Abstract
Although tick infestation is a significant health problem in livestock, there are limited studies on the dermatopathological aspects of natural tick infestation in cattle. This study aimed to describe the gross and histologic aspects of cutaneous lesions caused by tick infestation in cattle. Thirteen cases were selected based on necropsy data from a 10-year retrospective study. Predispositions were observed in beef cattle (P = .049) and the Angus breed (P = .012), and lesions occurred mainly in the fall (P = .007). Gross lesions included hypotrichosis (13/13; 100%), scales (12/13; 92%), alopecia (11/13; 85%), ulcers (7/13; 54%), crusts (7/13; 54%), and erosions (2/13; 15%). These gross lesions were mainly located in the thorax (12/13; 92%), head (11/13; 85%), abdomen (10/13; 77%), neck (9/13; 69%), limbs (9/13; 69%), and perineum (9/13; 69%). Histologically, all cases had ticks adhered to the epidermis with erosions (13/13; 100%), ulcers (11/13; 85%), orthokeratotic hyperkeratosis (13/13; 100%), irregular acanthosis (13/13; 100%), intraepidermal pustules (13/13; 100%), crusts (10/13; 77%), and ballooning degeneration (4/13; 31%). In the dermis, just below the tick insertion site, there was coagulation necrosis, fibrin deposition, and inflammatory infiltrate composed of mixed cells (neutrophils, lymphocytes, plasma cells, macrophages, and few eosinophils) (9/13; 69%), neutrophils (3/13; 23%), or eosinophils (1/13; 8%). This study reinforces the different patterns of cutaneous lesions caused by tick infestation in cattle, which should be considered as a potential cause of dermatitis in this species.
Ticks are obligate hematophagous ectoparasites of vertebrate animals and are considered important vectors of several bacterial, viral, protozoal, and rickettsial agents that cause diseases in humans and animals. 25 These ectoparasites belong to the phylum Arthropoda, class Arachnida, and order Ixodida and are divided into the families Argasidae (soft ticks) and Ixodidae (hard ticks). 16 The Ixodidae family comprises most species of ticks of veterinary importance and that are pathogenic for ruminants. 5 Among them, Rhipicephalus (Boophilus) microplus (popularly known as cattle tick, Southern cattle tick, or Asian blue tick) is the most important species for livestock, especially in subtropical and tropical areas. 12 In addition to being responsible for transmitting pathogens, high R. microplus infestation in cattle can also result in economic losses due to a significant reduction in the weight and productivity of affected animals, spoiling anemia, and condemnation of leather at slaughter. 16
Tick bites can cause different local reactions to the skin at the site of attachment, including direct lesions and predisposition to secondary bacterial infections and myiasis.9,13,16 The gross and histologic pattern of local cutaneous reaction depends on biting time, host factors, and tick properties, such as salivary secretions.13,14 The occurrence of these lesions in the skin is described in humans and animals.9,14,17,18,21,22,24 However, despite the widespread tick infestation in livestock worldwide, few studies characterize the dermatopathological aspects of natural tick infestation in cattle.9,17 Therefore, the aim of this study was to describe the gross and histologic aspects of cutaneous lesions caused by tick infestation in cattle.
Materials and Methods
Case Selection
Electronic reports of the cattle submitted to necropsy between January 2013 and February 2023 to the Department of Veterinary Pathology at the Universidade Federal do Rio Grande do Sul were searched for cases with cutaneous lesions caused by tick infestation. We included all cattle with gross and histologic descriptions of ticks attached to the skin, regardless of the anatomical location or tick life stage. Cases without ticks in histologic sections or with dermatitis associated with other infectious and parasitic agents were excluded. Some farms were visited for on-site observation, and the animals were euthanized mainly due to causes unrelated to cutaneous lesions. Information retrieved from the records of the selected cases included signalment (sex, breed, and age [less than or equal to 24 months or greater than 24 months]), breed aptitude (dairy or beef), clinical history of parasitic cutaneous lesions, geographical region of origin, cause of death, and season in which the necropsy was performed (spring, summer, fall, or winter). Data on the signalment, breed aptitude, and season of all cattle submitted for necropsy during the analyzed period were also obtained for comparative purposes.
Gross Evaluation
Necropsy records and photographs of the selected cases were re-evaluated for the presence of gross cutaneous lesions. These lesions were classified according to the anatomical site (head, neck, thorax, abdomen, limbs, back, perineum, scrotum, and tail), distribution (focal, multifocal, multifocal to coalescent, focally extensive, or diffuse), morphological pattern, and intensity of tick infestation. Based on current literature, 25 the morphological pattern of the gross lesions was classified as hypotrichosis (partial hair loss), alopecia (complete hair loss), scale (irregular, thin, dry, white fragments on the skin surface), erosion (shallow, moist, glistening depression of the epidermis), ulcer (depression of the epidermis with variable exposure of the dermis and subcutaneous tissue), crust (dry exudate, including fluid, blood, pustular debris, and scale, on the skin surface), pustule (superficial, small, circumscribed accumulation of purulent fluid within the epidermis), papule (elevated, firm, circumscribed area less than 1 cm in diameter), and nodule (elevated, often-firm, circumscribed, solid area greater than or equal to 1 cm in diameter). The intensity of tick infestation, determined by manually counting the number of ticks in all affected sites, was classified as mild (1–20 ticks), moderate (21–99), or marked (greater than or equal to 100).
