Abstract
Between September and November 2021, 5 snow leopards (Panthera uncia) and 1 lion (Panthera leo) were naturally infected with severe acute respiratory coronavirus 2 (SARS-CoV-2) and developed progressive respiratory disease that resulted in death. Severe acute respiratory syndrome coronavirus 2 sequencing identified the delta variant in all cases sequenced, which was the predominant human variant at that time. The time between initial clinical signs and death ranged from 3 to 45 days. Gross lesions in all 6 cats included nasal turbinate hyperemia with purulent discharge and marked pulmonary edema. Ulcerative tracheitis and bronchitis were noted in 4 cases. Histologically, there was necrotizing and ulcerative rhinotracheitis and bronchitis with fibrinocellular exudates and fibrinosuppurative to pyogranulomatous bronchopneumonia. The 4 cats that survived longer than 8 days had fungal abscesses. Concurrent bacteria were noted in 4 cases, including those with more acute disease courses. Severe acute respiratory syndrome coronavirus 2 was detected by in situ hybridization using probes against SARS-CoV-2 spike and nucleocapsid genes and by immunohistochemistry. Viral nucleic acid and protein were variably localized to mucosal and glandular epithelial cells, pneumocytes, macrophages, and fibrinocellular debris. Based on established criteria, SARS-CoV-2 was considered a contributing cause of death in all 6 cats. While mild clinical infections are more common, these findings suggest that some SARS-CoV-2 variants may cause more severe disease and that snow leopards may be more severely affected than other felids.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is associated with a wide range of symptoms in people, from mild illness to severe pulmonary disease that can include death or long-term health complications.12,33,60 Severe acute respiratory syndrome coronavirus 2 is also zoonotic and is capable of infecting a wide range of mammalian hosts. Natural and experimental SARS-CoV-2 infections in animals, including domestic felids, ferrets, rodents, nonhuman primates, and mink, vary widely in terms of disease severity, pathologic findings, and virus localization.9,10,21,35,39,43,44,49 With ongoing human infections, animals will continue to be exposed to SARS-CoV-2 through spillback. Therefore, understanding disease manifestations and progression in zoo species is necessary to improve clinical treatment and outcomes in these rare and sometimes endangered species. Nondomestic felids including tigers (Panthera tigris), snow leopards (Panthera uncia), and lions (Panthera leo), which are common in zoo collections, are susceptible to severe, acute respiratory infection with SARS-CoV-2 and were among the first confirmed natural animal infections in the United States.5,34
In reports of SARS-CoV-2 infections in nondomestic felids, transmission from human caretakers is considered a common route of infection, and SARS-CoV-2 variants sequenced from nondomestic felid cases are typically most closely related to the prevalent, local, human strains.14,26,34,36,40 In case reports with multiple animals infected and in experimental infections, cat-to-cat transmission has also been observed, and infections in socially housed species, such as snow leopards and lions, are more common than in individually housed animals.5,16,20,23,26,36,49
Among cases in nondomestic felids, lions, tigers, and snow leopards are reported to have similar, progressive clinical signs, including both upper respiratory disease and pneumonia.5,26,40 In animals that eventually recovered from SARS-CoV-2-associated disease, respiratory signs were associated with neutrophilic and histiocytic inflammation and degenerate epithelial cells on tracheal wash cytology and broncho-interstitial patterns and peripheral lung consolidation on radiographs.5,34 Some of these nondomestic felids also developed clinical gastrointestinal disease evidenced by vomiting and diarrhea. 5
In domestic cat experimental models, there is a shorter duration of viral RNA recovery (up to 2 weeks) than in nondomestic felids.5,16,49,57 Oral, nasal, intratracheal, and ocular inoculation all result in productive infections and viral shedding, and multiple routes of inoculation were commonly used in these studies to ensure infection.16,35,43 Severe acute respiratory syndrome coronavirus 2-associated disease is more severe in young cats. Direct inoculation into the lower trachea of young cats produces clinical and histological disease, including both acute respiratory infection and chronic airway remodeling.8,47 Domestic cat-to-cat transmission has been produced in experimental models via co-housing of cats inoculated with SARS-CoV-2 and uninfected sentinel cats. Sentinel cats had similar or less-severe disease than cats infected by direct inoculation.4,8,23,49 In studies of shelter cats, seropositivity was low, even when exposed to SARS-CoV-2-positive caretakers. 11 However, surveillance testing associated viral exposure with clinical signs in 48% of cases of pet cats and dogs exposed to humans diagnosed with SARS-CoV-2 infection.30,54 Factors including animal handling, exposure to respiratory droplets or contaminated fomites, aerosolization of feces during cleaning, animal age, and social housing may affect the duration and severity of disease in both natural and experimental felid SARS-CoV-2 infections.5,11,23,36
Nondomestic felids in zoos appear to represent a disproportionately high number of natural SARS-CoV-2 animal infections and in many cases have had more severe clinical disease with prolonged shedding in comparison with other species, including domestic cats.5,14,16,17,35,57 Nondomestic felid cases report prolonged, postinfection nasal (up to 7 weeks) and fecal (more than 4 weeks) shedding of viral RNA by reverse transcription quantitative polymerase chain reaction (PCR).5,26,57 Anti-SARS-CoV-2 neutralizing antibodies have been detected 4 months post initial detection in lions.5,14 To better understand the pathogenesis of these infections, we describe the gross and histologic lesions in 6 nondomestic felids, 5 snow leopards and 1 African lion, which died of respiratory disease after being naturally infected with SARS-CoV-2.
Materials and Methods
Case Descriptions
Five snow leopards (cases 1, 3, 4, 5, 6) and 1 lion (case 2) was housed at 4 different U.S. zoological institutions (A-D). The lion and 3 snow leopards were male; the other 2 snow leopards were female. Ages ranged from 2.5 to 14 years old.
Biosecurity for zoo A included the use of surgical masks in all felid areas (indoor and outdoor) prior to the index case. Zoo B required surgical masks to be used by zoo staff around all feline and primate species. At the time of the case, public visitors to the zoo were not required to wear any type of facemask while visiting the outdoor facility. At zoo C, a minimum distance of 6 feet was maintained between all members of the public and the animal enclosures, including outdoor spaces, by plexiglass barriers. All staff had daily temperature checks, were fully vaccinated against COVID-19, or underwent weekly testing and were required to wear an N95 mask or 2 surgical masks before entering a shared space with SARS-CoV-2-sensitive taxa (felids, chiropterans, primates, and mustelids). In addition, staff used footbaths during animal containment area entry and exit. At zoo D, all zoo staff were required to wear N95 facemasks and nitrile gloves when servicing felid exhibits and during food preparation. At the time of the case, public visitors to the zoo were required to wear a mask while visiting the indoor exhibits, but there was no mask requirement for the outdoor exhibit. A minimum distance of 6 feet was maintained between all members of the public and animal enclosures, including the outdoor space. Some animals had additional diagnostic testing including PCR for Chlamydia felis, feline calicivirus, feline herpes virus 1, Bordetella bronchiseptica, H7N2 influenza virus, influenza A virus, and Mycoplasma felis; these tests were negative.
