5, the dogs received 10 μg/kg filgrastim in the morning, followed by mononuclear cell collections and CD34+ cell enumerations that day. Two, 6 Gy doses of TBI were administered on days 6 and 7 for a total of 10 to 12 Gy, similar to previously reported TBI methods.24,25 Harvested mononuclear cells were infused immediately after TBI on day 7. One of the five dogs received a haploidentical alloHCT, which utilized CD34+ cells isolated from a DLA-matched donor dog whose DLA-88, DRB1, DQA1, and DAB1 MHC loci were matched at 7/8 alleles. In this setting, the donor dog’s stem cells were mobilized and collected as described above. The recipient dog received 8 Gy TBI before infusion of the donor dog’s mononuclear cells on day 7.
Post-HCT, all dogs were treated with a variety of antibiotics, antiemetics, and fluid therapy, similar to a previously documented regimen.24,25 All dogs experienced temporary grade IV neutropenia and thrombocytopenia, as has been previously reported post-HCT. 7 These five dogs declined clinically post-HCT, resulting in the death of 4/5 cases and humane euthanasia of one dog 8 to 12 days after the HCT procedure.
All organ systems were grossly evaluated by a pathology trainee and board-certified anatomic pathologist within 24 hours of death. At necropsy, 4/5 dogs had a serosanguinous to hemorrhagic effusion in the thoracic and abdominal cavities. Common findings included petechiae and ecchymoses of the costal pleura, pericardium, diaphragm, and lung (Table 1). Representative sections of each major organ system and gross lesions were evaluated histologically. Histologic lesions attributed to radiation toxicity included dilated crypts with loss or attenuation of enterocytes in the small intestine (3/5), hematopoietic tissue