Histologic Evaluation
Formalin-fixed paraffin-embedded tissue blocks of the selected cases were recovered from the archive. Skin sections stained with hematoxylin and eosin and by Luna’s stain were re-examined by 3 pathologists (SMPM, IRS, and SPP). Luna’s stain was performed to identify eosinophil granules. In cases with more than one section of skin, the slide with the most intense lesion was considered. Although all histologic sections evaluated were haired skin, the exact anatomical site of the collection was not available in some cases.
In the epidermis, the presence or absence of hyperkeratosis (thickening of the stratum corneum), acanthosis (thickening of the stratum spinosum), ballooning degeneration (intracytoplasmic vacuoles in epidermal cells), pustule (accumulations of neutrophils within the epidermis), erosion (partial loss of the epidermis keeping the basement membrane intact), ulcer (full loss of the epidermis and basement membrane), and crust (dried fluid and cellular debris on the epidermal surface) were evaluated. The type of hyperkeratosis was classified as orthokeratotic or parakeratotic, according to the loss or retention of keratinocyte nucleus, respectively. Acanthosis was classified as regular, irregular, papillary, or pseudocarcinomatous, according to the growth pattern of the epidermis. The intensity of both hyperkeratosis and acanthosis was evaluated by comparing the thickness of the adjacent epidermis without alteration and classified as mild (thickness less than 50% of the normal epidermis), moderate (thickness equal to 50%), or marked (thickness greater than 50%).
The dermis was evaluated for the presence or absence of inflammatory infiltrate, proliferation and ectasia of blood vessels, hemorrhage, pigmentary incontinence (melanin within macrophages or free in the upper dermis), and collagen degeneration (brightly eosinophilic, granular to amorphous material bordering the fibers). The inflammatory infiltrate was also classified according to the cell composition, location (superficial dermis, deep dermis, hair follicles, and perivascular location), distribution (focal, multifocal, multifocal to coalescent, focally extensive, and diffuse), and intensity. The inflammatory cells in the hair follicle were sub-classified as perifolliculitis, mural folliculitis, intraluminal folliculitis, and furunculosis. The intensity of the inflammatory infiltrate was determined by manual counting in a 0.237-mm2 area, using the field with the highest cell density, and classified as absent (less than 20 mononuclear cells, predominantly in perivascular areas without other histologic changes), mild (21–70 cells), moderate (71–150 cells), and marked (greater than 150 cells). The extension of these lesions to adjacent soft tissues was also evaluated.
Finally, the pattern of the inflammatory cutaneous lesions of each case was classified as (1) nodular to diffuse dermatitis, (2) perivascular/interstitial dermatitis, (3) folliculitis and furunculosis, (4) cytotoxic (interface) dermatitis, (5) intraepidermal vesicular/pustular dermatitis, (6) subepidermal vesicular/pustular dermatitis, (7) vasculitis, and (8) panniculitis based on the veterinary dermatopathology literature. 1
Statistical Analysis
The results were initially evaluated by descriptive statistics. The association between the signalment, breed aptitude, and season groups and the occurrence of tick infestation and cutaneous lesions due to tick infestation were evaluated by Pearson chi-square or Fisher’s exact tests, depending on characteristics of the contingency tables. A pairwise z test with Bonferroni correction was applied to discriminate the differences between the groups when the Pearson chi-square or Fisher’s exact tests were statistically significant. A P value <.05 was regarded as statistically significant. Analyses were performed using IBM SPSS Statistics for Windows, version 22.0 (International Business Machines Corporation, https://ibm.com).
Results
Cases Data
Of the 929 cattle submitted for necropsy during the study period, 182 (19.5%) had descriptions of tick infestation in the necropsy reports. The data on signalment, breed aptitude, and season of occurrence of these cases are available in Table 1. Thirteen cases (1.4%) were selected for the pathological study based on the inclusion criteria. The animals were from the metropolitan region of Porto Alegre, Rio Grande do Sul, South of Brazil. Selected 13 animals included 9 (69%) female and 4 (31%) male cattle, with ages ranging from 4 months to 6 years (62% [8/13] were less than or equal to 24 months of age, and 38% [5/13] were greater than 24 months of age). All cattle were beef breed, mainly Angus (10/13; 77%), followed by Brangus (1/13; 8%), Hereford (1/13; 8%), and mixed-breed (1/13; 8%). The tick infestations were more reported in animals necropsied in the fall (8/13; 61%), followed by the summer (4/13; 31%) and the spring (1/13; 8%). Based on the expected proportion of cattle submitted to necropsy in our department, there were significant statistical differences for the occurrence of cutaneous lesions caused by ticks in beef cattle, Angus breed, and the fall (Table 1). Unfortunately, clinical data about the temporal evolution of these cutaneous lesions were not available. Only three cases described similar cutaneous lesions associated with the tick infestation in other cattle from the same farms. No cattle had a cause of death directly associated with the cutaneous lesions. Individual data regarding signalment, breed aptitude, cause of death, and season in which cattle were observed with cutaneous lesions are available in Supplemental Table S1.