Gross and Histological Evaluation
Complete gross necropsies were conducted by facility veterinarians and/or board-certified veterinary pathologists (MD, JO, DS, BB, JS, LWr, or JY). Nasopharyngeal swabs were collected for SARS-CoV-2 testing at the time of necropsy. Necropsy tissues were immediately fixed in 10% neutral-buffered formalin. Following fixation for 24 to 72 hours, sections were processed routinely, paraffin-embedded and sectioned at 3 to 4 μm and stained with hematoxylin and eosin. Additional special stains including Grocott-Gomori’s methenamine silver, Brown and Brenn, and Lillie-Twort Gram stains were evaluated as warranted. Histologic tissues were evaluated by multiple board-certified veterinary pathologists (MD, KT, JS, JR, and JO).
SARS-CoV-2 Reverse Transcription-PCR Testing
Real-time reverse transcription polymerase chain reaction (rRT-PCR) testing on fresh lung and nasal swab for SARS-CoV-2 was conducted at University of Nebraska Veterinary Diagnostic Laboratory, University of Illinois College of Veterinary Medicine Veterinary Diagnostic Laboratory, and Kansas State University Veterinary Diagnostic Lab based on an in-house developed and validated multiplex assay using previously published methods.1,22,25,29,56 Following initial rRT-PCR testing of fresh samples, all positive samples were sent to the U.S. Department of Agriculture’s National Veterinary Services Laboratories for confirmatory testing and sequence analysis. Reverse transcription polymerase chain reaction testing for SARS-CoV-2 on formalin-fixed paraffin-embedded (FFPE) tissues was performed at the Infectious Diseases Pathology Branch, Centers for Disease Control and Prevention (Atlanta, Georgia), as previously described.6,45
In Situ Hybridization and Immunohistochemistry
Formalin-fixed paraffin-embedded lung, trachea, nasal tissue, and heart from all cats were tested for the presence of SARS-CoV-2 RNA using RNAScope in situ hybridization (ISH) as described previously using oligonucleotide probes targeting the SARS-CoV-2 spike (S) and nucleocapsid (N) genes (Advanced Cell Diagnostics # 848561 and 863831).6,45 All tissues were tested in parallel using a negative control probe targeting the DapB gene from the Bacillus subtilis strain SMY (Advanced Cell Diagnostics # 310043). Severe acute respiratory syndrome coronavirus 2-infected Vero cells embedded with fragments of normal human tissues were used as a positive control. An immunohistochemical assay targeting the SARS-CoV-2 nucleocapsid protein was performed on respiratory tissues from 5/6 cats (all except case 6), as previously described. 46 Immunohistochemistry (IHC) for Aspergillus spp. and Pseudomonas aeruginosa was also performed in select cases to confirm these coinfections. For all IHC assays, an immunoalkaline phosphatase detection system and Mach 4 Universal AP Polymer Kit (Biocare Medical, Pacheco, CA) with Permanent Red Chromogen (Cell Marque; MilliporeSigma, St. Louis, MO) were used as previously described. 46 Severe acute respiratory syndrome coronavirus 2 IHC used SARS-CoV-2-infected Vero cells embedded with fragments of normal human tissues as a positive control. Pseudomonas IHC used mouse anti-Pseudomonas (Biogenesis/Biorad Catalog# 7889-9007) diluted at 1:100 with proteinase K pretreatment. 46 Aspergillus IHC used mouse anti-Aspergillus (Dako Corp Catalog#M3564; discontinued) diluted at 1:000 with proteinase K pretreatment. 45 Positive controls were bacterial/fungal cultures embedded with fragments of normal human tissues. Negative controls had normal mouse serum in place of the primary antibody.
Bacterial Culture and PCR
For bacterial cultures, fresh lung tissues or respiratory fluid was sampled and plated following validated and approved standard operating procedures in an American Association of Veterinary Laboratory Diagnosticians fully accredited diagnostic laboratory. Aerobic culture plates, including tryptic soy agar with 5% sheep’s blood, chocolate agar, Columbia agar supplemented with nalidixic acid, and MacConkey agar (Remel, Lenexa, KS, USA), were inoculated. Plates were examined at 24 and 48 hours after inoculation. Identification of potential bacterial pathogens from the culture media was performed using MALDI-TOF mass spectrometry according to standard procedures. Salmonella sp. were serogrouped using a commercially available kit (WellcollexTM Color Salmonella Rapid Latex Agglutination Kit, Remel). Salmonella isolates were sent for serotyping to a reference laboratory (United States Department of Agriculture’s National Veterinary Services Laboratory, Ames, IA). For in vitro antimicrobial sensitivity, a broth microdilution system was used following Clinical Laboratory Standards Institute guidelines and quality control recommendations. 58 For cases with bacteria seen in tissue by Gram stain, bacterial 16S rRNA gene PCR assay was performed on FFPE tissue, as previously described.22,25
Fungal Culture and PCR
Fungal culture of lung samples (case 3) were inoculated into Sabouraud dextrose agar, Emmons and Saboraud dextrose agar, and Emmons agar supplemented with chloramphenicol and gentamicin (Remel, Lenexa, Kansas) prepared according to standard laboratory procedures and examined weekly for 4 weeks. Identification of fungal pathogens was performed using identifying characteristics by microscopy. Molecular identification of fungi in fresh or FFPE tissue samples was achieved by PCR and DNA sequence analysis of the fungal internal transcribed spacer region, as described by Lysen et al with the exception that total nucleic acid was purified from tissues by using the QIAamp DNA FFPE Advanced UNG Kit 50 RXN (Qiagen, Gathersburg, MD) according to the manufacturer’s instructions. For FFPE tissue, 5 to 10 scrolls of sections (5 μm) were used for DNA extraction. 32
Severe acute respiratory syndrome coronavirus 2 sequence information is available via the Global Initiative on Sharing All Influenza Data (GISAID) database. Medical records are available at the discretion of the zoo, and inquiries should be directed to the corresponding author. All other data analyzed in this study are available by a request to the author.
Results
Clinical Findings
The results of laboratory tests are summarized in Supplemental Table S1.
Case 1. A 2.5-year-old, female snow leopard at zoo A developed progressive respiratory signs including nasal hemorrhage, dyspnea, and small bowel diarrhea. This snow leopard was positive for SARS-CoV-2 via nasal rRT-PCR (cycle threshold value [Ct] = 21.7) and developed progressive respiratory clinical signs and hematochezia that was treated with antibiotics (cefovecin and ceftiofur), corticosteroids, and supportive care. Radiographs were interpreted as a severe, broncho-interstitial pattern. Bacterial genome sequencing of the nasal swab amplified a nonspecific Mycoplasma sp. Despite antibiotic treatment, death occurred 72 hours after the initial onset of clinical signs. Bacterial 16SrRNA gene PCR on FFPE lung tissue identified Pseudomonas spp. and Mycoplasma leonicaptiva; Pseudomonas spp. was also confirmed by IHC. Other nondomestic felids at the same zoo, including other snow leopards, an African leopard, and a tiger, similarly developed respiratory clinical signs and recovered within 21 days with supportive therapy (including broad-spectrum antibiotics, antiemetic medications, and supportive fluids).
