Abstract
An epidemic of highly pathogenic avian influenza (HPAI) began in North America in the winter of 2021. The introduced Eurasian H5N1 clade 2.3.4.4b virus subsequently reassorted with North American avian influenza strains. This postmortem study describes the lesions and influenza A virus antigen distribution in 3 species of raptors, including bald eagles (Haliaeetus leucocephalus, n = 6), red-tailed hawks (Buteo jamaicensis, n = 9), and great horned owls (Bubo virginianus, n = 8), naturally infected with this virus strain based on positive reverse transcriptase polymerase chain reaction and sequencing results from oropharyngeal swabs. The birds presented with severe neurologic signs and either died or were euthanized because of the severity of their clinical signs and suspected influenza virus infection. Gross lesions were uncommon and included forebrain hemorrhages in 2 eagles, myocarditis in 1 hawk, and multifocal pancreatic necrosis in 3 owls. Histological lesions were common and included encephalitis, myocarditis, multifocal pancreas necrosis, multifocal adrenal necrosis, histiocytic splenitis, and anterior uveitis in decreasing frequency. Influenza A viral antigen was detected in brain, heart, pancreas, adrenal gland, kidney, spleen, liver, and eye. In conclusion, bald eagles, red-tailed hawks, and great horned owls infected with the HPAI clade 2.3.4.4b virus strain and showing neurological signs of illness may develop severe or fatal disease with histologically detectable lesions in the brain that are frequently positive for viral antigen.
An outbreak of highly pathogenic avian influenza (HPAI) began in North America in the winter of 2021 and was caused by the introduction of Eurasian (EA) A/goose/Guangdong/1996-lineage H5 clade 2.3.4.4b via wild migratory birds initially in the Atlantic flyway.3,6,13 The introduced EA H5N1 virus subsequently reassorted with North American (AM) avian influenza strains. In 2015, an introduction of EA H5 HPAI (clade 2.3.4.4c; formerly known as clade 2.3.4.4icA) viruses occurred via the Pacific flyway and was responsible for what previously was the largest and most costly animal disease outbreak in the United States. 23 During the 2015 outbreak, the introduced H5 viruses were associated with wild bird mortality events. Virus was detected in 26 bird species in the Pacific, central, and Mississippi flyways during the 2015 outbreak, but there were no detections in the Atlantic flyway, and the total number of detections in wild birds reached only about 100 during that outbreak. 15 The last detection of that virus (EA H5 HPAI clade 2.3.4.4c) was in a wild mallard duck from Montana in December 2016 (U.S. Department of Agriculture [USDA] Animal & Plant Health Inspection Service [APHIS] 2014–2015 Highly Pathogenic Avian Influenza Outbreak). 22 The distribution and impacts of the 2022 H5 2.3.4.4b HPAI outbreak have far surpassed that of 2015 with numerous large-scale wild bird mortality events in nearly 150 bird species, and spillover into mammals as well as poultry across all 4 flyways.37,38 Scavenging and carnivorous mammals, such as foxes, coyotes, skunks, raccoons, bobcats, mountain lions, bears, and opossums, amongst others, infected with the H5 2.3.4.4b HPAI virus (HPAIV) typically show neurologic signs.2,11,36 Disease and death in raptors, specifically, have been reported previously due to H5 2.3.4.4 clade viruses and prior clades of the A/goose/Guangdong/1996-lineage (gs/GD) H5 HPAIV.8,9,12,18,26,27,29,39 Experimental infections with gs/GD viruses have resulted in neurologic disease and death in various raptor species.5,14,24,34 Our postmortem study describes the lesions and viral antigen distribution in 3 species of raptors, including bald eagles (Haliaeetus leucocephalus), red-tailed hawks (Buteo jamaicensis), and great horned owls (Bubo virginianus) naturally infected with H5N1 clade 2.3.4.4b HPAIV during the 2022 outbreak.
Materials and Methods
Selection Criteria
Two species of the Accipitridae family (bald eagles and red-tailed hawks) and 1 species of the Strigidae family (great horned owls) were included in this study (Supplemental Table S1). More specifically, the birds of this study were 6 bald eagles, 9 red-tailed hawks, and 7 great horned owls that were admitted alive to The Raptor Center of the University of Minnesota between 5 April and 6 May 2022. In addition, 1 great horned owl that was admitted dead on 1 September 2022 was included in this study. The age of the birds was determined by their size and plumage characteristics. All animals admitted alive showed neurologic signs including seizures, nonresponsiveness, and/or nystagmus. The birds were euthanized on the day of admission because of a poor prognosis for recovery. The euthanasia was performed with a pentobarbital overdose given intravenously when venous access was available. Alternatively, the birds were euthanized via intracardiac pentobarbital administration under deep anesthesia with inhalant isoflurane gas in animals without suitable venous access. The postmortem examinations were performed within a maximum of 48 hours after the death of the euthanized animals and within 12 hours of submission of the owl that was found dead. None of the animals were frozen prior to the postmortem examination. West Nile virus (WNV) infection was not considered a possible cause of the clinical signs because all birds were admitted outside of the WNV season in Minnesota (early June to the end of October), except for the owl that was submitted dead on 1 September 2022.
Necropsies and Histopathology
All necropsies were performed in a biosafety cabinet. Samples of brain, heart, liver, bone marrow, spleen, lung, kidney, adrenal glands, gonads, proventriculus, ventriculus, intestine, pancreas, and bursa of Fabricius (when present) were fixed for 72 hours in 10% neutral-buffered formalin and processed routinely to produce 4 μm thick, paraffin-embedded, hematoxylin and eosin-stained tissue sections. The degree of necrosis and inflammation in any particular tissue was subjectively semi-quantitatively graded as absent (0), mild (1), moderate (2), or marked (3) based on the estimated amount of the tissue section that was affect and the estimated number of foci of necrosis and/or inflammation. In the brain, a degree of 1 was assigned when few small areas of selective neuronal necrosis or pan-necrosis (necrosis of neurons and other components of the neuroparenchyma such as glial cells), few neuroparenchymal and/or meningeal perivascular cuffs with up to 2 layers, and/or small portions of inflamed ventricular system were observed in at least 1 compartment (forebrain, cerebellum, or brainstem). A degree of 3 was assigned when large or coalescing portions of the neuroparenchyma were necrotic, numerous vessels had perivascular cuffs, multiple cuffs had more than 3 layers, and/or large portions of the ventricular system were inflamed. Brains with a combination of lesions that was more severe than grade 1 but less severe than grade 3 fell into the grade 2 category. In the other organs (heart, adrenal gland, pancreas, liver, kidney, and spleen), a degree of 1 was assigned when up to 3 small foci of necrosis with or without lymphoplasmacytic inflammation were present, representing less than approximately 5% of the tissue section. In these organs, a degree of 2 constituted focal or multifocal necrosis and inflammation that affected up to approximately 25% of tissue while involvement of larger areas of tissue resulted in a degree of 3. In the eye, the presence of few scattered inflammatory cells in the anterior chamber/iris and the pecten and/or the presence of necrosis affecting a small portion of the retina was graded as 1. All lesions were scored by one of the authors (AW).