Association between signalment and cattle with tick infestation and cattle with cutaneous lesions caused by tick infestation. c
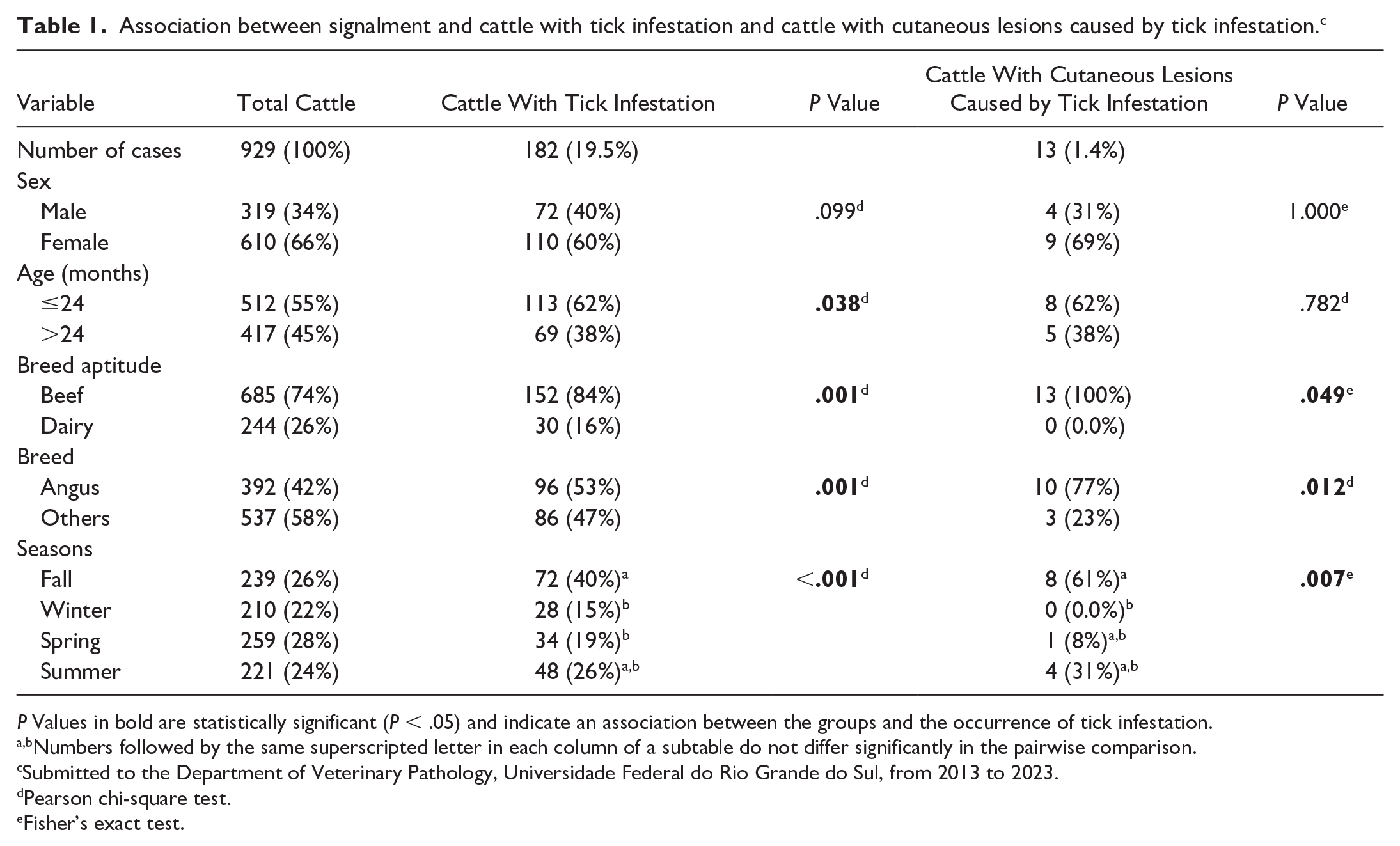
P Values in bold are statistically significant (P < .05) and indicate an association between the groups and the occurrence of tick infestation.
Numbers followed by the same superscripted letter in each column of a subtable do not differ significantly in the pairwise comparison.
Submitted to the Department of Veterinary Pathology, Universidade Federal do Rio Grande do Sul, from 2013 to 2023.
Pearson chi-square test.
Fisher’s exact test.