Case 2. A 14-year-old male lion housed at zoo B that had a history of idiopathic epilepsy controlled by medication presented with conjunctival swelling, thick mucoid nasal discharge, lethargy, and decreased appetite. The lion was treated with antibiotics (amoxicillin clavulanate), meloxicam, and supportive care. Antemortem SARS-CoV-2 rRT-PCR of nasal exudate was positive, with low Ct values indicating high viral load (Ct = 15). Clinical signs progressed to lower respiratory tract congestion, dyspnea, and death over the course of 7 days. Bronchial cytology had neutrophilic inflammation, increased squamous epithelial cells, and a mix of E coli and normal flora were cultured (Fig. 1). Pharyngeal and conjunctival swabs were PCR negative for Chlamydia felis, feline calicivirus, feline herpes virus 1, Bordetella bronchiseptica, H7N2 influenza virus, influenza A virus, and Mycoplasma felis. Enterobacteriaceae was identified by PCR on FFPE lung tissues.
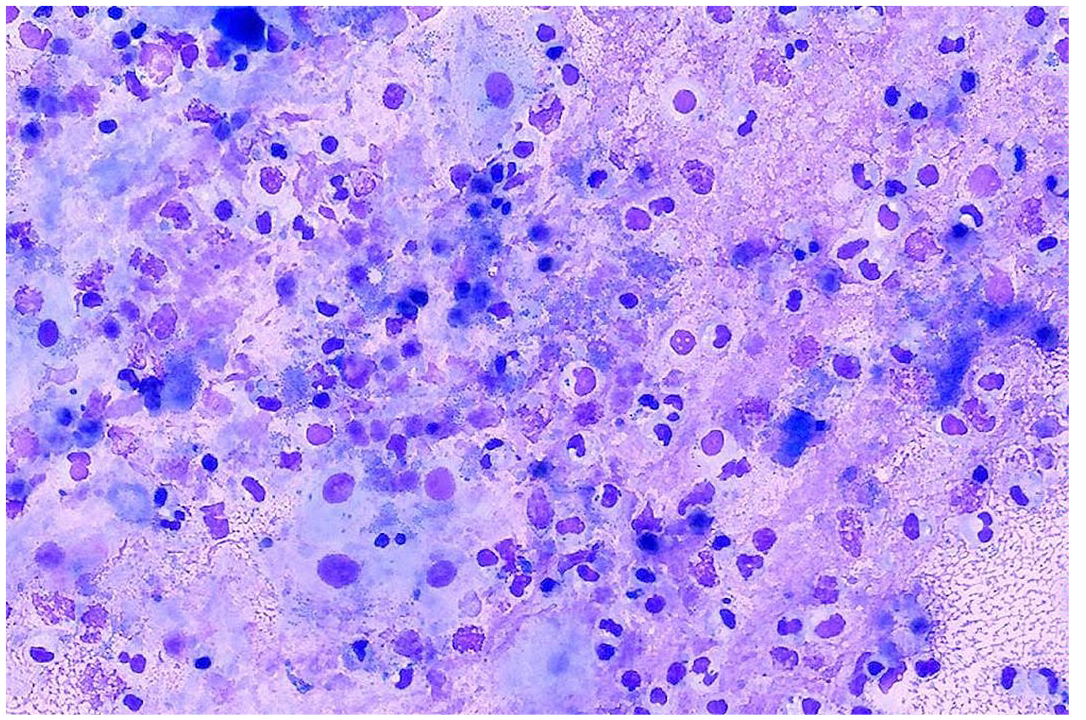
Bronchial cytology, lion. Neutrophilic and histiocytic inflammation, epithelial cells, and cellular debris with mixed bacteria. Case 2. Wright’s Giemsa.
Case 3: At zoo C, an 11-year-old, male snow leopard was observed coughing with oculonasal discharge. Severe acute respiratory syndrome coronavirus 2 fecal and nasal swabs were rRT-PCR positive (nasal rRT-PCR Ct = 19.5). At the same facility, other nondomestic felids including other snow leopards (cases 4 and 5), a clouded leopard, and tigers similarly developed respiratory clinical signs. The clouded leopard and tigers recovered. This snow leopard developed progressive respiratory clinical signs and hematochezia that was treated with antibiotics (clavamox, clindamycin, and enrofloxacin), corticosteroids, and supportive care including supplemental oxygen. On an endoscopy on day 16, 1.1 L of blood, purulent fluid, and mucus were suctioned from the airways, and tracheal ulcers were observed. Owing to poor prognosis, the snow leopard was euthanized 17 days after initial presentation. Postmortem lung culture identified Salmonella enterica subspecies enterica serovar Dublin and Actinomyces hyovaginalis.
Case 4: At zoo C, an 8-year-old, male snow leopard co-housed with cases 3 and 5 was observed to have an oculonasal discharge and cough 4 days after clinical signs were observed for case 3. Severe acute respiratory syndrome coronavirus 2 rRT-PCR from fecal and nasal swabs were positive (nasal rRT-PCR Ct = 18.6). Clinical signs progressed over the course of 15 days to severe hematochezia, epistaxis, dyspnea, and death. This snow leopard was treated with antibiotics (clavamox, enrofloxacin, and clindamycin), corticosteroids, and supportive care including supplemental oxygen and endotracheal suction.
Case 5: At zoo C, an 8-year-old female snow leopard (cohoused with cases 3 and 4) was observed to have an oculonasal discharge 4 days after the first snow leopard (case 3). Severe acute respiratory syndrome coronavirus 2 rRT-PCR from fecal and nasal swabs were positive (nasal rRT-PCR Ct = 23.6), and the snow leopard developed clinical signs similar to the other snow leopards at the same zoo, with additional thrombocytopenia. Clinical signs progressed over the course of 15 days to severe hematochezia, epistaxis, and dyspnea. This snow leopard was treated with antibiotics (clavamox, enrofloxacin, and clindamycin), corticosteroids, and supportive care including supplemental oxygen and endotracheal suction. The snow leopard died 15 days after initial clinical signs were observed.
Case 6: An 11-year-old male snow leopard at zoo D was housed adjacent to 2 other snow leopards and were cared for by the same keeper. The snow leopards presented with clinical signs consistent with respiratory disease; the other 2 snow leopards recovered. Empiric therapy was initiated, including ondansetron, prednisolone, azithromycin, and itraconazole. This snow leopard had minimal response to therapy and was positive for SARS-CoV-2 RNA by rRT-PCR from a fecal sample (Ct = 29.8). Respiratory and gastrointestinal signs improved 14 days after initial presentation, but respiratory disease never fully resolved. The cat’s condition worsened abruptly 36 days after initial presentation, and the cat died 4 days later (day 40 from presentation).