An oropharyngeal swab for influenza virus-specific testing via polymerase chain reaction (PCR) was collected from each bird during the postmortem examination.
Because infection with HPAIV was considered a possibility at the outset of the necropsies, no ancillary testing (aside from histopathology) for alternate or concurrent pathogens was pursued in any of the cases in an attempt to minimize the possibility of exposure of lab personnel to the virus.
Immunohistochemistry
Sections of brain, heart, liver, bone marrow, spleen, lung, kidney, adrenal glands, gonads, proventriculus, ventriculus, intestine, pancreas, and bursa of Fabricius (when present) underwent immunohistochemistry. A peroxidase-based polymer system was used, as previously described. 44 A commercially available mouse monoclonal antibody (IgG2a isotype) targeting the nucleoprotein of influenza A viruses was used at a 1:4000 dilution (clone 1331, Meridian Biosciences, catalog C65331M). 1 Chicken serum was used as blocking component in a 2% solution. Antigen retrieval was achieved with proteinase K pretreatment of the sections. Formalin-fixed, paraffin-embedded brain tissue from a known influenza A virus positive domestic turkey was used as positive control. Negative controls included substitution of the primary antibody by ascites fluid from nonimmunized Balb/c mice.
The amount of viral antigen expression in the various organs was semi-quantitatively graded as being absent (0), mild (1), moderate (2), or marked (3) based on the estimated number of influenza virus antigen-positive cells and estimated area with immunopositive cells in any given tissue section. In either organ, the presence of less than approximately 50 immunopositive cells and/or immunopositive cells in less than approximately 5% of the tissue section was graded as 1. A grade 2 was assigned when up to approximately 20% of a tissue section had immunopositve cells. Sections with immunopositive cells in areas covering more than approximately 20% of the tissue sections were graded as 3. All immunohistologic sections were scored by one of the authors (AW).
Real-time reverse transcriptase (RT)-PCR
The oropharyngeal swabs were examined for the matrix gene of influenza A virus via a semi-quantitative real time RT-PCR assay, as previously described.30,31,46 The use of oropharyngeal swabs is preferred over the use of cloacal swabs in raptors based on the observation that viral shedding via the oral cavity lasted longer than the shedding via the cloacal route. 14 Positive samples were further analyzed at the National Veterinary Service Laboratories for the H5 and N1 subtype using RT-PCR and whole genome sequencing as previously reported.6,11,32,35 Ct values below 35 were considered to be unequivocally positive in all 3 RT-PCR assays. In addition, the pathotype was determined and whole genome sequencing was attempted. 46
Results
Necropsy
Gross lesions included acute unilateral forebrain hemorrhages in 2 bald eagles, widespread pale myocardial discoloration in 1 red-tailed hawk, and distinct dark red foci in the pancreas of 3 great horned owls (Fig. 1a–c). The spleens of 3 eagles, 4 hawks, and 4 owls were enlarged and diffusely red. Since all but 1 bird in this study were euthanized, this finding was interpreted as euthanasia-related splenic congestion. The spleen of 1 owl had numerous, beige pinpoint specks.
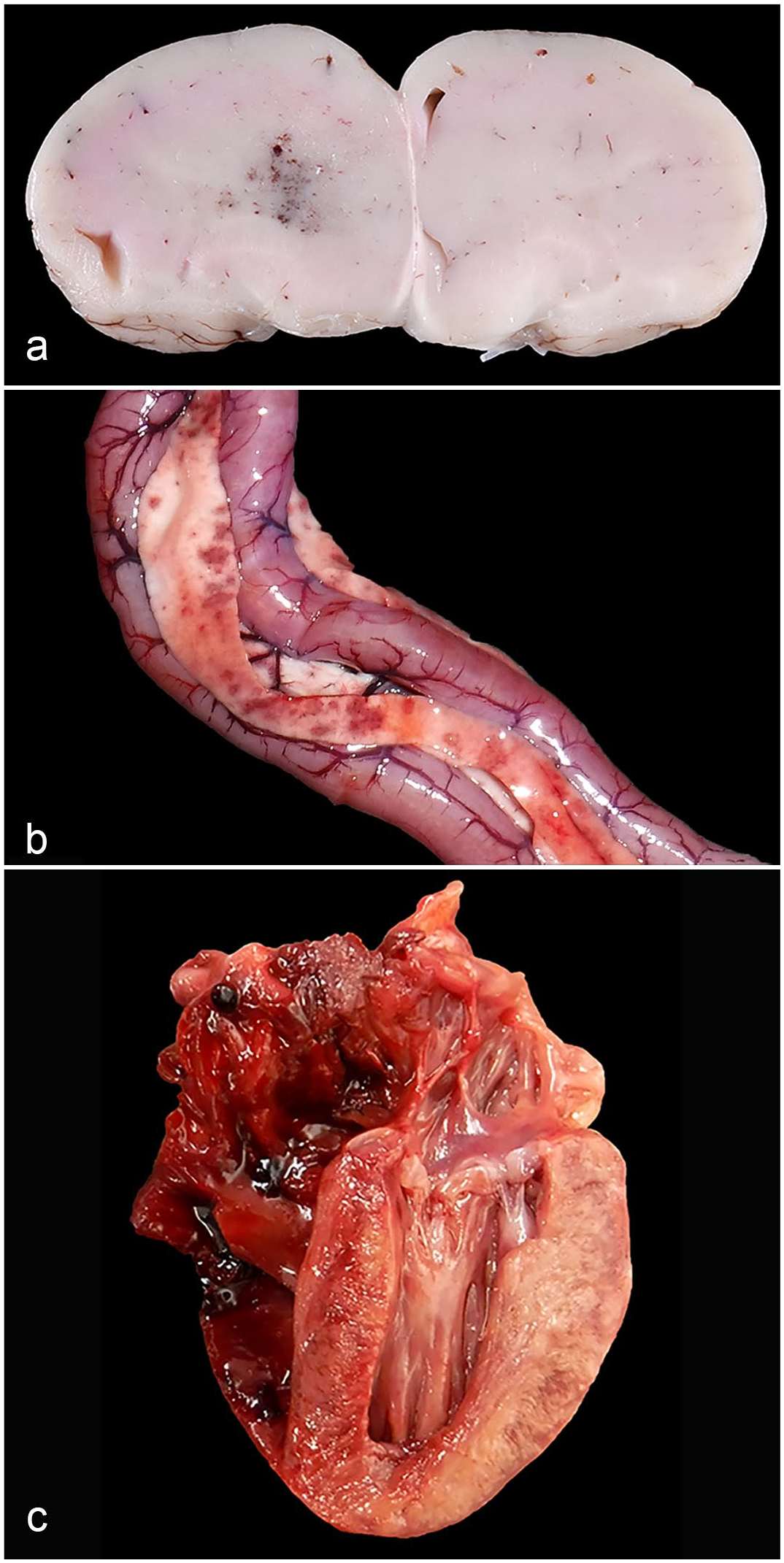
Gross lesions in raptors infected with H5 clade 2.3.4.4b highly pathogenic avian influenza virus. (a) Forebrain, bald eagle. One hemisphere is slightly swollen and has hemorrhage at its center. Case 2. (b) Pancreas, great horned owl. Multiple distinct red foci are scattered within the pancreas. Case 20. (c) Heart, red-tailed hawk. The myocardium of the left ventricular free wall and septum is beige discolored. Case 9.