Gross Findings
The gross lesions were multifocal (7/13; 54%) or multifocal to coalescent (6/13; 46%) and were characterized by hypotrichosis (13/13; 100%), scales (12/13; 92%), alopecia (11/13; 85%), ulcers (7/13; 54%), crusts (7/13; 54%), and erosions (2/13; 15%). These lesions were located mainly on the thorax (12/13; 92%), head (11/13; 85%), abdomen (10/13; 77%), neck (9/13; 69%), thoracic and pelvic limbs (9/13; 69%), and perineum (9/13; 69%). Less frequently affected sites included the dorsum (1/13; 8%), scrotum (1/13; 8%), and tail base (1/13; 8%). On the head, the lesions were predominantly on the lateral regions of the face (Fig. 1a) and the internal and external regions of the ears (Fig. 1b). In the neck, thorax, and abdomen, lesions were observed mainly in ventral areas, such as in the dewlap (Fig. 1c) and axillary and inguinal regions (Fig. 1d). In these cattle, the tick infestation was observed at different stages of development and with intensity ranging from moderate (10/13; 77%) to marked (3/13; 23%). Individual data regarding the gross lesions are available in Supplemental Table S2.
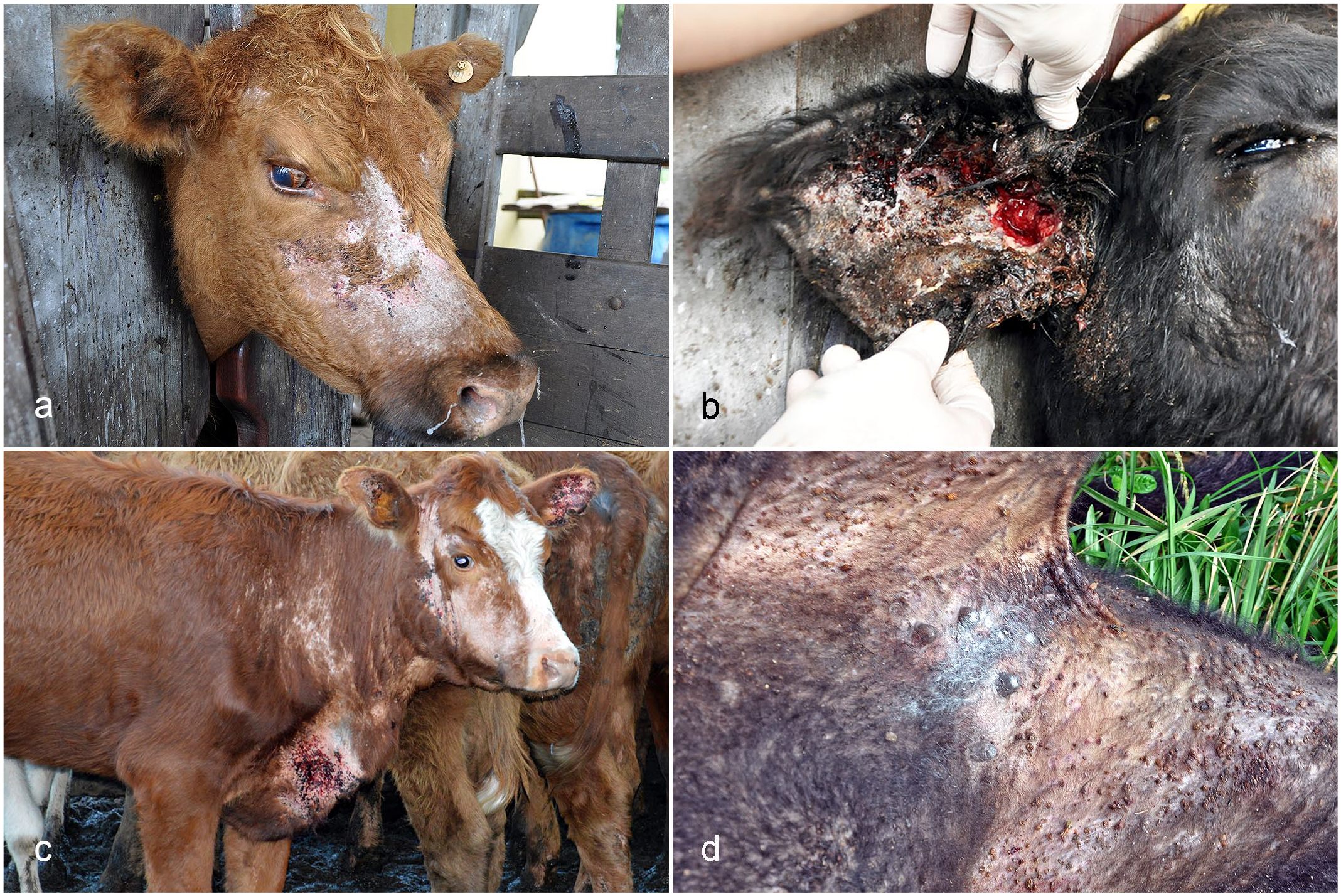
Tick bite dermatitis, skin, cattle. (a) Skin on the lateral region of the face with hypotrichosis, alopecia, scales, ulcers, crusts, and ticks. (b) Skin inside the ear with hypotrichosis, ulcers, crusts, and ticks. (c) Skin on the dewlap, lateral region of the face, and ears with hypotrichosis, alopecia, ulcers, crusts, and ticks. (d) Skin in the inguinal region with hypotrichosis, crusts, and ticks.