depletion of the bone marrow (5/5), diffuse alveolar damage (1/5), dermal/pannicular necrosis (1/5), saponification of peripancreatic fat (1/5), and cystitis with hemorrhage (1/5) (Fig. 1). Interestingly, small intestinal crypts displayed signs of epithelial injury but lacked numerous inflammatory cells, possibly owing to the neutropenia in these patients. Additional histologic findings considered unrelated to treatment included glomerulosclerosis (1/5) and hepatocellular glycogen-type vacuolation (1/5).
Gross postmortem and histopathological findings in 5 dogs with B-cell lymphoma treated with hematopoietic cell transplantation (HCT).
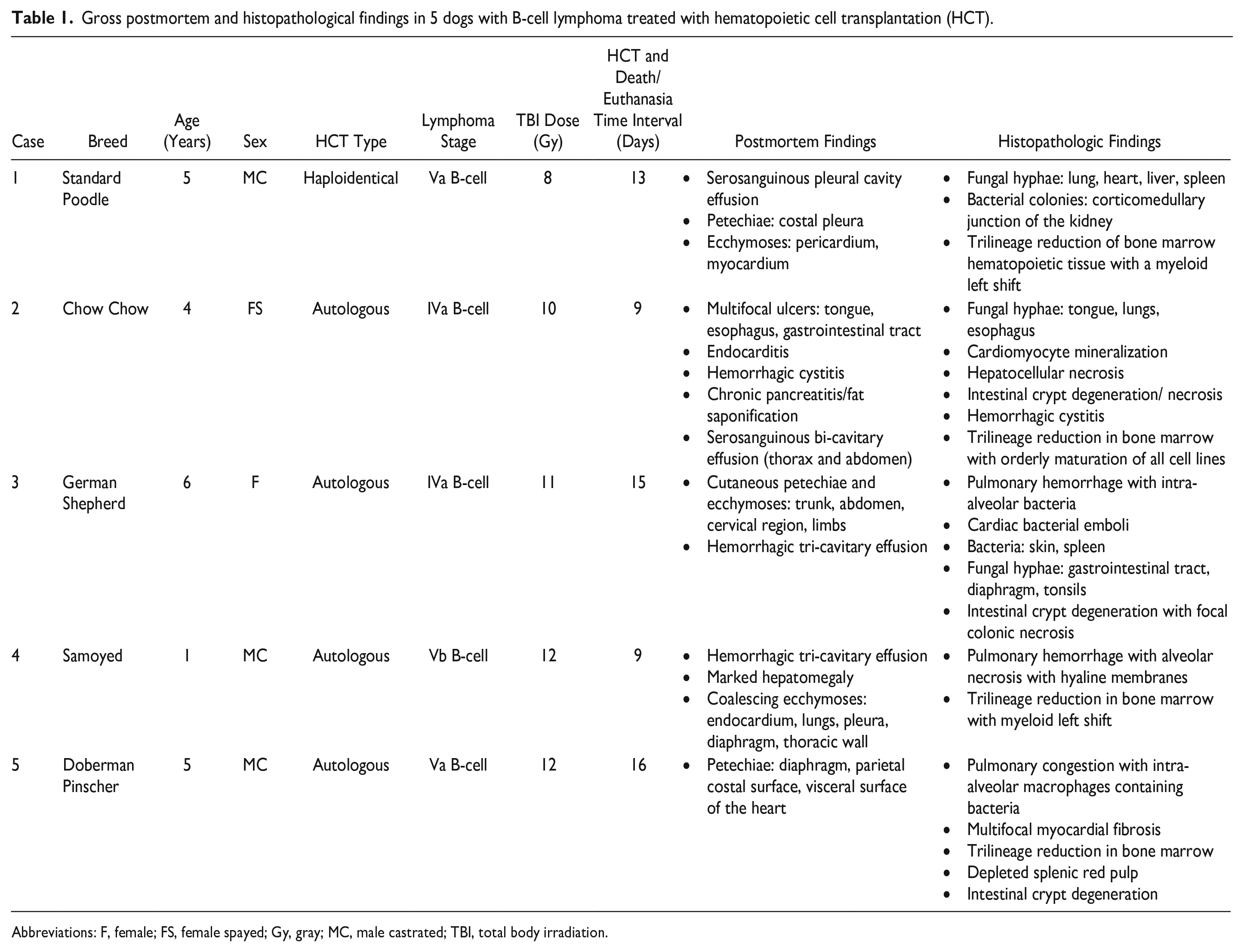
Abbreviations: F, female; FS, female spayed; Gy, gray; MC, male castrated; TBI, total body irradiation.
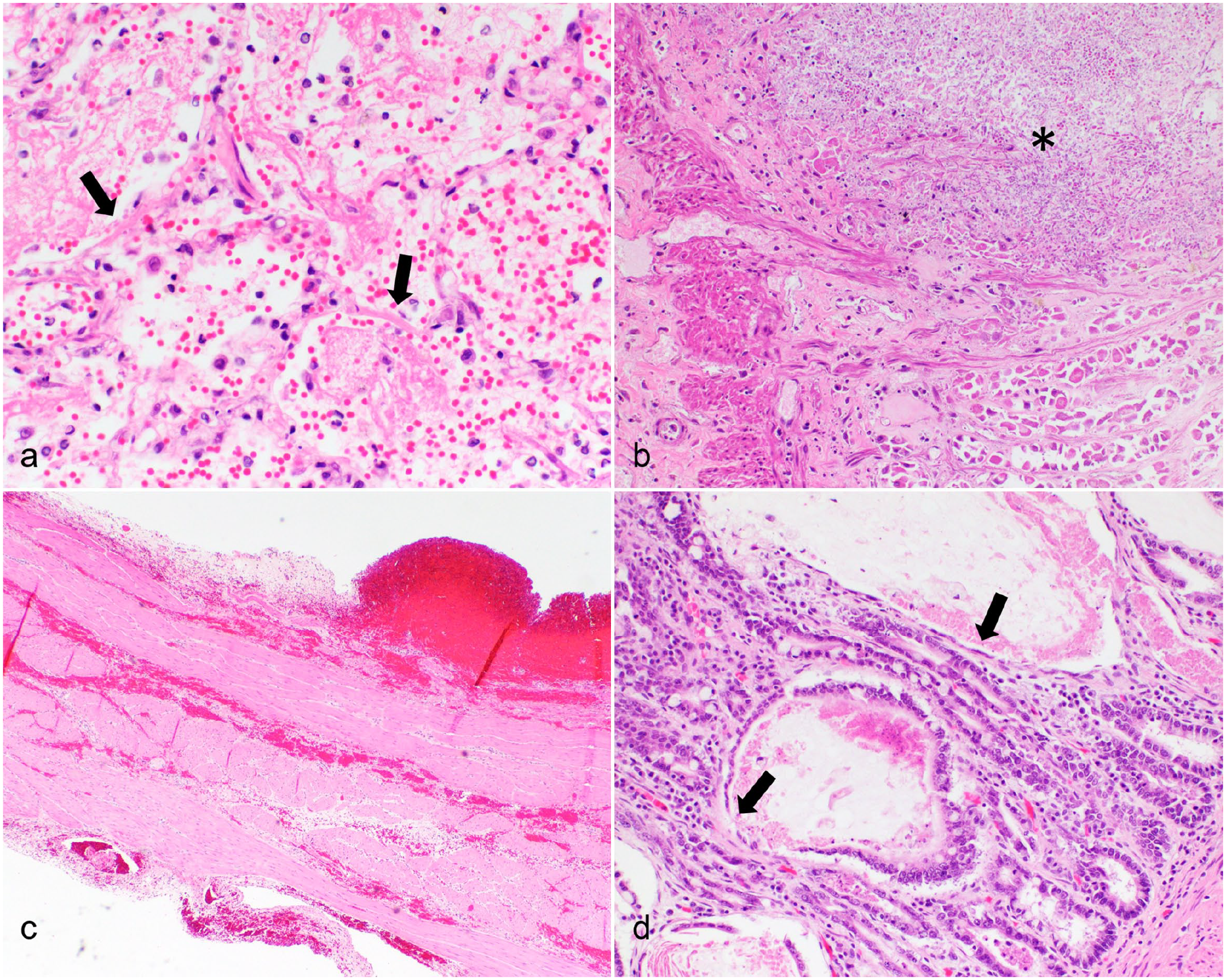
Histopathologic features of total body irradiation toxicity in dogs with B-cell multicentric lymphoma. Hematoxylin and eosin. (a) Lung. There is diffuse alveolar injury with hyaline membranes (arrows), fibrin exudation, and intra-alveolar hemorrhage. Case 1. (b) Stomach. There is coagulative necrosis of the gastric mucosa adjacent to an ulcer filled with numerous fungal organisms (asterisk). Case 3. (c) Bladder. There is transmural interstitial hemorrhage of the bladder wall, which is considered secondary to thrombocytopenia. Case 2. (d) Small intestine. Crypts are dilated, contain cellular debris, and are multifocally lined by necrotic to attenuated epithelium (arrows). The lack of neutrophilic infiltrates may reflect neutropenia. Case 2.