Histopathology
All birds of this study had brain lesions attributable to infection with a virus (Table 1, Supplemental Table S2). On average, the brain lesions tended to be more severe in owls compared to hawks and eagles. The lesions included fibrinoid necrosis of capillary walls with or without thrombosis, meningoencephalitis, selective acidophilic neuronal necrosis, focal or multifocal pan-necrosis, and ependymitis (Fig. 2a–d). In all 3 species, the cellular infiltrates consisted of mononuclear cells, including lymphocytes and macrophages, and, in the majority of the cases, granulocytes. Mononuclear inflammatory cells were the predominant cell component in the meninges and perivascular infiltrates. The nuclei of necrotic neurons were fine granular in some cases. Granulocytes infiltrated areas of parenchymal necrosis and were present to a lesser extent in the perivascular infiltrates. In all 3 species, the lesions were prominent in the forebrain and were present to a lesser degree in the mesencephalon, cerebellum, and brainstem. In the brainstem and cerebellum, areas of gray matter pan-necrosis were occasionally limited to the neuroparenchyma that was nearest to the 4th ventricle. The ependymitis was characterized by multifocal necrosis of the ependymal cells and infiltration of the subependymal neuroparenchyma with predominantly granulocytes and macrophages. These cells also were present in the ventricular lumen. All owls and 7 of the hawks had ependymitis of variable severity, while only 2 eagles had ependymitis. The choroid plexus of a lateral ventricle had areas of acute necrosis with fibrin exudation and hemorrhage in 2 hawks. Fibrinoid necrosis of capillary walls (with or without thrombosis) and granulocytic vasculitis was present in the brains of 2 eagles, 4 hawks, and all owls. Histological heart lesions were detected in 5 eagles, 7 hawks, and 6 owls. In some cases, multiple foci of acute cardiomyocyte necrosis were present without any significant inflammatory reaction. In other cases, multifocal infiltration of the myocardium by lymphocytes and fewer granulocytes was associated with areas of coagulative or lytic myocardial necrosis (Fig. 3a).
Median degree (and range) of histologic lesions and mean intensity (and range) of immunohistochemically (IHC) detected viral antigen presence in select tissues of raptors that were polymerase chain reaction-positive for highly pathogenic influenza virus (Eurasian/American 2.3.4.4b H5N1) based on oropharyngeal swabs.
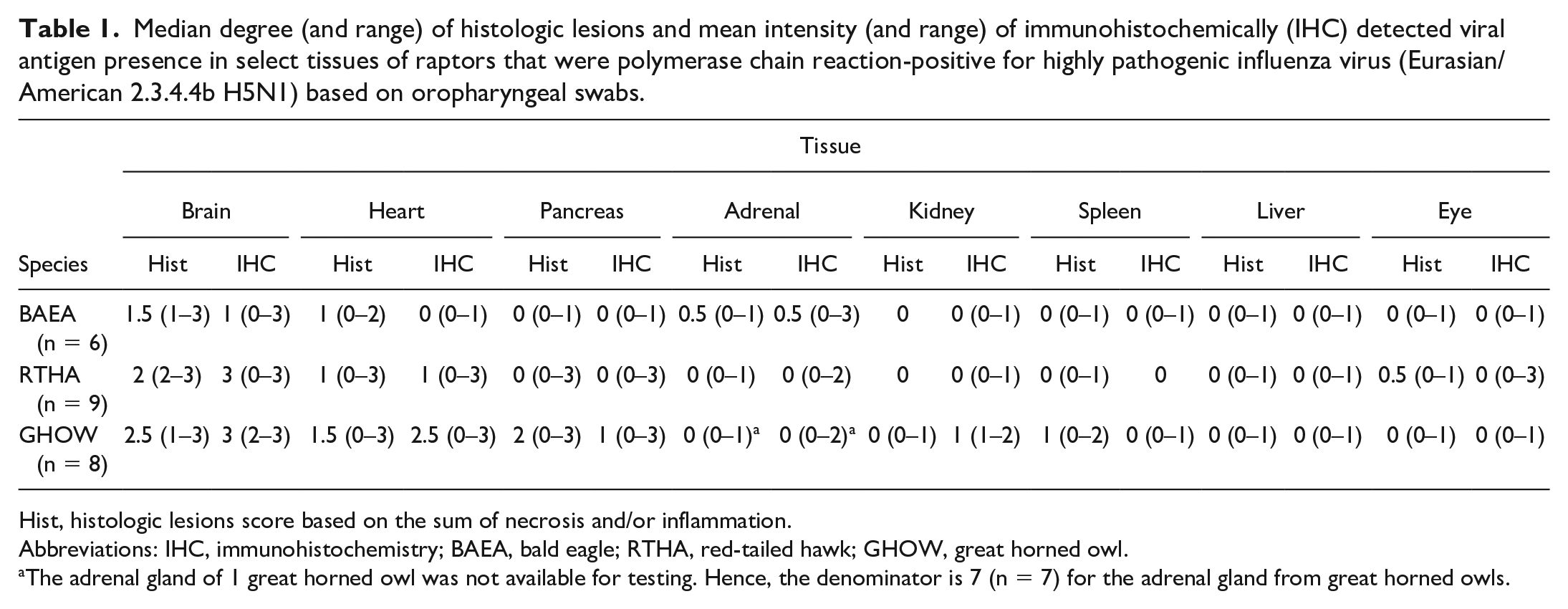
Hist, histologic lesions score based on the sum of necrosis and/or inflammation.
Abbreviations: IHC, immunohistochemistry; BAEA, bald eagle; RTHA, red-tailed hawk; GHOW, great horned owl.
The adrenal gland of 1 great horned owl was not available for testing. Hence, the denominator is 7 (n = 7) for the adrenal gland from great horned owls.