Histological Findings
The main histologic characteristic was the epidermis penetrated by the tick’s mouthparts (chelicerae and hypostome). The mouthparts were located within a vertical tube that crossed the epidermis and often reached the subjacent dermis, forming a feeding cavity (Fig. 2a) with erosions (13/13; 100%) and ulcers (11/13; 85%). The tube was composed of a highly eosinophilic, amorphous material (external cement), firmly adhered to the top of the epidermis, and had serrated structures in the inner surface (internal cement) (Fig. 2b, c). These serrated structures were probably formed by the shape of the tick’s hypostome teeth. The external cement spreads over the surface of the adjacent epidermis, becoming less thick distally (wing-like part of the external cement). In most cases, the tick’s body (idiosoma) or body part was present above the mouthparts. The idiosoma was characterized by oval structures (2.4–7.9 mm in length), delineated by a thick, hypereosinophilic outer layer (highly chitinized body wall), and hemocoel filled by internal structures (salivary glands, tracheal ring, striated muscle, reproductive system that sometimes contained eggs, and a digestive system with digested blood) (Fig. 2d). The adjacent epidermis had orthokeratotic hyperkeratosis (13/13; 100%), irregular acanthosis (13/13; 100%) (Fig. 3a), intraepidermal pustules (13/13; 100%) (Fig. 3b), crusts (10/13; 77%) (Fig. 3c), and ballooning degeneration (4/13; 31%). Both hyperkeratosis and acanthosis were predominantly moderate (6/13; 46%), followed by mild (4/13; 31% [hyperkeratosis] and 5/13; 38% [acanthosis]) and severe (3/13; 23% [hyperkeratosis] and 2/13; 15% [acanthosis]).
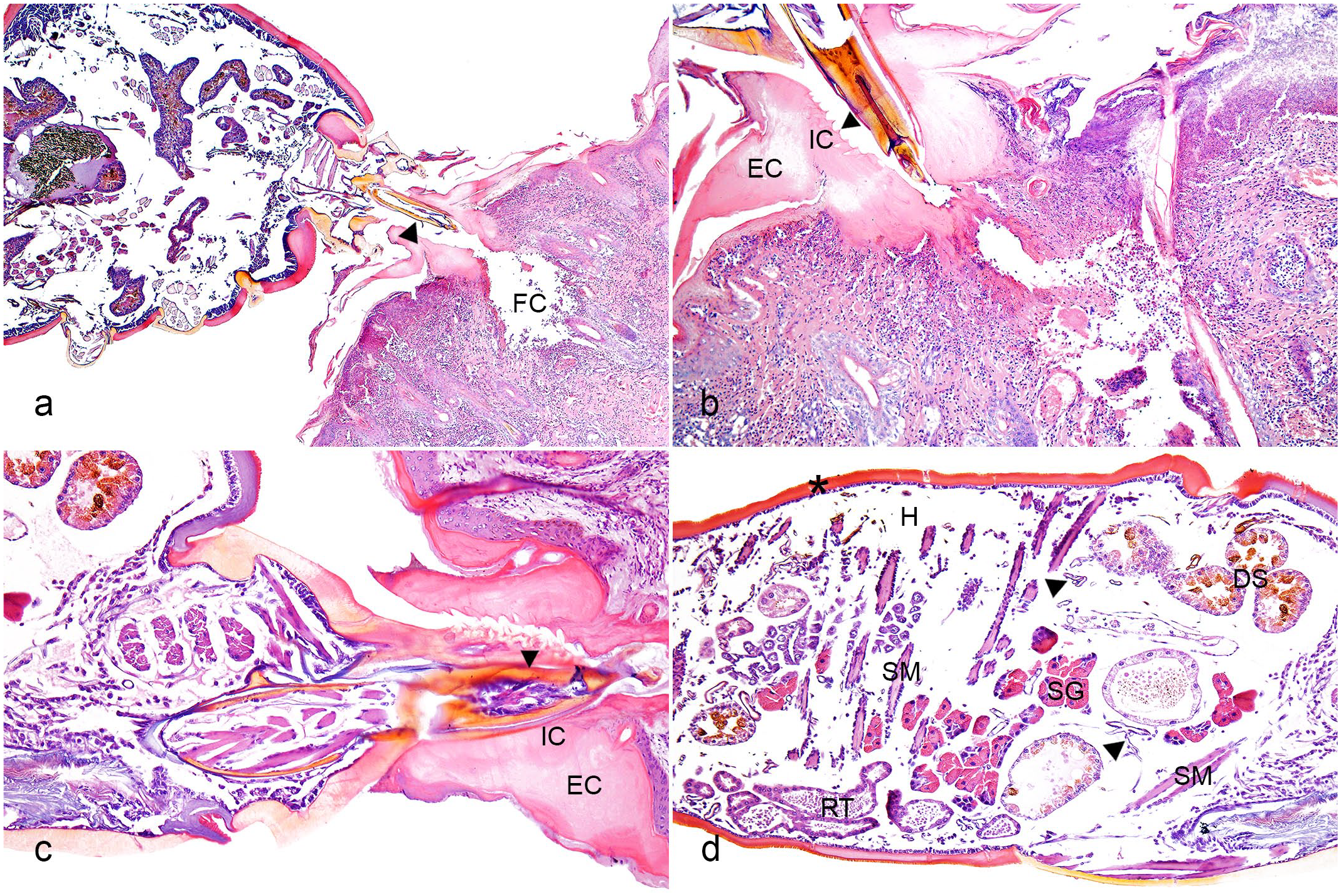
Tick bite dermatitis, skin, cattle. Hematoxylin and eosin. (a) Tick chelicerae and hypostome (arrowhead) located within a vertical tube that traversed the epidermis and reached the underlying dermis, forming a feeding cavity (FC). (b) Vertical tube composed of external cement (EC) firmly adhered to the surface of the epidermis and forming the wing-like part over the adjacent epidermis. The internal cement (IC) had serrated structures (arrowhead) on the inner surface. There is coagulative necrosis with fibrin deposition, cell debris, and inflammatory infiltrates in the dermis just below the tick attachment site. (c) The anterior portion of the tick with chelicerae and hypostome (arrowhead) inside a conical tube of external (EC) and internal cement (IC). (d) Tick idiosome lined by a highly chitinized (asterisk) body wall and hemocoel (H) filled by longitudinal and cross-sections of salivary glands (SG), tracheal ring (arrowhead), striated muscle (SM), reproductive tract (RT), and digestive system (DS).