Three of the 5 dogs had histologic evidence of multisystemic fungal infection. Fungal morphologies ranged from 3 to 5 µm diameter ovoid yeast, sometimes forming short chains (pseudohyphae), to 2 to 4 µm wide parallel-wall hyphae with acute-angle branching, consistent with candidiasis. Organs affected included the tongue/esophagus (1/5), liver (2/5), lungs (2/5), and heart (2/5). In 2/3 cases, fungal cultures yielded Candida albicans and Candida tropicalis; the third case was putatively diagnosed with candidiasis based on fungal histomorphology. Bacterial infection was also evident, with colonies of gram-positive cocci present in alveolar spaces in 3/5 dogs. Emboli of gram-positive cocci were present in the lung, kidney, heart, and spleen of one dog and the kidney of another. One dog had both fungi and bacteria present in the lungs; gram-positive cocci were present in alveolar spaces and within interstitial capillaries (Supplemental Figure S1). When fungi or bacteria were present, there was minimal macrophagic inflammation or complete lack of an inflammatory response. Only one dog did not have fungal or bacterial organisms noted in any organ.
Immunohistochemistry (Supplemental Table S1) for CD3 (T-cells, rabbit polyclonal Dako A0452), Pax5 (B-cells, mouse monoclonal Dako M7037), MUM1 (plasma cells, mouse monoclonal Dako M7259), and Iba-1 (histiocytes, rabbit Wako 19-19741) was performed on sections of heart (case 1), lung (case 2), and liver (case 3) with histologic evidence of fungal colonies to characterize the host response to fungal infection (Fig. 2). Normal canine lymph nodes were used as a positive control, while negative controls did not have the primary antibody applied to the slide. Immunohistochemistry did not identify any MUM1 or Pax5-positive B-cells surrounding fungal colonies in the tissues examined. Rare CD3+ T-lymphocytes and Iba-1+ histiocytes were present at the periphery of fungal colonies.