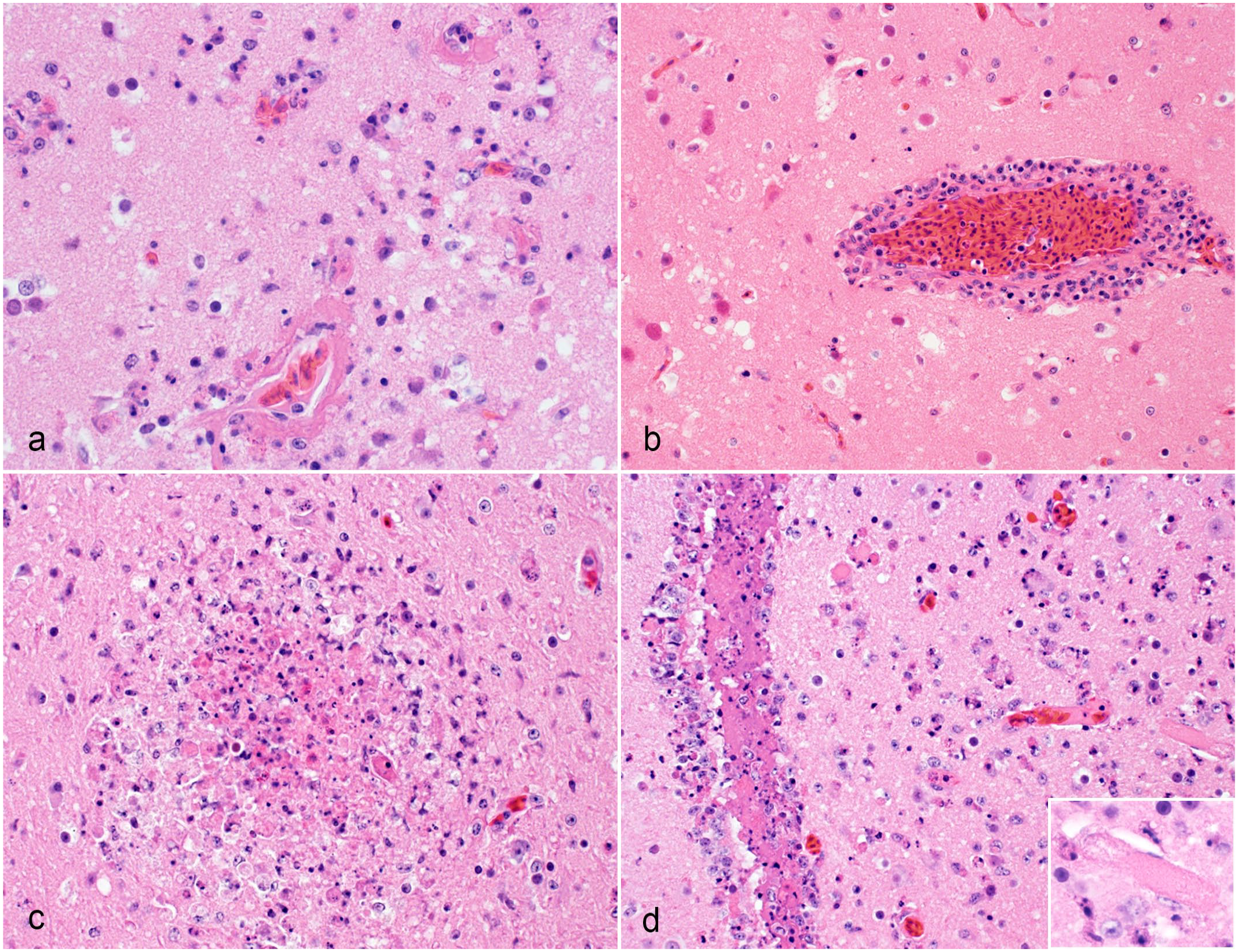
Histological brain lesions in raptors infected with H5 clade 2.3.4.4b highly pathogenic avian influenza virus. Hematoxylin and eosin. (a) Forebrain, great horned owl. Multiple capillaries have fibrinoid necrosis of the walls. They are infiltrated by degenerate granulocytes. The adjacent neuroparenchyma is edematous and hypercellular largely due to the infiltration by granulocytes. Case 20. (b) Forebrain, red-tailed hawk. The wall of a capillary is infiltrated by inflammatory cells and has a prominent perivascular cuff that is largely composed of lymphocytes but also granulocytes and macrophages. The neurons in the adjacent neuroparenchyma are angular and acidophilic (selective acidophilic neuronal necrosis). Case 15. (c) Forebrain, great horned owl. The forebrain has a distinct focus of necrosis. The core of the necrotic focus consists of degenerate granulocytes and macrophages. Case 21. (d) Forebrain, great horned owl. The ependymal lining of a lateral ventricle is disrupted and infiltrated by numerous granulocytes. The ventricular lumen contains eosinophilic homogenous material and debris of granulocytes and macrophages. Several capillaries of the periventricular neuroparenchyma are occluded by thrombi. The neuroparenchyma is hypercellular due to the infiltration by viable and degenerate inflammatory cells, including numerous granulocytes. Inset: A capillary is occluded by a thrombus. Case 18.
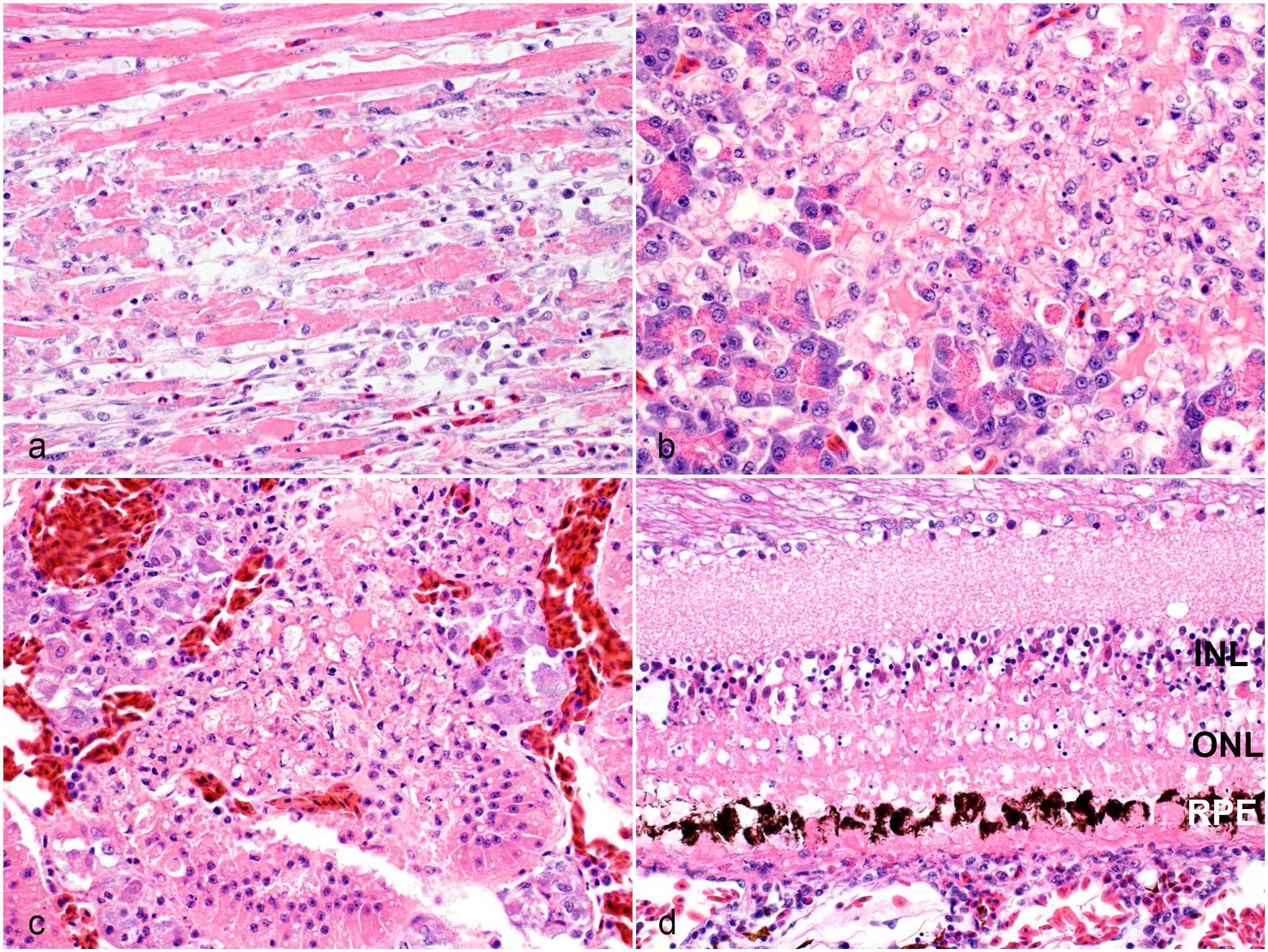
Histological lesions in extraneural organs of raptors infected with H5 clade 2.3.4.4b highly pathogenic avian influenza virus. Hematoxylin and eosin. (a) Heart, red-tailed hawk. A focus of hyaline degeneration and necrosis of cardiomyocytes is infiltrated by few inflammatory cells including macrophages, lymphocytes, and granulocytes. Case 9. (b) Pancreas, red-tailed hawk. The pancreas has an area of necrosis. Case 15. (c) Adrenal gland, great horned owl. The adrenal gland has an area of necrosis that is largely limited to the inter-renal cells. Case 21. (d) Eye, red-tailed hawk. The retinal pigmented epithelial cells (RPE), photoreceptor layer, outer nuclear layer (ONL), outer plexiform layer, and outer portion of the inner nuclear layer (INL) are necrotic. The outer plexiform layer is collapsed due to the necrosis. The choroid is infiltrated by few lymphocytes and granulocytes. Case 9.