Gross Findings
In all cases, nasal sinuses were dark red, thickened or edematous, and contained frothy to purulent fluid that variably obstructed nasal cavities, trachea, and mainstem bronchi (Fig. 2a). Tracheas frequently contained additional fluid and were congested, edematous, and had multifocal mucosal ulcers. Acute cases (cases 1 and 2) had diffusely red to dark red wet lungs that exuded cloudy, serosanguinous to tan fluid (Fig. 2b). In cases with longer duration (cases 3–6), nasal cavities, trachea, and mainstem bronchi contained hemorrhagic and purulent exudate (Supplemental Fig. S1). Lungs contained multifocal abscesses and locally extensive necrosis in addition to consolidation and edema (Fig. 2c, d). In these cases and in case 1, there were pericardial, abdominal, and thoracic fibrinous, serous, or serosanginous effusions. Myocardial petechia and multifocal myocardial pallor were histologically associated with lpyogranulomatous to granulomatous inflammation. Additional findings included renal abscesses (case 6) and multifocal, mucosal ulcers in the distal esophagus (case 6), stomach (cases 3 and 6), small intestine (cases 2, 5, and 6), and colon (cases 4 and 5). These ulcers were associated with hemorrhage and partially digested blood (Supplemental Fig. S2).
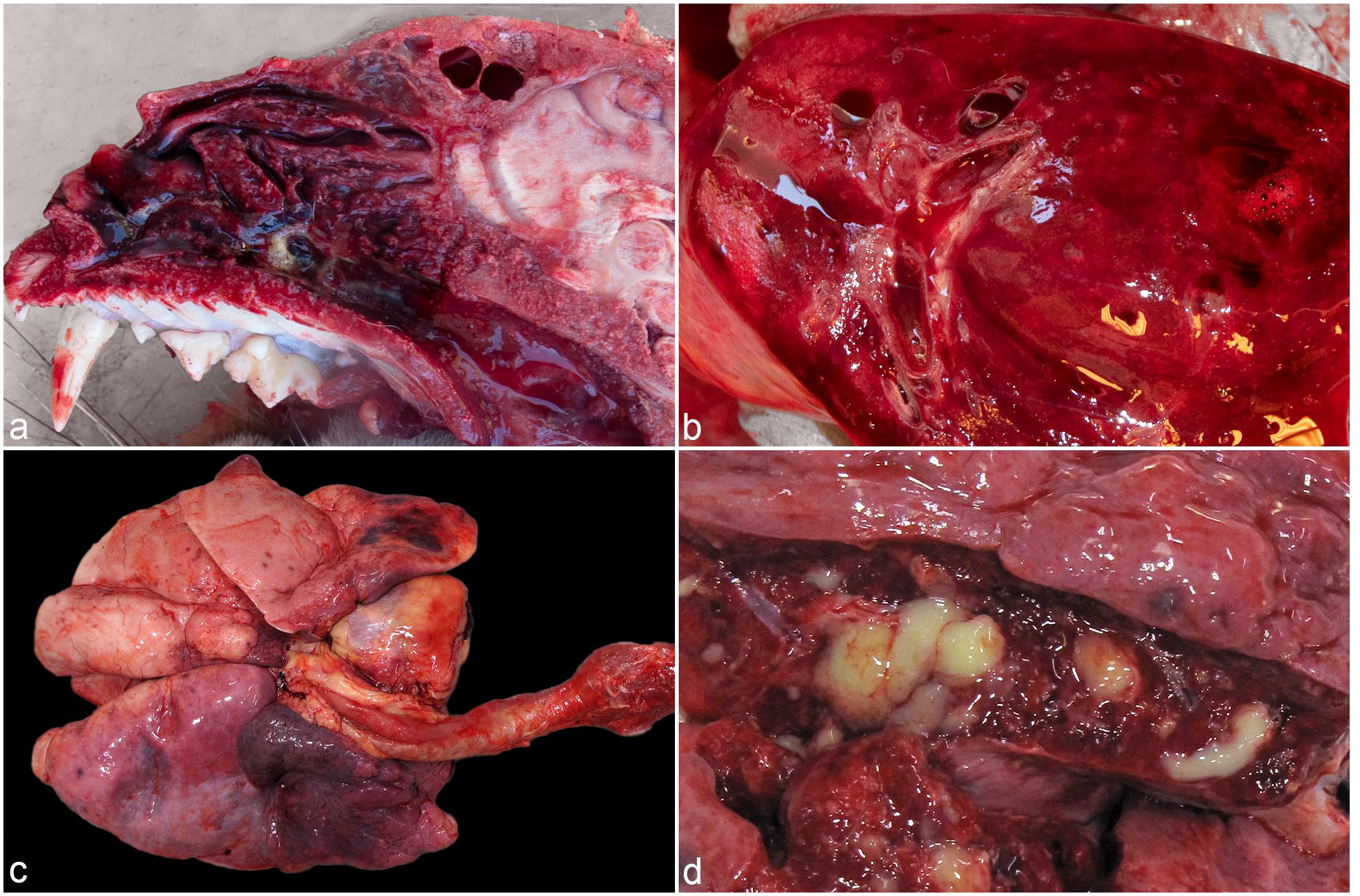
Figure 2
Histology Findings
The nasal cavity and sinuses contained abundant cellular debris, numerous foamy macrophages, neutrophils, bacteria, hemorrhage, cellular debris, and abundant fibrin (Fig. 3a). Nasal mucosa was absent or attenuated with variably disorganized epithelial cell piling and squamous metaplasia. In cases 3 to 6, the fibrinocellular exudate contained mats of fungal hyphae with nondichotomous branching, infrequent septation, and nonparallel walls (Fig. 3b). In these cats, there was thrombosis of small submucosal vessels, submucosal lymphoplasmacytic and histiocytic inflammation, edema, and variable fibrosis. In all cats, the trachea mucosa was absent, attenuated or disorganized with multifocal ulcers (Fig. 3c). Consistent with secondary bacterial and fungal infections, ulcers were frequently covered with fibrinosuppurative and hemorrhagic exudate that contained bacilli and/or fungal hyphae (Supplemental Fig. S3a). Moderate numbers of macrophages, neutrophils, and few lymphocytes and plasma cells infiltrated the exposed submucosa. Squamous metaplasia and submucosal pyogranulomatous inflammation and granulation tissue with fibrinoid necrosis of small, submucosal vessels were present in cats that survived the initial acute infection. Remaining submucosal glands were distended with cell debris and thickened by squamous metaplasia.