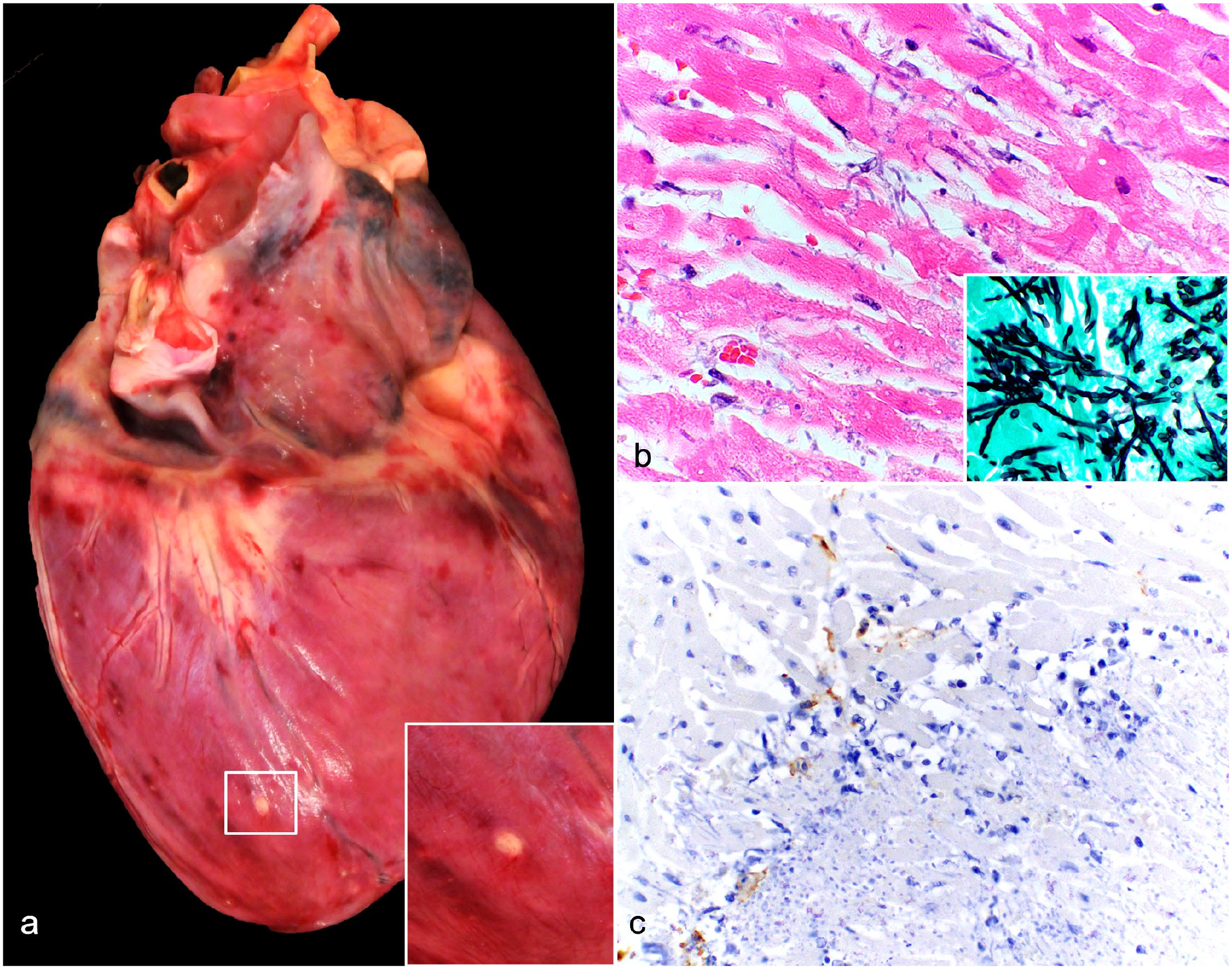
Gross and histopathological features of fungal infection without the presence of inflammation following total body irradiation. Dog, case 1. (a) Heart. There are epicardial ecchymoses and a white to tan, well-circumscribed nodule (white box) toward the apex of the heart. Inset: higher magnification of the white to tan epicardial nodule. (b) The myocardium in the area of the nodule in (a) infiltrated by numerous fungal hyphae without inflammation. Hematoxylin and eosin. Inset: Grocott methenamine silver highlights yeast and pseudohyphae characteristic of Candida species. (c) Mats of fungi are associated with rare immunolabeled cells (macrophages), but lack mature granuloma formation, indicating an inadequate immune response to fungal pathogens. Iba-1 immunohistochemistry.
While prolonged survival times can be achieved in a subset of patients undergoing TBI/HCT, the effects of treatment, particularly due to TBI, can be a double-edged sword. The mortality rate associated with TBI/HCT is variable due to patient response to therapy, with graft-vs-host disease and radiation toxicity being the main contributing factors.9,13 Moreover, patients presenting for HCT can be clinically challenging cases, many having advanced disease despite having undergone various antineoplastic therapies, including chemotherapy and radiation. For some dogs with lymphoma, HCT is a chance to achieve complete remission. However, the myeloablative effects of TBI, a component of most HCT protocols, can result in life-threatening conditions, including sepsis and thromboembolic disease.3,13 These sequelae can pose greater risk to dogs with already dysregulated immune systems, as is the case in dogs with lymphoma.