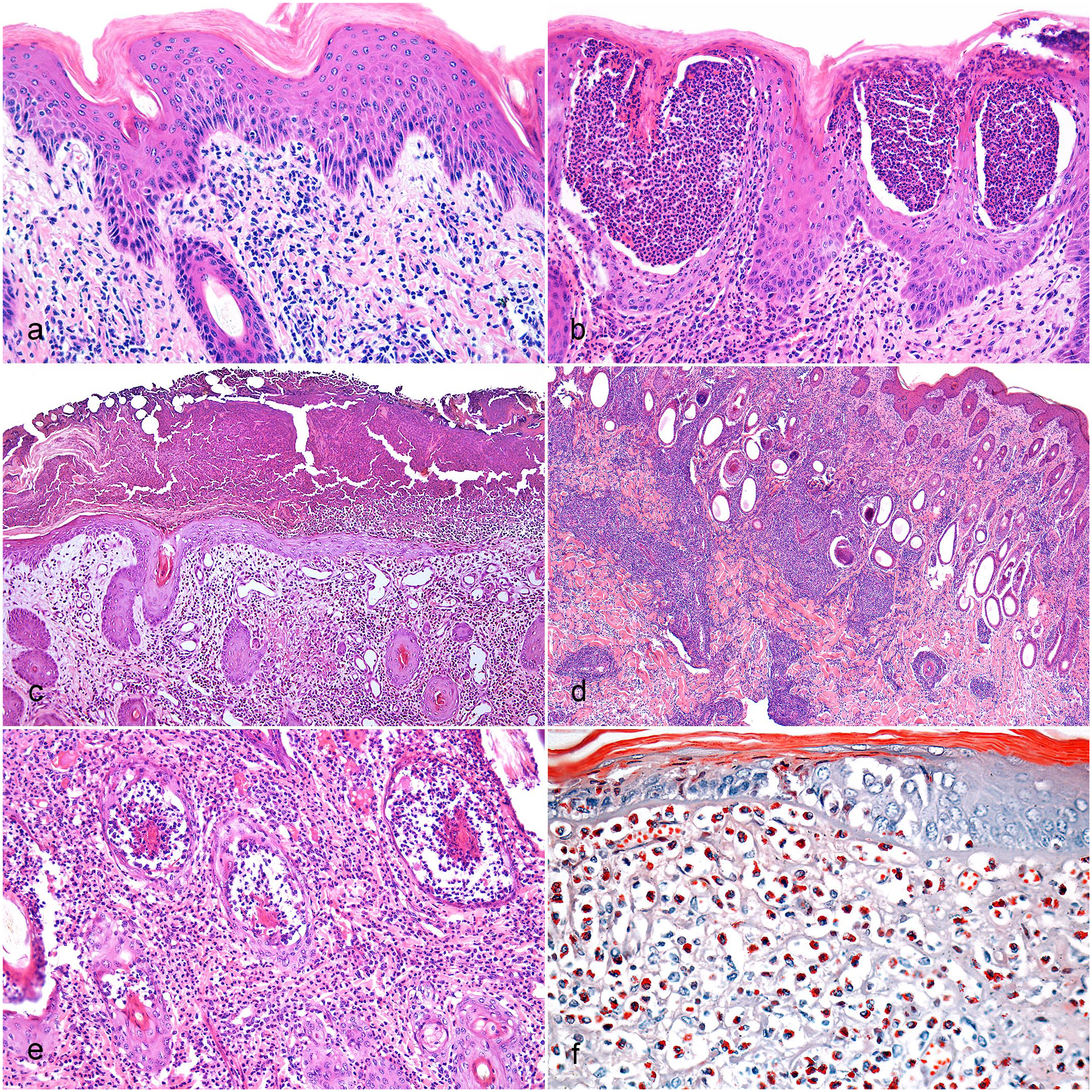
Tick bite dermatitis, skin, cattle. (a) Epidermis with irregular acanthosis and orthokeratotic hyperkeratosis. Hematoxylin and eosin (HE). (b) Epidermis with multifocal pustular areas containing intact and degenerated neutrophils. HE. (c) Epidermis covered by crusts formed by cell debris interspersed and eosinophilic amorphous material. Dermis with mixed inflammatory infiltrates, fibrin deposition, and proliferation and ectasia of blood vessels. HE. (d) Mixed inflammatory infiltrates in the superficial dermis, deep dermis, and perivascular area. HE. (e) Neutrophilic inflammatory infiltrates in the superficial dermis and affecting hair follicles (mural folliculitis). HE. (f) Eosinophilic inflammatory infiltrates in the superficial dermis with positive cytoplasmic granules. Luna’s stain.