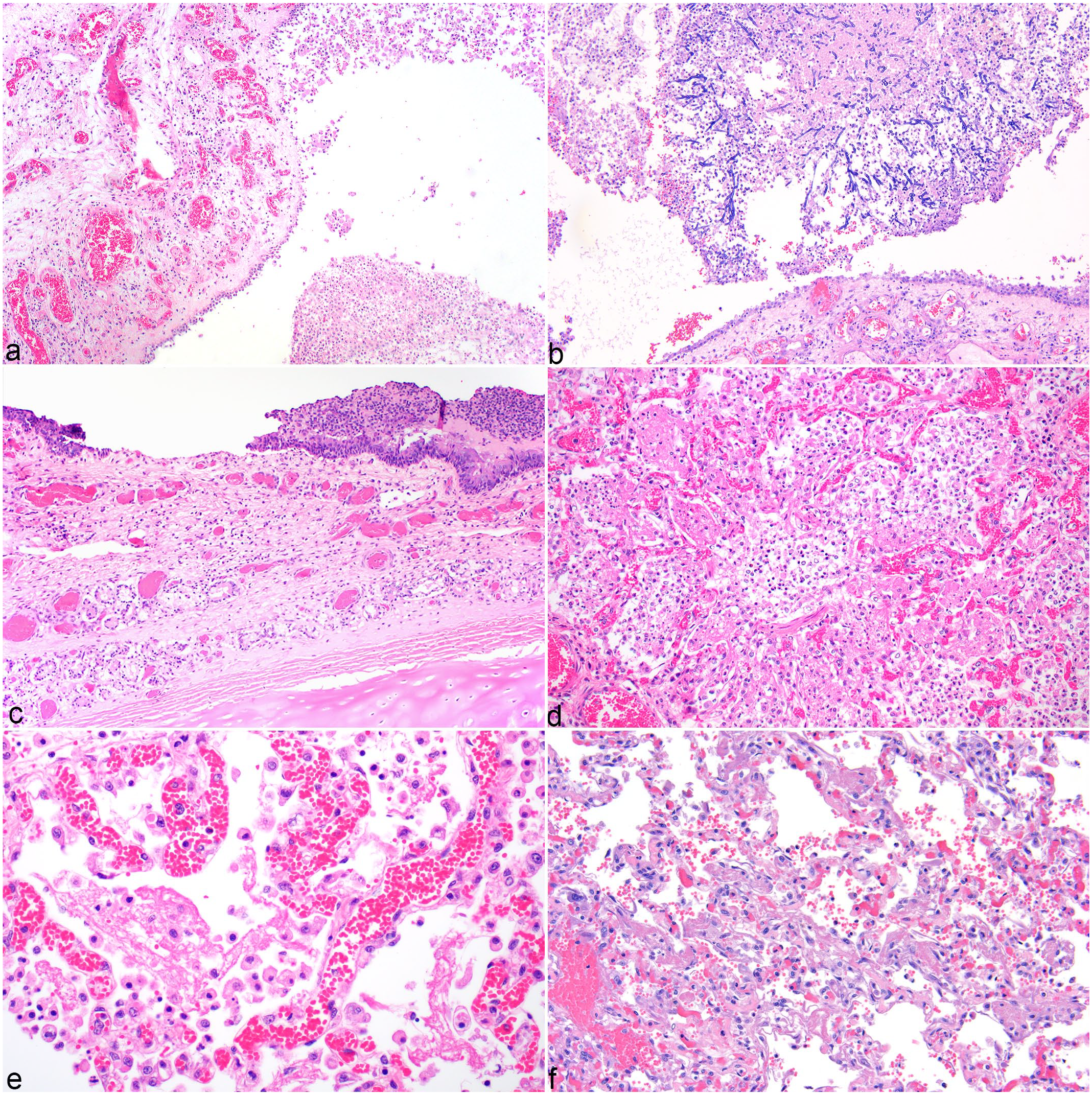
SARS-CoV-2 infection, hematoxylin and eosin.
Pulmonary lesions were the most severe in cases 1 and 2, which had rapid disease course, and centered on bronchi and bronchioles but affected greater than 90% of some sections. Large and mid-sized airways had attenuated to eroded epithelium, abundant intraluminal fibrin, pyogranulomatous inflammation, necrotic respiratory epithelial cells, and small amounts of blood (Fig. 3d). Alveoli and bronchioles were frequently denuded of pneumonocytes, lined by fibrin exudate, and contained numerous neutrophils, macrophages, fibrin, and mixed bacteria (Fig. 3e). Alveolar septae were widened by edema, fibrin, and neutrophils. Alveolar capillaries were congested.
In cases 3 to 6, bronchi and bronchioles were extensively obstructed with suppurative to pyogranulomatous inflammation, abundant cell debris, fibrin, fungal hyphae, and bacteria. Intact respiratory epithelium had extensive squamous metaplasia with submucosal edema, congestion, and mixed inflammation. A plurality of bronchi and bronchioles had concurrent, transmural necrosis, and distal airways had multifocal nodules of pyogranulomatous inflammation and necrosis associated with mats of fungal hyphae (Supplemental Fig. S3b). Alveolar septa were expanded by congestion, fibrin, edema, foamy macrophages, and smooth muscle hypertrophy. Often, alveolar capillaries contained few erythrocytes and were obscured by intraluminal fibrin and edema. Alveolar septa were lined by hyperplastic type II pneumocytes, and variably organized and compacted fibrin (Fig. 3f).
Additional histological findings in case 1 included fibrin in glomerular capillaries. Additional histological findings in longer duration infections (cases 3–6) include multifocal to coalescing, pyogranulomatous inflammation and necrosis associated with fungal emboli in the lungs (Supplemental Fig. S3c), brain, thyroid gland, myocardium (Supplemental Fig. S3d), kidneys, stomach, eyes, cerebrum, and colon. In case 3, multifocal, gastric ulcers and the submucosa granulation tissue were not associated with fungal organisms.
SARS-CoV-2 Detection in Tissues by ISH and IHC
The distribution of SARS-CoV-2 viral RNA by ISH and antigen by IHC is summarized in Supplemental Table S2. RNA and antigen localization had similar distributions, with stronger and more extensive labeling by ISH than IHC (Fig. 4a, b). For ISH, there was stronger signal using the N gene probes compared to S gene probe. In most cats, nasal turbinates had moderate to abundant, strong, punctate labeling within luminal exudate and within macrophages (Fig. 4a). In half of the cats, there was additional, rare, punctate labeling within nasal epithelial cells (Fig. 4c). Case 4 had abundant labeling in nasal submucosal glands. Most cats had absent or rare labeling of tracheal epithelial cells or macrophages. All cats had ISH labeling in tracheal and bronchial submucosal glands (Fig. 4d), and in material in airway lumens. Multiple cases had punctate labeling at the margins of alveolar septa and within pneumocytes as well as extracellularly in alveoli and in alveolar macrophages (Fig. 4e, f). Rare blood vessels within the heart and lungs had sporadic, intraluminal cell-associated labeling.