Aside from immunosuppression, radiation effects on epithelia, leading to gastrointestinal ulceration within the gastrointestinal tract, can contribute to morbidity and predispose to opportunistic infections. 15 In the cases of systemic fungal and bacterial infections in this study, the infections most commonly occurred in the alimentary tract and respiratory tract, respectively. Given that these are both epithelial lined organ systems, it follows that deleterious effects of TBI on epithelia may contribute to colonization by opportunistic pathogens and allow for microbial translocation into the systemic vasculature. Other histologic changes observed, namely, small intestinal crypt dilation with enterocyte injury, diffuse alveolar damage, and dermal/pannicular necrosis, further support some degree of epithelial radiotoxicity in these patients.1,4,12,15,16,23 Once these epithelial barriers are breached, the body relies on the immune system to eliminate pathogens.
As previously stated, TBI will adversely affect immune responses due to bone marrow toxicity and subsequent pancytopenia. Other causes for pancytopenia in patients that have undergone TBI and HCT include failure of engraftment, slow recovery, and/or disease relapse. 13 Dogs that have undergone TBI and HCT typically have depleted bone marrow due to radiation toxicity, but should rebound in the following days to months. 20 On average, it takes approximately 3 weeks for hematocrits to recover to baseline, 15 days for neutrophils to reach reference range values, and several months for platelet counts to recover to normal limits post-TBI. 7 In the 5 cases of this report, there was an initial mobilization of myeloid cells, but the number of myeloid cells quickly declined approximately 3 days post HCT/TBI (Supplemental Table S2). Given that 4/5 dogs received autologous-HCT, graft-vs-host disease is not considered a likely cause for the decline in blood cells and platelets, and radiation-induced myeloablation is favored. 28
Impairment of the bone marrow’s ability to produce mature leukocytes significantly dampens the immune system so the host is unable to mount an appropriate immune response to pathogenic stimuli. 28 In this case series, 4 of 94 (4%) dogs with lymphoma undergoing HCT and TBI were diagnosed with systemic mycoses and/or bacterial infections with minimal host responses, as evidenced by the minimal leukocyte infiltration of infected tissues. Candida spp., including C albicans and C tropicalis, have been documented as causes of opportunistic systemic mycoses in human patients undergoing TBI.5,21 Similarly, TBI in dogs has been shown to cause opportunistic cutaneous fungal infections. 11 However, systemic mycosis in dogs after TBI has not yet been reported.
An adequate immune response to candidiasis and bacterial infections requires functional neutrophils.10,27 The neutropenia observed in these cases likely contributed to colonization by these opportunistic pathogens. Moreover, the minimal T-lymphocytic and histiocytic infiltration, identified immunohistochemically, and lack of mature granuloma formation suggest an ineffective immune response toward fungal pathogens. To reduce the possibility of bacterial sepsis, prophylactic wide-spectrum antibiotic therapy was given to these dogs. Altering TBI doses and increasing fractionation or utilizing radiation-free myeloablation therapies, such as busulfan, could also be considered to minimize radiation toxicity.6,13,22
This case series discusses and provides valuable insight into gross and histologic complications associated with TBI and HCT therapy in five dogs with multicentric lymphoma. All five dogs exhibited severe pancytopenia, which subsequently reduced their ability to resist opportunistic bacterial and fungal infections. Three dogs developed systemic mycoses. The occurrence of such infections in dogs undergoing TBI and HCT is rare and has not been previously reported. Although canine HCT is in its infancy, it is imperative to understand and document the potential pathologic side effects of TBI, since it is the main myeloablative procedure in both auto- and alloHCT. This knowledge can help improve the outcomes of canine HCT, reducing the incidence of secondary infections, mortality, and enhancing the overall success of the procedure.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241249114 – Supplemental material for Postmortem pathologic findings in dogs that underwent total body irradiation and hematopoietic cell transplant: A case series of five dogs with B-cell multicentric lymphoma
Supplemental material, sj-pdf-1-vet-10.1177_03009858241249114 for Postmortem pathologic findings in dogs that underwent total body irradiation and hematopoietic cell transplant: A case series of five dogs with B-cell multicentric lymphoma by William Benedict, Steven Suter and Danielle Meritet in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