Histologic lesions were variably present in the pancreas, adrenal gland, liver, and kidney. Necrosis of acinar cells of the pancreas affected 2 eagles, 3 hawks and 7 owls (Fig. 3b). Necrosis of adrenal gland cells was present in 3 eagles, 2 hawks, and 1 of the 7 owls for which adrenal gland was available for the histologic examination (Fig. 3c). Necrosis of individual hepatocytes or groups of hepatocytes occurred in 2 eagles, 2 hawks and 3 owls. In 1 owl, renal tubular epithelial cells were multifocally necrotic.
Ocular lesions in the form of pectenitis were diagnosed in 1 eagle, 2 hawks, and 2 owls. Furthermore, anterior uveitis was present in 1 eagle, 2 hawks, and 1 owl. One hawk had focal necrosis of the outer segment of the retina (Fig. 3d).
Foci of lytic necrosis were present in the spleens of 2 eagles, 2 hawks, and 3 owls. Histiocytic splenitis, characterized by an increased cellularity of the parenchyma with foamy macrophages or macrophages containing brownish cytoplasmic pigment, was observed in 4 owls.
Immunohistochemistry
The immunoreactivity for influenza A nucleoprotein antigen was nuclear or nuclear and cytoplasmic in the various infected cell types. Target cells in the nervous system were neurons of the brain (4 eagles, 7 hawks, and all owls; Fig. 4a), ependymal cells of the brain (1 eagle, 5 hawks, and all owls; Fig. 4b, c), and ganglion cells and neurons of the inner nuclear layer of the retina (1 eagle, 2 hawks, and 1 owl; Fig. 4d). In extraneural organs, cardiomyocytes (2 eagles, 6 hawks, and 6 owls; Fig. 5a), acinar epithelial cells of the pancreas (1 eagle, 2 hawks, and 7 owls; Fig. 5b), adrenal and/or inter-renal cells of the adrenal gland (3 eagles, 2 hawks, and 1 out of 7 owls; Fig. 5c), and tubular epithelial cells of the kidney (2 eagles, 3 hawks, and all owls; Fig. 5d) were infected. Relatively rarely immunolabled cells in some species were arterial smooth muscle cells and macrophages of the spleen (2 eagles and 3 owls), Mueller cells of the retina (1 eagle, 2 hawks, and 1 owl), and hepatocytes (1 eagle, 1 hawk, and 1 owl). The Mueller cells were identified by their elongated nuclei that form a palisading row at approximately the junction of the inner and middle thirds of the inner nuclear layer of the retina. 41