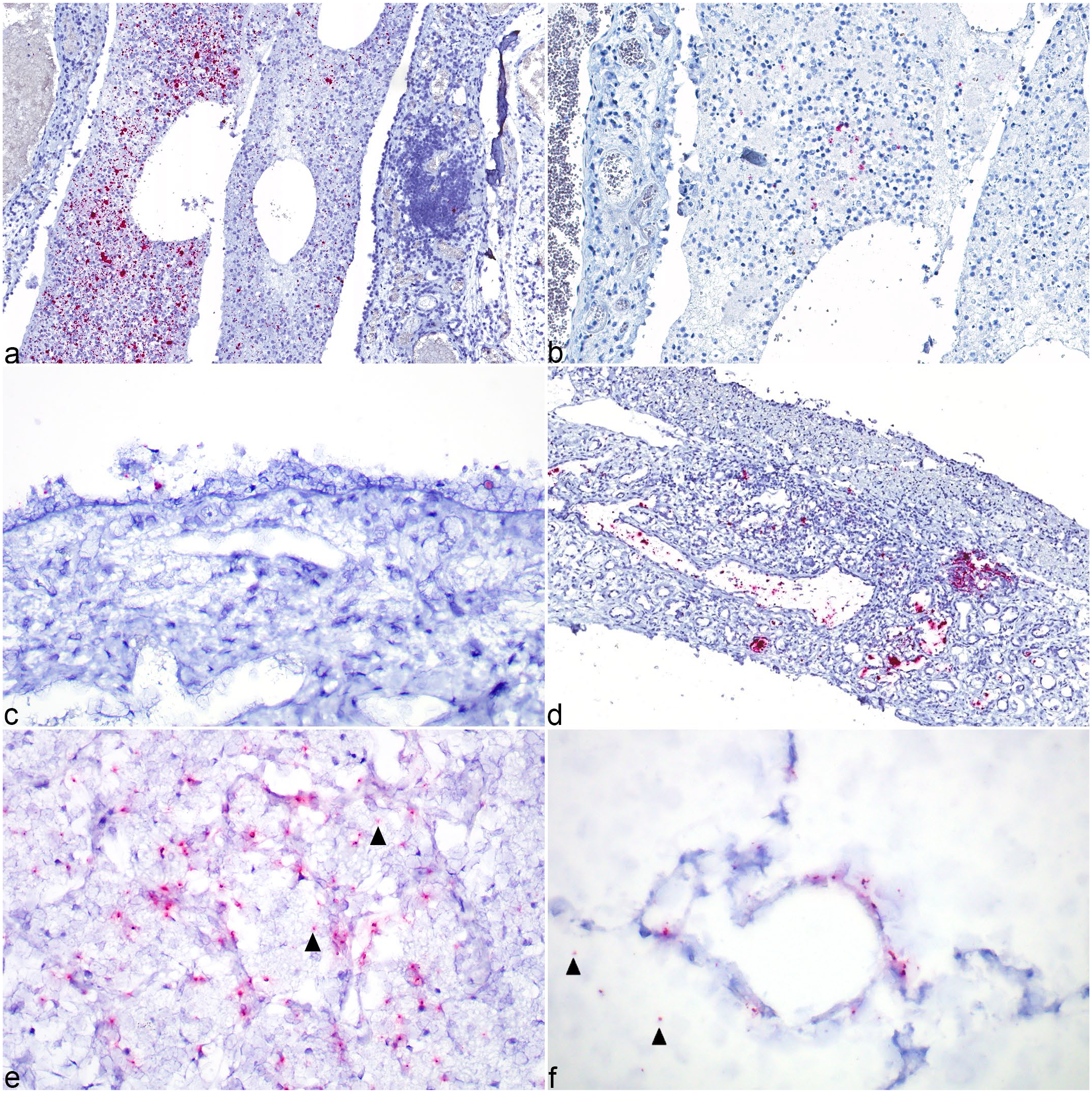
SARS-CoV-2 infection, in situ hybridization (ISH), N gene probe and immunohistochemistry (IHC).
Viral Sequence Phylogenic Analysis
Phylogenetic tree analysis of spike gene of SARS-CoV-2 strains in cases 1, 2, 3, and 5 indicated that four 2021 strains (3 snow leopard strains SD-21-030290-001, NE-NCOV21-37805, NE-NCOV21-37807, and 1 lion strain HI-21-031374-001) identified in 3 zoos of this study were closely related and were all part of the delta variant cluster (Fig. 5). Prior to outbreaks caused by delta variant, the nonvariant (B and B.1.177.21 for lions, B.1.2 for snow leopard) and other variant (alpha for lions, mu GH for snow leopards) strains were previously identified.
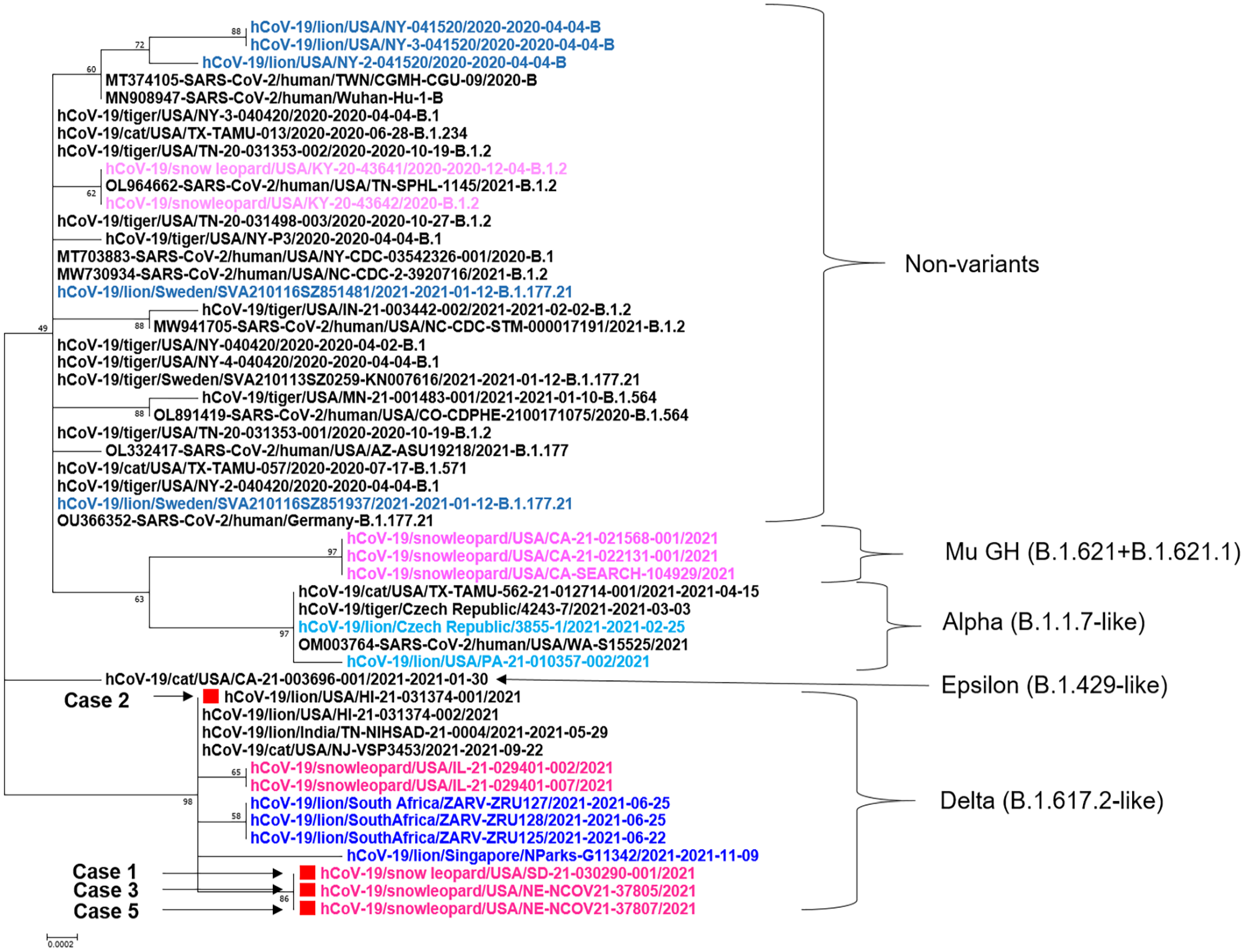
Phylogenetic analysis of SARS-CoV-2 spike genes of animal and human strains including 4 cases from this study (Cases 1, 2, 3, and 5) marked with red squares. Nonvariant and variant strains of lions and snow leopards are highlighted with blue and pink, respectively. Abbreviation: SARS-CoV-2, severe acute respiratory coronavirus 2.