In the dermis just below the sites of tick attachment, there was coagulative necrosis with fibrin deposition, cell debris, and inflammatory infiltrates. Inflammatory infiltrates were mainly mixed (neutrophils, lymphocytes, plasma cells, macrophages, and few eosinophils) (9/13; 69%) (Fig. 3d) and less frequently composed of only neutrophils (3/13; 23%) (Fig. 3e) or eosinophils (1/13; 8%) (Fig. 3f). Inflammatory infiltrates were observed in the superficial dermis (13/13; 100%), deep dermis (12/13; 92%), and perivascular locations (10/13; 77%). Occasionally, there was mural folliculitis (5/13; 38%), intraluminal folliculitis (4/13; 31%), and furunculosis (3/13; 23%). The distribution of the inflammatory infiltrate was predominantly multifocal to coalescent (11/13; 85%) or multifocal (2/13; 15%). The intensity of the inflammatory infiltrate ranged from mild (5/13; 38%) to moderate (3/13; 23%) to severe (5/13; 38%). Other dermal findings included hemorrhage (7/13; 54%), proliferation and ectasia of blood vessels (6/13; 46%), pigmentary incontinence (5/13; 38%), fibrin thrombi (3/13; 23%), and collagen degeneration (2/13; 15%). No lesions were observed in the adjacent subcutaneous, muscular, or cartilaginous tissues.
Considering the predominant distribution and location of these inflammatory lesions, more than one overlapping inflammatory pattern was observed in the same skin section. Nodular to diffuse (13/13; 100%) and vesicular/pustular intraepidermal (13/13; 100%) dermatitis patterns predominated, followed by perivascular/interstitial dermatitis (10/13; 77%) and folliculitis and furunculosis (5/13; 38%). Individual data regarding epidermal lesions, dermal lesions, and inflammatory lesion patterns are available in Supplemental Tables S3–S5, respectively.
Discussion
Limited investigations of the epidemiologic and pathologic details of dermatitis caused by natural tick infestation in cattle are available in the literature.5,7,9,17 The frequency of tick infestation in cattle in our study (19.5%) was similar to the frequency previously described (10.7%–40.0%).5,7,23 However, cutaneous lesions caused by ticks were observed in only 1.4% of the evaluated animals. This result may be underestimated since the presence of cutaneous lesions caused by tick infestation is not typically considered a significant finding during necropsy, especially when the lesions are mild. Thus, as they are not directly related to the cause of death of the animals, skin sample collection for histological analysis might not occur.
In our study, most of the animals with tick infestation were 24 months of age or younger and female. These findings are similar to those described by other authors and suggest that they could be attributed to lower sanitary control against ectoparasites in very young animals and also to hormonal stress, which would make females more susceptible to parasitism by ticks.6,7 Most cattle with tick infestation were beef breeds, mainly Angus. This breed is the most used breed for beef production in Southern Brazil and is the most susceptible European breed (Bos taurus) to tick infestation, 11 which consequently justifies the higher frequency of this breed in our study.
A limitation of this retrospective case series is that parasitological identification of the tick species involved in each case was not performed. Histological characteristics of the tick also do not allow this identification. R. microplus is the main tick species responsible for parasitizing and compromising productivity in Brazilian livestock. 2 Although this tick species is widely distributed in the studied region, it was not possible to confirm that R. microplus was involved in the cases in this study. Other species (e.g., Amblyomma cajennense and A. sculptum) are occasionally reported parasitizing cattle; however, in these cases, cattle are not the definitive host, and the parasitic load is low. 2 The occurrence of ticks is generally seasonal, and 3–4 generations can be observed in a year (1 generation at each season), which depends on the presence of favorable climatic conditions (e.g., heat and humidity) for their development. 15 In our study, the highest number of cattle with tick infestations and associated cutaneous lesions occurred in the fall, which had a statistically significant difference compared to the other seasons. This finding can be justified by the fact that the fall marks the development of the third generation of ticks in Southern Brazil, when there is a higher concentration of teleogenous ticks in the environment and parasitizing cattle. 15
Gross lesions were characterized by areas with hypotrichosis, alopecia, and scales to erosions, ulcers, and crusts distributed mainly in the axillary region and lateral regions of the face, ears, inguinal region, neck, limbs, and perineum. These findings are similar to those described in cattle and other domestic animals. The clinical course of these lesions varies from weeks to months.9,13 Although the bite is painless in most cases, intense irritation and local itching can occur, which can result in more severe lesions with ulcerations and crusts. 9 Unfortunately, information about the temporal evolution and clinical signs was absent in the clinical history of the animals included in this study. However, extensive and ulcerated gross lesions that usually affected some sites where the animals could scratch themselves (e.g., lateral regions of the face and neck) suggest chronicity associated with intense pruritus and self-trauma. Some histopathological findings (e.g., compact hyperkeratosis, irregular acanthosis, crusts, erosions, ulcers, and mixed inflammatory infiltrate) commonly found in traumatic dermatitis in companion animals 10 were also observed in our cases, which also corroborates with this hypothesis of self-trauma.