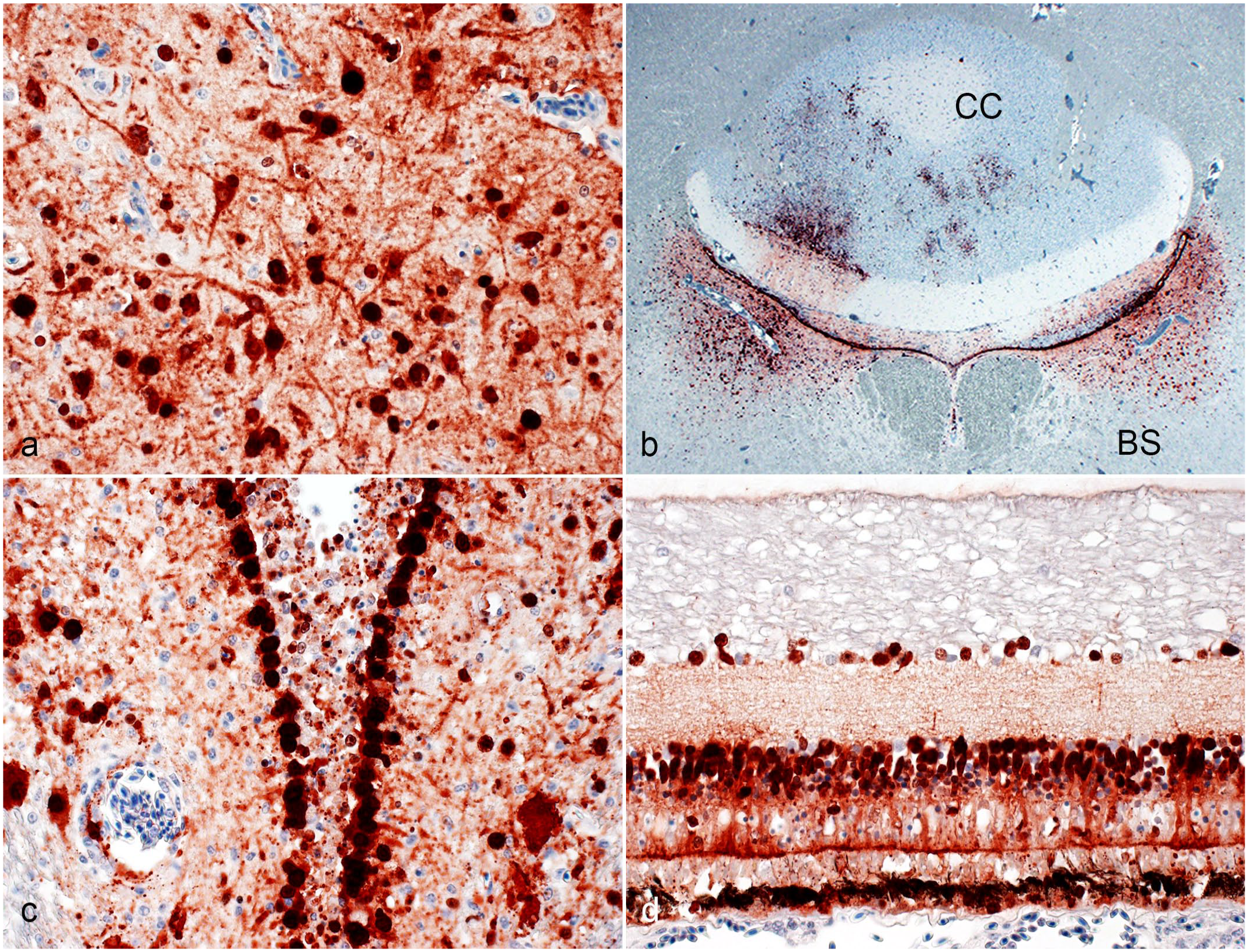
H5 clade 2.3.4.4b highly pathogenic avian influenza virus infection of the nervous system in raptors. Immunohistochemistry for nucleoprotein of influenza A virus. (a) Forebrain, red-tailed hawk. The nuclei and bodies of numerous neurons and their processes in the forebrain are strongly immunopositive. Case 8. (b) Cerebellum and brainstem, red-tailed hawk. The ependymal cells lining the 4th ventricle are strongly immunopositive. Neurons in 2 bilateral symmetric brainstem (BS) nuclei and of the cerebellar cortex (CC, including Purkinje cells and neurons of the granule layer of the folium adjacent to the 4th ventricle) are immunopositive. Case 8. (c) Forebrain, great horned owl. The nuclei and cytoplasm of the ependymal cells lining the 3rd ventricle are strongly immunopositive. Cell processes and the nuclei and bodies of numerous neurons in the periventricular neuroparenchyma are immunopositive. Forebrain. Case 20. (d) Eye, red-tailed hawk. The cytoplasm and nuclei of pigmented epithelial cells as well as the nuclei and cytoplasm of neurons of the inner nuclear layer and ganglion cell layer are immunopositive. Case 9.
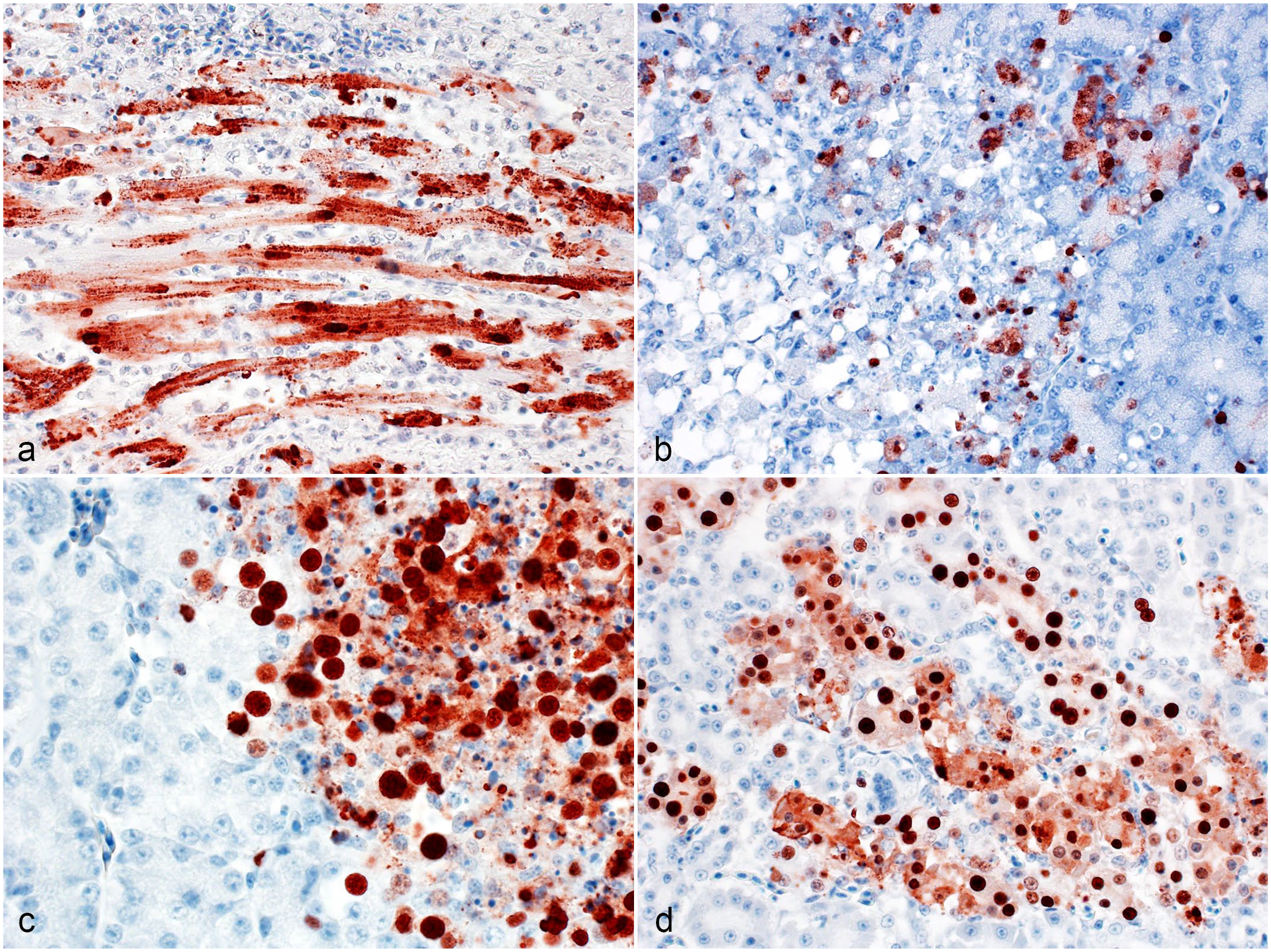
H5 clade 2.3.4.4b highly pathogenic avian influenza virus infection of extraneural organs in raptors. Immunohistochemistry for nucleoprotein of influenza A virus. (a) Heart, great horned owl. The nuclei and sarcoplasm of numerous cardiomyocytes are strongly immunopositive. Case 18. (b) Pancreas, great horned owl. The nuclei and cytoplasm of several acinar cells near an area of necrosis are immunopositive. Case 21. (c) Adrenal gland, red-tailed hawk. The nuclei and cytoplasm of adrenal and/or inter-renal cells near an area of necrosis are immunopositive. Case 14. (d) Kidney, great horned owl. The nuclei and cytoplasm of the epithelial cells of several renal tubules are immunopositive. Case 21.