Discussion
Consistent with established pathoepidemiological models for determining the contribution of SARS-CoV-2 in animal disease, all described felid cases supported that SARS-CoV-2 infection was a contributing factor in the animals’ deaths. 7 All cats had progressive respiratory disease and lacked significant previously existing comorbidities that would have predisposed them to severe disease. Antemortem testing, including bronchial lavage, radiographs, and SARS-CoV-2 rRT-PCR, are similar to previously documented infections in nondomestic felids.18,26,34 Histopathologic lesions in all cats are consistent with direct viral damage to the respiratory tract, with secondary infections. However, some of the key histopathological changes associated with SARS-CoV-2 lung infection in other species, such as lymphoplasmacytic inflammation and diffuse alveolar damage (hyaline membranes and type II pneumocyte hyperplasia) were not prominent. Whether these changes were obliterated by changes associated with secondary fungal and bacterial infections, were absent due to uneven tissue distribution, or were absent due to differences in species response to infection is uncertain. Prolonged corticosteroid treatment, as described for most of these cases, has been associated with decreased proliferation and differentiation of pneumocytes in animal models of acute lung injury. 38 Corticosteroid treatment may have modified histological lesions and increased the risk of gastrointestinal hemorrhage and secondary bacterial and fungal infections.
Domestic cats are an experimental animal model of SARS-CoV-2 infection with similarities and differences to these natural infections in nondomestic felids. When young (susceptible) domestic cats are inoculated with a high, intratracheal dose of virus, tracheal lesions are similar to those observed in these nondomestic felid cases. In addition, fibrin exudation and distal airway inflammation is similar to acute nondomestic felid cases.43,46 Some predictive factors, including high ACE-2 expression and protein sequence identity in the upper respiratory tract, are likely similar between nondomestic felids and domestic cats.2,27 However, in multiple experimental domestic cat studies, clinical disease is mild or absent despite the presence of viral RNA and histological disease in respiratory tissues. In domestic cats, viral RNA is decreased in respiratory tissues after 7 days and not detected in studies that are extended to 21 days.16,43 In comparison, the prevalence of SARS-CoV-2 IHC and ISH-positive labeling in respiratory tissues was similar in both acute and chronic, nondomestic felid cases. Compared to the reported peribronchial fibrosis reported in cats 21 and 28 days after experimental infection, nondomestic felids have histological changes typical of acute-stage infection despite a prolonged course of disease.8,42 These findings may indicate that reduced viral clearance contributes to increased risk of severe SARS-CoV-2 disease nondomestic felids and increases concerns that nondomestic felids that recover from SARS-CoV-2 may have long-term respiratory injury. Prolonged viral shedding may have implications for disease transmission in nondomestic felids that use feces to mark territory, including snow leopards. 15 In zoos, prolonged fecal contamination of equipment and environments could increase the risk of viral spread between enclosures or require lengthened quarantine of infected animals or potentially asymptomatic transported animals as viral shedding continues for more than a month after the resolution of clinical disease.5,14,26 Both prolonged viral shedding and high levels of virus within tissues, as observed via ISH in this study, increase concerns for zoonotic spread of disease. 50
Severe acute respiratory syndrome coronavirus 2 viral RNA was detected in multiple sample types (nasal swab, bronchial fluid, and lung tissue) and by multiple methods (RT-PCR, ISH, and IHC). Labeling of viral RNA and antigen was most commonly seen intra- and extracellularly within necrotic and inflammatory debris throughout the respiratory tract, and less consistently within bronchial glands and intra-alveolar cells of the lungs. Whether labeling within cells in areas of abundant debris was within macrophages, epithelial cells, or both was difficult to discern. In situ hybridization also identified rare labeling in intact respiratory epithelial and glandular epithelial cells.
Similar to the findings in these snow leopards and lion, SARS-CoV-2 IHC and ISH in naturally infected mink localizes viral RNA and antigen in nasal exudate, nasal epithelial cells and glands, tracheal and bronchial epithelium, and pneumocytes.13,39,46 While bacterial rhinitis in mink with SARS-CoV-2 infection has been reported, secondary bacterial bronchopneumonia is comparatively rare. 46 In the Syrian hamster and domestic cat infection model, which have less severe disease and faster viral clearance compared to mink and nondomestic felids, viral antigen is observed within bronchi and bronchioles and adjacent distal airways, which is a pattern most consistent with gravity dispersion of infected cells and fluids.21,43 Viral accumulation in bronchial glands and the upper respiratory tract may allow for subsequent rounds of distal airway seeding after extensive tracheal mucosal necrosis and pulmonary viral clearance. Similar viral accumulation in bronchial glands has been seen in humans that died from severe SARS-CoV-2 disease. 33 In addition, altered distal airway mucus secretion is a proposed contributor to severe SARS-CoV-2 respiratory disease in mouse and macaque experimental models. 31 Sustained infection of bronchial glands may contribute to airway obstruction and hypoxia in nondomestic felids and contribute to disease severity. In humans, inflammation associated with bacterial infections or aspirated oral bacteria can also increase the expression of the viral receptor ACE2 in respiratory epithelial cells. 51 Secondary bacterial and fungal infections in both the upper and lower airways may contribute to more severe, prolonged SARS-CoV-2 disease via cell receptor modification and re-infection of lower airways from aspirated cell debris.