In the literature, it has been suggested that host and parasite factors, including the substances in tick salivary secretions, host resistance level, previous contact with the agent, and time of evolution of the lesions, might result in variations in the histopathological lesions in infested humans and animals.4,13,14,17 Tick salivary secretions contain several components, including anti-hemostatic factors (antiplatelet, anticoagulant, anti-vasoconstrictor, and vasodilator), cement, immunosuppressive substances, enzymes, and proteins. 22 Among these components, cement is a substance composed of proteins, lipids, and glycoproteins, secreted in the saliva of most species of ticks belonging to the Ixodidae family, including R. microplus.14,17,20,22 This substance is histologically characterized by amorphous hypereosinophilic material around the mouthparts, seen in all our cases, and is responsible for strong attachment to the host epidermis and forming a more efficient channel for fluid exchange.14,20 The cement also induces a less-intense initial inflammatory response at the attachment site due to the release of anti-inflammatory and immunosuppressive substances that neutralize the defense response by the host, thus restricting the development of lesions only to the dermis and epidermis, 14 which is similar to what was observed in our study.
In our study, more than one pattern of inflammatory lesions were observed in the same animal, which is similar to what is described in the literature in cases of arthropod bites. 1 Both nodular to diffuse and pustular dermatitis were observed in all cases; however, these patterns often coexisted with the perivascular/interstitial dermatitis pattern. The inflammatory infiltrate was predominantly composed of neutrophils and mononuclear cells despite the inflammatory lesions being of parasitic origin. A pure eosinophilic infiltrate was observed in only one case, and in the remaining cases, eosinophils composed a smaller proportion than other cell types. These findings could be associated with the level of host resistance or indicate that these animals have not been previously exposed to ticks.3,19 Tick-susceptible breeds tend to develop an inflammatory response with low eosinophils,3,8,19 which can explain our results. On the other hand, an intense eosinophilic inflammatory reaction is usually observed in resistant breeds and those previously exposed, which interferes with the survival of ticks, since the degranulation of these cells results in the release of toxic products that compromise their feeding. 19 Another lesion described in animals and humans is “tick granulomas,” which consist of persistent nodular lesions.4,13 These granulomas are characterized by diffuse nodular dermatitis composed of lymphocytes, histiocytes, and fewer eosinophils and plasma cells. 13 This condition occurs due to a delayed hypersensitivity reaction, and lymphoid nodules with developing follicles, resembling pseudolymphoma, also may occur.4,9,13 This type of alteration is infrequent and was not observed in our cases.
Another inherent limitation of this study was that the entire body of ticks was not always observed in the histological sections evaluated. Sometimes, only a part of the tick’s body or mouthparts was present next to the cutaneous lesion. Routine histological preparation of a whole tick is challenging due to the tick’s chitinous exoskeleton, which is rigid and impermeable. 26 Specialized techniques and equipment, not always available in laboratories, are more suitable for a detailed evaluation of the tick’s histological structures. 26 A recent study demonstrated that Bouin’s solution is more effective than 10% neutral-buffered formalin for tick fixation, allowing for the generation of sections with more histological details of the parasite’s internal structures. 26 However, in the cases evaluated in this study, tick fixation was performed with 10% neutral-buffered formalin, which probably interfered with obtaining sections of the whole tick.
In conclusion, this study reinforces the different patterns of cutaneous lesions associated with tick infestation in cattle. The lesions occurred mainly in the Angus breed and in the fall. Gross lesions were primarily characterized by hypotrichosis, scales, alopecia, ulcers, crusts, and erosions in the axillary region and lateral regions of the face, ears, inguinal region, neck, limbs, and perineum. The histological lesions were characterized by mixed, neutrophilic, or occasionally eosinophilic dermatitis with intralesional tick mouthparts. These variations in the inflammatory type may be due to different factors related to the type of tick and host. Parasitism by this arthropod should be included in the differential diagnosis of causes of dermatitis in cattle.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231217207 – Supplemental material for Characterization of cutaneous lesions caused by natural tick infestation in cattle
Supplemental material, sj-pdf-1-vet-10.1177_03009858231217207 for Characterization of cutaneous lesions caused by natural tick infestation in cattle by Stella Maris P. Melo, Igor R. Santos, Bruno A. Almeida, Luciana Sonne, Welden Panziera, David Driemeier and Saulo P. Pavarini in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) supported this study.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