Eagles
With the exception of 1 eagle (case 4), the quantity of viral antigen was usually low in the eagles with a more limited tissue distribution than in the owls (Table 1, Supplemental Table S2). One of the eagles was negative for influenza A nucleoprotein antigen in all examined tissue sections (case 3).
Hawks
The brains of 7 of the 9 hawks were moderately or markedly influenza A virus antigen positive (Table 1, Supplemental Table S2). Approximately 40% of the forebrain area was positive in 1 case (case 15). The degree of immunopositivity of 6 of the 9 hearts varied between mild and marked. Approximately 50% of the myocardium of 1 hawk was viral antigen positive (case 9). One hawk was negative for viral antigen in all tissues examined (case 12). Pancreas, adrenal gland, liver, eye and kidney were only positive for viral antigen in few hawks. Viral antigen was not detected in the spleen of any of the hawks.
Owls
Influenza A virus antigen was abundant with a widespread organ distribution in the owls (Table 1, Supplemental Table S2). The brains of all owls were positive for influenza A nucleoprotein antigen. Approximately 20% to 25% of the forebrain area was positive in 4 owls while in 3 more cases the positive areas comprised approximately 40% to 75% of the forebrain. The hearts of 6 owls were positive for influenza virus antigen. In 3 of these 6 owls, approximately 20% of the myocardium harbored viral antigen positive cardiomyocytes, while approximately 50% of the myocardium of 1 more of these 6 owls was positive for viral antigen. Renal tubular epithelial cells were positive in all owls. Acinar cells of the pancreas were positive in 7 owls. With 1 exception (case 21), the adrenal gland of owls was negative for viral antigen. Few cells, morphologically resembling macrophages, were positive in the spleens of 2 owls, while few smooth muscle cells of an artery were positive in the spleen of another owl. Few hepatocytes adjacent to a focus of hepatocellular necrosis were positive in 1 owl. Few neurons of the ganglion cell layer and inner nuclear layer of the retina were positive in 1 owl.
RT-PCR
Oropharyngeal swabs from all birds were positive for the matrix gene of influenza A virus and H5 (Supplemental Tables S3 and S4). The whole genome sequence data of the viruses infecting 3 eagles, 7 hawks, and 5 owls were obtained. All birds were infected with EA/AM 2.3.4.4b H5N1 representing 5 different reassortant genotypes including B1.1, B1.2, B2, B3.1, and B3.2 (Supplemental Table S3).
Discussion
This study describes gross, microscopic, and immunohistochemical findings in a wide variety of organs of bald eagles, red-tailed hawks, and great horned owls that were positive for gs/GD H5 clade 2.3.4.4b HPAIV. Clinically, neurologic signs were hallmark findings in these birds, and lesions and viral antigen were most consistently and most abundantly detected in the brain across the 3 species. Viral antigen had a more widespread tissue antigen distribution in the owls compared with eagles and hawks.
The 2022 H5N1 clade 2.3.4.4b event in North America is distinct from the 2015 outbreak with gs/GD H5 clade 2.3.4.4c HPAIV in the overall number of deaths among wild birds, and consequently, the amount of virus in the flyways as represented by frequency of detection of deceased animals.37,38 In addition, the 2015 wild bird mortality events occurred largely along the Pacific flyway, where the virus was introduced to North America, with few detections in the Central and Mississippi flyways, and no detections in the Atlantic flyway, while in 2022, wild bird mortality events occurred from coast to coast across all 4 flyways.37,38 The genotypes detected among the 23 raptors of our study and across North America are consistent with the genotypes detected in waterfowl populations and sporadic spillover events in mammals from the upper Midwest of the United States of America. This suggests that the raptor cases are spillover infections from prey rather than sustained transmission events.2,11
Gross lesions were rare in our study. This finding is consistent with previous studies of raptors naturally and experimentally infected with HPAIVs.5,8,9,14,18,24,26,29,34,39 Splenic lesions have been commonly reported in HPAIV-infected raptors, but were rare in our study. 29 Euthanasia-related splenic congestion and enlargement may have masked virus induced splenic enlargement and subtle splenic lesions. Pancreatic necrosis was a feature of infection of raptors during previous outbreaks of highly pathogenic influenza. 26 The foci of pancreatic necrosis are reminiscent of the pancreatic lesions described in birds with velogenic avian paramyxovirus-1 infection (Newcastle disease virus). 40 Notably, gross lung lesions, such as pulmonary congestion, edema, and hemorrhage or overt pneumonia, were not detected in any of the examined raptors of our study but have been described in raptors in previous studies.26,29
All birds had histologic lesions in the brain and the majority of birds had cardiac lesions. Pancreatic, adrenal, and ocular lesions were present in a subpopulation of the birds. Lesions found in the brain, heart, pancreas, and adrenal glands have been previously reported in HPAIV-infected raptors.5,9,14,18,24,26,29,34 While lesions occurred in all species, they tended to be more severe in the owls of our study, and were fairly subtle in some of the eagles. The small number of individuals of each of the 3 examined species did not allow for a comprehensive analysis of trends. Influenza A virus-associated encephalitis, myocarditis, and pancreatic necrosis are extremely rare occurrences in humans, while adrenal gland necrosis has not been described in humans.17,19,20,21 Pulmonary lesions are fairly common in humans with an aggravated course of influenza but were absent at the microscopic level in any of the birds of our study.17,19,20
The pattern of the brain lesions was rather unique compared with the lesions of other viral encephalitidies in raptors. The unique aspects included (1) an emphasis on forebrain involvement, (2) capillary necrosis with pan-necrosis (ischemic necrosis/infarction) of the neuropil, (3) a granulocytic component of the cellular infiltrate, and (4) a prominent ependymitis. When present, the combination of these brain lesions, particularly the presence of foci of pan-necrosis with a prominent granulocytic component and the ependymitis, allows for the distinction of HPAIV induced brain lesions from brain lesions induced by WNV, a major differential diagnosis for raptors with neurologic disease in the midwestern United States during the mosquito season.42,43,45 Furthermore, adrenal and pancreatic necrosis are not described in eagles, hawks, and great horned owls with WNV infection.42,43,45 In contrast, the gross and microscopic cardiac lesions of H5 2.3.4.4b HPAIV are morphologically similar to WNV-associated myocarditis.42,43,45 Ocular lesions were rare in birds in this study but are common in WNV-infected hawks and eagles.28,41,42,43,45 The necrotizing character of HPAIV-induced lesions has previously been described.5,8,9,14,18,24,26,29,34,39 Ependymitis was found in a peregrine falcon naturally infected with HPAIV and experimentally infected gyr-saker falcon hybrids but has not been described in owls, hawks, or eagles.5,8,9,29,41