Secondary, disseminated fungal and bacterial infections or co-infections were present in all 4 cats that survived beyond 8 days. While secondary bacterial and fungal infections are not uncommon sequelae of viral infections, secondary bacterial rhinitis has only been reported in natural SARS-CoV-2 infections in mink. 46 In contrast, the high prevalence of bacterial and fungal infections in these cases is more similar to severe and prolonged human SARS-CoV-2 infections that are treated with immune-modulatory, oxygen, and antibiotic therapies. In humans, additional factors associated with high levels of therapeutic intervention including intubation and prolonged intensive care increase secondary infection risk, including risk for angioinvasive Aspergillus infections.19,55 Unlike experimental infections in domestic cats or natural infections in mink, these snow leopards and other captive, nondomestic felids experience high levels of therapeutic intervention that potentially increased the risk of secondary infections. Often, clinical intervention for large felids necessitates anesthesia and intubation, and similar practices in humans with COVID-19 are associated with an increased risk of secondary infections. 62 With these commonalities, a high frequency of secondary bacterial and fungal infections that significantly increases disease severity has currently only been consistently identified in snow leopards. In humans, secondary infections were commonly reported (up to 50% of fatalities) early in the pandemic and have decreased with improved therapeutic interventions and vaccines.28,33 The finding of serious secondary infections in these cats indicates that antibiotic and antifungal therapies should accompany treatment for SARS-CoV-2 infection in big cats.
While neuroinvasion of SARS-CoV-2 has been recognized in humans, rhesus macaques, and mice with humanized ACE2, neuronal degeneration, axonal damage, or demyelination consistent with SARS-CoV-2 neurological disease was not observed in these cases.24,41,48 These findings are consistent with experimental studies in cats and suggest a low prevalence of neuroinvasive SARS-CoV-2 in felids.16,48 Although rare, intracellular viral material was observed in the lumens of pulmonary and cardiac vessels. However, the primary localization of viral RNA in respiratory epithelial cells, macrophages, and mucosal debris, and the absence of endothelial cell viral infection in sections with vascular degeneration support a respiratory route of infection as the primary cause and route of dissemination in these cases. While intraluminal viral labeling is present in intestine at low levels, swallowing of respiratory debris may be a more likely source of virus as enterocyte infection was not observed (data not shown).
To date, SARS-CoV-2 infection has been confirmed in approximately 5% of felids housed in Association of Zoos and Aquariums accredited zoos, and nondomestic felids were the first animals in the United States to have documented disease associated with infection. Globally, nondomestic felids naturally infected with SARS-CoV-2 include tigers (Malayan and Amur subspecies), African lions, snow leopards, puma, lynx, and fishing cats. 42 Snow leopards have particularly high rates of severe disease (38% of infected animals) when infected with SARS-CoV-2. 54 According to a recent longitudinal study of snow leopard mortality, there is no evidence that snow leopards have an increased risk of respiratory infections in general, and the most common causes of death in adult snow leopards are degenerative diseases, such as chronic renal disease and neoplasia. 59 Among the animals in these cases, only the lion had a pre-existing disease, idiopathic epilepsy. In humans, epilepsy and antiseizure medication do not increase risk of SARS-CoV-2 infection nor is there evidence for increased adverse outcomes in people with epilepsy. 3 Therefore, it is considered unlikely that epilepsy contributed to SARS-CoV-2 in this case. At the zoos where the snow leopard cases originated, other conspecific felids were positive for SARS-CoV-2 infection, yet the snow leopards developed severe respiratory disease. In addition, cheetahs cohoused at zoo C routinely tested negative for SARS-CoV-2, despite their increased susceptibility to feline enteric coronaviruses compared to other nondomestic felids. 53 Thus, the increased susceptibility of snow leopards to SARS-CoV-2 is likely independent of other respiratory disease risks. Unfortunately, there are large gaps in our understanding of immune function and disease susceptibility of nondomestic felids, and these findings highlight the need for further studies into snow leopards, in particular.
In all of these described cases where RNA sequence was available, cats were infected with the delta variant of SARS-CoV-2, suggesting that this variant may have had high zoonotic transmissibility.37,61 Delta-variant-associated nondomestic felid infections also may be more likely to have an unfavorable outcome, as none of the prior nondelta strains were associated with nondomestic felid deaths, including infections in snow leopards with D614G (prevariant clade B.1.2) and mu variants.54,57 While all zoos had instituted biosafety measures, including the use of surgical or N95 masks by zoo staff per the Association of Zoos and Aquariums Felid Taxon Advisory Group guidelines, transmission either from caretakers or visitors remains the most likely source of viral exposure in these cats.14,36,52 In some locations, recommendations for the use of masks was limited by local and state regulations.
Knowledge of host susceptibility and viral pathogenesis is critical to controlling and managing the ongoing SARS-CoV-2 pandemic. Nondomestic felids were the first animals in the United States to develop documented disease associated with infection. The cases described herein confirm that SARS-CoV-2 can also be a primary and/or contributory factor in severe disease and death in these species. While none of the cats in this study were vaccinated at the time of infection, it remains uncertain as to whether future variants will pose a similar threat and whether vaccination will prevent similar outcomes. Understanding why the large nondomestic felids, and in particular snow leopards, are disproportionately susceptible and have succumbed to SARS-CoV-2 infection is an area for future study. As SARS-CoV-2 becomes endemic, the role of animals in the epidemiology of this virus will remain a critical research need.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231225500 – Supplemental material for Mortality associated with SARS-CoV-2 in nondomestic felids
Supplemental material, sj-pdf-1-vet-10.1177_03009858231225500 for Mortality associated with SARS-CoV-2 in nondomestic felids by Mary Drozd, Jana M. Ritter, Jonathan Peter Samuelson, Maryanna Parker, Leyi Wang, Samantha J. Sander, Jill Yoshicedo, Louden Wright, Jenee Odani, Trent Shrader, Elizabeth Lee, Shawn R. Lockhart, Ria R. Ghai and Karen A. Terio in Veterinary Pathology
Footnotes
Acknowledgements
The authors would like to thank Dr. Bruce Brodersen and Dr. David Steffen for the assistance with snow leopard necropsies from Zoo C. Dr. Dustin Loy for performing microbiological testing from Zoo C cases, Gretchen Anchor and Dr. Martha Delaney for their assistance with the in situ hybridization assays, Lindsey Estetter for performing PCR assays, Brian Min for performing fungal PCR assays, Luciana Flannery for performing immunohistochemical assays, Lindsey Estetter and Colleen Olmstead for performing SARS-CoV-2 RT-PCR assays, and Julu Bhatnagar for molecular pathology support, Steven Rekant for epidemiologic and case investigation support, the USDA National Veterinary Services Laboratories for confirmatory testing and sequencing, and Dr. Seth Harris for his editorial advice and mentorship.
Supplemental Material for this article is available online.
Disclaimer
The findings and conclusions are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention (CDC). The mention of company names or products does not constitute endorsement by the CDC.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