Influenza A virus antigen was detected in at least one of the examined organs (brain, heart, pancreas, adrenal gland, kidney, spleen, eye, and liver) in all great horned owls, all but 1 bald eagle, and all but 1 red-tailed hawk of our study. Attachment of hemagglutinin to the target cell via sialic acid receptors is a pre-requisite of the viral infection. 20 HPAIVs and low pathogenic avian influenza viruses differ at the hemagglutinin cleavage site motif. Low pathogenic avian influenza viruses with their monobasic hemagglutinin cleavage site bind exclusively to epithelial cells of the alimentary and respiratory tract, while HPAIVs with their multibasic hemagglutinin cleavage site bind to a wide variety of target cells, including endothelial cells.7,25 In gyr-saker falcon hybrids, possibly suitable sialic acid cell surface receptors have been identified in the nasal cavity, trachea, bronchi, rectal enterocytes, endothelial cells, macrophages, and lymphocytes based on lectin histochemistry. 5 Based on morphologic findings, small and large neurons, including Purkinje cells and ependymal cells, appear to be target cells of gs/GD H5 clade 2.3.4.4b HPAIV in the brain and eyes of the raptors of our study. These cells sustain viral replication as evidenced by the expression of viral nucleoprotein. Double-labeling studies and cell culture studies will be necessary to investigate whether glial cells including astrocytes and oligodendrocytes also can become infected. Cardiomyocytes and exocrine cells of the pancreas also are susceptible to viral infection and express viral antigen. Cells of adrenal gland can be infected with HPAIV, but it is unclear whether HPAIV has a tropism for adrenal cells or inter-renal cells (or both). In addition, viral antigen was detected in renal tubular epithelial cells, most consistently in the owls of our study. The tropism of HPAIVs for neurons, cardiomyocytes, exocrine pancreas cells, adrenal gland cells, and renal tubular epithelial cells has been demonstrated in previous studies of naturally and experimentally infected raptors.5,8,9,14,18,24,26,29,34,39
Raptors, similar to terrestrial carnivores, likely become infected when they ingest infected prey or infected carcasses.2,5,9,11,15 The presence of lesions and viral antigen in the proventriculus of 2 buzzards naturally infected with an H5N8 HPAIV, and successful experimental infection of falcons by feeding infected meat serve as evidence supporting this assumption.5,9 However, the esophagus, proventriculus, ventriculus, and intestines did not have any significant lesions in any of the examined birds of our study nor was viral antigen detected in the mucosa at these sites. Alternatively, the respiratory route may be suspected, but neither lesions nor viral antigen were present in trachea or lungs in the raptors of our study. Unfortunately, the heads (nasal and oral cavity) of the birds were not examined histologically and were destroyed along with all other fixed samples from the birds shortly after confirmation of HPAIV infection, according to the Federal Select Agent Program regulations and the regulations implemented by the University of Minnesota Biosafety and Occupational Health Department. Viral antigen was demonstrated via immunohistochemistry in the nasal cavity, trachea, and gastrointestinal tract in experimentally infected gyr-saker falcon hybrids. 24 Once the virus enters the host, it likely spreads within the body hematogenously. Endothelial cell infection is a key feature of the pathogenesis of birds with HPAIV.20,25 Endothelial cell infection has been demonstrated by immunohistochemistry in gallinaceous birds, waterfowl, and raptors.16,29,33 The brain infection and the spread of the infection within the brain may have occurred hematogenously in the birds of our study. The presence of vascular necrosis in the brains of quite a few of the cases supports this notion. However, endothelial cell infection or infection of circulating blood cells was not apparent in neural or extraneural tissue in any of the cases of our study. It is possible that the phase of viremia and endothelial cell infection is only short-lived in raptors with HPAIV infection and has ended by the time the birds are moribund. Alternatively, a neurogenic infection of the brain may be considered. The olfactory route is a plausible scenario. In most birds, apart from vultures, kiwis, and members of the order Procellariiformes, the olfactory system has historically been considered to be negligible, but more recently, it has been recognized that the olfactory system plays an important role (eg, for reproduction) even in birds that heavily rely on the visual system rather than their sense of smell. 4 Based on the prominent involvement of the ventricular system in the present cases, we speculate that the cerebrospinal fluid plays a role in disseminating the virus within the central nervous system. The observation that areas of selective neuronal necrosis and pan-necrosis in the brainstem and cerebellum were closely associated with the ventricular system in some cases supports this speculation. In chickens infected with H7N1 HPAIV, encephalitis developed 1 to 2 days after intranasal inoculation. The encephalitis had a bilateral symmetrical pattern, and endothelial cells, neurons, and ependymal cells were infected. 10 The authors concluded that the virus infected the brain hematogenously and that the virus spreads early throughout the brain via the cerebrospinal fluid. Ependymal cells positive for HPAIV antigen were also reported in 1 naturally infected peregrine falcon and experimentally infected kestrels and gyr-saker falcon hybrids.5,34,39
In conclusion, bald eagles, red-tailed hawks, and great horned owls naturally infected with gs/GD H5 clade 2.3.4.4b HPAIV may develop severe neurologic disease. Affected birds with severe neurologic signs commonly have lesions in brain and heart and occasionally have lesions in pancreas, adrenal gland, eye, liver, and spleen. The presence of ependymitis in most of the raptors along with the presence of lesions in pancreas and adrenal gland helped to distinguish HPAI from WNV infection in our study from a diagnostic pathology standpoint. PCR positive oropharyngeal swabs, and in many cases the detection of viral antigen in the brain and other tissues, confirmed that the birds were infected with influenza A virus. Serological studies will be beneficial in understanding how commonly bald eagles, red-tailed hawks, and great horned owls survive the infection.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231222227 – Supplemental material for Lesions and viral antigen distribution in bald eagles, red-tailed hawks, and great horned owls naturally infected with H5N1 clade 2.3.4.4b highly pathogenic avian influenza virus
Supplemental material, sj-pdf-1-vet-10.1177_03009858231222227 for Lesions and viral antigen distribution in bald eagles, red-tailed hawks, and great horned owls naturally infected with H5N1 clade 2.3.4.4b highly pathogenic avian influenza virus by Arno Wünschmann, Dana Franzen-Klein, Mia Torchetti, Michele Confeld, Michelle Carstensen and Victoria Hall in Veterinary Pathology
Footnotes
Acknowledgements
We are indebted to the public for submitting the cases, to the staff of The Raptor Center of the University of Minnesota for caring for the animals, to the necropsy staff, histology lab, Ellie LaChance from the immunohistochemistry lab, and the PCR lab of the Minnesota Veterinary Diagnostic Laboratory and finally many at the National Veterinary Services Laboratories for their dedication and invaluable technical expertise. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official U.S. Department of Agriculture (USDA) or U.S. Government determination or policy.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
